Exercise across the Lung Cancer Care Continuum: An Overview of Systematic Reviews
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Literature Search
2.3. Data Extraction and Quality Assessment
2.4. Synthesis of Results
2.5. Protocol Deviations
3. Results
3.1. Search Results
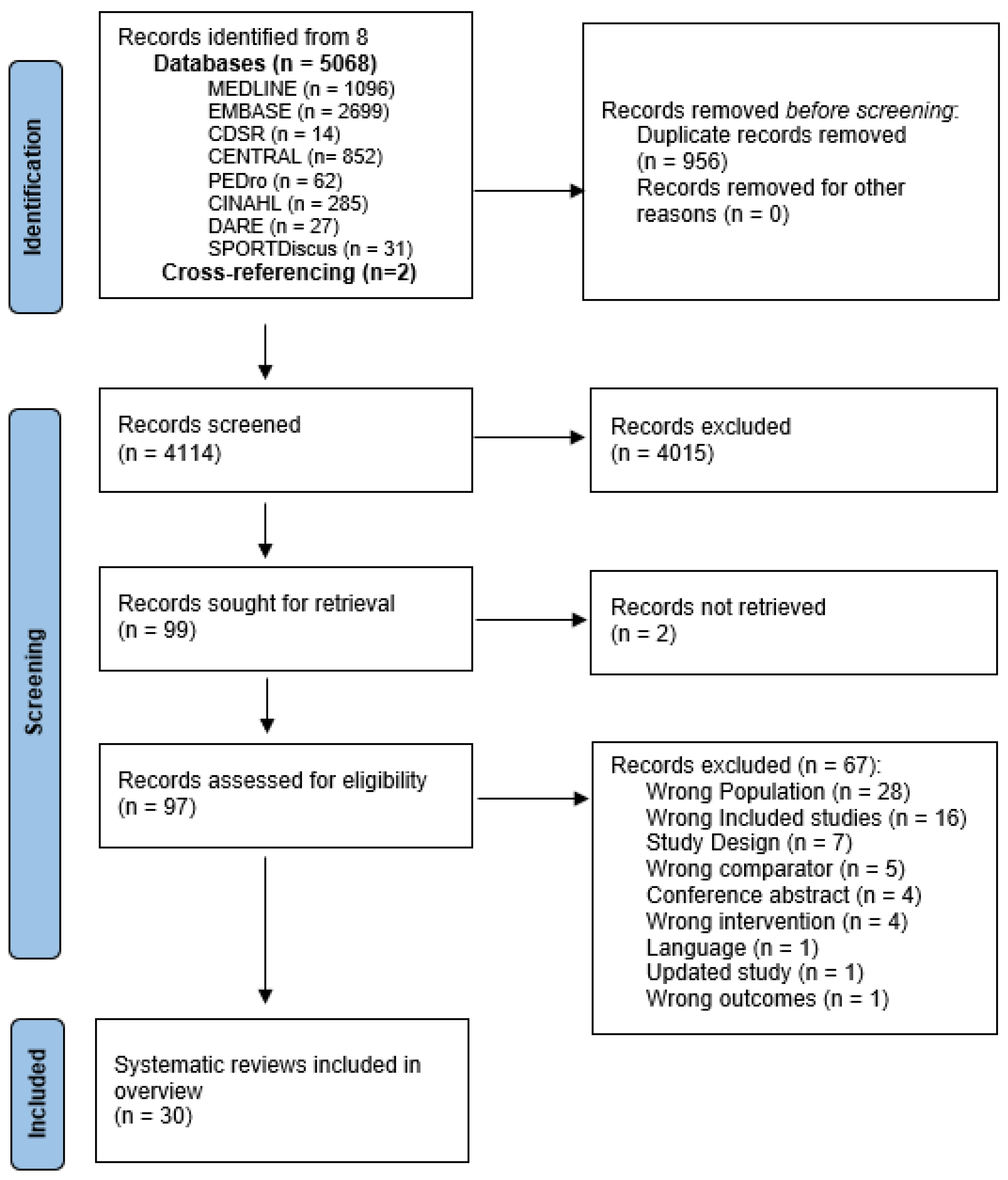
3.2. Methodological Rigour and Quality of Reporting
3.3. Characteristics of Included Studies
3.3.1. Participants, Interventions and Outcomes
| Author Year, SR Focus and Search Dates | Primary Studies (Number; Number Pre, During or Post Treatment (if Mixed)) | Participant Characteristics (n, Treatment Type (if Mixed)) | Type (n NSCLC/SCLC/Mesothelioma); Stage (TNM Classification) | Exercise Intervention Types, Prescription Ranges (F.I.T.T.), Supervised and/or Unsupervised) | Additional Intervention/s (Number of Studies, Combined with What?) | Comparator | Outcomes (* Primary, if Reported) | Languages Searched |
|---|---|---|---|---|---|---|---|---|
| Pre-treatment only (surgical) | ||||||||
| Cavalheri 2017 [4] Lung cancer exercise training. Inception—Nov 2016 | 5 RCTs | 203, surgical | NSCLC (one study (n = 19) did not specify type); Stage: 100 I–IIIB, 103 NR | Type: aerobic, resistance, respiratory (including IMT), education. FITT: 3× per day for 1 week to 5× per week for 4 weeks. Intensity: reported in 2 studies (70–80% max work rate). Supervision: NR. | 1× trial—both groups received education re energy conservation techniques, relaxation and stress management and focus on nutrition. ×2 trials described as ‘pulmonary rehabilitation’ components in addition to exercise NR | Non-exercise training or usual care | PPC *, days with ICC post-op *, LOS, fatigue, exercise capacity, pulmonary function, postoperative mortality, dyspnoea | No language restriction |
| Steffens 2018 [16] Cancer (any type) exercise interventions Inception—Nov 2016 | 5 RCTs + 1 qRCT (of 13 included studies) | 434, surgical | Type and stage NR | Type: aerobic, resistance, respiratory (including IMT). FITT: 20–60 min, 3× daily for 1 week to 5× weekly for 2 weeks. 1× NR duration, 1× NR dosage. Supervision: NR. | Exercise only | Usual care | Post-operative complications, LOS | No language restriction |
| Treanor 2018 [17] Prehabilitation interventions for newly diagnosed cancer. Inception—Apr 2017 | 7 RCTs (of 18 included studies) | 449, surgical | 197 NSCLC, 252 NR; Stage: 36 I-IIIa, 88 NR. | Type: aerobic, resistance, respiratory. FITT: 1–3× per day to 1 day per week for 1–3 weeks. 1× NR dosage. Intensity: reported by 1 study (Licker 2016) 80–100% peak work rate. Supervision: combined supervised hospital and home based. | ×3 trials described as ‘pulmonary rehabilitation’—components in addition to exercise NR | Usual care (×1 study included daily walking advice) | LOS, post-op complications, time intubated, pulmonary function, QoL and feelings of hope and power | English |
| Rosero 2018 [18] Physical exercise for people with NCSLC. Jan 1970–Feb 2018 | 10 RCTs | 676, surgical | NSCLC; Stage: 625 I–II, 51 III-IV | Type: aerobic, resistance, respiratory (including IMT), PNF, stretching FITT: 20–60 min, 1–2× per day to 3–7 days per week, 1–4 weeks. Intensity: ×4 studies—70–100% PWC, ×6 NR. Supervision: supervised. | Psychological educational guidance (×1 trial), pharmacotherapy (×1 trial) | Usual care | Exercise capacity *, lung function, dyspnoea, fatigue, PPC, post-op days in hospital, HRQoL, RPE | English |
| Li 2019 [19] Exercise therapy effects on surgical outcomes in lung cancer with or without COPD. Inception–June 2017 | 7 RCTs + 1 prospective cohort with retrospective control | 404, surgical | NSCLC; Stage: 25 I–III, 130 I–IV, 40 NR | Type: aerobic (including HIIT), resistance, respiratory. FITT: 15–30 min, 1–3× per day, 5–7× per week over 1–4 weeks. Intensity: NR. Supervision: combined supervised hospital and home-based. | Pharmacotherapy (×1 trial) | Usual care (including chest physiotherapy and breathing exercises) | PPC *, duration of ICC *, LOS *, pulmonary function, exercise capacity, dyspnoea | No language restriction |
| Pu 2021 [20] Impact of respiratory exercises for people with NSCLC. Inception -Mar 2021 | 10 RCTs | 768, surgical | NSCLC; Stage NR. | Type: aerobic, respiratory (including IMT). FITT: 3 days–4 weeks IMT: between 10–20 min, 2–4× per day. AT + RT:15–30 min, daily to 3× per week. Intensity: modified Borg ‘light’—7, Borg 13–16, 70% CPET max score. Supervision: NR | Exercise only | Standard of care | PPC *, LOS *, HRQoL, mortality, surgical complications, lung function and exercise capacity | English |
| Gravier 2021 [21] Effects of exercise training on post-operative complications and other outcomes. Inception—Dec 2020 | 14 RCTs | 791, surgical | Type NR (eligibility states NSCLC); Stage: 60 IA-IIIB, 73 NR | Type: aerobic, resistance, respiratory (including IMT), stretching, balance. FITT: 1–8 weeks, ×2–3/week to ×3/day. Intensity: 50–100% Wpeak, 13–16 Borg RPE. Supervision: supervised IP or OP, ×2 studies unsupervised home-based. | Pulmonary rehabilitation (×1 trial), education (×2 trials), psychology (×1 trial), psychology + nutrition (×1 trial) | Usual care or only chest physiotherapy or education | Post-op complications (POC) *, 30-day mortality *, LOS, exercise capacity, respiratory function, QoL, anxiety, depression, program adherence, program completion, AEs | No language restriction |
| de Oliveira Vacchi 2022 [22] Effects of IMT +/− rehabilitation on functional and pulmonary capacity. Inception—Nov 2019 | 6 RCTs | 219, surgical | Type and stage NR | Type: aerobic, resistance, respiratory (including IMT). FITT: ×2/day to ×5/week, 1–4 weeks. Intensity: IMT: 15–20% MIP, increased by 5–10% to max 60%. AT: ‘high’ ×2 studies, 80% max load × 2 studies. Supervision: NR. | Exercise only | Usual care or only chest physio or education | Functional capacity, pulmonary function, quality of life, post-operative complications, LOS, mortality | No language restriction |
| Post-treatment only (surgical) | ||||||||
| Li 2017 [23] Efficacy of exercise of training following lung resection surgery. Inception -Feb 2017 | 6 RCTs | 438 l, surgical | NSCLC 370, mixed 68 (NSCLC + metastatic tumour + other type); Stage NR | Type: aerobic, resistance, respiratory (including IMT). FITT: 2× daily to 2× per week, 5–60 min for 2–20 weeks. Intensity: 60–95% MHR. Supervision: NR. | Exercise only | Usual care or standard physiotherapy treatment | Quality of life *, exercise capacity, physical activity, lung function, POC, PPC, muscle strength, symptoms (pain, dyspnoea) | No language restriction |
| Sommer 2018 [24] Post-surgical rehabilitation for lung cancer patients. Inception—Feb 2016 | 4 RCTs (within 1 year of surgery) | 262, surgical | NSCLC NR (states ‘mainly’); Stage NR (‘evenly distributed between groups’) | Type: aerobic, resistance, respiratory (IMT), whole body vibration. FITT: 1× weekly for 10 weeks to 3× weekly for 12–20 weeks. Initiated between 5 days–8 weeks following lung resection. Intensity: 60–95% MHR, 70% WMax, RPE 11–16. Supervision: supervised outpatient. 1× study also included inpatient and unsupervised. | Exercise only | Usual care (including home exercise education), general information and discouraged to improve exercise tolerance. | Exercise capacity *, HRQoL | English, one of the Scandinavian languages, or German |
| Cavalheri 2019 [25] Post-operative rehabilitation for lung cancer patients. Inception—Feb 2019 | 8 RCTs (within 1 year of surgery) | 450, surgical | NSCLC; Stage: 72 I, 33 II, 13 III, 6 IV, NR 350. | Type: aerobic, resistance, respiratory (including IMT), balance. FITT: 5–60 min, 2× day–5× per week over 4–20 weeks. Intensity: 60–90% HRR, 3–4 Borg dyspnoea, 13–15 RPE, 50–80% Wmax, 80–95% max HR, 80% 6 MWT speed. Supervision: supervised inpatient and outpatient +/− home-based component. | Exercise group had pain treatment adjusted by anaesthiologist (×1 trial). Nurse counselling, up to 3× 60 min (×1 trial) | Usual care | Exercise capacity *, safety *, pulmonary function, HRQoL, muscle strength, anxiety and depression | No language restriction |
| Larsen 2019 [26] Respiratory physiotherapy following lung resection. Inception—Mar 2017 | 11 RCTs and 2 qRCTs (5RCT and 1qRCT included non-LC). | 1280 (618 LC), surgical | Type and stage NR. | Type: aerobic, resistance, respiratory. FITT: 5–30 mins, 2× day, for up to 5 days. Intensity: NR. Supervision: supervised inpatient or NR. | Exercise / respiratory PT only | Standard care (Included chest PT in 2× studies) | Mortality, PPC, LOS | No language restriction |
| Pre and post-treatment (surgical) | ||||||||
| Rueda 2011 [27] Non-invasive interventions to improve wellbeing for people with lung cancer. Inception—Feb 2011 | 2 RCTs (1× pre- and 1× post-op) of 15 included studies | 157; pre (104) and post-operative (53) | NCSLC; Stage: 104 IA–IIIA, 53 NR | Type: aerobic, resistance, respiratory FITT: Pre-op: 7–10 days aerobic/resistance (1× per day) + respiratory (10× per day). Post-op: inpatient aerobic/resistance ×2 per day for 1–5 days—followed by home exercise program. Intensity: NR Supervision: supervised inpatient, unsupervised/home visits home program. | Exercise only | No exercise training | Exercise capacity, Quality of life, hope (Hearth Hope Index) and power (PKPCT), quadriceps strength | NR |
| Mainini 2016 [28] Pre and post operative physical exercise interventions for patients with NSCLC. May 2013–May 2016 | 6 RCTs (1× pre- and 5× post-op) | 414; pre (40), and post-operative (374), | NSCLC; Stage NR | Type: aerobic, resistance, respiratory (including IMT), balance FITT: Pre-op: aerobic and respiratory, 5× weekly for 3 weeks. Post-op: aerobic, resistance, respiratory (including IMT), balance, range: daily to 1× weekly, 5 days–20 weeks. Intensity: 60–70% CPET work max or 70–80% max 6 MWT speed, RPE 11–16, 60–90% HRR, 80–95% MHR. Supervision: pre-op supervised outpatient, post-op supervised and unsupervised. | Exercise combined with dyspnoea management—both groups 3 × 1 h post-op nurse counselling (×1 trial) | Usual care | Exercise capacity, HRQoL, and lung function (FEV1, FVC and DLCO) | English, French, Italian, Portuguese and Spanish |
| Wang 2018 [29] Preoperative breathing exercises for patients with operable lung cancer. Inception—Dec 2017 | 16 RCTs (8 pre-, 4 periop, 4 post-op) | 1234 | 166 NSCLC, 1068 NR; Stage: 398 Ia-IIIb (not receiving radio/chemotherapy), 836 NR. | Type: aerobic (including HIIT), resistance, respiratory (including IMT), balance FITT: 2×day–5× per week over 4–20 weeks. Intensity: ×1 HIIT (Huang 2017), ×1 20–60% MIP, ×7 NR. Supervision: NR. | Exercise only | NR | Pulmonary function *, PPCs *, LOS and exercise capacity | English and Chinese |
| Himbert 2020 [30] Intervention effects on pulmonary and physical function pre and post, lung cancer surgery. Jan 1946–Mar 2020 | 22 RCTs (11 pre-, 11 post-op) | 893; 560 pre, 333 post-operative (only reported from 5RCTs) | 674 NSCLC; Stage: 151 I–IIIa, 742 NR. | Type: aerobic (including HIIT), resistance, respiratory (including IMT), balance FITT: Pre-surgery: 10–60 min, 1–7 days a week over 5 days–8 weeks, moderate-high intensity. Combination of inpatient, outpatient, supervised and home-based. Post-surgery: 5–60 min, 1–7 days a week over 1–20 weeks, moderate-high intensity. Supervised (6 studies), home-based (1 study), both supervised and unsupervised (2 studies). | Exercise only | Usual care | Pulmonary (lung volume and capacity) and physical function (cardiorespiratory fitness, functional capacity, physical performance) domains | English |
| Machado 2021 [31] Effects of exercise pre and post lung cancer surgery on HRQoL. Inception—Mar 2021 | 10 RCTs (1 pre, 9 post-op (4 during adjuvant chemo)) | 651; pre (22), 629 post-op (140 received adjuvant Rx, mainly chemotherapy) | NSCLC; Stage: 454 I-II, 71 NR | Type: aerobic (including HIIT), resistance, respiratory (including IMT), balance, mobility and stretching. FITT: 5–60 mins/session, ×2–3/week (centre-based) and ×5/week (home-based), 4 weeks (pre-op), 6–20 weeks (post-op). Intensity: HIIT (80% Wpeak or 85–100% HRmax) ×5 studies, continuous ×6 studies (light ×1, mod ×5). Supervision: largely supervised. | Relaxation (×1 trial) | Usual care with no exercise training, general PA advice | HRQoL *, fatigue | English |
| Xu 2022 [32] Effects of exercise pre and post lung cancer surgery on PPCs and LOS. Inception—Jun 2021 | 23 RCTs (12 pre-, 10 post- and 1 peri-op) | 2068; (809 pre-, 1189 post-, 70 peri-operative) | 1054 NSCLC, 1014 NR; Stage: 140 I-II, 343 I-III, 164 I-IIIA, 60 I-IIIB, 749 I-IV, 612 NR. | Type: aerobic (including HIIT), resistance, respiratory (including IMT), balance, mobility and shoulder ROM. FITT: Pre-op: ×2/week–×4/day (IMT). Duration: 1–4 weeks, 20–40 mins/session. Post-op: ×1/day-hourly, during I/P admission–12 weeks, 5–30 min. Peri-op: ×2/day, during I/P admission. Intensity: IMT: 15–60% MIP, aerobic 60–80% Wpeak/60–90% HRR, Borg 13–16/11–13, <6. Supervision: Pre-op—largely supervised or supervised/home unsupervised. Post-op—largely supervised. Periop—supervised. | Nutrition and psychosocial (relaxation)—×1 trial | Usual care | Hospital LOS *, post-operative pulmonary complications *, post-operative complications, chest tube duration, mortality | English |
| Pre, during and/or post-treatment (surgical and non-surgical) | ||||||||
| Hsieh 2017 [33] Supportive care interventions on depressive symptoms for people with lung cancer. Inception—Sept 2015 | 3 RCTs (1× post-treatment, 2× NR) of 12 included studies | 187, treatment type NR | 24 NSCLC early stage; 163 type/stage NR | Type: ‘pulmonary rehabilitation’, aerobic, respiratory (including IMT), stretching. FITT: 3–5× per week, 30–40 min/session for 4–12 weeks. Intensity: NR. Supervision: supervised and unsupervised home based. | Exercise only | Standard care | Depressive symptoms * | English or Chinese |
| Papadopolous 2018 [34] Effects of nonpharmacologic interventions on sleep quality of people with lung cancer. Inception—Dec 2016 | 3×RCT, 2×qRCT (mixed post-op and during chemo and/or RT) | 364 (109 post op, 95 chemo, 4 RT, 3 chemoRT, 153 NR) | Type NR; Stage 72 I, 24 II, 48 III, 149 IV, 71 NR | Type: aerobic, resistance, respiratory, Tai Chi FITT: ×2/day–×1/week, 30–45 min/session. ×2 inpatient, during inpatient admission only—12 weeks. Intensity: NR. Supervision: ×2 home based, ×1 supervised and home-based, ×1 during inpatient admission supervision NR in 3 studies. | 15–20 min/week behavioural support sessions (×1 trial) | Usual care or wait-list | Sleep quality * | No language restriction (sensitivity analysis excluded non-English) |
| Liu 2019 [35] Respiratory exercise effects on exercise capacity and mental health in people with lung cancer. Inception—Apr 2018 | 15 RCTs (2 pre-op, 2 pre and post-op, 5 post-op, 3 during non-surgical Rx (2 chemotherapy, 1 chemoradiotherapy), 3 unknown) | 930 (532 surgery, 149 surgery/chemo/radiotherapy, 20 chemo/radiotherapy, 49 chemo, 180 NR) | Type: 149 NSCLC/SCLC/mesothelioma, 69 NSCLC/SCLC, 82 NSCLC, 630 NR. Stage: 342 I -IV, 588 NR | Type: respiratory (including IMT), aerobic (including HIIT) and resistance. FITT: 15 minutes–3 h, 2–3× per day to 3–5× per week for 1–12 weeks. Intensity NR. Supervision: unsupervised and supervised (inpatient and outpatient). | Oxygen therapy, aerosol inflation (×1 trial), counselling, teaching coping and adaption strategies (×1 trial), acupressure, modified swallow technique and cough easing techniques (×1 trial), education (×1 trial), goal setting (×1 trial) | Conventional care or no treatment | Dyspnea *, exercise capacity *, anxiety, depression, | English and Chinese |
| Singh 2020 [36] Adverse events, feasibility and effectiveness of lung cancer exercise. Inception—May 2020 | 32 RCTs (27 pre-op, 5 post-op) | 2109 (1695 pre-op, 414 post-op). 8 RCTs during, 17 RCTs post and 7 RCTs during or post adjuvant chemotherapy | NSCLC 1351, NSCLC + SCLC 758; Stage: 137 I/II, 446 III/IV and 1526 mixed. | Type: aerobic, resistance, respiratory, Tai Chi. FITT: Session duration/frequency/intensity: NR. Program duration 1–20 weeks (21 studies < 12 weeks). Supervision: supervised (27 RCTs) and unsupervised (5 RCTs). | Exercise only | Non-exercise control or usual care | Safety (adverse events), feasibility (recruitment, retention and exercise adherence) and health-related outcomes (quality of life, exercise capacity, fatigue, strength, anxiety, depression, sleep, lung function, dyspnoea, pain and hospital LOS) | English |
| Ma 2020 [37] Effects of exercise on quality of life for people with lung cancer. Inception—Sept 2019 | 16 RCTs (3× preoperative, 7× post-operative, 6× during and following non-surgical Rx) | 758 participants (106 pre-op, 361 post op, 291 advanced/treatment NR) | 704 NSCLC, 49 NSCLC or SCLC, 5 SCLC; Stage 165 I, 79 II, 81 III, 81 IV; 37 I-II, 155 III-IV, 71 I-IV, 6 recurrent, 5 no malignancy, 38 NR. | Type: aerobic, resistance, respiratory FITT: 5–60 min, 1–7 x per week, 1–20 weeks. Supervision: supervised (12 RCTs), unsupervised (1 RCT) and both (3 RCTs)—home-based and outpatient. | Exercise only | Usual care/ no intervention (3× trials included chest physiotherapy/conventional physiotherapy) | QoL * | English |
| Yang 2020 [38] Home-based exercise effects on exercise capacity, symptoms, and quality of life in people with lung cancer. Inception—Dec 2018 | 7 RCTs (4× post-op, 3 NR) | 559 (175 post-op, 88 post-op + during adjuvant chemotherapy, 296 NR) | 175 NSCLC, 384 NR; Stage NR | Type: aerobic, resistance, balance, Tai Chi, Chinese Medicine Baduanjin FITT: 5–60 min, 2× daily to 3× per week, for 6 weeks to 3 months. Intensity: light (1 RCT), 60–80% HR max (2 RCTs), NR 4 RCTs. Supervision: all home-based, level of supervision NR. | Exercise only | Routine guidance | Exercise capacity, cancer-related fatigue, pain, insomnia, appetite loss, coughing, anxiety, depression and HRQoL | English and Chinese |
| Codima 2021 [39] Exercise for symptom management and quality of life for people with lung cancer. Jan 1998–Jan 2019 | 10 RCTs (4× pre-op, 3× post-op, 2× during chemotherapy or targeted therapy, 1× mixed) | 835 (225 pre-op, 374 post-op, 120 systemic therapy, 116 diverse/no Rx) | 657 NSCLC, 178 NR; Stage: 259 I-IIIa, 24 IIIa-IV, 434 I-IV, 78 I-II, 40 NR | Type: aerobic, resistance, respiratory, Tai Chi, stretching. FITT: 30–60 min, ×1/day–×1 per week for 1 to 20 weeks. Intensity: 60–100% HRmax or VO2 peak, Borg 11–16 (5 RCTs). Supervision: Supervised (7 RCTs), partially supervised (2 RCTs), and unsupervised (1 RCT). Inpatient, outpatient and home-based. | Group and individual counselling (×1 trial), relaxation and counselling (×1 trial) | Usual care | HRQoL and symptoms | English |
| Ma 2021 [40] Nonpharmacological interventions for cancer related fatigue for people with lung cancer. Inception—Jun 2020 | 6 RCTs (1× post-op, 1× post-op/post chemo, 2× during chemo, 1× during targeted therapy, 1× NR) of 18 included studies | 364 (72 post-op, 164 during treatment incl target therapy and chemotherapy, 17 curative intent treatment, 111 NR) | Type: NR; Stage: 163 I-IV, 17 I-III, 135 III-IV, 49 IV. | Type: aerobic, resistance, Tai Chi and electrical muscle stimulation. FITT: 30–60 min, 1–7× per week over 6–12 weeks. Intensity: 1×high, others NR. Supervision: 1×RCT home based, 1× RCT supervised, others: setting/supervision NR. | Exercise only | Usual Care, ×1 RCT included low impact exercise (ROM, stretching and breathing) (×1 trial) | Fatigue * | English |
| Heredia-Ciuro 2021 [41] Effects of HIIT in lung cancer survivors. Inception—Mar 2021 | 3 RCTs, 2 pilot RCTs, 3 prospective randomised open, blinded end-point (PROBE) studies (3× pre-op, 2× post-op, 1× post-op/post chemo, 1× during RT, 1× during targeted therapy) | 305 (151 pre-op, 98 post-op, 17 post-op/post chemo, 15 during RT, 24 during targeted therapy) | Type: 281 NSCLC, 24 adenocarcinoma; Stage: 37 I-II, 168 I-IIIA, 15 ‘advanced’, 85 NR. | Type: aerobic (HIIT), resistance, respiratory (IMT). FITT: ×3–5/week, 20–60 min, 2–20 weeks. Intensity: HIIT 80–95% iPPO (patient’s peak power)/60–100% Wpeak/70–80% 6 MWT speed/80% VO2 peak/RPE 15–17; resistance: 6–12 RM (reported in only 1 study); IMT: 50% Pimax/PEmax (reported in only 1 study). Supervision: All supervised. | Exercise only | Usual care (standard medical Rx, routine post-Rx physio, general info and monitoring from hospital) | Cardiorespiratory fitness (VO2 peak) | English and non-English (if a translation was available) |
| Zhou 2021 [42] Effects of exercise on fatigue in lung cancer. Inception—Mar 2020 | 8 RCTs (2× post-op, 1× post-op/post chemo, 2× during chemo, 2× during chemoRT, 1× during chemo or RT or targeted therapy, 1× during targeted therapy) | 570 (283 post-op, 17 post-op +/− post adjuvant chemo, 111 chemo or RT or targeted therapy, 15 chemoRT, 120 chemo, 24 targeted therapy) | Type: 430 NSCLC, 140 NR. Stage: 228 I-IIIA, 179 III-IV, 163 NR. | Type: aerobic, resistance, balance, Tai Chi. FITT: ×1–5/week, 20–60 min (1 RCT commenced @ 5 mins), 6–12 weeks. Intensity: 1×RCT light, 1×RCT moderate, 1×RCT mod-high, 5×RCT NR. Supervision: 1×RCT supervised, 2×RCT home-based. | Behaviour support and education (×1 trial) | Usual care/conventional physio, general health education materials (×1 trial), daily stretching (×1 trial) | Fatigue *, depression, anxiety, HRQoL, functional capacity | English |
| During and/or post-treatment (non-surgical) | ||||||||
| Peddle-Mc Intyre 2019 [43] Exercise training for advanced lung cancer. Inception—July 2018 | 6 RCTs (all non-surgical–3× during Rx, 2× palliative Rx or scheduled/eligible for Rx, 1× post Rx) | 221 (48 chemotherapy, 24 EGFR inhibitors, 11 unspecified treatment, 27 no treatment, 111 NR) | 160 NSCLC/SCLC, 24 NSCLC, 27 NSCLC/mesothelioma, 11 NR; Stage 187 IIIA-IV, 27 ‘advanced’, NR 8. | Type: aerobic, resistance, respiratory (including IMT) FITT: 30–65 min, 1–5 days per week over 6–12 weeks. Intensity: 55–75% HRR, 60–80% VO2 peak (RPE 11–17), 60–70% max HR, 40–70% PImax. Supervision: supervised +/− home-based. | 15–20 min/week behavioural support sessions (×1 trial) | Usual care (including conventional physiotherapy). Qigong for 6 weeks (×1 trial) | Exercise capacity *, muscle strength, HRQoL, dyspnoea, fatigue, anxiety and depression, lung function, physical activity, adverse events, overall survival, performance status | No language restriction |
| Lee 2021 [44] Exercise intervention effects for people with lung cancer during chemotherapy. Jan 2000–May 2020 | 6 RCTs (all during chemotherapy Rx) | 244, receiving chemotherapy (both radical and palliative) | 201 NSCLC or SCLC, 43 NSCLC; Stage NR. | Type: aerobic, resistance, respiratory FITT: 20–75 mins, 2–5× per week over 6–12 weeks. Intensity: 60% HRR, 80–95% incremental peak power output, 30–80% peak work rate and 40–70% 1RM. Supervision: supervised and home-based. | Exercise only | Usual care | Pulmonary function, quality of life, pain, exercise capacity, strength, anxiety and depression, physiologic measurements (BP, max and resting HR, WBC, RBC) | English |
Pre-Treatment Only (Surgical)
Post-Treatment Only (Surgical)
Pre and Post-Treatment (Surgical)
Pre, during and/or Post-Treatment (Surgical and Non-Surgical)
During and/or Post-Treatment (Non-Surgical)
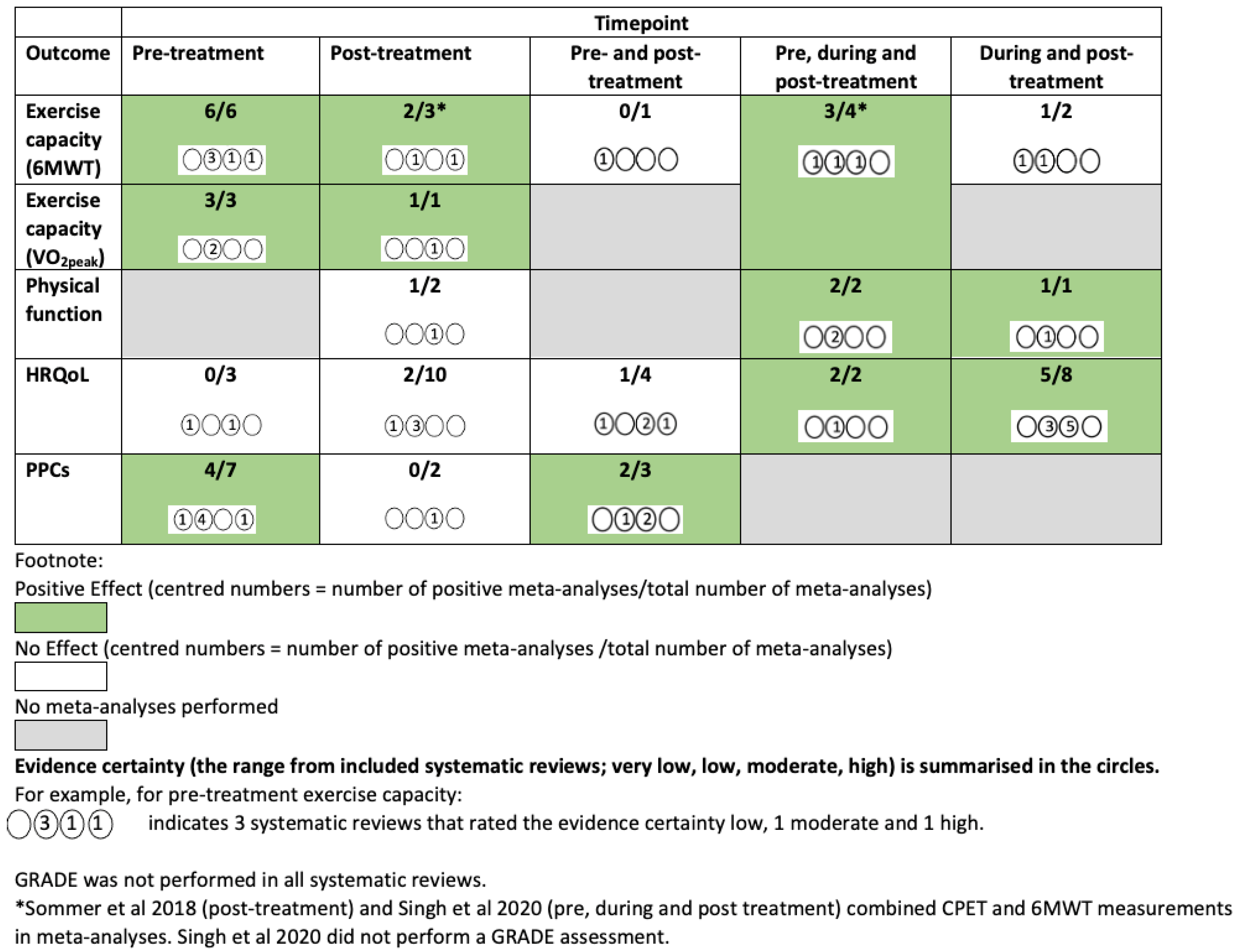
3.4. Intervention Effects
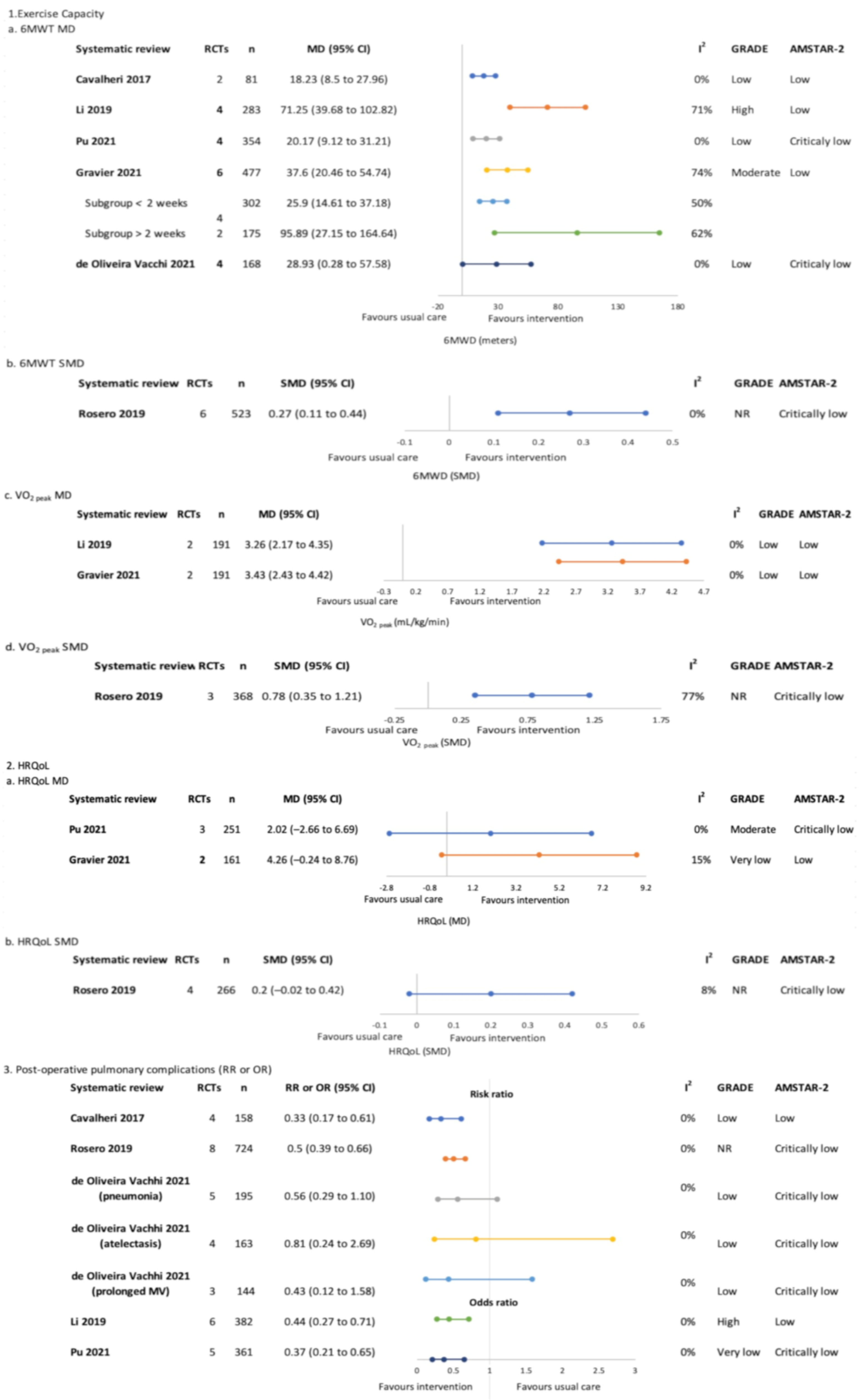
3.4.1. Pre-Treatment Only (Surgical)—Figure 3
Exercise Capacity
Health-Related Quality of Life
Post-Operative Pulmonary Complications
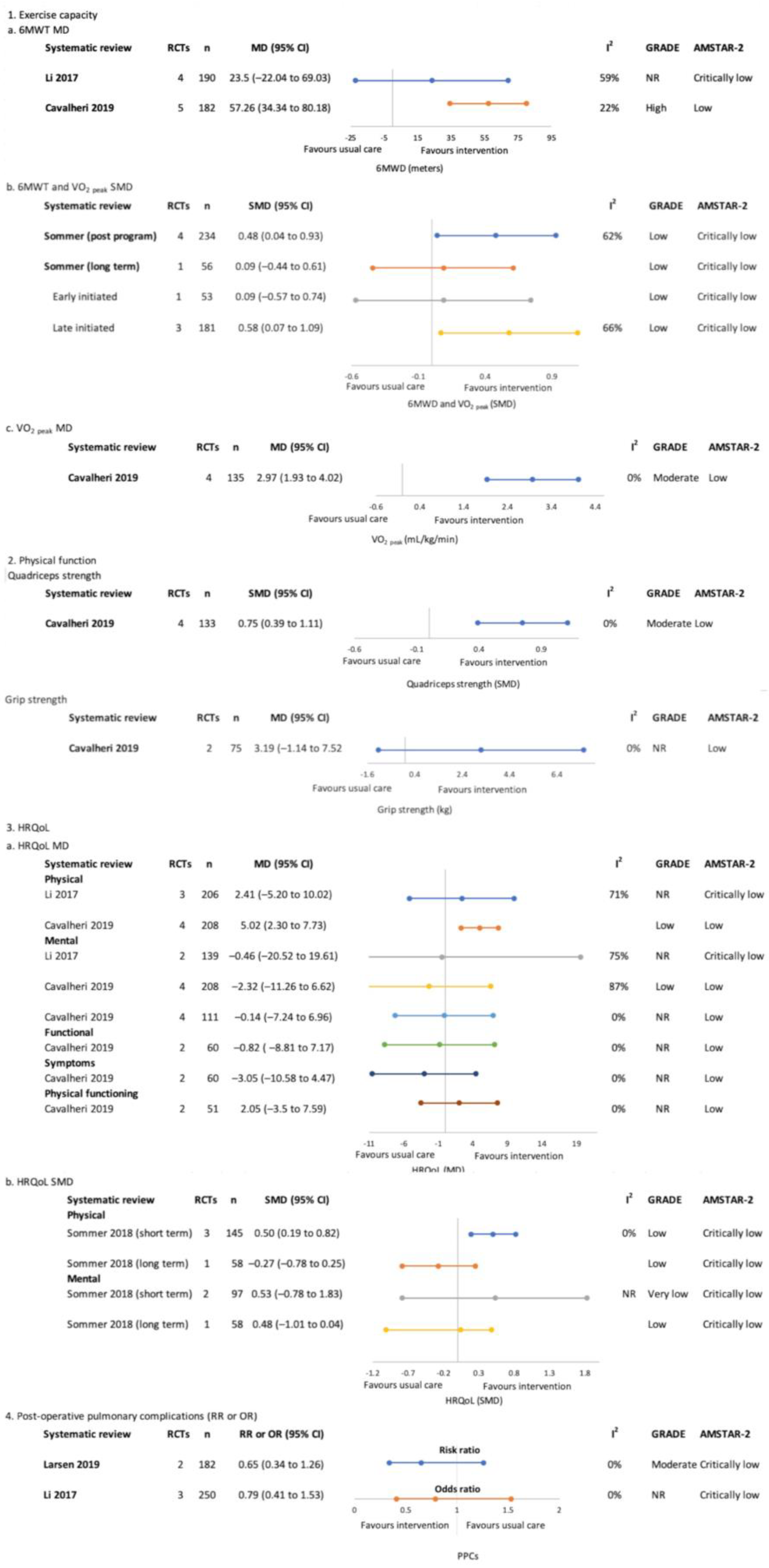
3.4.2. Post-Treatment Only (Surgical)—Figure 4
Exercise Capacity
Physical Function
Health-Related Quality of Life
Post-Operative Pulmonary Complications
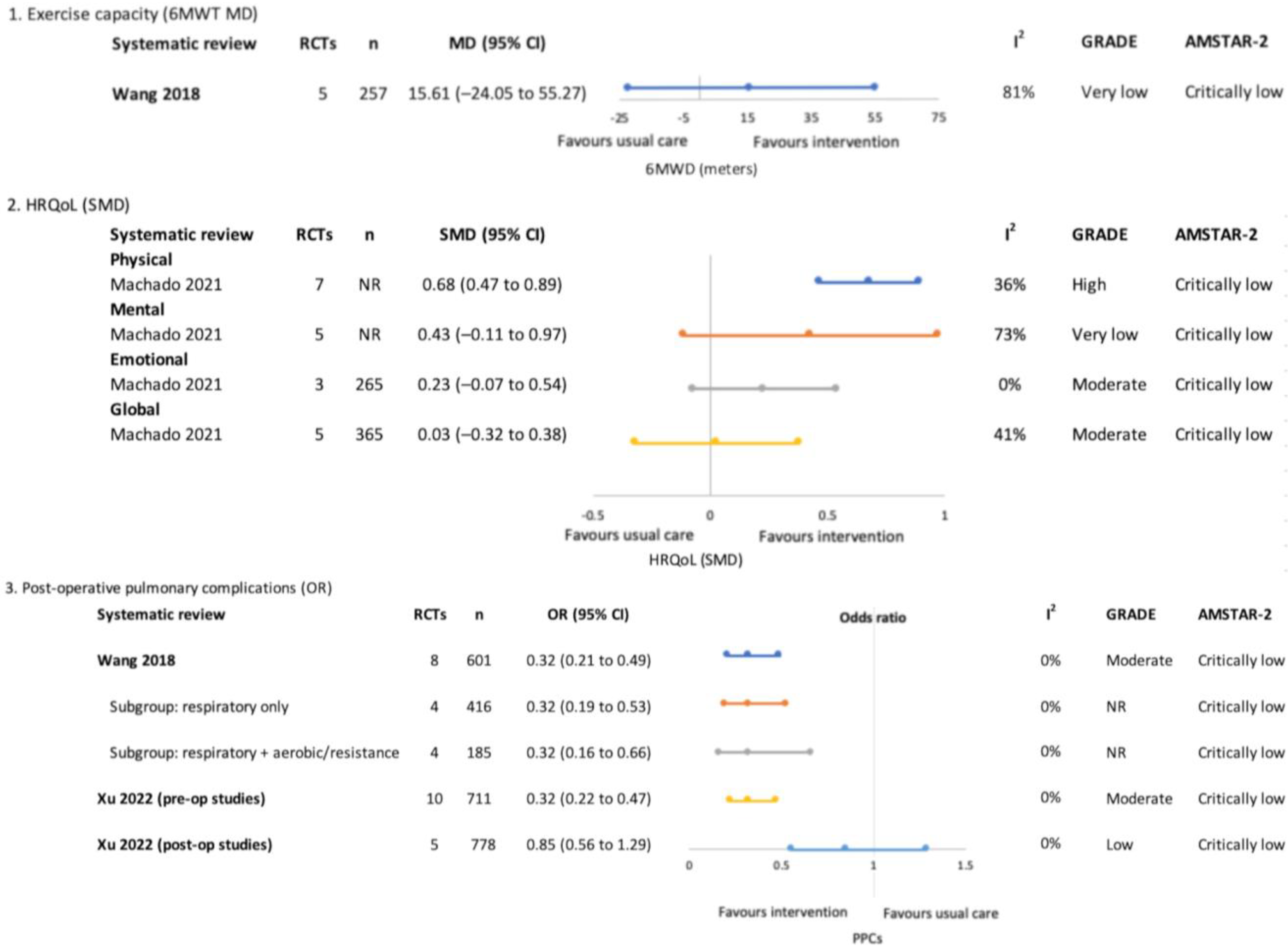
3.4.3. Pre and Post-Treatment (Surgical)—Figure 5
Exercise Capacity
Health-Related Quality of Life
Post-Operative Pulmonary Complications
3.4.4. Pre, during and/or Post-Treatment (Surgical and Non-Surgical)—Figure S2
Exercise Capacity
Physical Function
Health-Related Quality of Life
3.4.5. During and/or Post-Treatment (Non-Surgical)—Figure S3
Exercise Capacity
Physical Function
Health-Related Quality of Life
3.4.6. Safety
3.5. Overlap of Included Systematic Reviews
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute. Cancer Statistics Review 1975–2018; Howlader, N., Noone, A.M., Krapcho, M., Miller, D., Brest, A., Yu, M., Ruhl, J., Tatalovich, Z., Mariotto, A., Lewis, D.R., et al., Eds.; National Cancer Institute: Bethesda, MD, USA, 2021. [Google Scholar]
- Luo, Q.; O’Connell, D.L.; Yu, X.Q.; Kahn, C.; Caruana, M.; Pesola, F.; Sasieni, P.; Grogan, P.B.; Aranda, S.; Cabasag, C.J.; et al. Cancer incidence and mortality in Australia from 2020 to 2044 and an exploratory analysis of the potential effect of treatment delays during the COVID-19 pandemic: A statistical modelling study. Lancet Public Health 2022, 7, e537–e548. [Google Scholar] [CrossRef] [PubMed]
- Cavalheri, V.; Granger, C. Preoperative exercise training for patients with non-small cell lung cancer. Cochrane Database Syst. Rev. 2017, 6, CD012020. [Google Scholar] [CrossRef] [PubMed]
- Granger, C.L.; McDonald, C.F.; Berney, S.; Chao, C.; Denehy, L. Exercise intervention to improve exercise capacity and health related quality of life for patients with Non-small cell lung cancer: A systematic review. Lung Cancer 2011, 72, 139–153. [Google Scholar] [CrossRef]
- Shipton, D.A.; Dennett, A.M.; Sayner, A.M.; Tang, C.Y. A national survey of oncology physiotherapy services for cancer survivors in Australia. Asia Pac. J. Clin. Oncol. 2022, 18, e404–e413. [Google Scholar] [CrossRef]
- Zhou, W.; Woo, S.; Larson, J.L. Effects of perioperative exercise interventions on lung cancer patients: An overview of systematic reviews. J. Clin. Nurs. 2020, 29, 4482–4504. [Google Scholar] [CrossRef]
- Driessen, E.J.; Peeters, M.E.; Bongers, B.C.; Maas, H.A.; Bootsma, G.P.; van Meeteren, N.L.; Janssen-Heijnen, M.L. Effects of prehabilitation and rehabilitation including a home-based component on physical fitness, adherence, treatment tolerance, and recovery in patients with non-small cell lung cancer: A systematic review. Crit. Rev. Oncol. Hematol. 2017, 114, 63–76. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: New York, NY, USA, 2022. [Google Scholar]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. PLoS Med. 2021, 18, e1003583. [Google Scholar] [CrossRef]
- Veritas Health Innovation. Covidence Systematic Review Software; Veritas Health Innovation: Melbourne, Australia, 2021. [Google Scholar]
- Granholm, A.; Alhazzani, W.; Møller, M.H. Use of the GRADE approach in systematic reviews and guidelines. Br. J. Anaesth. 2019, 123, 554–559. [Google Scholar] [CrossRef]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, 358, j4008. [Google Scholar] [CrossRef] [PubMed]
- Pieper, D.; Antoine, S.L.; Mathes, T.; Neugebauer, E.A.; Eikermann, M. Systematic review finds overlapping reviews were not mentioned in every other overview. J. Clin. Epidemiol. 2014, 67, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Steffens, D.; Beckenkamp, P.R.; Hancock, M.; Solomon, M.; Young, J. Preoperative exercise halves the postoperative complication rate in patients with lung cancer: A systematic review of the effect of exercise on complications, length of stay and quality of life in patients with cancer. Br. J. Sport. Med. 2018, 52, 344. [Google Scholar] [CrossRef] [PubMed]
- Treanor, C.; Kyaw, T.; Donnelly, M. An international review and meta-analysis of prehabilitation compared to usual care for cancer patients. J. Cancer Surviv. Res. Pract. 2018, 12, 64–73. [Google Scholar] [CrossRef]
- Rosero, I.D.; Ramírez-Vélez, R.; Lucia, A.; Martínez-Velilla, N.; Santos-Lozano, A.; Valenzuela, P.L.; Morilla, I.; Izquierdo, M. Systematic Review and Meta-Analysis of Randomized, Controlled Trials on Preoperative Physical Exercise Interventions in Patients with Non-Small-Cell Lung Cancer. Cancers 2019, 11, 944. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, S.; Yan, S.; Wang, Y.; Wang, X.; Sihoe, A.D.; Yang, Y.; Wu, N. Impact of preoperative exercise therapy on surgical outcomes in lung cancer patients with or without COPD: A systematic review and meta-analysis. Cancer Manag. Res. 2019, 11, 1765–1777. [Google Scholar] [CrossRef] [PubMed]
- Pu, C.Y.; Batarseh, H.; Zafron, M.L.; Mador, M.J.; Yendamuri, S.; Ray, A.D. Effects of Preoperative Breathing Exercise on Postoperative Outcomes for Lung Cancer Patients Undergoing Curative Intent Lung Resection: A Meta-Analysis. Arch. Phys. Med. Rehabil. 2021, 102, 2416–2427. [Google Scholar] [CrossRef]
- Gravier, F.E.; Smondack, P.; Prieur, G.; Medrinal, C.; Combret, Y.; Muir, J.F.; Baste, J.M.; Cuvelier, A.; Boujibar, F.; Bonnevie, T. Effects of exercise training in people with non-small cell lung cancer before lung resection: A systematic review and meta-analysis. Thorax 2021, 77, 486–496. [Google Scholar] [CrossRef]
- De Oliveira Vacchi, C.; Martha, B.; Macagnan, F. Effect of inspiratory muscle training associated or not to physical rehabilitation in preoperative anatomic pulmonary resection: A systematic review and meta-analysis. Support Care Cancer 2021, 30, 1079–1092. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Guo, N.N.; Jin, H.R.; Yu, H.; Wang, P.; Xu, G.G. Effects of exercise training on patients with lung cancer who underwent lung resection: A meta-analysis. World J. Surg. Oncol. 2017, 15, 158. [Google Scholar] [CrossRef]
- Sommer, M.S.; Staerkind, M.E.; Christensen, J.; Vibe-Petersen, J.; Larsen, K.R.; Pedersen, J.H.; Langberg, H. Effect of postsurgical rehabilitation programmes in patients operated for lung cancer: A systematic review and meta-analysis. J. Rehabil. Med. 2018, 50, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Cavalheri, V.; Burtin, C.; Formico, V.R.; Nonoyama, M.L.; Jenkins, S.; Spruit, M.A.; Hill, K. Exercise training undertaken by people within 12 months of lung resection for non-small cell lung cancer. Cochrane Database Syst. Rev. 2019, 2019, CD009955. [Google Scholar] [CrossRef] [PubMed]
- Larsen, K.S.; Skoffer, B.; Gregersen Oestergaard, L.; Van Tulder, M.; Petersen, A.K. The effects of various respiratory physiotherapies after lung resection: A systematic review. Physiother. Theory Pract. 2020, 36, 1201–1219. [Google Scholar] [CrossRef] [PubMed]
- Rueda, J.R.; Solà, I.; Pascual, A.; Subirana Casacuberta, M. Non-invasive interventions for improving well-being and quality of life in patients with lung cancer. Cochrane Database Syst. Rev. 2011, 2011, CD004282. [Google Scholar] [CrossRef] [PubMed]
- Mainini, C.; Rebelo, P.F.; Bardelli, R.; Kopliku, B.; Tenconi, S.; Costi, S.; Tedeschi, C.; Fugazzaro, S. Perioperative physical exercise interventions for patients undergoing lung cancer surgery: What is the evidence? SAGE Open Med. 2016, 4, 2050312116673855. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Liu, X.; Jia, Y.; Xie, J. Impact of breathing exercises in subjects with lung cancer undergoing surgical resection: A systematic review and meta-analysis. J. Clin. Nurs. 2018, 28, 717–732. [Google Scholar] [CrossRef]
- Himbert, C.; Klossner, N.; Coletta, A.M.; Barnes, C.A.; Wiskemann, J.; LaStayo, P.C.; Varghese, T.K., Jr.; Ulrich, C.M. Exercise and lung cancer surgery: A systematic review of randomized-controlled trials. Crit. Rev. Oncol. Hematol. 2020, 156, 103086. [Google Scholar] [CrossRef]
- Machado, P.; Pimenta, S.; Oliveiros, B.; Ferreira, J.P.; Martins, R.A.; Cruz, J. Effect of Exercise Training on Quality of Life after Colorectal and Lung Cancer Surgery: A Meta-Analysis. Cancers 2021, 13, 4975. [Google Scholar] [CrossRef]
- Xu, X.; Cheung, D.S.T.; Smith, R.; Lai, A.Y.K.; Lin, C.C. The effectiveness of pre- and post-operative rehabilitation for lung cancer: A systematic review and meta-analysis on postoperative pulmonary complications and length of hospital stay. Clin. Rehabil. 2022, 36, 172–189. [Google Scholar] [CrossRef]
- Hsieh, C.C.; Hsiao, F.H. The effects of supportive care interventions on depressive symptoms among patients with lung cancer: A meta-analysis of randomized controlled studies. Palliat. Support. Care 2017, 15, 710–723. [Google Scholar] [CrossRef]
- Papadopoulos, D.; Papadoudis, A.; Kiagia, M.; Syrigos, K. Nonpharmacologic Interventions for Improving Sleep Disturbances in Patients with Lung Cancer: A Systematic Review and Meta-analysis. J. Pain Symptom Manag. 2018, 55, 1364–1381.e5. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, Y.-Q.; Xie, J. Effects of Breathing Exercises on Patients with Lung Cancer. Oncol. Nurs. Forum 2019, 46, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Spence, R.; Steele, M.L.; Hayes, S.; Toohey, K. Exercise for Individuals with Lung Cancer: A Systematic Review and Meta-Analysis of Adverse Events, Feasibility, and Effectiveness. Semin. Oncol. Nurs. 2020, 36, 151076. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.C. Effect of Exercise Interventions on Quality of Life in Patients with Lung Cancer: A Systematic Review of Randomized Controlled Trials. Oncol. Nurs. Forum 2020, 47, E58–E72. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Liu, L.; Gan, C.E.; Qiu, L.H.; Jiang, X.J.; He, X.T.; Zhang, J.E. Effects of home-based exercise on exercise capacity, symptoms, and quality of life in patients with lung cancer: A meta-analysis. Eur. J. Oncol. Nurs. 2020, 49, 101836. [Google Scholar] [CrossRef]
- Codima, A.; das Neves Silva, W.; de Souza Borges, A.P.; de Castro, G. Exercise prescription for symptoms and quality of life improvements in lung cancer patients: A systematic review. Support. Care Cancer 2021, 29, 445–457. [Google Scholar] [CrossRef]
- Ma, R.C.; Yin, Y.Y.; Wang, Y.Q.; Liu, X.; Xie, J. Systematic Review and Meta-analysis of Nonpharmacological Interventions for Lung Cancer Fatigue. West. J. Nurs. Res. 2021, 43, 392–402. [Google Scholar] [CrossRef]
- Heredia-Ciuró, A.; Fernández-Sánchez, M.; Martín-Núñez, J.; Calvache-Mateo, A.; Rodríguez-Torres, J.; López-López, L.; Valenza, M.C. High-intensity interval training effects in cardiorespiratory fitness of lung cancer survivors: A systematic review and meta-analysis. Support Care Cancer 2022, 30, 3017–3027. [Google Scholar] [CrossRef]
- Zhou, L.; Chen, Q.; Zhang, J. Effect of Exercise on Fatigue in Patients with Lung Cancer: A Systematic Review and Meta-Analysis of Randomized Trials. J. Palliat. Med. 2021, 24, 932–943. [Google Scholar] [CrossRef]
- Peddle-McIntyre, C.J.; Singh, F.; Thomas, R.; Newton, R.U.; Galvão, D.A.; Cavalheri, V. Exercise training for advanced lung cancer. Cochrane Database Syst. Rev. 2019, 2, CD012685. [Google Scholar] [CrossRef]
- Lee, J. Physiologic and psychologic adaptation to exercise interventions in lung cancer patients undergoing chemotherapy: A systematic review and meta-analysis of randomized controlled trials. Support. Care Cancer 2021, 29, 2863–2873. [Google Scholar] [CrossRef] [PubMed]
- Granger, C.; Cavalheri, V. Preoperative exercise training for people with non-small cell lung cancer. Cochrane Database Syst. Rev. 2022, 9. [Google Scholar] [CrossRef]
- Bradley, P.; Merchant, Z.; Rowlinson-Groves, K.; Taylor, M.; Moore, J.; Evison, M. Feasibility and outcomes of a real-world regional lung cancer prehabilitation programme in the UK. Br. J. Anaesth. 2023, 130, e47–e55. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Lin, R.; Zhang, C.; Zhang, M.; Wang, Y.; Zhang, Y. Exercise training modalities in patients with lung cancer: A protocol for systematic review and network meta-analysis. BMJ Open 2022, 12, e058788. [Google Scholar] [CrossRef]
- Scott, J.M.; Thomas, S.M.; Herndon, J.E.; Douglas, P.S.; Yu, A.F.; Rusch, V.; Huang, J.; Capaci, C.; Harrison, J.N.; Stoeckel, K.J.; et al. Effects and tolerability of exercise therapy modality on cardiorespiratory fitness in lung cancer: A randomized controlled trial. J. Cachexia Sarcopenia Muscle 2021, 12, 1456–1465. [Google Scholar] [CrossRef]
- Buffart, L.M.; Kalter, J.; Sweegers, M.G.; Courneya, K.S.; Newton, R.U.; Aaronson, N.K.; Jacobsen, P.B.; May, A.M.; Galvão, D.A.; Chinapaw, M.J.; et al. Effects and moderators of exercise on quality of life and physical function in patients with cancer: An individual patient data meta-analysis of 34 RCTs. Cancer Treat. Rev. 2017, 52, 91–104. [Google Scholar] [CrossRef]
- Edbrooke, L.; Denehy, L.; Granger, C.L.; Kapp, S.; Aranda, S. Home-based rehabilitation in inoperable non-small cell lung cancer-the patient experience. Support Care Cancer 2020, 28, 99–112. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Edbrooke, L.; Bowman, A.; Granger, C.L.; Burgess, N.; Abo, S.; Connolly, B.; Denehy, L. Exercise across the Lung Cancer Care Continuum: An Overview of Systematic Reviews. J. Clin. Med. 2023, 12, 1871. https://doi.org/10.3390/jcm12051871
Edbrooke L, Bowman A, Granger CL, Burgess N, Abo S, Connolly B, Denehy L. Exercise across the Lung Cancer Care Continuum: An Overview of Systematic Reviews. Journal of Clinical Medicine. 2023; 12(5):1871. https://doi.org/10.3390/jcm12051871
Chicago/Turabian StyleEdbrooke, Lara, Amy Bowman, Catherine L. Granger, Nicola Burgess, Shaza Abo, Bronwen Connolly, and Linda Denehy. 2023. "Exercise across the Lung Cancer Care Continuum: An Overview of Systematic Reviews" Journal of Clinical Medicine 12, no. 5: 1871. https://doi.org/10.3390/jcm12051871
APA StyleEdbrooke, L., Bowman, A., Granger, C. L., Burgess, N., Abo, S., Connolly, B., & Denehy, L. (2023). Exercise across the Lung Cancer Care Continuum: An Overview of Systematic Reviews. Journal of Clinical Medicine, 12(5), 1871. https://doi.org/10.3390/jcm12051871