Histopathologic Evaluation of Corneal Tissue After Adjunctive Rose Bengal Photodynamic Antimicrobial Therapy and Keratoplasty in Advanced Acanthamoeba Keratitis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Study Population
2.3. Study Definitions and Outcome Measures
2.4. Rose Bengal Photodynamic Antimicrobial Therapy Procedure
2.5. Histopathologic Analysis
2.6. Microbiologic Analysis
2.7. Statistical Analysis
3. Results
3.1. Participant Characteristics
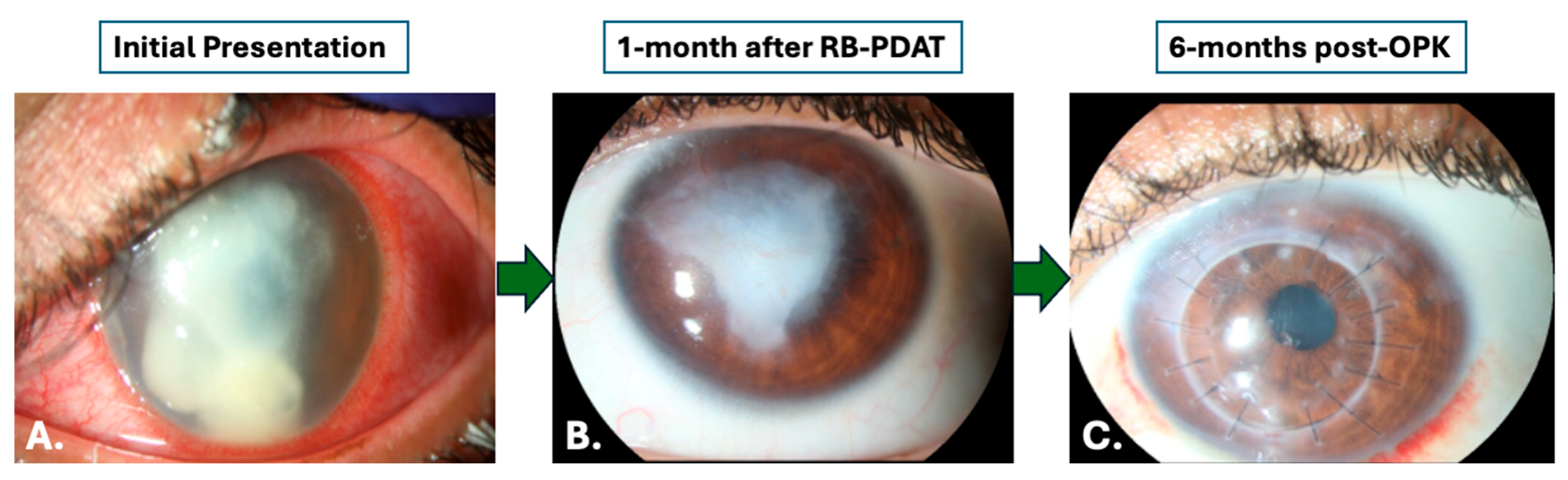
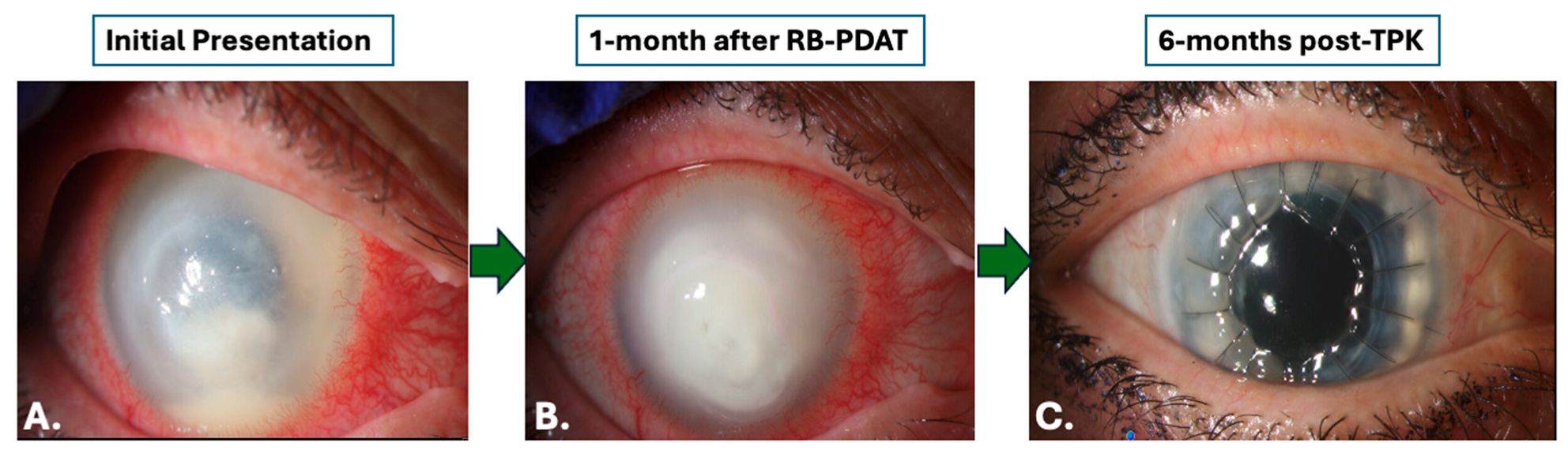
3.2. Treatment
3.3. Microbiologic and Histopathologic Outcomes
4. Discussion
5. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tu, E.Y.; Joslin, C.E.; Sugar, J.; Shoff, M.E.; Booton, G.C. Prognostic factors affecting visual outcome in Acanthamoeba keratitis. Ophthalmology 2008, 115, 1998–2003. [Google Scholar] [CrossRef]
- De Lacerda, A.G.; Lira, M. Acanthamoeba keratitis: A review of biology, pathophysiology and epidemiology. Ophthalmic Physiol. 2021, 41, 116–135. [Google Scholar] [CrossRef]
- Yodar, J.S.; Verani, J.; Heidman, N.; Hoppe-Bauer, J.; Alfonso, E.C.; Miller, D.; Jones, D.B.; Bruckner, D.; Langston, R.; Jeng, B.H.; et al. Acanthamoeba keratitis: The persistence of cases following a multistate outbreak. Ophthalmic Epidemiol. 2012, 19, 221–225. [Google Scholar] [CrossRef]
- Kaufman, A.R.; Tu, E.Y. Advances in the management of Acanthamoeba keratitis: A review of the literature and synthesized algorithmic approach. Ocul. Surf. 2022, 25, 26–36. [Google Scholar] [CrossRef]
- Varacalli, G.; Di Zazzo, A.; Mori, T.; Dohlman, T.H.; Spelta, S.; Coassin, M.; Bonini, S. Challenges in Acanthamoeba keratitis: A review. J. Clin. Med. 2021, 10, 942. [Google Scholar] [CrossRef]
- Tu, E.Y.; Joslin, C.E.; Sugar, J.; Booton, G.C.; Shoff, M.E.; Fuerst, P.A. The relative value of confocal microscopy and superficial corneal scrapings in the diagnosis of Acanthamoeba keratitis. Cornea 2008, 27, 764–772. [Google Scholar] [CrossRef]
- Goh, J.W.Y.; Harrison, R.; Hau, S.; Alexander, C.L.; Tole, D.M.; Avadhanam, V.S. Comparison of in vivo confocal microscopy, PCR and culture of corneal scrapes in the diagnosis of Acanthamoeba keratitis. Cornea 2018, 37, 480–485. [Google Scholar] [CrossRef] [PubMed]
- Yera, H.; Ok, V.; Kuet, F.L.K.; Dahane, N.; Ariey, F.; Hasseine, L.; Delaunay, P.; Martiano, D.; Marty, P.; Bourges, J.L. PCR and culture for diagnosis of Acanthamoeba keratitis. Br. J. Ophthalmol. 2021, 105, 1302–1306. [Google Scholar] [CrossRef] [PubMed]
- Papa, V.; Rama, P.; Radford, C.; Minassian, D.C.; Dart, J.K.G. Acanthamoeba keratitis therapy: Time to cure and visual outcome analysis for different antiamoebic therapies in 227 cases. Br. J. Ophthalmol. 2020, 104, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Naranjo, A.; Arboleda, A.; Martinez, J.D.; Durkee, H.; Aguilar, M.C.; Relhan, N.; Nikpoor, N.; Galor, A.; Dubovy, S.R.; Leblanc, R.; et al. Rose bengal photodynamic antimicrobial therapy for patients with progressive infectious keratitis: A pilot study. Am. J. Ophthalmol. 2019, 208, 387–396. [Google Scholar] [CrossRef]
- Sepulveda-Beltran, P.; Levine, H.; Altamirano, D.S.; Martinez, J.D.; Durkee, H.; Mintz, K.; Leblanc, R.; Tothova, J.D.; Miller, D.; Parel, J.M.; et al. Rose Bengal photodynamic antimicrobial therapy: A review of the intermediate-term clinical and surgical outcomes. Am. J. Ophthalmol. 2022, 243, 125–134. [Google Scholar] [CrossRef]
- Martinez, J.D.; Arrieta, E.; Naranjo, A.; Monsalve, P.; Mintz, K.J.; Peterson, J.; Arboleda, A.; Durkee, H.; Aguilar, M.C.; Palaez, D.; et al. Rose bengal photodynamic antimicrobial therapy: A pilot safety study. Cornea 2021, 40, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- Yokagawa, H.; Kobayashi, A.; Yamazaki, N.; Ishibashi, Y.; Oikawa, Y.; Tokoro, M.; Sugiyama, K. Bowman’s layer encystment in cases of persistent Acanthamoeba keratitis. Clin. Ophthalmol. 2012, 6, 1245–1251. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Huang, P.; Tepelus, T.; Vickers, L.A.; Baghdasaryan, E.; Huang, J.; Irvine, J.; Hsu, H.Y.; Saddda, S.; Lee, O.L. Quantitative analysis of depth, distribution, and density of cysts in Acanthamoeba keratitis using confocal microscopy. Cornea 2017, 36, 927–932. [Google Scholar] [CrossRef] [PubMed]
- Amescua, G.; Arboleda, A.; Nikpoor, N.; Durkee, H.; Relhan, N.; Aguilar, M.C.; Flynn, H.W.; Miller, D.; Parel, J.M. Rose Bengal Photodynamic Antimicrobial Therapy: A novel treatment for resistant fusarium keratitis. Cornea 2017, 36, 1141–1144. [Google Scholar] [CrossRef] [PubMed]
- Chopra, R.; Mulholland, P.J.; Hau, S.C. Invivo confocal microscopy morphologic features and cyst density in Acanthamoeba keratitis. Am. J. Ophthalmol. 2020, 217, 38–48. [Google Scholar] [CrossRef]
- Kobayashi, A.; Ishibashi, Y.; Oikawa, Y.; Yokagawa, H.; Sugiyama, K. In vivo and ex vivo laser confocal microscopy findings in patients with early-stage Acanthamoeba keratitis. Cornea 2008, 27, 439–445. [Google Scholar] [CrossRef]
- Thulasi, P.; Saeed, H.N.; Rapuano, C.J.; Hou, J.H.; Appenheimer, A.B.; Chodosh, J.; Kang, J.J.; Morrill, A.M.; Vyas, N.; Zegans, M.E.; et al. Oral miltefosine as salvage therapy for refractory acanthamoeba keratitis. Am. J. Ophthalmol. 2021, 223, 75–82. [Google Scholar] [CrossRef]
- Naranjo, A.; Martinez, J.D.; Miller, D.; Tonk, R.; Amescua, G. Systemic miltefosine as an adjunct treatment of progressive acanthamoeba keratitis. Ocul. Immunol. Inflamm. 2021, 29, 1576–1584. [Google Scholar] [CrossRef]
- Cherfan, D.; Verter, E.E.; Melki, S.; Gisel, T.E.; Doyle, F.J., Jr.; Scarcelli, G.; Yun, S.H.; Redmond, R.W.; Kochevar, I.E. Collagen cross-linking using rose bengal and green light to increase corneal stiffness. Investig. Ophthalmol. Vis. Sci. 2013, 54, 3426–3433. [Google Scholar] [CrossRef]
- Wertheimer, C.M.; Mendes, B.; Pei, Q.; Brandt, K.; Kochevar, I.E. Arginine as an enhancer in rose bengal photosensitized corneal crosslinking. Transl. Vis. Sci. Technol. 2020, 9, 24. [Google Scholar] [CrossRef] [PubMed]
- Naranjo, A.; Pelaez, D.; Arrieta, E.; Salero-Coca, E.; Martinez, J.D.; Sabater, A.L.; Amescua, G.; Parel, J.M. Cellular and molecular assessment of rose Bengal photodynamic antimicrobial therapy on keratocytes, corneal endothelium and limbal stem cell niche. Exp. Eye Res. 2019, 188, 1078080. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.D.; Naranjo, A.; Amescua, G.; Dubovy, S.R.; Arboleda, A.; Durkee, H.; Aguilar, M.C.; Flynn, H.W., Jr.; Miller, D.; Parel, J.M. Human corneal changes after rose bengal photodynamic antimicrobial therapy for treatment of fungal keratitis. Cornea 2018, 37, e46–e48. [Google Scholar] [CrossRef]
- Aribas, A.B.T.; Sarikaya, B.; Bilgihan, K. Iontophoresis-assisted rose bengal and green light corneal cross-linking. Cornea 2020, 39, 1533–1540. [Google Scholar] [CrossRef] [PubMed]


| Parameter | |
|---|---|
| Gender, no. (%) | |
| Male | 5 (29) |
| Female | 12 (71) |
| Affected eye, no. (%) | |
| Right | 7 (41) |
| Left | 10 (59) |
| Age at time of surgical intervention (years) | |
| Mean, (Standard Deviation) | 44 (16) |
| Median | 44 |
| Range | 17–65 |
| Type of surgical intervention, no. (%) | |
| Therapeutic Penetrating Keratoplasty | 8 (47) |
| Optical Penetrating Keratoplasty | 9 (41) |
| Deep Anterior Lamellar Keratoplasty | 2 (12) |
| Follow-up time, months | |
| Mean, (Standard Deviation) | 30 (15) |
| Contact lens user, no. (%) | 15 (88) |
| Patient Group | Treatment, No. (%) | |||
|---|---|---|---|---|
| Chlorhexidine 0.02% Ophthalmic Solution | Polyhexamethylene Biguanide 0.02% Ophthalmic Solution | Voraconazole 50/200 mg Tablet | Miltefosine 50 mg Caps | |
| All patients (n = 17) | 16 (94) | 13 (77) | 7 (41) | 7 (41) |
| TPK only (n = 8) | 7 (88) | 6 (75) | 4 (50) | 5 (63) |
| OPK/DALK only (n = 9) | 9 (100) | 7 (78) | 3 (33) | 2 (22) |
| Parameter | |
|---|---|
| Method of Diagnosis of Acanthamoeba Keratitis, no. (%) | |
| Corneal scrape | 14 (82) |
| Corneal biopsy | 3 (18) |
| Therapeutic Penetrating Keratoplasty Group, no. (%) | |
| Confocal microscopy findings: | |
| Positive for cysts | 3 (38) |
| Unclear/non-diagnostic | 1 (13) |
| Negative | 1 (13) |
| Confocal not performed | 3 (38) |
| Positive for cysts on corneal tissue pathology analysis | 6 (75) |
| Culture of corneal host button at time of surgery: | |
| Positive for Acanthamoeba | 3 (38) |
| Negative for Acanthamoeba | 5 (63) |
| Culture not performed | 0 (0) |
| Average Acanthamoeba cyst depth from Descemet’s layer | 42.0 ± 52.5 μm |
| Average number of grading cysts | 1.3 ± 0.7 |
| Average corneal button thickness | 661.7 ± 106.5 μm |
| Reinfection following surgical intervention | 1 (13) |
| Time from symptom onset to AK diagnosis | 35.0 ± 35.1 days |
| Optical Penetrating Keratoplasty and Deep Anterior Lamellar Keratoplasty Group, no. (%) | |
| Confocal microscopy findings: | |
| Positive for cysts | 1 (11) |
| Unclear/non-diagnostic | 1 (11) |
| Negative | 0 (0) |
| Confocal not performed | 7 (78) |
| Positive for cysts on corneal tissue pathology analysis | 3 (33) |
| Culture of corneal host button at time of surgery: | |
| Positive for Acanthamoeba | 0 (0) |
| Negative for Acanthamoeba | 2 (22) |
| Culture not performed | 7 (78) |
| Average Acanthamoeba cyst depth from Descemet’s layer | 261.7 ± 222.7 μm |
| Average number of grading cysts | 1.3 ± 0.9 |
| Average corneal button thickness | 474.2 ± 126.6 μm |
| Reinfection following surgical intervention | 0 (0) |
| Time from symptom onset to AK diagnosis | 78.1 ± 114.3 days |
| p-values following unpaired t-tests comparing TPK to OPK/DALK | |
| Average Acanthamoeba cyst depth from Descemet’s layer | 0.016 |
| Average number of grading cysts | 1.000 |
| Average corneal button thickness | 0.005 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, J.J.; Navia, J.C.; Huang, J.M.; Camacho, M.; Tan, C.H.; Sepulveda-Beltran, P.A.; Mustafa, S.; Durkee, H.; Arboleda, A.; Aguilar, M.C.; et al. Histopathologic Evaluation of Corneal Tissue After Adjunctive Rose Bengal Photodynamic Antimicrobial Therapy and Keratoplasty in Advanced Acanthamoeba Keratitis. J. Clin. Med. 2025, 14, 6104. https://doi.org/10.3390/jcm14176104
Huang JJ, Navia JC, Huang JM, Camacho M, Tan CH, Sepulveda-Beltran PA, Mustafa S, Durkee H, Arboleda A, Aguilar MC, et al. Histopathologic Evaluation of Corneal Tissue After Adjunctive Rose Bengal Photodynamic Antimicrobial Therapy and Keratoplasty in Advanced Acanthamoeba Keratitis. Journal of Clinical Medicine. 2025; 14(17):6104. https://doi.org/10.3390/jcm14176104
Chicago/Turabian StyleHuang, Jordan J., Juan Carlos Navia, Joshua M. Huang, Matthew Camacho, Charissa H. Tan, Paula A. Sepulveda-Beltran, Sara Mustafa, Heather Durkee, Alejandro Arboleda, Mariela C. Aguilar, and et al. 2025. "Histopathologic Evaluation of Corneal Tissue After Adjunctive Rose Bengal Photodynamic Antimicrobial Therapy and Keratoplasty in Advanced Acanthamoeba Keratitis" Journal of Clinical Medicine 14, no. 17: 6104. https://doi.org/10.3390/jcm14176104
APA StyleHuang, J. J., Navia, J. C., Huang, J. M., Camacho, M., Tan, C. H., Sepulveda-Beltran, P. A., Mustafa, S., Durkee, H., Arboleda, A., Aguilar, M. C., Miller, D., Parel, J.-M., Amescua, G., Dubovy, S. R., & Martinez, J. D. (2025). Histopathologic Evaluation of Corneal Tissue After Adjunctive Rose Bengal Photodynamic Antimicrobial Therapy and Keratoplasty in Advanced Acanthamoeba Keratitis. Journal of Clinical Medicine, 14(17), 6104. https://doi.org/10.3390/jcm14176104







