Feasibility and Safety of Adopting a New Approach in Delivering a 450 nm Blue Laser with a Flattop Beam Profile in Vital Tooth Whitening. A Clinical Case Series with an 8-Month Follow-Up
Abstract
1. Introduction
1.1. The Photoactivation of Bleaching Agents: Mechanism of Action
1.2. The Controversial Link between the Bleaching Agent and Its Adverse Effects
1.3. Chosen Bleaching Agent Properties
1.4. Laser versus Light-Emitted Diode (LED)-Assisted Bleaching
2. Materials and Methods
2.1. Study Design
2.2. Eligibility Criteria
- Inclusion Criteria
- Fit and healthy subjects of both genders aged ≥18 years old presenting with discoloured upper and lower teeth in the region of UR5-UL5 and LR5-LL5, respectively, based on the VITA colour guide (Vita Easyshade®; VITA Zahnfabrik H. Rauter GmbH & Co., KG, Bad Säckingen, Germany);
- Subjects with no active intraoral lesions, active carious lesions, or any previous tooth sensitivity; subjects with external vital tooth discolouration were in the inclusion criteria.
- Exclusion Criteria
- Subjects with allergic reactions to any components in the commercial bleaching agents, such as peroxides, latex, and dyes;
- Subjects with systematic diseases;
- Subjects who could not commit to follow-up appointments;
- Pregnant and lactating women;
- Subjects with severe periodontal conditions;
- Subjects who had hypersensitivity to light.
2.3. Patient Protocol Prior to Bleaching Treatment
- At least one week prior to the treatment, patients underwent a professional dental hygiene session (ultrasonic and guided biofilm therapy protocols);
- Full medical and social histories were taken from the patient on the day of the bleaching treatment;
- The oral soft and hard tissues were fully examined to verify any possible problems or risks in performing the bleaching treatment;
- The patient’s initial colour shade was recorded using the VITA colour guide (Vita Easyshade®; VITA Zahnfabrik H. Rauter GmbH & Co., KG, Bad Säckingen, Germany);
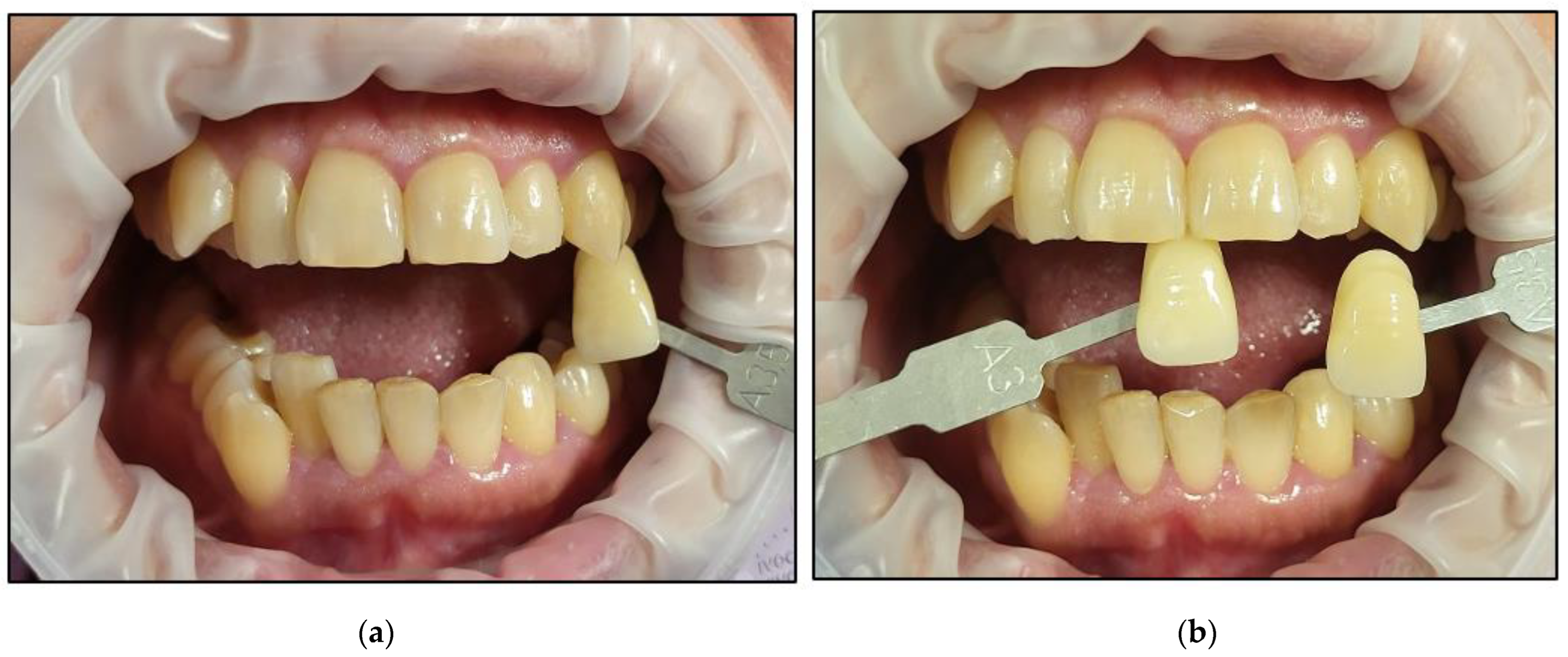
- The colour of a darkened tooth was identified (Figure 1). The upper left canine was used as a colour reference because it is the most saturated tooth in the arch (greater dentin mass and volume of intrinsic pigment). Hence, the canine could be considered a good reference point in selecting a shade because they have the highest chroma (intensity) of the dominant hue (colour) of the teeth [44];
- Pre-treatment photos were taken;
- The patients were provided with a full explanation of the bleaching treatment, including all its associated steps. Informed written consent was obtained from all the patients prior to the treatment in relation to the bleaching treatment, photographs, and publishing of the study in a scientific journal.
2.4. The Bleaching Agent
2.4.1. BlancOne® ULTRA+
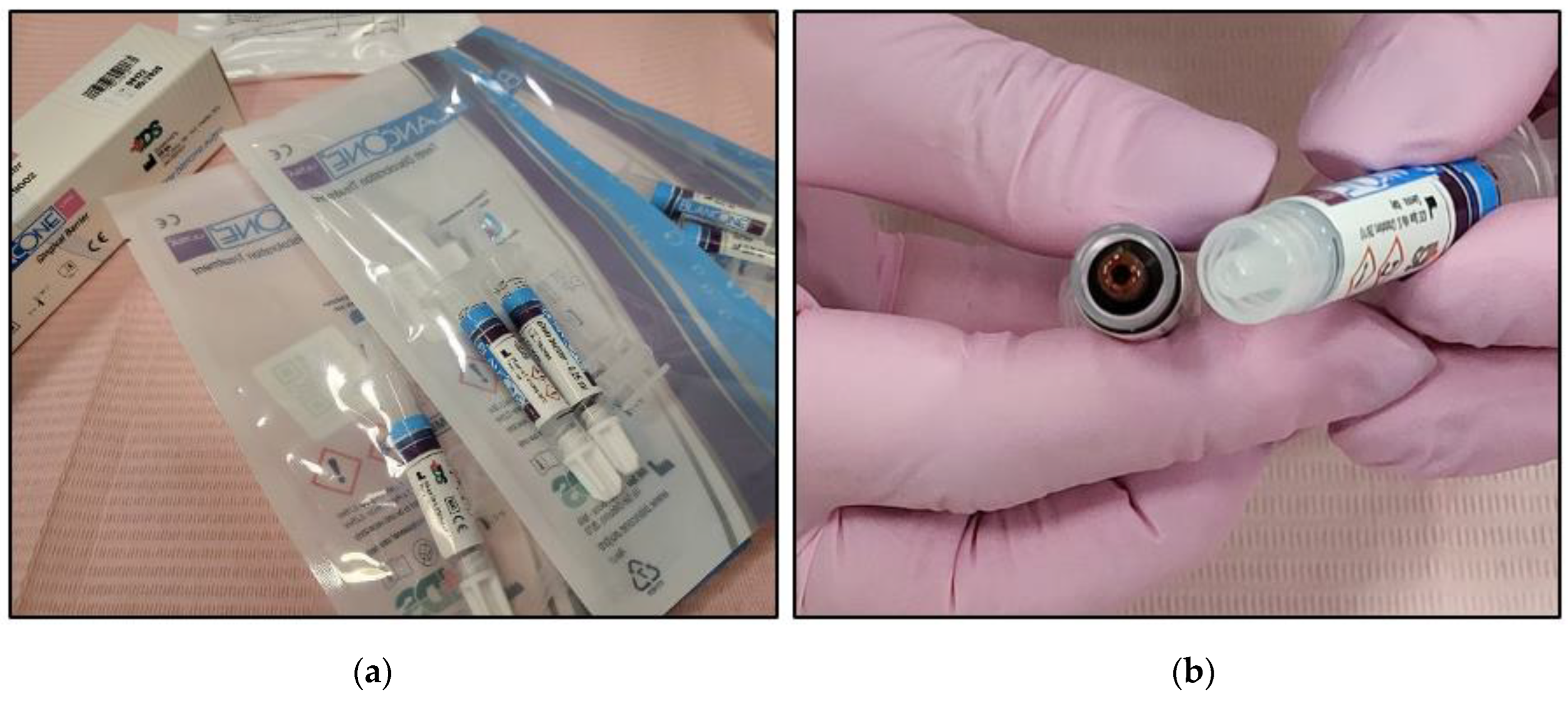
2.4.2. BlancOne ULTRA+ Preparation
- The two syringes’ caps were removed and then they were connected via a connector;
- The two components were mixed by transferring them several times from one syringe to the other until a homogeneous mix was achieved (approximately 20 passages back and forth) (Figure 3);
- The full amount of the mixed gel was transferred into one of the two syringes;
- Transparent applicator tip was inserted.
2.5. Steps for the Bleaching Treatment Protocol
2.5.1. Patient Preparation Protocol
- The cheeks and lips were prepared by isolating them with a silicon mouth retractor “OptraGate”-latex-free (Ivovlar Vivadent, Opfikon, Switzerland). OptraGate is a single-use, latex-free lip and cheek retractor that provides increased visibility and accessibility during dental procedures. It is highly flexible and elastic in all directions of movement for patient comfort and assists in keeping the patient’s mouth open;
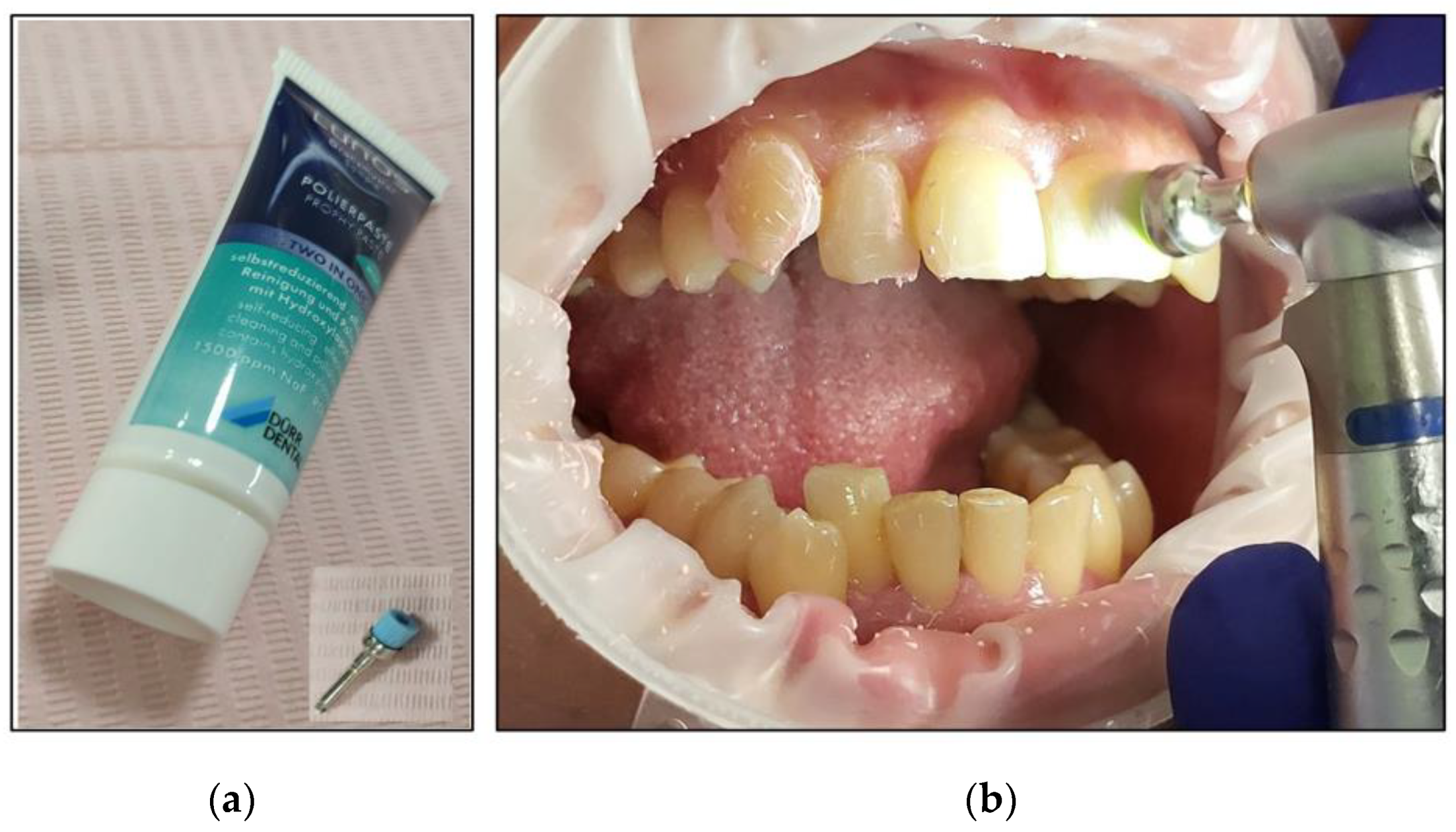
- The salivary biofilm was removed from the buccal surfaces of the recruited teeth with a brush and prophylactic paste (Lunos® Prophy Paste Super Soft, DÜRR DENTAL, Kettering, UK) (Figure 4a). The utilised brush (Figure 4a) was applied perpendicular to the tooth surface and rotated to clean in a circulating motion (Figure 4b);
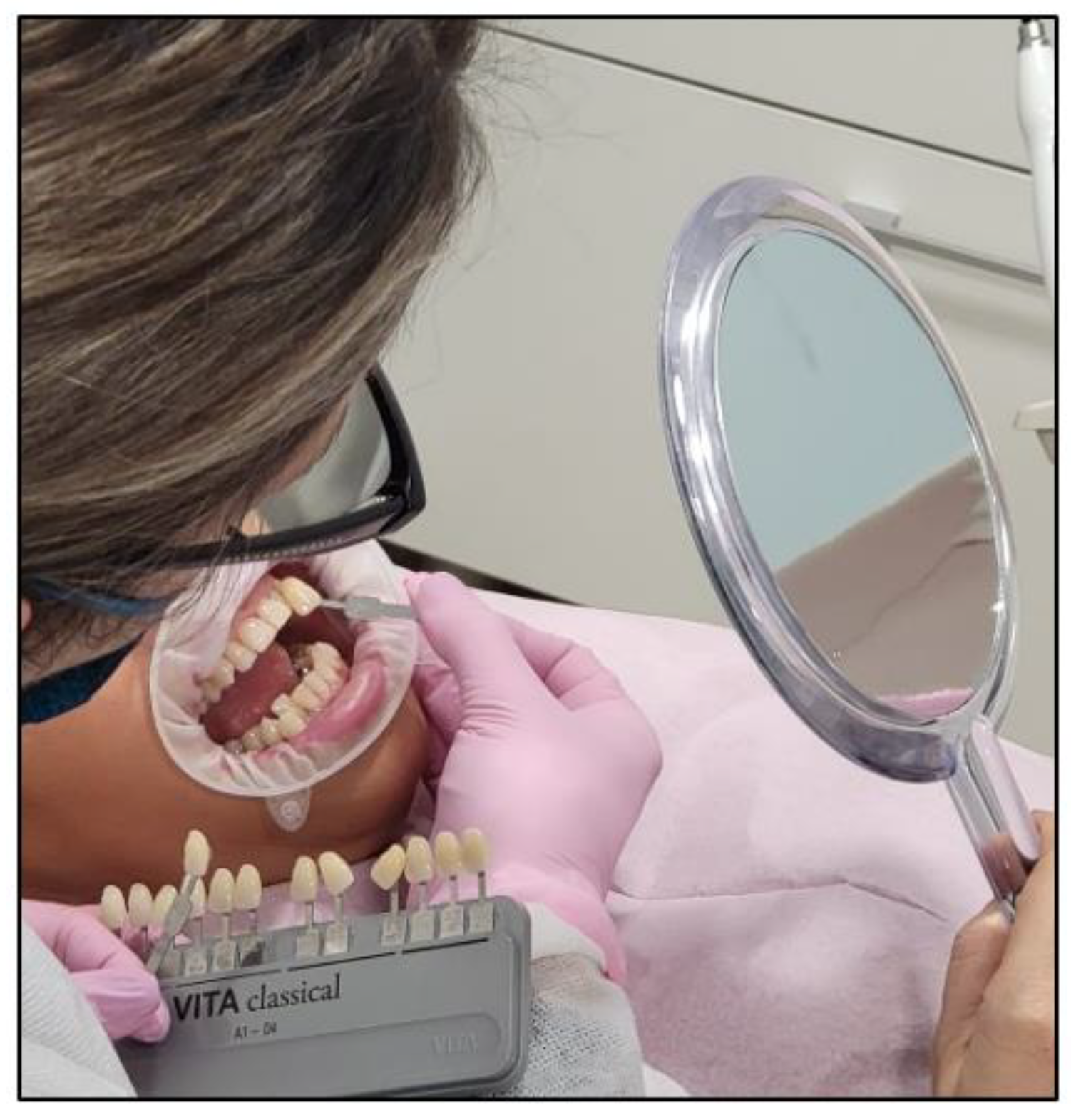
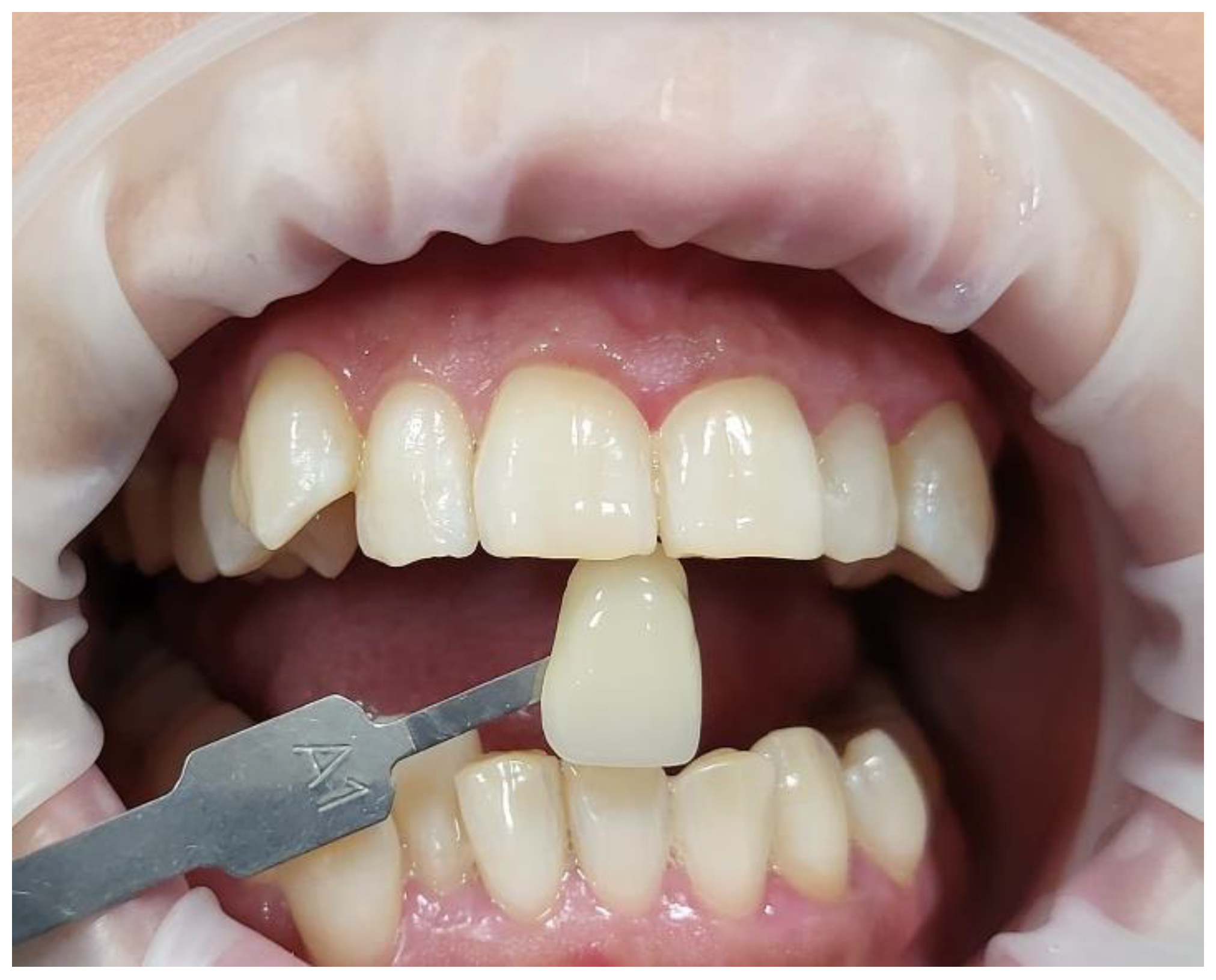
- The pre-treatment colour shade was identified using the VITA classical A1-D4® shade guide (VITA Zahnfabrik, Bad Säckingen, Germany), which is used especially for the whitening treatment. The tooth shade was placed near the buccal surface of the intended tooth that required bleaching (Figure 5);
- Pre-treatment photos were taken;
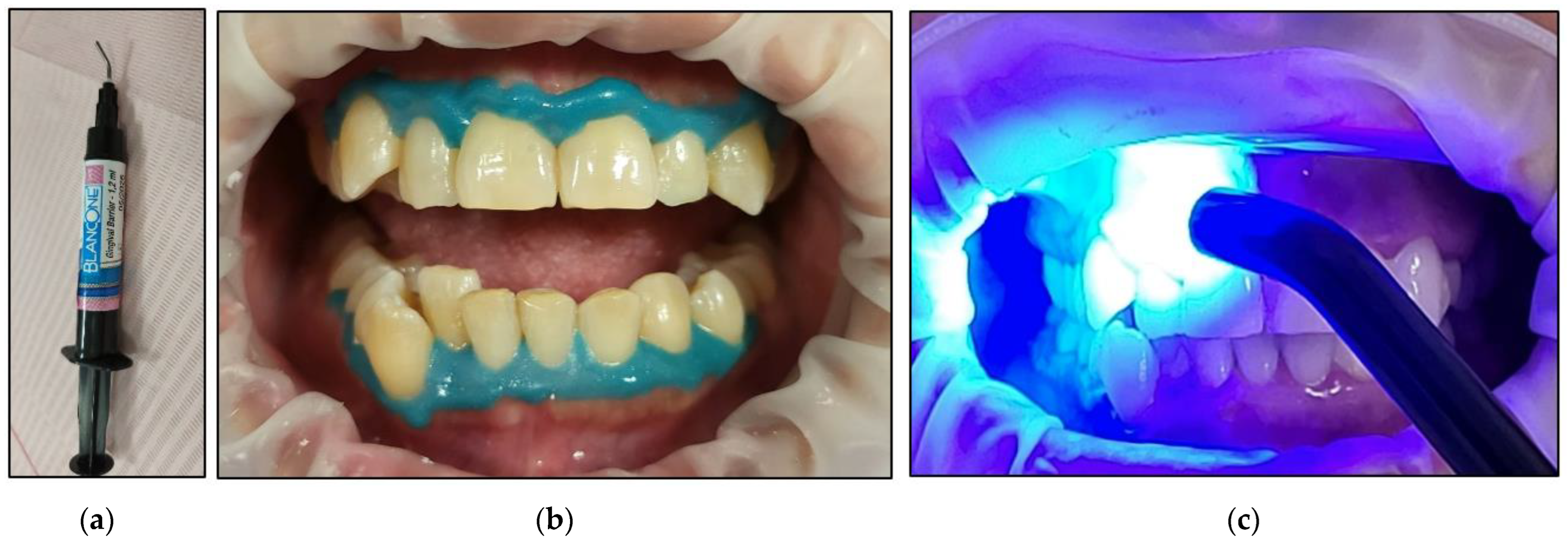
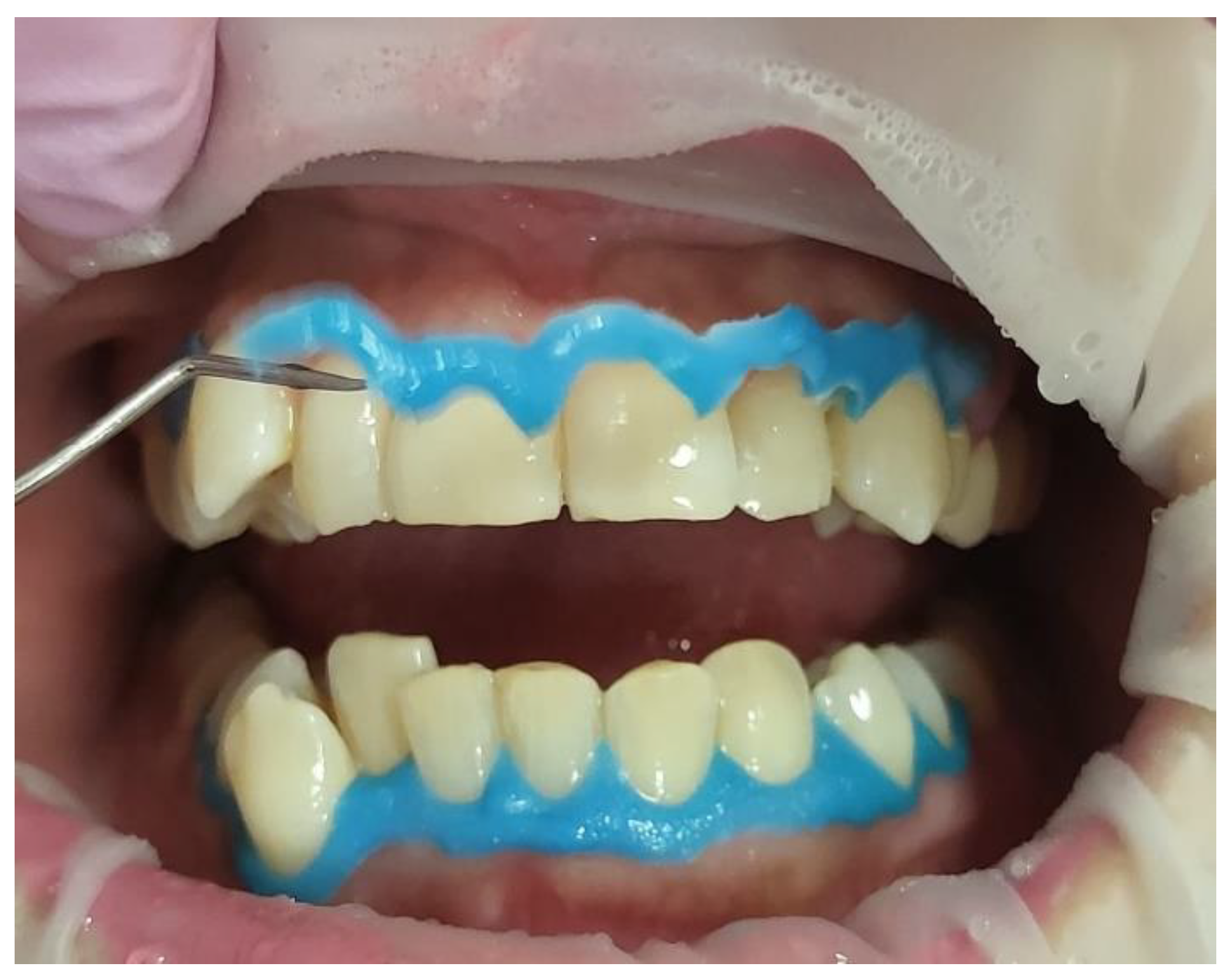
- Prior to applying the bleaching agent, the gingivae surround UR5-UL5 and LR5-LL5 (i.e., the tissue-free gingival margin and the papillae between the treated teethy) were isolated with a light-curing BLANCONE gingival-barrier liquid dam (BlancOne® CARE, IDS, Savona, Italy) (Figure 6a,b), after achieving a completely dry field. Then, a Woodpecker LED-B photopolymer lamp (Woodpecker, Beijing, China) was used to photopolymerise the gingival barrier (Figure 6c), which took between 20 s and 23 s. Suitable protective eyewear was worn.
2.5.2. Whitening Gel Application and Photoactivation Protocols
- All of the health and safety protocols were implemented. The patient, the operator, and the dental nurse wore the appropriate protective eyewear for the λ 450 nm laser during the entire bleaching treatment;
- The BlancOne ULTRA+ whitening gel was applied on the outer surface of the following teeth that required whitening (UR5-UL5 and LR5-LL5) by gently pushing the gel syringe piston in a thin layer ~2–3 mm in thickness (Figure 7);
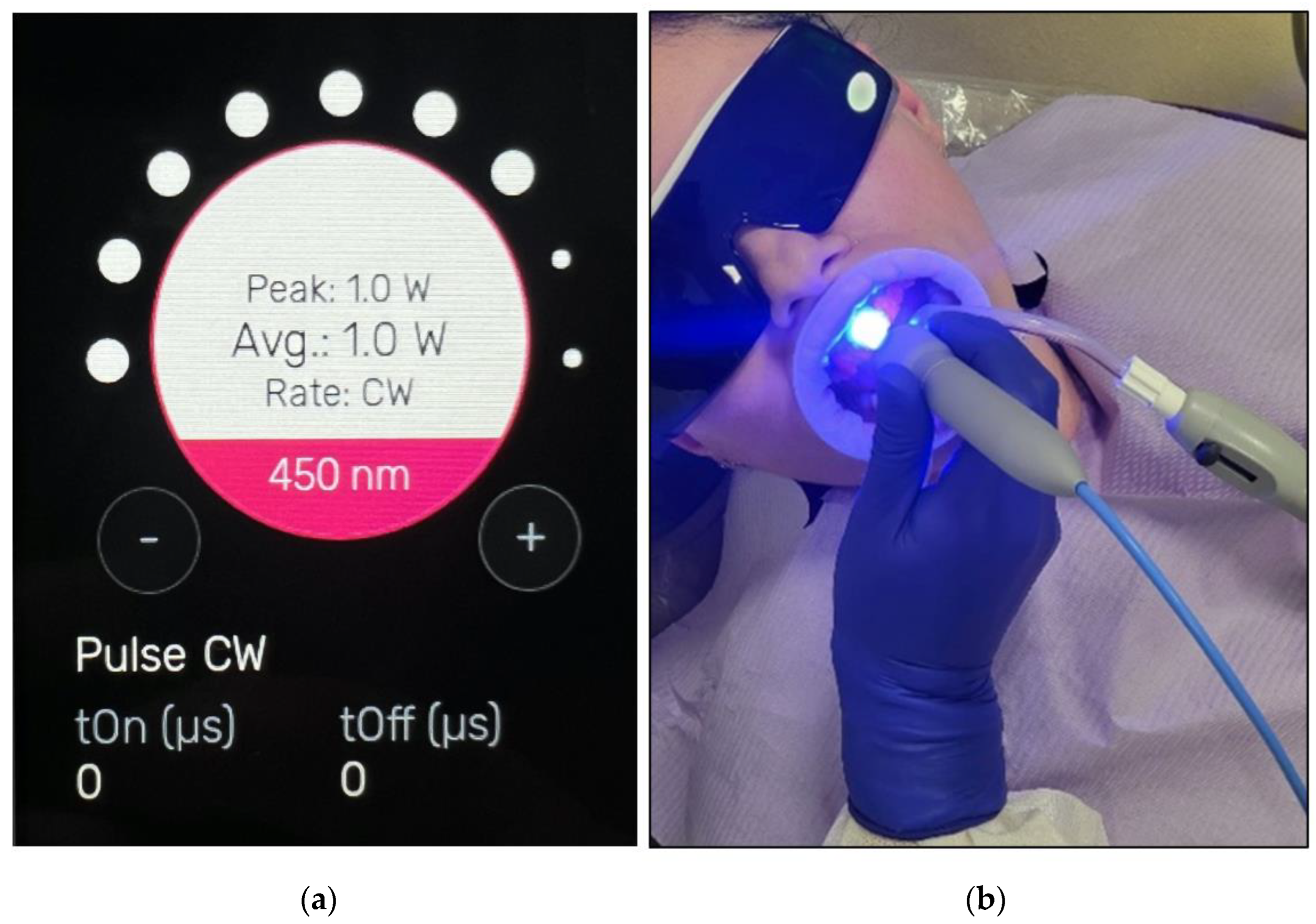
- The buccal surfaces of the treated teeth were photoactivated with a λ 450 nm laser with photonic energy delivered with a flattop handpiece immediately after the gel was applied for 15 s (Figure 8). Figure 8 clearly shows the laser’s light interaction with the gel, indicating an effective photoactivation process whereby photonic energy is equally distributed on the buccal surface of the treated tooth via a flattop delivery handpiece.
- Whitening Cycle Protocol
- The bleaching gel was applied to the teeth in a thin layer;
- Fifteen seconds irradiation time per spot was used to photoactivate the bleaching gel with λ 450 nm photonic energy delivered using a flattop handpiece. There were five spots per arch. The time required for the photoactivation of both arches was ~three minutes (min);
- The gel was rested on the teeth’s surface for 8 min between each whitening cycle.
- The gel was removed using dental suction (Figure 9a), and then the dental surfaces were thoroughly rinsed (Figure 9b) with continuous suction. Then, all the teeth were dried with cotton rolls. This process was repeated at the end of each cycle. Freshly prepared gel applications for each whitening cycle were performed;
- A new layer of whitening gel was applied;
- The whitening cycle was repeated three times in one session with an 8 min gel resting period (thermal relaxation) between each cycle.
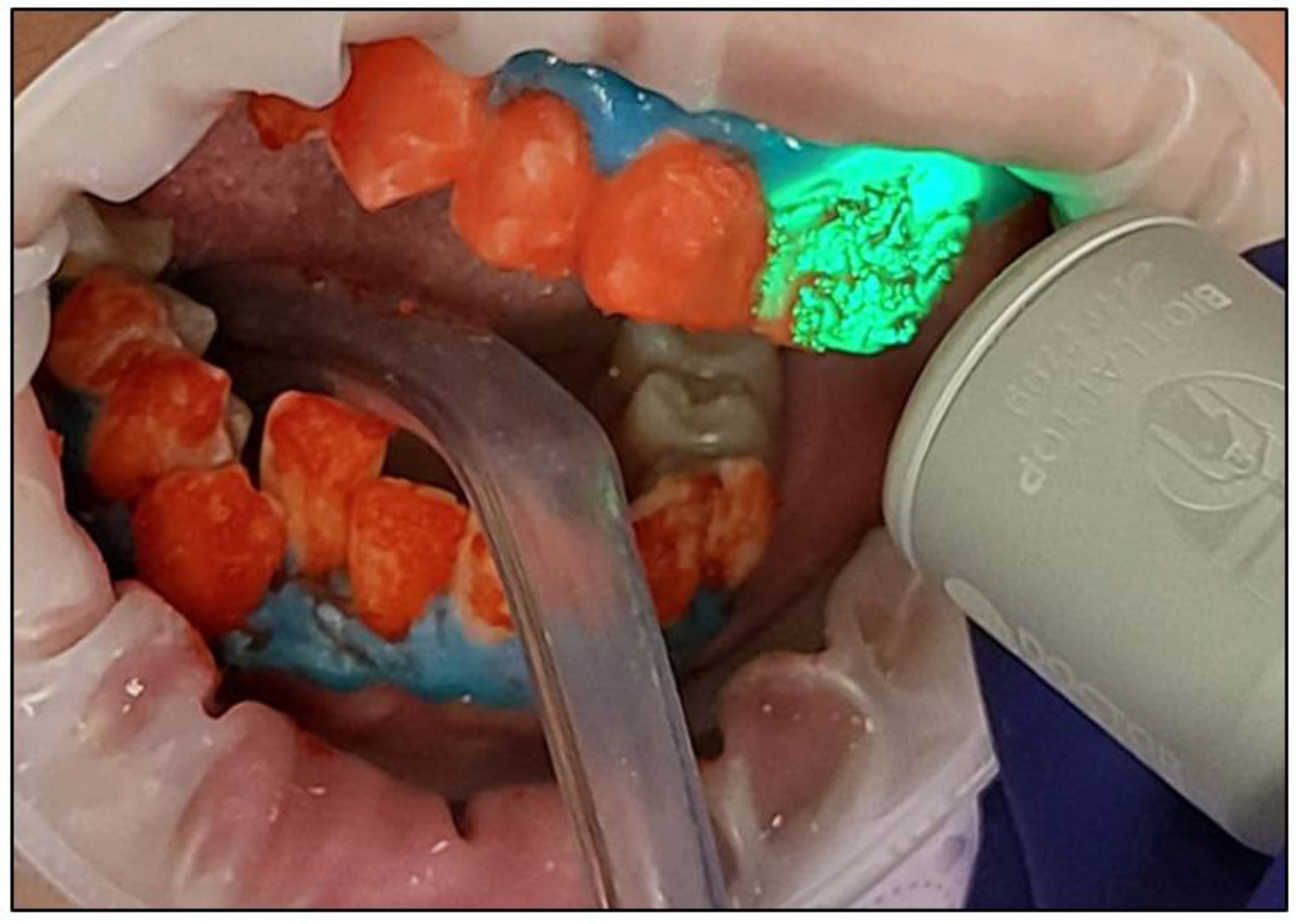
- After three whitening cycles, the gingival protective barrier was gently removed with a scaler, as shown in Figure 10;
- Figure 11 shows a clinical photo of case #1 immediately after three whitening cycles, obtaining the A1 shade colour;
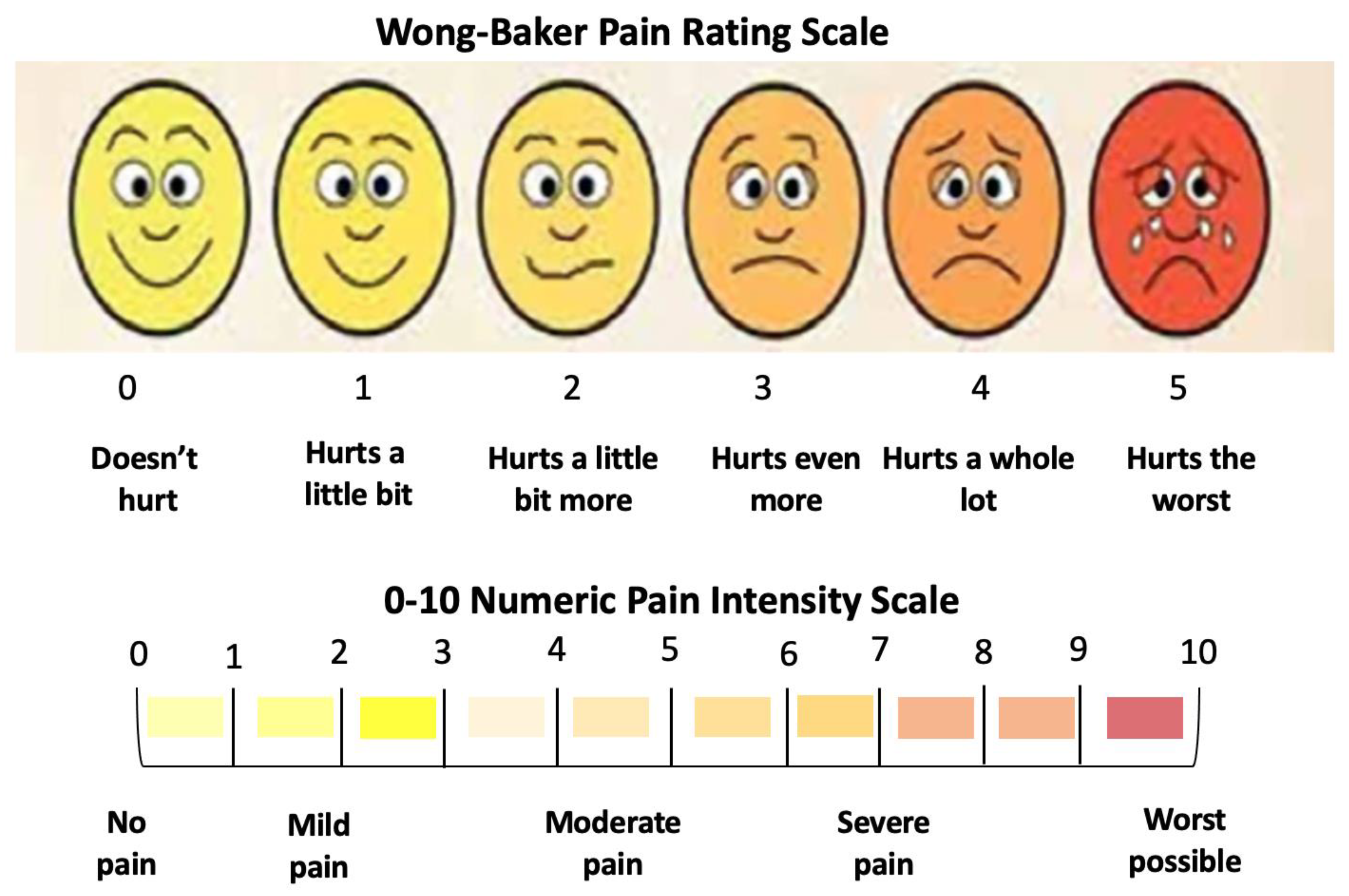
- The dental hypersensitivity/pain was reported based on the patient’s self-reporting of the VAS at pre-treatment (T0), during the three whitening cycles, at the end of three whitening cycles (T1), 24 h (h) (T2) and 48 h (T3) post-treatment and 8-month follow-up (T4);
- The colour shade at T0, T1, and T4 were recorded by two experienced independent assessors;
- The gingival irritation was assessed during the whitening cycles, at T1 in the clinic, and at T2 and T3 via a telephone call;
- The patient’s treatment satisfaction was evaluated at T1 at the clinic and at T2 and T3 via a telephone call;
- Post-treatment instruction leaflets were provided to all the patients to ensure good treatment maintenance.
2.6. Photoactivation Protocol and Laser Dosimetry
2.7. Outcome Assessment Tools
2.7.1. Colour Shade Guide
2.7.2. Visual Analogue Scale
2.7.3. Löe and Silness Gingival Index
2.7.4. Patient Satisfaction
2.8. Statistical Analysis
3. Results
3.1. Demographic Characteristics
3.2. Assessment of Whitening Outcomes
- Case #1
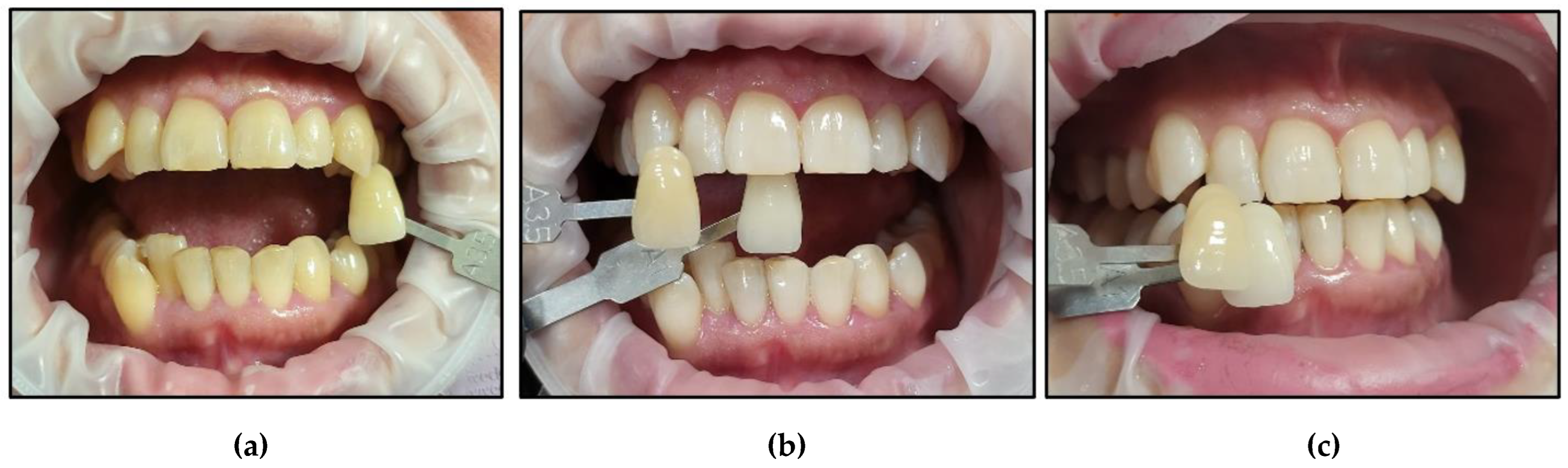
- Case #2

- Case #3
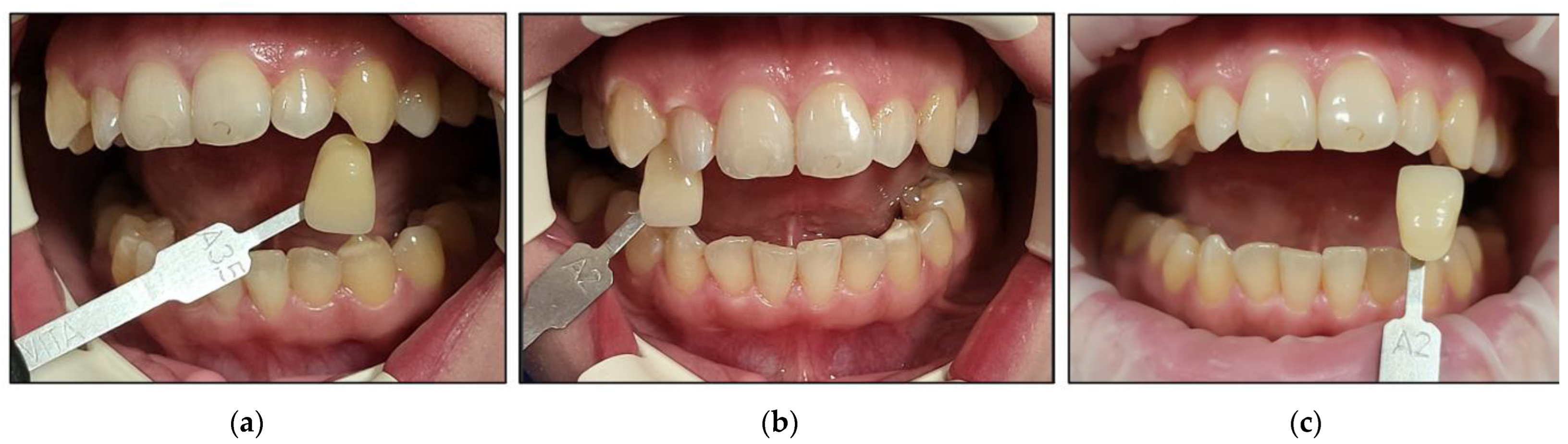
- Case #4
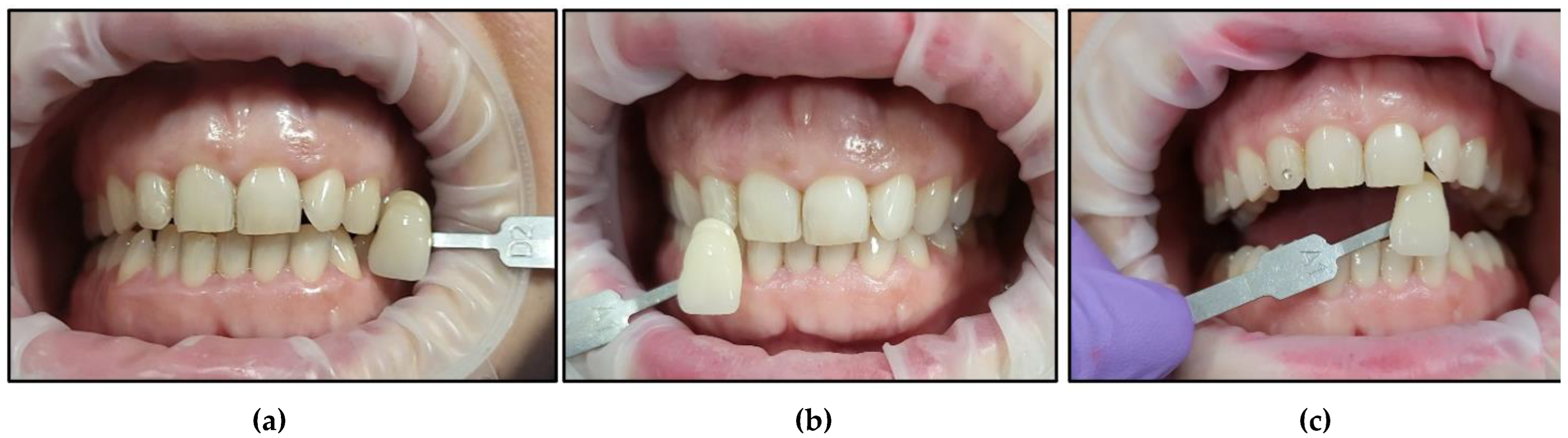
- Case #5

- Case #6
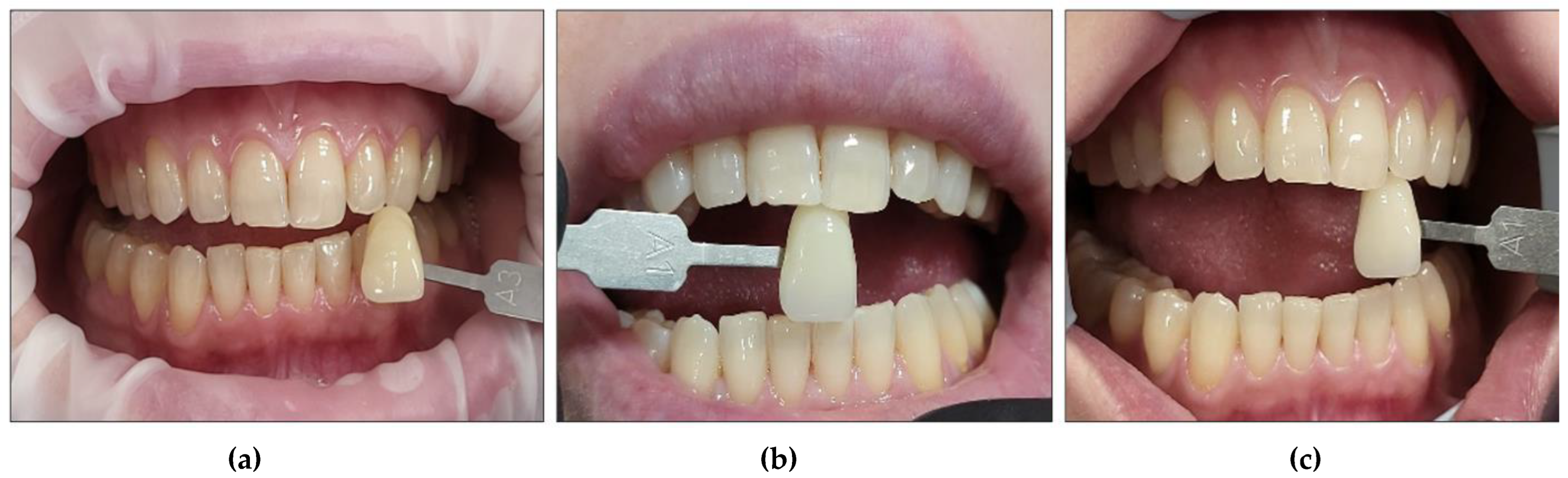
3.3. Pain and Bleaching-Induced Tooth Sensitivity Assessment
3.4. Assessment of Gingival Irritation
3.5. Evaluation of Patient Satisfaction
4. Discussion
4.1. Appraising the Optimised Whitening Outcomes
4.1.1. Evaluating the Concept of H2O2 Concentration in the Bleaching Agent
4.1.2. Laser vs. LED Assessment in Dental Bleaching
4.1.3. Bleaching Application Time
4.1.4. Temperature
4.2. Evaluation of Post-Operative Complications
4.3. Bleaching Treatment Safety, Feasibility, and Satisfaction
4.4. Study Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bezerra-Júnior, D.M.; Silva, L.M.; Martins, L.M.; Cohen-Carneiro, F.; Pontes, D.G. Esthetic rehabilitation with tooth bleaching, enamel microabrasion, and direct adhesive restorations. Gen. Dent. 2016, 64, 60–64. [Google Scholar] [PubMed]
- Mon, T.; Norkhafizah, S.; Nurhidayati, H. Factor influencing patient satisfaction with dental appearance and treatments they desire to improve aesthetics. BMC Oral Health 2011, 11, 6. [Google Scholar] [CrossRef]
- Pavicic, D.; Kolceg, M.; Lajnert, V.; Pavlic, A.; Brumini, M.; Spalj, S. Changes in Quality of Life Induced by Tooth Whitening are Moderated by Perfectionism: A Randomized, Double-Blind, Placebo-Controlled Trial. Int. J. Prosthodont. 2018, 31, 394–396. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.R.; Wertz, P.W. Review of the Mechanism of Tooth Whitening. J. Esthet. Restor. Dent. 2015, 27, 240–257. [Google Scholar] [CrossRef] [PubMed]
- Kashima-Tanaka, M.; Tsujimoto, Y.; Kawamoto, K.; Senda, N.; Ito, K.; Yamazaki, M. Generation of Free Radicals and/or Active Oxygen by Light or Laser Irradiation of Hydrogen Peroxide or Sodium Hypochlorite. J. Endod. 2003, 29, 141–143. [Google Scholar] [CrossRef]
- Torres, C.R.; Wiegand, A.; Sener, B.; Attin, T. Influence of chemical activation of a 35% hydrogen peroxide bleaching gel on its penetration and efficacy--in vitro study. J. Dent. 2010, 38, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Batista, G.R.; Barcellos, D.C.; Torres, C.R.; Goto, E.H.; Pucci, C.R.; Borges, A.B. The influence of chemical activation on tooth bleaching using 10% carbamide peroxide. Oper. Dent. 2011, 36, 162–168. [Google Scholar] [CrossRef]
- Abdel-Halim, E.S.; Al-Deyab, S.S. One-step bleaching process for cotton fabrics using activated hydrogen peroxide. Carbohydr. Polym. 2013, 92, 1844–1849. [Google Scholar] [CrossRef]
- Carey, C.M. Tooth whitening: What we now know. J. Evid. Dent. Pract. 2014, 14, 70–76. [Google Scholar] [CrossRef]
- Baroudi, K.; Hassan, N.A. The effect of light-activation sources on tooth bleaching. Niger. Med. J. 2014, 55, 363–368. [Google Scholar] [CrossRef]
- Qi, F.; Otsuki, M.; Hiraishi, N.; Hatayama, T.; Wijethunge, C.L.; Tagami, J. Effect of photo-thermal acceleration on in-office bleaching. Odontology 2021, 109, 828–835. [Google Scholar] [CrossRef] [PubMed]
- De Moor, R.J.G.; Verheyen, J.; Verheyen, P.; Diachuk, A.; Meire, M.A.; De Coster, P.J.; De Bruyne, M.; Keulemans, F. Laser teeth bleaching: Evaluation of eventual side effects on enamel and the pulp and the efficiency in vitro and in vivo. Sci. World J. 2015, 2015, 835405. [Google Scholar] [CrossRef]
- Sun, L.; Liang, S.; Sa, Y.; Wang, Z.; Ma, X.; Jiang, T.; Wang, Y. Surface alteration of human tooth enamel subjected to acidic and neutral 30% hydrogen peroxide. J. Dent. 2011, 39, 686–692. [Google Scholar] [CrossRef]
- Epple, M.; Meyer, F.; Enax, J. A Critical Review of Modern Concepts for Teeth Whitening. Dent. J. 2019, 7, 79. [Google Scholar] [CrossRef] [PubMed]
- He, L.B.; Shao, M.Y.; Tan, K.; Xu, X.; Li, J.Y. The effects of light on bleaching and tooth sensitivity during in-office vital bleaching: A systematic review and meta-analysis. J. Dent. 2012, 40, 644–653. [Google Scholar] [CrossRef] [PubMed]
- Martini, E.C.; Parreiras, S.O.; Szesz, A.L.; Coppla, F.M.; Loguercio, A.D.; Reis, A. Bleaching-induced tooth sensitivity with application of a desensitizing gel before and after in-office bleaching: A triple-blind randomized clinical trial. Clin. Oral Investig. 2020, 24, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Moghadam, F.V.; Majidinia, S.; Chasteen, J.; Ghavamnasiri, M. The degree of color change, rebound effect and sensitivity of bleached teeth associated with at-home and power bleaching techniques: A randomized clinical trial. Eur. J. Dent. 2013, 7, 405–411. [Google Scholar] [CrossRef]
- Li, Y.; Greenwall, L. Safety issues of tooth whitening using peroxide-based materials. Br. Dent. J. 2013, 215, 29–34. [Google Scholar] [CrossRef]
- Tredwin, C.J.; Naik, S.; Lewis, N.J.; Scully, C. Hydrogen peroxide tooth-whitening (bleaching) products: Review of adverse effects and safety issues. Br. Dent. J. 2006, 200, 371–376. [Google Scholar] [CrossRef]
- Karadas, M.; Demirbuga, S. Influence of a short-time antioxidant application on the dentin bond strength after intracoronal bleaching. Microsc. Res. Tech. 2019, 82, 1720–1727. [Google Scholar] [CrossRef]
- Cakir, F.Y.; Korkmaz, Y.; Firat, E.; Oztas, S.S.; Gurgan, S. Chemical analysis of enamel and dentin following the application of three different at-home bleaching systems. Oper. Dent. 2011, 36, 529–536. [Google Scholar] [CrossRef]
- Faraoni-Romano, J.J.; Da Silveira, A.G.; Turssi, C.P.; Serra, M.C. Bleaching agents with varying concentrations of carbamide and/or hydrogen peroxides: Effect on dental microhardness and roughness. J. Esthet. Restor. Dent. 2008, 20, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Joiner, A.; Thakker, G.; Cooper, Y. Evaluation of a 6% hydrogen peroxide tooth whitening gel on enamel and dentine microhardness in vitro. J. Dent. 2004, 32, 27–34. [Google Scholar] [CrossRef]
- Maia, E.; Baratieri, L.N.; Caldeira de Andrada, M.A.; Monteiro, S., Jr.; Vieira, L.C. The influence of two home-applied bleaching agents on enamel microhardness: An in situ study. J. Dent. 2008, 36, 2–7. [Google Scholar] [CrossRef]
- Polydorou, O.; Hellwig, E.; Auschill, T.M. The effect of different bleaching agents on the surface texture of restorative materials. Oper. Dent. 2006, 31, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Polydorou, O.; Mönting, J.S.; Hellwig, E.; Auschill, T.M. Effect of in-office tooth bleaching on the microhardness of six dental esthetic restorative materials. Dent. Mater. 2007, 23, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Toteda, M.; Philpotts, C.J.; Cox, T.F.; Joiner, A. Evaluation of a 6% hydrogen peroxide tooth-whitening gel on enamel microhardness after extended use. Quintessence Int. 2008, 39, 853–858. [Google Scholar]
- Türker, S.B.; Biskin, T. The effect of bleaching agents on the microhardness of dental aesthetic restorative materials. J. Oral Rehabil. 2002, 29, 657–661. [Google Scholar] [CrossRef]
- Unlü, N.; Cobankara, F.K.; Altinoz, C.; Ozer, F. Effect of home bleaching agents on the microhardness of human enamel and dentin. J. Oral Rehabil. 2004, 31, 57–61. [Google Scholar] [CrossRef]
- Elfallah, H.M.; Bertassoni, L.E.; Charadram, N.; Rathsam, C.; Swain, M.V. Effect of tooth bleaching agents on protein content and mechanical properties of dental enamel. Acta Biomater. 2015, 20, 120–128. [Google Scholar] [CrossRef]
- Magalhaes, J.G.; Marimoto, A.R.; Torres, C.R.; Pagani, C.; Teixeira, S.C.; Barcellos, D.C. Microhardness change of enamel due to bleaching with in-office bleaching gels of different acidity. Acta Odontol. Scand. 2012, 70, 122–126. [Google Scholar] [CrossRef]
- Pinto, C.F.; Oliveira, R.D.; Cavalli, V.; Giannini, M. Peroxide bleaching agent effects on enamel surface microhardness, roughness and morphology. Braz. Oral Res. 2004, 18, 306–311. [Google Scholar] [CrossRef] [PubMed]
- Markovic, L.; Jordan, R.A.; Lakota, N.; Gaengler, P. Micromorphology of enamel surface after vital tooth bleaching. J. Endod. 2007, 33, 607–610. [Google Scholar] [CrossRef] [PubMed]
- Elfallah, H.M.; Swain, M.V. A review of the effect of vital teeth bleaching on the mechanical properties of tooth enamel. Dent. J. 2013, 109, 87–96. [Google Scholar]
- Ozkan, P.; Kansu, G.; Ozak, S.T.; Kurtulmus-Yilmaz, S.; Kansu, P. Effect of bleaching agents and whitening dentifrices on the surface roughness of human teeth enamel. Acta Odontol. Scand. 2013, 71, 488–497. [Google Scholar] [CrossRef] [PubMed]
- Sa, Y.; Sun, L.; Wang, Z.; Ma, X.; Liang, S.; Xing, W.; Jiang, T.; Wang, Y. Effects of two in-office bleaching agents with different pH on the structure of human enamel: An in situ and in vitro study. Oper. Dent. 2013, 38, 100–110. [Google Scholar] [CrossRef]
- Abe, A.T.; Youssef, M.N.; Turbino, M.L. Effect of bleaching agents on the nanohardness of tooth enamel, composite resin, and the tooth-restoration interface. Oper. Dent. 2016, 41, 44–52. [Google Scholar] [CrossRef]
- Minoux, M.; Serfaty, R. Vital tooth bleaching: Biologic adverse effects-a review. Quintessence Int. 2008, 39, 645–659. [Google Scholar]
- Alkahtani, R.; Stone, S.; German, M.; Waterhouse, P. A review on dental whitening. J. Dent. 2020, 100, 103423. [Google Scholar] [CrossRef]
- Pasquale, C.; De Angelis, N.; Barberis, F.; Lagazzo, A.; Dellacasa, E.; Biggio, D.; Schiaffino, M.; Raiteri, R.; Ceseracciu, L.; Benedicenti, S.; et al. Safety and Effectiveness of Conventional Commercial Products for Professional Tooth Bleaching: Comparative Ex Vivo Study Using AFM Microscopy and Nanoindentation. Appl. Sci. 2023, 13, 9371. [Google Scholar] [CrossRef]
- Gurgan, S.; Cakir, F.Y.; Yazici, E. Different light-activated in-office bleaching systems: A clinical evaluation. Lasers Med. Sci. 2010, 25, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Heiskanen, V.; Hamblin, M.R. Photobiomodulation: Lasers: Vs. light emitting diodes? Photochem. Photobiol. Sci. 2018, 17, 1003–1017. [Google Scholar] [CrossRef]
- Benedicenti, A.; Benedicenti, S. Atlas of Laser Therapy: State of the Art, 4th ed.; Teamwork Media Srl: Villa Carcina, Italy, 2016. [Google Scholar]
- Pop-Ciutrila, I.S.; Colosi, H.A.; Dudea, D.; Badea, M.E. Spectrophotometric color evaluation of permanent incisors, canines and molars. A cross-sectional clinical study. Clujul Med. 2015, 88, 537–544. [Google Scholar] [CrossRef]
- Elamin, H.; Aboubakr, N.; Ibrahim, Y. Identifying the tooth shade in group of patients using vita easy shade. Eur. J. Dent. 2015, 9, 213–217. [Google Scholar] [PubMed]
- DeLoach, L.J.; Higgins, M.S.; Caplan, A.B.; Stiff, J.L. The Visual Analog Scale in the Immediate Postoperative Period. Anesth. Analg. 1998, 86, 102–106. [Google Scholar] [CrossRef]
- Martin, J.; Fernandez, E.; Banamondes, V.; Werner, A.; Elphick, K.; Oliveira, O.B., Jr.; Moncada, G. Dentin hypersensitivity after teeth bleaching with in-office systems. Randomized clinical trial. Am. J. Dent. 2013, 26, 10–14. [Google Scholar] [PubMed]
- McClanahan, S.F.; Bartizek, R.D.; Biesbrock, A.R. Identification and consequences of distinct Löe-Silness gingival index examiner styles for the clinical assessment of gingivitis. J. Periodontol. 2001, 72, 383–392. [Google Scholar] [CrossRef]
- Hanna, R.; Parker, S. The advantages of carbon dioxide laser applications in paediatric oral surgery. A prospective cohort study. Lasers Med. Sci. 2016, 31, 1527–1536. [Google Scholar] [CrossRef]
- Ameri, H.; Ghavamnasiri, M.; Abed, A. Effects of different bleaching time interval on fracture toughness of enamel. J. Conserv. Dent. Endod. 2011, 14, 73–75. [Google Scholar] [CrossRef]
- Grobler, S.R.; Majeed, A.; Moola, M.H.; Rossow, R.J.; van Wyk Kotze, T. In vivo Spectrophotometric assessment of the tooth Whitening Effectiveness of Nite White 10% with Amorphous Calcium Phosphate, Potassium Nitrate and Fluoride, Over a 6-month Period. Open Dent. J. 2011, 5, 18–23. [Google Scholar] [CrossRef]
- De Souza Costa, C.A.; Riehl, H.; Kina, J.F.; Sacono, N.T.; Hebling, J. Human pulp responses to in-office tooth bleaching. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 109, e59–e64. [Google Scholar] [CrossRef] [PubMed]
- Trindade, F.Z.; Ribeiro, A.P.D.; Sacono, N.T.; Oliveira, C.F.; Lessa, F.C.; Hebling, J.; Costa, C.A. Trans-enamel and trans-dentinal cytotoxic effects of a 35% H2O2 bleaching gel on cultured odontoblast cell lines after consecutive applications. Int. Endod. J. 2009, 42, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Palo, R.M.; Bonetti-Filho, I.; Valera, M.C.; Camargo, C.H.R.; Camargo, S.E.A.; Moura-Netto, C.; Pameijer, C. Quantification of peroxide ion passage in dentin, enamel, and cementum after internal bleaching with hydrogen peroxide. Oper. Dent. 2012, 37, 660–664. [Google Scholar] [CrossRef]
- Zimmerman, B.; Datko, L.; Cupelli, M.; Alapati, S.; Dean, D.; Kennedy, M. Alteration of dentin-enamel mechanical properties due to dental whitening treatments. J. Mech. Behav. Biomed. Mater. 2010, 3, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Polydorou, O.; Scheitza, S.; Spraul, M.; Vach, K.; Hellwig, E. The effect of long-term use of tooth bleaching products on the human enamel surface. Odontology 2018, 106, 64–72. [Google Scholar] [CrossRef]
- Roderjan, D.A.; Stanislawczuk, R.; Soares, D.G.; de Souza Costa, C.A.; Favoreto, M.W.; Reis, A.; Loguercio, A.D. Effects of medium or high concentrations of in-office dental bleaching gel on the human pulp response in the mandibular incisors. Restor Dent Endod. 2023, 48, e12. [Google Scholar] [CrossRef]
- Strobl, A.; Gutknecht, N.; Franzen, R.; Hilgers, R.D.; Lampert, F.; Meister, J. Laser-assisted in-office bleaching using a neodymium: Yttrium–aluminum–garnet laser: An in vivo. Lasers Med. Sci. 2010, 25, 503–509. [Google Scholar] [CrossRef]
- Maran, B.M.; Burey, A.; de Paris Matos, T.; Loguercio, A.D.; Reis, A. In-office dental bleaching with light vs. without light: A systematic review and meta-analysis. J Dent. 2018, 70, 1–13. [Google Scholar] [CrossRef]
- Wetter, N.U.; Barroso, M.C.; Pelino, J.E. Dental bleaching efficacy with diode laser and LED irradiation: An in vitro study. Lasers Surg. Med. 2004, 35, 254–258. [Google Scholar] [CrossRef]
- Li, Z.; Wu, Z.; Wang, J.; Huang, M.; Lin, M. Expanding the applications of photodynamic therapy-Tooth bleaching. Clin. Oral Investig. 2022, 26, 2175–2186. [Google Scholar] [CrossRef]
- Bistey, T.; Nagy, I.P.; Simo, A.; Hegedus, C. In vitro FT-IR study of the effects of hydrogen peroxide on superficial tooth enamel. J. Dent. 2007, 35, 325–330. [Google Scholar] [CrossRef]
- de Carvalho, A.C.; de Souza, T.F.; Liporoni, P.C.; Pizi, E.C.; Matuda, L.A.; Catelan, A. Effect of bleaching agents on hardness, surface roughness and color parameters of dental enamel. J. Clin. Exp. Dent. 2020, 12, e670–e675. [Google Scholar] [CrossRef] [PubMed]
- Soares, D.G.; Basso, F.G.; Hebling, J.; de Souza Costa, C.A. Concentrations and application protocols for hydrogen peroxide bleaching gels: Effects on pulp cell viability and whitening efficacy. J. Dent. 2014, 42, 185–198. [Google Scholar] [CrossRef]
- Kina, J.F.; Huck, C.; Riehl, H.; Martinez, T.C.; Sacono, N.T.; Ribeiro, A.P.; Costa, C.A. Response of human pulps after professionally applied vital tooth bleaching. Int. Endod. J. 2010, 43, 572–580. [Google Scholar] [CrossRef]
- Dapadopoulou, A.; Dionysopoulos, D.; Strakas, D.; Kouros, P.; Kolokitha, O.E.; Tolidis, K. Temperature changes in the pulp chamber and bleaching gel during tooth bleaching assisted by diode laser (445 nm) using different power settings. Lasers Med. Sci. 2023, 38, 209. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; An, Q.; Zou, F.; Yu, D.; Chen, M. Analysis of low-frequency vibration-assisted bone drilling in reducing thermal injury. Mater. Manuf. Process. 2021, 36, 27–38. [Google Scholar] [CrossRef]
- Morsi, A.; Haidary, D.; Franzen, R.; Gutknecht, N. Intra-pulpal temperature evaluation during diode laser (445 nm) irradiation for treatment of dentine hypersensitivity: In vitro a pilot study. Laser Dent. Sci. 2020, 4, 139–144. [Google Scholar] [CrossRef]
- Al-Hamd, R.K.H.; Al-Janabi, A. Evaluation of the laser wavelength role on tooth bleaching in terms of color change, roughness, and microhardness with pulpal rapid temperature monitoring: An in vitro study. Appl. Opt. 2023, 62, 5475–5486. [Google Scholar] [CrossRef]
- Amengual, J.; Forner, L. Dentine hypersensitivity in dental bleaching: Case report. Minerva Stomatol. 2009, 58, 181–185. [Google Scholar]
- Solé-Magdalena, A.; Martinez-Alonso, M.; Coronado, C.A.; Junquera, L.M.; Cobo, J.; Vega, J.A. Molecular basis of dental sensitivity: The odontoblasts are multisensory cells and express multifunctional ion channels. Ann. Anat.-Anat. Anz. 2018, 215, 20–29. [Google Scholar] [CrossRef]
- Reuss, A.M.; Groos, D.; Scholl, R.; Schröter, M.; Maihöfner, C. Blue-light treatment reduces spontaneous and evoked pain in a human experimental pain model. Pain Rep. 2021, 6, e968. [Google Scholar] [CrossRef] [PubMed]
- Hanna, R.; Dalvi, S.; Bensadoun, R.J.; Raber-Durlacher, J.E.; Benedicenti, S. Role of Photobiomodulation Therapy in Neurological Primary Burning Mouth Syndrome. A Systematic Review and Meta-Analysis of Human Randomised Controlled Clinical Trials. Pharmaceutics 2021, 13, 1838. [Google Scholar] [CrossRef]
- Hanna, R.; Bensadoun, R.J.; Beken, S.V.; Burton, P.; Carroll, J.; Benedicenti, S. Outpatient Oral Neuropathic Pain Management with Photobiomodulation Therapy: A Prospective Analgesic Pharmacotherapy-Paralleled Feasibility Trial. Antioxidants 2022, 11, 533. [Google Scholar] [CrossRef]
- Hanna, R.; Dalvi, S.; Bensadoun, R.J.; Benedicenti, S. Role of Photobiomodulation Therapy in Modulating Oxidative Stress in Temporomandibular Disorders. A Systematic Review and Meta-Analysis of Human Randomised Controlled Trials. Antioxidants 2021, 10, 1028. [Google Scholar] [CrossRef]
- Hanna, R.; Dalvi, S.; Tomov, G.; Hopper, C.; Rebaudi, F.; Rebaudi, A.L.; Bensadoun, J.R. Emerging potential of phototherapy in management of symptomatic oral lichen planus: A systematic review of randomised controlled clinical trials. J. Biophotonics 2023, 16, e202300046. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Li, Q.; Wang, Y. Effects of pH values of hydrogen peroxide bleaching agents on enamel surface properties. Oper. Dent. 2011, 36, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.X.; Tenenbaum, H.C.; Wilder, R.S.; Quock, R.; Hewlett, E.R.; Ren, Y.F. Pathogenesis, diagnosis and management of dentin hypersensitivity: An evidence-based overview for dental practitioners. BMC Oral Health 2020, 6, 220. [Google Scholar] [CrossRef]
- Al-Harbi, A.; Ardu, S.; Bortolotto, T.; Krejci, I. Effect of extended application time on the efficacy of an in-office hydrogen peroxide bleaching agent an in vitro study. Eur. J. Esthet. Dent. 2013, 8, 226–236. [Google Scholar]
- Basting, R.; Amaral, F.; Françe, F.; Flório, F. Clinical comparative study of the effectiveness of and tooth sensitivity to 10% and 20% carbamide peroxide home-use and 35% and 38% hydrogen peroxide in-office bleaching material containing desensitizing agents. Oper. Dent. 2012, 37, 464–473. [Google Scholar] [CrossRef]
- Rebelo, M.A.B.; de Queiroz, A.C. Gingival Indices: State of Art. In Gingival Diseases—Their Aetiology, Prevention and Treatment; Panagakos, F., Ed.; IntechOpen: London, UK, 2011; ISBN 978-953-307-376-7. Available online: http://www.intechopen.com/books/gingival-diseases-their-aetiology-prevention-and-treatment/gingival-indices-state-of-art (accessed on 13 December 2023).
- Kothari, S.; Gray, A.R.; Lyons, K.; Wen Tan, X.; Brunton, P.A. Vital bleaching and oral-health-related quality of life in adults: A systematic review and meta-analysis. J. Dent. 2019, 84, 22–29. [Google Scholar] [CrossRef]



| Product Name | % Hydrogen Peroxide | pH | Working Type Mode | Photoactivation Wavelength | Photoactivation Time | Gel Resting Time between Each Cycle | Number of Applications/ Sessions |
|---|---|---|---|---|---|---|---|
| BlancOne ULTRA+ | 35 | 5.5 | light | 430–490 nm | 15 s | 8 min | 3 |
| Device Specifications | Manufacturer | Doctor Smile-Lambda-Italy |
| Model identifier | Wiser 3 | |
| Emitter type | Diode laser | |
| Medical/laser class | IV | |
| Beam delivery system | Fibre | |
| Probe design | Single probe | |
| Beam profile | Flattop | |
| Beam divergence full angle | 0° | |
| Irradiation Parameters | Wavelength (nm) | 450 |
| Therapeutic power output (W) | 1 | |
| Emission mode | CW | |
| Beam spot size at target (cm2) | 1 | |
| Irradiance at target (W/cm2) | 1 | |
| Energy per spot (J) | 15 | |
| Total energy | 150 | |
| Fluence (J/cm2) per point | 15 | |
| Irradiation time (s) | 15 | |
| Treatment Protocol | Total number of irradiated points | 10 |
| Laser-tissue distance | ~2 cm (No loss of energy due to the unique properties of the flattop delivery system) | |
| Application technique | Static | |
| Total number of treatments/sessions | 3 | |
| Frequency of session | Once |
| Case # | Gender | Age | Systemic Disease | Medication | Smoking Status | No. of Cigarettes/Day |
|---|---|---|---|---|---|---|
| 1 | F | 43 | Nil | Nil | Yes | 20 |
| 2 | F | 25 | Nil | Nil | Yes | 15 |
| 3 | F | 37 | Nil | Nil | No | - |
| 4 | F | 43 | Nil | Nil | Yes | 5 |
| 5 | F | 38 | Nil | Nil | No | - |
| 6 | F | 34 | Nil | Nil | No | - |
| Case # | Colour Shade at T0 | Colour Shade at T1 | Colour Shade at T4 | Colour Shade Fold Improvement at T1 and Maintained at T4 compared to T0 |
|---|---|---|---|---|
| 1 | A3.5 | A1 | A1 | 10 |
| 2 | A1 | A1 | A1 | 3 |
| 3 | A3.5 | A2 | A2 | 7 |
| 4 | D2 | A1 | A1 | 2 |
| 5 | A2 | A1 | A1 | 3 |
| 6 | A3 | A1 | A1 | 7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hanna, R.; Miron, I.C.; Benedicenti, S. Feasibility and Safety of Adopting a New Approach in Delivering a 450 nm Blue Laser with a Flattop Beam Profile in Vital Tooth Whitening. A Clinical Case Series with an 8-Month Follow-Up. J. Clin. Med. 2024, 13, 491. https://doi.org/10.3390/jcm13020491
Hanna R, Miron IC, Benedicenti S. Feasibility and Safety of Adopting a New Approach in Delivering a 450 nm Blue Laser with a Flattop Beam Profile in Vital Tooth Whitening. A Clinical Case Series with an 8-Month Follow-Up. Journal of Clinical Medicine. 2024; 13(2):491. https://doi.org/10.3390/jcm13020491
Chicago/Turabian StyleHanna, Reem, Ioana Cristina Miron, and Stefano Benedicenti. 2024. "Feasibility and Safety of Adopting a New Approach in Delivering a 450 nm Blue Laser with a Flattop Beam Profile in Vital Tooth Whitening. A Clinical Case Series with an 8-Month Follow-Up" Journal of Clinical Medicine 13, no. 2: 491. https://doi.org/10.3390/jcm13020491
APA StyleHanna, R., Miron, I. C., & Benedicenti, S. (2024). Feasibility and Safety of Adopting a New Approach in Delivering a 450 nm Blue Laser with a Flattop Beam Profile in Vital Tooth Whitening. A Clinical Case Series with an 8-Month Follow-Up. Journal of Clinical Medicine, 13(2), 491. https://doi.org/10.3390/jcm13020491









