Effects of Investigational Moisturizers on the Skin Barrier and Microbiome following Exposure to Environmental Aggressors: A Randomized Clinical Trial and Ex Vivo Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design and Protocol
2.3. Investigational Products
2.4. Endpoints and Assessments
2.5. Statistical Analysis
2.6. Exploratory Endpoints of Skin Microbiome
2.7. Microbiome Diversity Analysis
2.8. Skin Explant Chamber Stimuli Model Assay
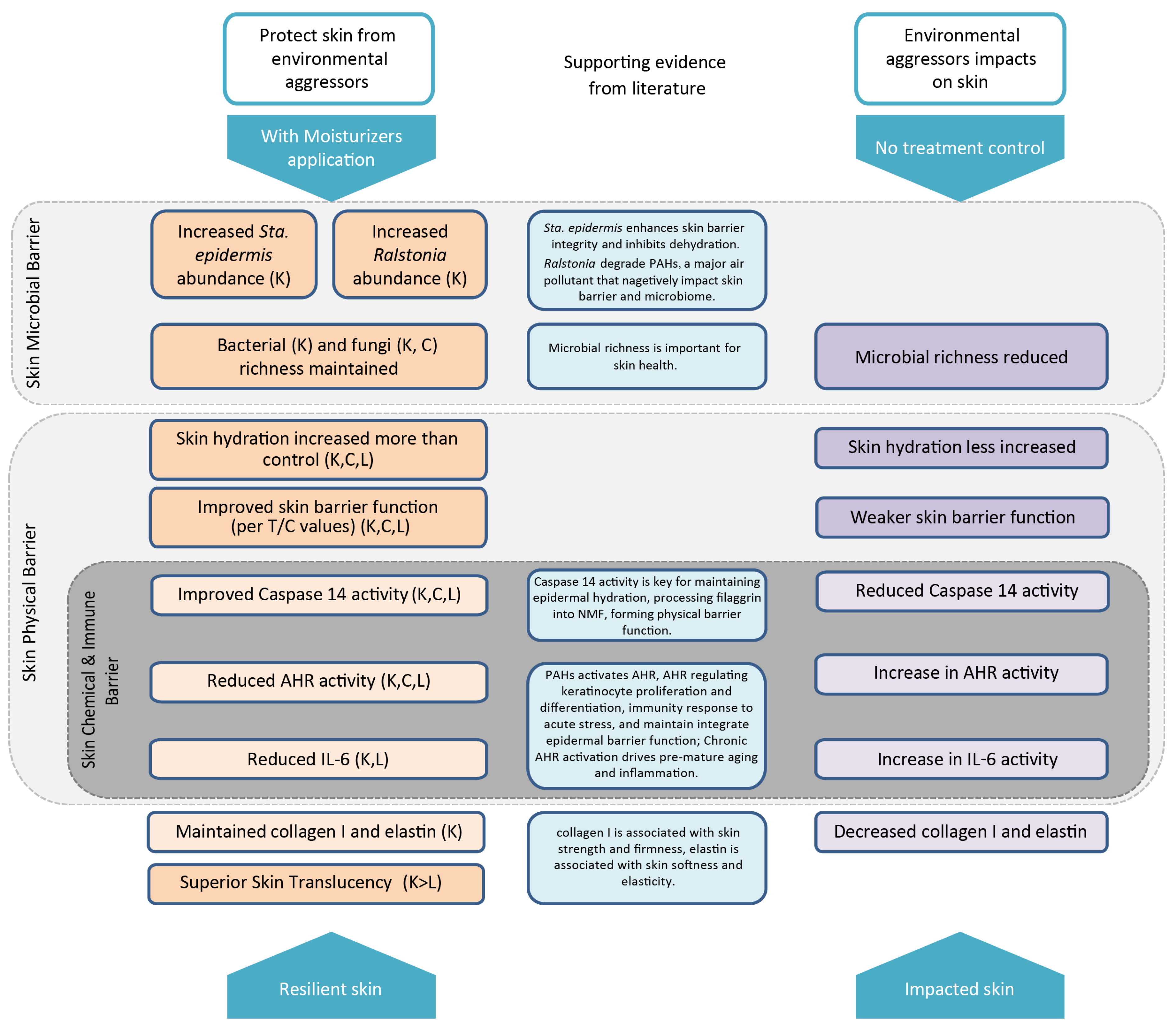
3. Results
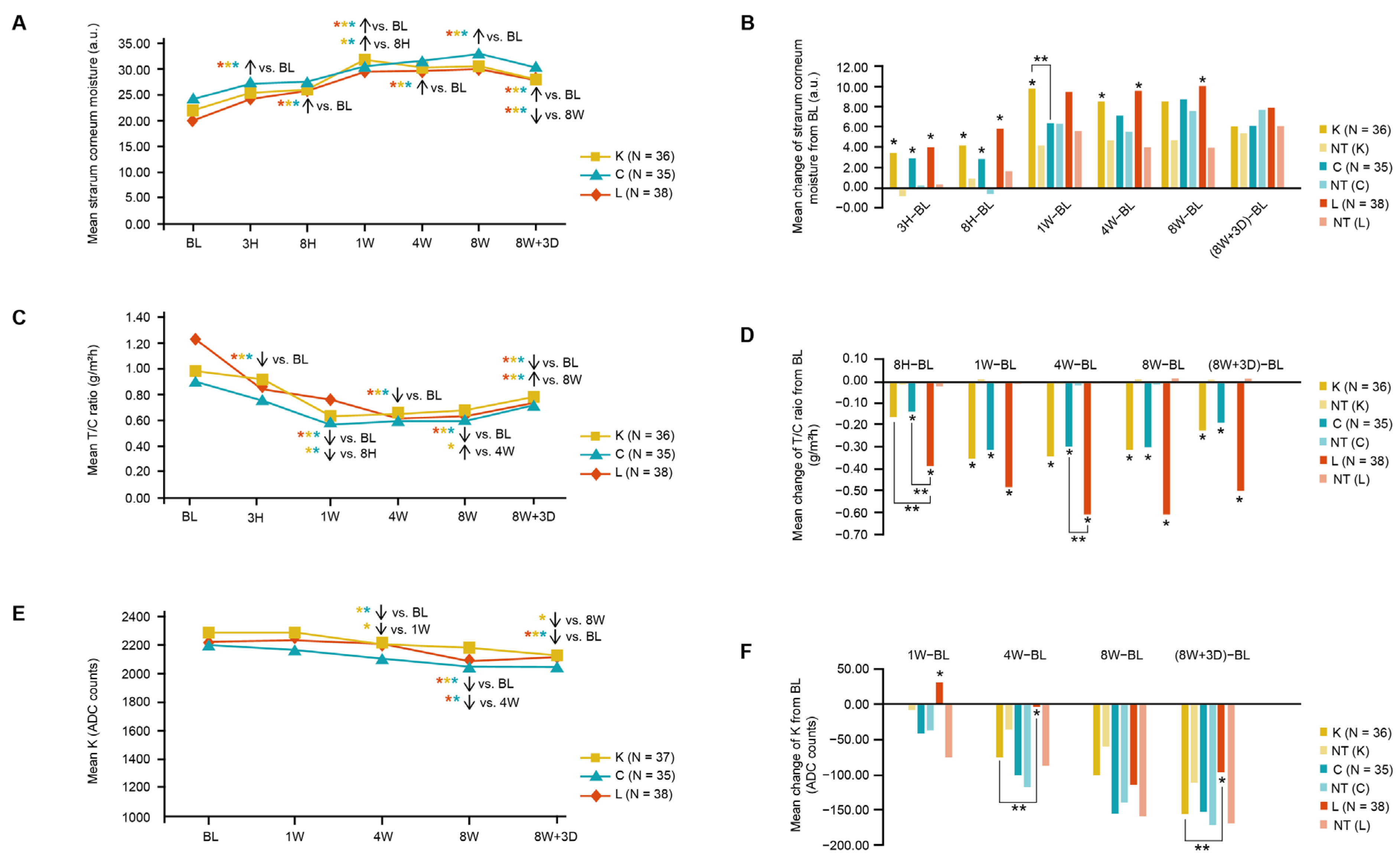
3.1. Skin Clinical Parameters in Three Moisturizer Groups
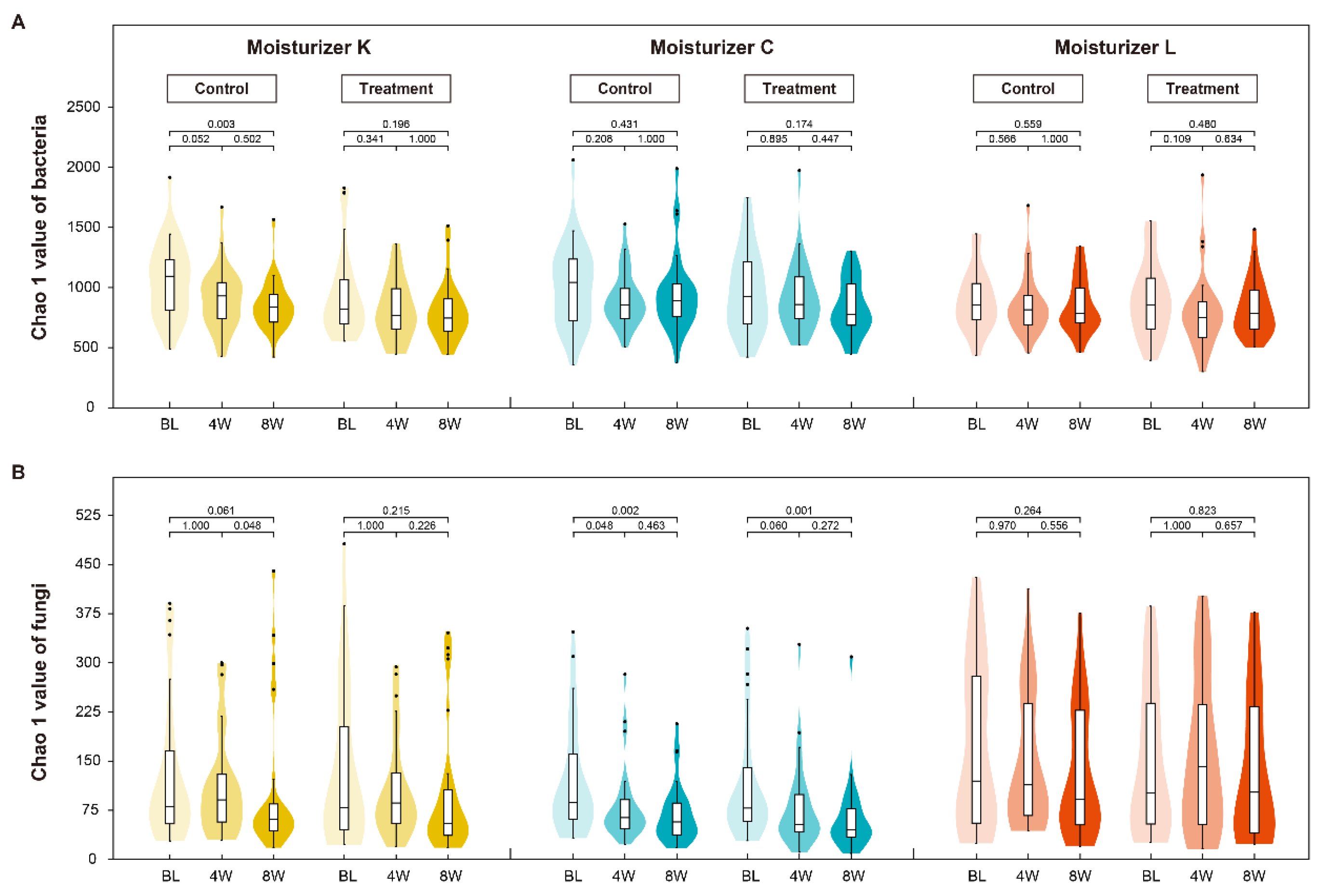
3.2. Skin Microbiome Composition
3.3. Factors Affecting Microbiome Composition and Diversity
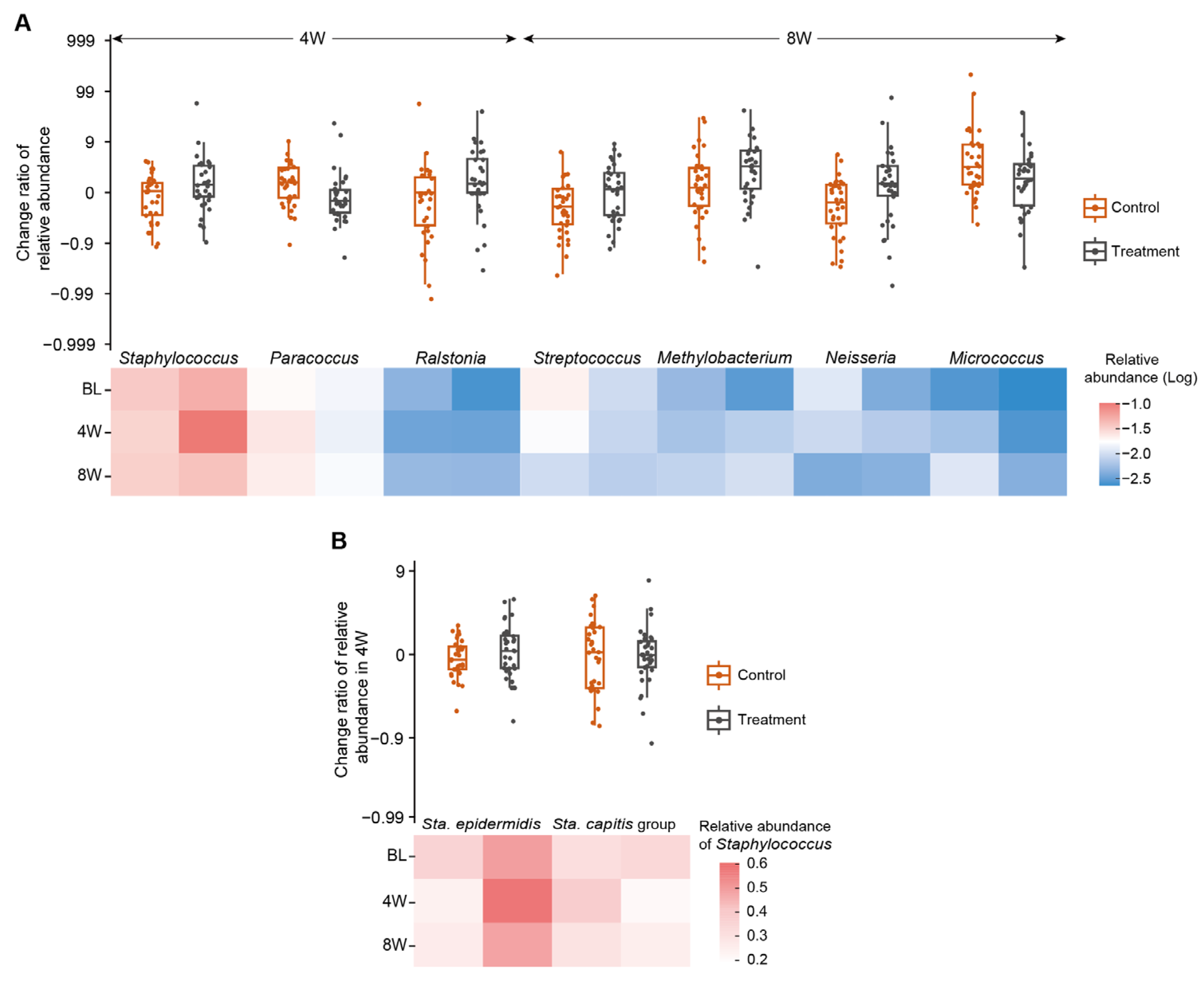
3.4. Impact of Investigational Moisturizers on the Skin Microbiome
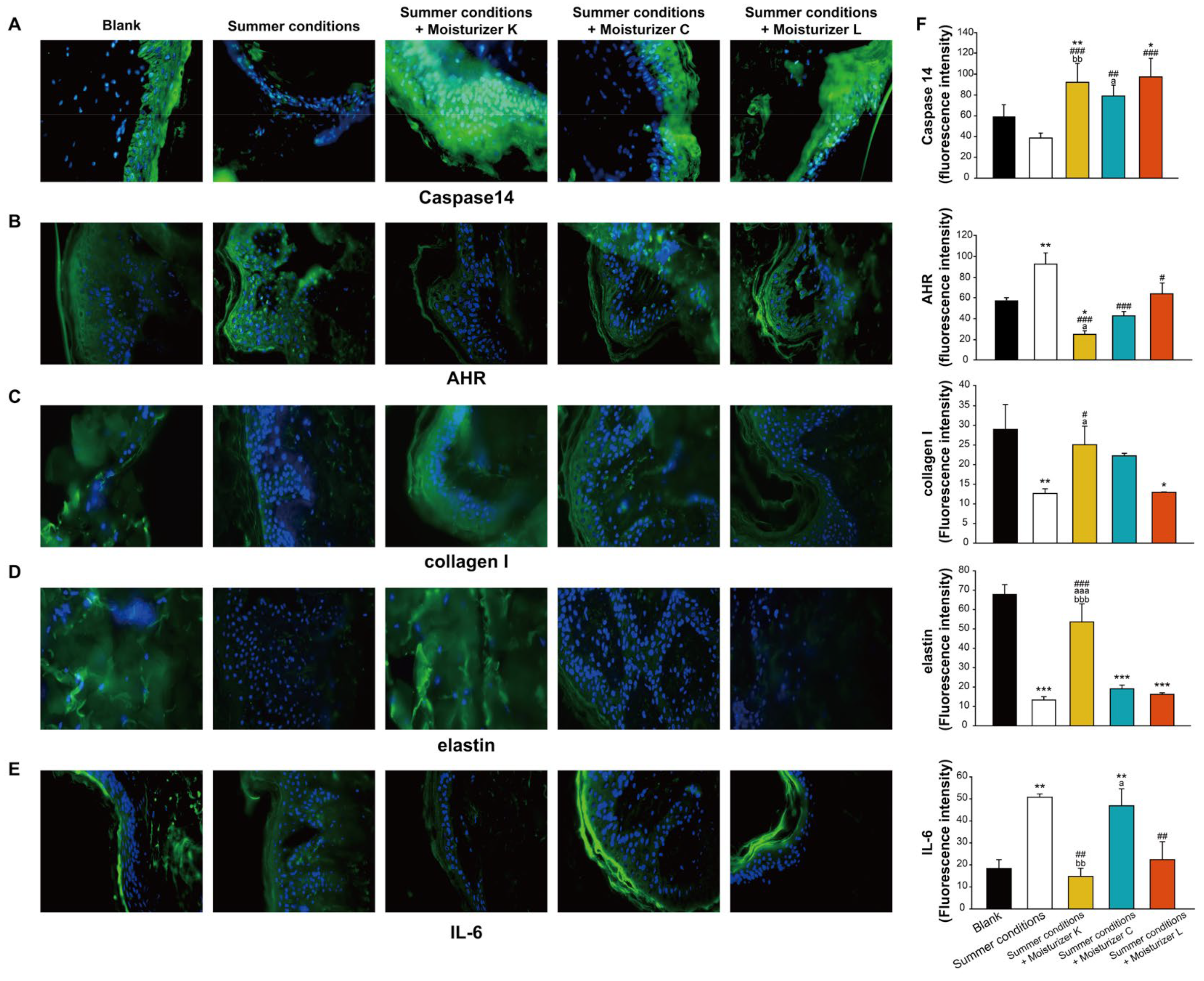
3.5. Ex Vivo Chamber Stimulus Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, X.; Xing, L.; Lai, R.; Yuan, C.; Humbert, P. Literature mapping: Association of microscopic skin microflora and biomarkers with macroscopic skin health. Clin. Exp. Dermatol. 2021, 46, 21–27. [Google Scholar] [CrossRef]
- Byrd, A.L.; Belkaid, Y.; Segre, J.A. The human skin microbiome. Nat. Rev. Microbiol. 2018, 16, 143–155. [Google Scholar] [CrossRef]
- Huse, S.M.; Ye, Y.; Zhou, Y.; Fodor, A.A. A core human microbiome as viewed through 16S rRNA sequence clusters. PLoS ONE 2012, 7, e34242. [Google Scholar] [CrossRef] [PubMed]
- Richmond, J.M.; Harris, J.E. Immunology and skin in health and disease. Cold Spring Harb. Perspect. Med. 2014, 4, a015339. [Google Scholar] [CrossRef] [PubMed]
- Krutmann, J.; Bouloc, A.; Sore, G.; Bernard, B.A.; Passeron, T. The skin aging exposome. J. Dermatol. Sci. 2017, 85, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Krutmann, J.; Liu, W.; Li, L.; Pan, X.; Crawford, M.; Sore, G.; Seite, S. Pollution and skin: From epidemiological and mechanistic studies to clinical implications. J. Dermatol. Sci. 2014, 76, 163–168. [Google Scholar] [CrossRef]
- Parrado, C.; Mercado-Saenz, S.; Perez-Davo, A.; Gilaberte, Y.; Gonzalez, S.; Juarranz, A. Environmental stressors on skin aging. Mechanistic insights. Front. Pharmacol. 2019, 10, 759. [Google Scholar] [CrossRef]
- Jansen van Rensburg, S.; Franken, A.; Du Plessis, J.L. Measurement of transepidermal water loss, stratum corneum hydration and skin surface pH in occupational settings: A review. Skin Res. Technol. 2019, 25, 595–605. [Google Scholar] [CrossRef]
- Purnamawati, S.; Indrastuti, N.; Danarti, R.; Saefudin, T. The role of moisturizers in addressing various kinds of dermatitis: A review. Clin. Med. Res. 2017, 15, 75–87. [Google Scholar] [CrossRef]
- Loden, M. The clinical benefit of moisturizers. J. Eur. Acad. Dermatol. Venereol. 2005, 19, 672–688. [Google Scholar] [CrossRef]
- Kim, B.; Cho, H.E.; Moon, S.H.; Ahn, H.J.; Bae, S.; Cho, H.D.; An, S. Transdermal delivery systems in cosmetics. Biomed. Dermatol. 2020, 4, 10. [Google Scholar] [CrossRef]
- Robinson, M.; Visscher, M.; Laruffa, A.; Wickett, R. Natural moisturizing factors (NMF) in the stratum corneum (SC). I. Effects of lipid extraction and soaking. J. Cosmet. Sci. 2010, 61, 13–22. Available online: https://www.ncbi.nlm.nih.gov/pubmed/20211113 (accessed on 13 November 2021). [PubMed]
- Bouslimani, A.; da Silva, R.; Kosciolek, T.; Janssen, S.; Callewaert, C.; Amir, A.; Dorrestein, K.; Melnik, A.V.; Zaramela, L.S.; Kim, J.N.; et al. The impact of skin care products on skin chemistry and microbiome dynamics. BMC Biol. 2019, 17, 47. [Google Scholar] [CrossRef]
- Velasco, M.V.; Sauce, R.; Oliveira, C.A.; Pinto, C.A.; Martinez, R.M.; Baah, S.; Almeida, T.S.; Rosado, C.; Baby, A.R. Active ingredients, mechanisms of action and efficacy tests of antipollution cosmetic and personal care products. Braz. J. Pharm. Sci. 2018, 54, e0103. [Google Scholar] [CrossRef]
- Marques, G.A.; Hiraishi, C.F.; Macedo, P.I.S.; Pinto, C.A.S.O.; Gregório, J.; Rosado, C.; Velasco, M.V.R.; Baby, A.R. HPLC-TBARS-EVSC (high-performance liquid chromatography-thiobarbituric acid reactive substances-ex vivo stratum corneum) protocol: Selection of the subjects and approach to present the results. Int. J. Cosmet. Sci. 2023. Online ahead of print. [Google Scholar] [CrossRef]
- Li, C.; Jiang, Y.; Wang, D.Q. 28,140 How a lotion with prebiotic yeast extract regulates the environmental aggressors impact on the skin microbiome. J. Am. Acad. Dermatol. 2021, 85, AB175. [Google Scholar] [CrossRef]
- Manpreet, R. Topical Composition Containing Glycerin and Yeast Extract. US Patent No. US-2018161267-A1, 2018. Available online: https://www.freepatentsonline.com/y2018/0161267.html (accessed on 22 February 2018).
- Li, X.; Yuan, C.; Xing, L.; Humbert, P. Topographical diversity of common skin microflora and its association with skin environment type: An observational study in Chinese women. Sci. Rep. 2017, 7, 18046. [Google Scholar] [CrossRef]
- Li, X.; Galzote, C.; Yan, X.; Li, L.; Wang, X. Characterization of Chinese body skin through in vivo instrument assessments, visual evaluations, and questionnaire: Influences of body area, inter-generation, season, sex, and skin care habits. Skin Res. Technol. 2014, 20, 14–22. [Google Scholar] [CrossRef]
- Beijing Moji Fengyun Technology Co., Ltd. Moji Weather: Shanghai Weather Information; Beijing Moji Fengyun Technology Co., Ltd.: Beijing, China, 2017; Available online: https://tianqi.moji.com/weather/china/shanghai/shanghai (accessed on 25 July 2017).
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Kong, F.; Galzote, C.; Duan, Y. Change in skin properties over the first 10 years of life: A cross-sectional study. Arch. Dermatol. Res. 2017, 309, 653–658. [Google Scholar] [CrossRef]
- Kim, G.; Kim, M.; Kim, M.; Park, C.; Yoon, Y.; Lim, D.H.; Yeo, H.; Kang, S.; Lee, Y.G.; Beak, N.I.; et al. Spermidine-induced recovery of human dermal structure and barrier function by skin microbiome. Commun. Biol. 2021, 4, 231. [Google Scholar] [CrossRef] [PubMed]
- Dorrestein, P.C.; Gallo, R.L.; Knight, R. Microbial skin inhabitants: Friends forever. Cell 2016, 165, 771–772. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Byrd, A.L.; Park, M.; Program, N.C.S.; Kong, H.H.; Segre, J.A. Temporal stability of the human skin microbiome. Cell 2016, 165, 854–866. [Google Scholar] [CrossRef]
- Hoste, E.; Kemperman, P.; Devos, M.; Denecker, G.; Kezic, S.; Yau, N.; Gilbert, B.; Lippens, S.; De Groote, P.; Roelandt, R.; et al. Caspase-14 is required for filaggrin degradation to natural moisturizing factors in the skin. J. Investig. Dermatol. 2011, 131, 2233–2241. [Google Scholar] [CrossRef]
- Markiewicz, A.; Sigorski, D.; Markiewicz, M.; Owczarczyk-Saczonek, A.; Placek, W. Caspase-14—From biomolecular basics to clinical approach. A review of available data. Int. J. Mol. Sci. 2021, 22, 5575. [Google Scholar] [CrossRef] [PubMed]
- Southall, M.; Bianchini, J.; Mahmood, K.; Li, W.H.; Parsa, R.; Brillouet, A.S. 18294 Topical treatment with kiwi-derived yeast extract increases hyaluronic acid resulting in improved skin hydration and clinically perceived facial radiance. J. Am. Acad. Dermatol. 2020, 83, AB209. [Google Scholar] [CrossRef]
- Egert, M.; Simmering, R. The microbiota of the human skin. Adv. Exp. Med. Biol. 2016, 902, 61–81. [Google Scholar] [CrossRef]
- Prescott, S.L.; Larcombe, D.L.; Logan, A.C.; West, C.; Burks, W.; Caraballo, L.; Levin, M.; Etten, E.V.; Horwitz, P.; Kozyrskyj, A.; et al. The skin microbiome: Impact of modern environments on skin ecology, barrier integrity, and systemic immune programming. World Allergy Organ. J. 2017, 10, 29. [Google Scholar] [CrossRef]
- Iwase, T.; Uehara, Y.; Shinji, H.; Tajima, A.; Seo, H.; Takada, K.; Agata, T.; Mizunoe, Y. Staphylococcus epidermidis Esp. inhibits Staphylococcus aureus biofilm formation and nasal colonization. Nature 2010, 465, 346–349. [Google Scholar] [CrossRef]
- Zipperer, A.; Konnerth, M.C.; Laux, C.; Berscheid, A.; Janek, D.; Weidenmaier, C.; Burian, M.; Schilling, N.A.; Slavetinsky, C.; Marschal, M.; et al. Human commensals producing a novel antibiotic impair pathogen colonization. Nature 2016, 535, 511–516. [Google Scholar] [CrossRef]
- Nakatsuji, T.; Chen, T.H.; Narala, S.; Chun, K.A.; Two, A.M.; Yun, T.; Shafiq, F.; Kotol, P.F.; Bouslimani, A.; Melnik, A.V.; et al. Antimicrobials from human skin commensal bacteria protect against Staphylococcus aureus and are deficient in atopic dermatitis. Sci. Transl. Med. 2017, 9, 378. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Hunt, R.L.; Villaruz, A.E.; Fisher, E.L.; Liu, R.; Liu, Q.; Cheung, G.Y.C.; Li, M.; Otto, M. Commensal Staphylococcus epidermidis contributes to skin barrier homeostasis by generating protective ceramides. Cell Host Microbe 2022, 30, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Cau, L.; Williams, M.R.; Butcher, A.M.; Nakatsuji, T.; Kavanaugh, J.S.; Cheng, J.Y.; Shafiq, F.; Higbee, K.; Hata, T.R.; Horswill, A.R.; et al. Staphylococcus epidermidis protease EcpA can be a deleterious component of the skin microbiome in atopic dermatitis. J. Allergy Clin. Immunol. 2021, 147, 955–966. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Jeong, S.E.; Lee, S.; Kim, S.; Han, H.; Jeon, C.O. Effects of cosmetics on the skin microbiome of facial cheeks with different hydration levels. MicrobiologyOpen 2018, 7, e00557. [Google Scholar] [CrossRef]
- Ghosal, D.; Ghosh, S.; Dutta, T.K.; Ahn, Y. Current state of knowledge in microbial degradation of polycyclic aromatic hydrocarbons (PAHs): A review. Front. Microbiol. 2016, 7, 1369. [Google Scholar] [CrossRef]
- Zhou, N.Y.; Fuenmayor, S.L.; Williams, P.A. nag genes of Ralstonia (formerly Pseudomonas) sp. strain U2 encoding enzymes for gentisate catabolism. J. Bacteriol. 2001, 183, 700–708. [Google Scholar] [CrossRef]
- Alalaiwe, A.; Lin, Y.K.; Lin, C.H.; Wang, P.W.; Lin, J.Y.; Fang, J.Y. The absorption of polycyclic aromatic hydrocarbons into the skin to elicit cutaneous inflammation: The establishment of structure-permeation and in silico-in vitro-in vivo relationships. Chemosphere 2020, 255, 126955. [Google Scholar] [CrossRef]
- Leung, M.H.Y.; Tong, X.; Bastien, P.; Guinot, F.; Tenenhaus, A.; Appenzeller, B.M.R.; Betts, R.J.; Mezzache, S.; Li, J.; Bourokba, N.; et al. Changes of the human skin microbiota upon chronic exposure to polycyclic aromatic hydrocarbon pollutants. Microbiome 2020, 8, 100. [Google Scholar] [CrossRef]
- Esser, C.; Bargen, I.; Weighardt, H.; Haarmann-Stemmann, T.; Krutmann, J. Functions of the aryl hydrocarbon receptor in the skin. Semin. Immunopathol. 2013, 35, 677–691. [Google Scholar] [CrossRef]
- Furue, M.; Takahara, M.; Nakahara, T.; Uchi, H. Role of AhR/ARNT system in skin homeostasis. Arch. Dermatol. Res. 2014, 306, 769–779. [Google Scholar] [CrossRef]
- Vogeley, C.; Esser, C.; Tuting, T.; Krutmann, J.; Haarmann-Stemmann, T. Role of the aryl hydrocarbon receptor in environmentally induced skin aging and skin carcinogenesis. Int. J. Mol. Sci. 2019, 20, 6005. [Google Scholar] [CrossRef] [PubMed]
- Uberoi, A.; Bartow, M.K.C.; Zheng, Q.; Flowers, L.; Campbell, A.; Knight, S.A.B.; Chan, N.; Wei, M.; Lovins, V.; Bugayev, J.; et al. Commensal microbiota regulates skin barrier function and repair via signaling through the aryl hydrocarbon receptor. Cell Host Microbe 2021, 29, 1235–1248. [Google Scholar] [CrossRef] [PubMed]
- Ying, S.; Zeng, D.N.; Chi, L.; Tan, Y.; Galzote, C.; Cardona, C.; Lax, S.; Gilbert, J.; Quan, Z.X. The Influence of Age and Gender on Skin-Associated Microbial Communities in Urban and Rural Human Populations. PLoS ONE 2015, 10, e0141842. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Liu, X.; Kong, F.Q.; Duan, Y.Y.; Yee, A.L.; Kim, M.; Galzote, C.; Gilbert, J.A.; Quan, Z.X. Age and Mothers: Potent Influences of Children’s Skin Microbiota. J. Investig. Dermatol. 2019, 132, 2497–2505. [Google Scholar] [CrossRef]
- Toju, H.; Tanabe, A.S.; Yamamoto, S.; Sato, H. High-coverage ITS primers for the DNA-based identification of ascomycetes and basidiomycetes in environmental samples. PLoS ONE 2012, 7, e40863. [Google Scholar] [CrossRef]
- Zhu, T.; Duan, Y.Y.; Kong, F.Q.; Galzote, C.; Quan, Z.X. Dynamics of Skin Mycobiome in Infants. Front. Microbiol. 2020, 11, 1790. [Google Scholar] [CrossRef]
- Hamady, M.; Walker, J.J.; Harris, J.K.; Gold, N.J.; Knight, R. Error-correcting barcoded primers for pyrosequencing hundreds of samples in multiplex. Nat. Methods 2008, 5, 235–237. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glockner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Wang, Q.; Garrity, M.G.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef]
- Yoon, S.H.; Ha, S.M.; Kwon, S.; Lim, J.; Kim, Y.; Seo, H.; Chun, J. Introducing EzBioCloud: A taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int. J. Syst. Evol. Microbiol. 2017, 67, 1613–1617. [Google Scholar] [CrossRef]
- Nilsson, R.H.; Larsson, K.H.; Taylor, A.F.S.; Bengtsson-Palme, J.; Jeppesen, T.S.; Schigel, D.; Kennedy, P.; Picard, K.; Glockner, F.O.; Tedersoo, L.; et al. The UNITE database for molecular identification of fungi: Handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res. 2019, 47, D259–D264. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
| Moisturizer Type | ||||
|---|---|---|---|---|
| K a | C a | L a | Total | |
| Enrolled participants (n) | 40 | 41 | 41 | 122 |
| Completed (drop out) participants for clinical study (n) | 37 (3) | 35 (6) | 38 (3) | 110 (12) |
| Completed (drop out) participants for microbiome study (n) | 32 (8) | 32 (9) | 33 (8) | 97 (25) |
| Age (mean ± SD) a | 32.6 ± 5.5 | 33.2 ± 5.2 | 32.3 ± 5.6 | |
| Skin type (clinical study; n = 110) | ||||
| Normal (n) | 5 | 2 | 6 | 13 |
| Dry (n) | 24 | 26 | 29 | 79 |
| Oily (n) | 0 | 0 | 0 | 0 |
| Combination (n) | 8 | 7 | 3 | 18 |
| Sensitive (n) | 5 | 5 | 1 | 11 |
| Non-sensitive (n) | 32 | 30 | 37 | 99 |
| Lifestyle/occupation (clinical study; n = 110) | ||||
| Student (n) | 4 | 2 | 5 | 11 |
| Housewife (n) | 5 | 6 | 7 | 18 |
| Office worker (n) | 23 | 22 | 20 | 65 |
| Outdoor worker (n) | 4 | 5 | 2 | 11 |
| Other (n) | 1 | 0 | 4 | 5 |
| Lifestyle/occupation (microbiome study; n = 97) | ||||
| Student (n) | 3 | 2 | 4 | 9 |
| Housewife (n) | 5 | 4 | 5 | 14 |
| Office worker (n) | 20 | 21 | 19 | 60 |
| Outdoor worker (n) | 3 | 5 | 2 | 10 |
| Other (n) | 1 | 0 | 3 | 4 |
| Factor | Bacterial Community | Fungal Community | ||||||
|---|---|---|---|---|---|---|---|---|
| Weighted UniFrac | Unweighted UniFrac | Bray–Curtis | Binary Jaccard | |||||
| F Value | p-Value | F Value | p-Value | F Value | p-Value | F Value | p-Value | |
| Site | 1.438 | 0.181 | 2.574 | 0.001 | 52.67 | 0.001 | 2.309 | 0.001 |
| Environment-dependent lifestyle | 4.360 | 0.001 | 1.542 | 0.001 | 1.548 | 0.037 | 1.522 | 0.001 |
| Time | 0.976 | 0.417 | 1.608 | 0.001 | 11.68 | 0.001 | 4.127 | 0.001 |
| Individual | 4.665 | 0.001 | 1.732 | 0.001 | 2.089 | 0.001 | 1.921 | 0.001 |
| Moisturizer b | 2.650 | 0.019 | 1.192 | 0.068 | 1.700 | 0.044 | 1.702 | 0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, D.-Q.; Li, X.; Zhang, R.-Y.; Yuan, C.; Yan, B.; Humbert, P.; Quan, Z.-X. Effects of Investigational Moisturizers on the Skin Barrier and Microbiome following Exposure to Environmental Aggressors: A Randomized Clinical Trial and Ex Vivo Analysis. J. Clin. Med. 2023, 12, 6078. https://doi.org/10.3390/jcm12186078
Wang D-Q, Li X, Zhang R-Y, Yuan C, Yan B, Humbert P, Quan Z-X. Effects of Investigational Moisturizers on the Skin Barrier and Microbiome following Exposure to Environmental Aggressors: A Randomized Clinical Trial and Ex Vivo Analysis. Journal of Clinical Medicine. 2023; 12(18):6078. https://doi.org/10.3390/jcm12186078
Chicago/Turabian StyleWang, Dan-Qi, Xi Li, Ru-Yi Zhang, Chao Yuan, Bo Yan, Philippe Humbert, and Zhe-Xue Quan. 2023. "Effects of Investigational Moisturizers on the Skin Barrier and Microbiome following Exposure to Environmental Aggressors: A Randomized Clinical Trial and Ex Vivo Analysis" Journal of Clinical Medicine 12, no. 18: 6078. https://doi.org/10.3390/jcm12186078
APA StyleWang, D.-Q., Li, X., Zhang, R.-Y., Yuan, C., Yan, B., Humbert, P., & Quan, Z.-X. (2023). Effects of Investigational Moisturizers on the Skin Barrier and Microbiome following Exposure to Environmental Aggressors: A Randomized Clinical Trial and Ex Vivo Analysis. Journal of Clinical Medicine, 12(18), 6078. https://doi.org/10.3390/jcm12186078









