Predictive Value of Motor Evoked Potentials in the Resection of Intradural Extramedullary Spinal Tumors in Children
Abstract
1. Introduction
2. Materials and Methods
2.1. Clinical Evaluation
2.2. Motor Evoked Potentials
2.3. Statistical Analysis
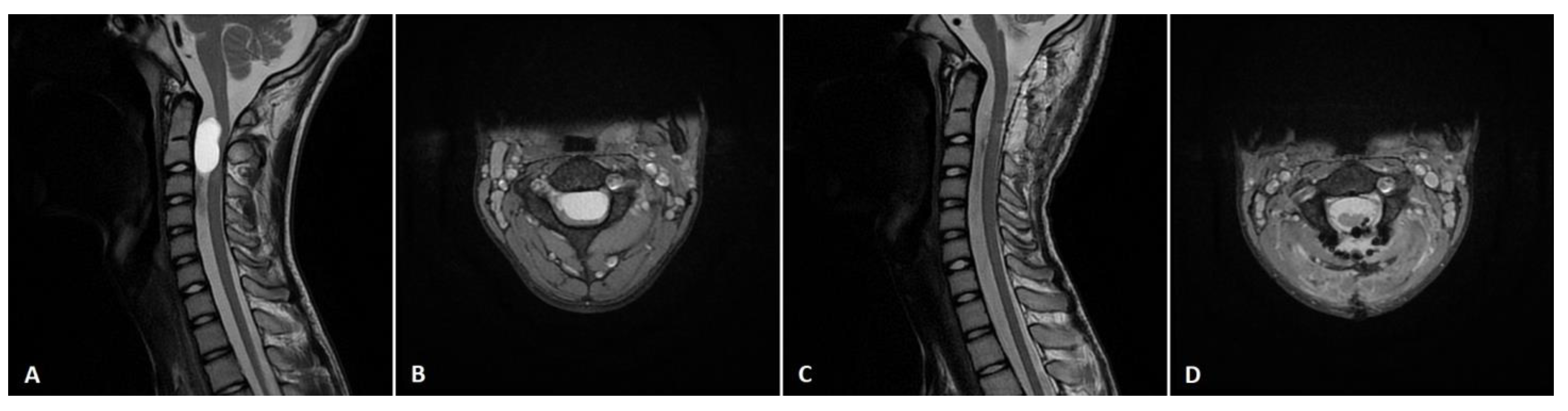
3. Results
The Impact of MEP on the Extent of Tumor Resection
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Baysefer, A.; Akay, K.M.; Izci, Y.; Kayali, H.; Timurkaynak, E. The clinical and surgical aspects of spinal tumors in children. Pediatr. Neurol. 2004, 31, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Binning, M.; Klimo, P.; Gluf, W.; Goumnerova, L. Spinal Tumors in Children. Neurosurg. Clin. N. Am. 2007, 18, 631–658. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.; Gandolfo, C.; Morana, G.; Tortori-Donati, P. Tumors of the Spine in Children. Neuroimaging Clin. N. Am. 2007, 17, 17–35. [Google Scholar] [CrossRef] [PubMed]
- van der Wal, E.C.; Klimek, M.; Rijs, K.; Scheltens-de Boer, M.; Biesheuvel, K.; Harhangi, B.S. Intraoperative Neuromonitoring in Patients with Intradural Extramedullary Spinal Cord Tumor: A Single-Center Case Series. World Neurosurg. 2021, 147, e516–e523. [Google Scholar] [CrossRef] [PubMed]
- Bidkar, P.U.; Thakkar, A.; Manohara, N.; Rao, K.S. Intraoperative neurophysiological monitoring in paediatric neurosurgery. Int. J. Clin. Pract. 2021, 75, e14160. [Google Scholar] [CrossRef]
- Costa, P.; Peretta, P.; Faccani, G. Relevance of intraoperative D wave in spine and spinal cord surgeries. Eur. Spine J. 2013, 22, 840–848. [Google Scholar] [CrossRef]
- Hsu, W.; Bettegowda, C.; Jallo, G.I. Intramedullary spinal cord tumor surgery: Can we do it without intraoperative neurophysiological monitoring? Child’s Nerv. Syst. 2010, 26, 241–245. [Google Scholar] [CrossRef]
- Rashad, S.; Elwany, A.; Farhoud, A. Surgery for spinal intramedullary tumors: Technique, outcome and factors affecting resectability. Neurosurg. Rev. 2018, 41, 503–511. [Google Scholar] [CrossRef]
- Tobin, M.K.; Geraghty, J.R.; Engelhard, H.H.; Linninger, A.A.; Mehta, A.I. Intramedullary spinal cord tumors: A review of current and future treatment strategies. Neurosurg. Focus 2015, 39, E14. [Google Scholar] [CrossRef]
- Azad, T.; Pendharkar, A.V.; Nguyen, V.; Pan, J.; Connolly, I.D.; Veeravagu, A.; Popat, R.; Ratliff, J.K.; Grant, G.A. Diagnostic Utility of Intraoperative Neurophysiological Monitoring for Intramedullary Spinal Cord Tumors: Systematic Review and Meta-Analysis. Clin. Spine Surg. 2018, 31, 112–119. [Google Scholar] [CrossRef]
- Kimchi, G.; Knoller, N.; Korn, A.; Eyal-Mazuz, Y.; Sapir, Y.; Peled, A.; Harel, R. Delayed variations in the diagnostic accuracy of intraoperative neuromonitoring in the resection of intramedullary spinal cord tumors. Neurosurg. Focus 2021, 50, E21. [Google Scholar] [CrossRef]
- Nuwer, M.R.; Emerson, R.G.; Galloway, G.; Legatt, A.D.; Lopez, J.; Minahan, R.; Yamada, T.; Goodin, D.S.; Armon, C.; Chaudhry, V.; et al. Evidence-Based Guideline Update: Intraoperative Spinal Monitoring with Somatosensory and Transcranial Electrical Motor Evoked Potentials. J. Clin. Neurophysiol. 2012, 29, 101–108. [Google Scholar] [CrossRef]
- Rijs, K.; Klimek, M.; Scheltens-de Boer, M.; Biesheuvel, K.; Harhangi, B.S. Intraoperative Neuromonitoring in Patients with Intramedullary Spinal Cord Tumor: A Systematic Review, Meta-Analysis, and Case Series. World Neurosurg. 2019, 125, 498–510.e2. [Google Scholar] [CrossRef]
- Sala, F.; Palandri, G.; Basso, E.; Lanteri, P.; Deletis, V.; Faccioli, F.; Bricolo, A. Motor Evoked Potential Monitoring Improves Outcome after Surgery for Intramedullary Spinal Cord Tumors: A Historical Control Study. Neurosurgery 2006, 58, 1129–1143. [Google Scholar] [CrossRef]
- Verla, T.; Fridley, J.S.; Khan, A.B.; Mayer, R.R.; Omeis, I. Neuromonitoring for Intramedullary Spinal Cord Tumor Surgery. World Neurosurg. 2016, 95, 108–116. [Google Scholar] [CrossRef]
- Zuccaro, M.; Zuccaro, J.; Samdani, A.F.; Pahys, J.M.; Hwang, S.W. Intraoperative neuromonitoring alerts in a pediatric deformity center. Neurosurg. Focus 2017, 43, E8. [Google Scholar] [CrossRef]
- Ghadirpour, R.; Nasi, D.; Iaccarino, C.; Giraldi, D.; Sabadini, R.; Motti, L.; Sala, F.; Servadei, F. Intraoperative neurophysiological monitoring for intradural extramedullary tumors: Why not? Clin. Neurol. Neurosurg. 2015, 130, 140–149. [Google Scholar] [CrossRef]
- Ghadirpour, R.; Nasi, D.; Iaccarino, C.; Romano, A.; Motti, L.; Sabadini, R.; Valzania, F.; Servadei, F. Intraoperative neurophysiological monitoring for intradural extramedullary spinal tumors: Predictive value and relevance of D-wave amplitude on surgical outcome during a 10-year experience. J. Neurosurg. Spine 2018, 30, 259–267. [Google Scholar] [CrossRef]
- Harel, R.; Schleifer, D.; Appel, S.; Attia, M.; Cohen, Z.R.; Knoller, N. Spinal intradural extramedullary tumors: The value of intraoperative neurophysiologic monitoring on surgical outcome. Neurosurg. Rev. 2017, 40, 613–619. [Google Scholar] [CrossRef]
- Ishida, W.; Casaos, J.; Chandra, A.; D’Sa, A.; Ramhmdani, S.; Perdomo-Pantoja, A.; Theodore, N.; Jallo, G.; Gokaslan, Z.L.; Wolinsky, J.-P.; et al. Diagnostic and therapeutic values of intraoperative electrophysiological neuromonitoring during resection of intradural extramedullary spinal tumors: A single-center retrospective cohort and meta-analysis. J. Neurosurg. Spine 2019, 30, 839–849. [Google Scholar] [CrossRef]
- Korn, A.; Halevi, D.; Lidar, Z.; Biron, T.; Ekstein, P.; Constantini, S. Intraoperative neurophysiological monitoring during resection of intradural extramedullary spinal cord tumors: Experience with 100 cases. Acta Neurochir. 2015, 157, 819–830. [Google Scholar] [CrossRef] [PubMed]
- Antkowiak, L.; Putz, M.; Sordyl, R.; Pokora, S.; Mandera, M. Relevance of intraoperative motor evoked potentials and D-wave monitoring for the resection of intramedullary spinal cord tumors in children. Neurosurg. Rev. 2022, 45, 2723–2731. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.S.; Ivan, M.E.; Stapleton, C.J.; Quinones-Hinojosa, A.; Gupta, N.; Auguste, K.I. Intraoperative changes in transcranial motor evoked potentials and somatosensory evoked potentials predicting outcome in children with intramedullary spinal cord tumors. J. Neurosurg. Pediatr. 2014, 13, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Morota, N.; Deletis, V.; Constantini, S.; Kofler, M.; Cohen, H.; Epstein, F.J. The Role of Motor Evoked Potentials during Surgery for Intramedullary Spinal Cord Tumors. Neurosurgery 1997, 41, 1327–1336. [Google Scholar] [CrossRef] [PubMed]
- Fulkerson, D.H.; Satyan, K.B.; Wilder, L.M.; Riviello, J.J.; Stayer, S.A.; Whitehead, W.E.; Curry, D.J.; Dauser, R.C.; Luerssen, T.G.; Jea, A. Intraoperative monitoring of motor evoked potentials in very young children. J. Neurosurg. Pediatr. 2011, 7, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Lieberman, J.A.; Lyon, R.; Feiner, J.; Diab, M.; Gregory, G.A. The Effect of Age on Motor Evoked Potentials in Children under Propofol/Isoflurane Anesthesia. Anesth. Analg. 2006, 103, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.G.; Kim, K.; Shin, H.-I.; Bang, M.S.; Kim, H.-S.; Choi, J.; Wang, K.-C.; Kim, S.-K.; Lee, J.Y.; Phi, J.H.; et al. Feasibility of intraoperative monitoring of motor evoked potentials obtained through transcranial electrical stimulation in infants younger than 3 months. J. Neurosurg. Pediatr. 2019, 23, 758–766. [Google Scholar] [CrossRef]
- Sala, F.; Manganotti, P.; Grossauer, S.; Tramontanto, V.; Mazza, C.; Gerosa, M. Intraoperative neurophysiology of the motor system in children: A tailored approach. Child’s Nerv. Syst. 2010, 26, 473–490. [Google Scholar] [CrossRef]
- Marrazzo, A.; Cacchione, A.; Rossi, S.; Carboni, A.; Gandolfo, C.; Carai, A.; Mastronuzzi, A.; Colafati, G.S. Intradural Pediatric Spinal Tumors: An Overview from Imaging to Novel Molecular Findings. Diagnostics 2021, 11, 1710. [Google Scholar] [CrossRef]
- Fan, F.; Zhou, J.; Zheng, Y.; Liu, S.; Tang, Z.; Wang, Y. Clinical Features, Treatments, and Prognostic Factors of Spinal Myxopapillary Ependymoma. World Neurosurg. 2021, 149, e1105–e1111. [Google Scholar] [CrossRef]
- Cofano, F.; Giambra, C.; Costa, P.; Zeppa, P.; Bianconi, A.; Mammi, M.; Monticelli, M.; Di Perna, G.; Junemann, C.V.; Melcarne, A.; et al. Management of Extramedullary Intradural Spinal Tumors: The Impact of Clinical Status, Intraoperative Neurophysiological Monitoring and Surgical Approach on Outcomes in a 12-Year Double-Center Experience. Front. Neurol. 2020, 11, 598619. [Google Scholar] [CrossRef]
- Kang, H.; Gwak, H.S.; Shin, S.H.; Woo, M.K.; Jeong, I.H.; Yoo, H.; Kwon, J.W.; Lee, S.H. Monitoring rate and predictability of intraoperative monitoring in patients with intradural extramedullary and epidural metastatic spinal tumors. Spinal Cord 2017, 55, 906–910. [Google Scholar] [CrossRef][Green Version]
| No. | Sex | Age (Years) | McCormick Grade at Admission | Neurological Deficits | Tumor Location (Vertebral Levels) | Histology | MEP Events | McCormick Grade at Discharge | Postoperative Deficits |
|---|---|---|---|---|---|---|---|---|---|
| 1 | F | 17 | 1 | - | S1-S3, posterolateral | Medulloblastoma (CSF seeding) | 1 | ||
| 2 | F | 16 | 1 | Sensory | L5-S2, anterior | Myxopapillary ependymoma | 1 | ||
| 3 | M | 15 | 2 | Motor | T12-L2, L5-S3, posterior | Myxopapillary ependymoma | 2 | ||
| 4 | M | 9 | 2 | Sensory, motor | L2-L4, lateral | Schwannoma | 2 | ||
| 5 | F | 5 | 4 | Motor | C4-C7, anterolateral | Rhabdoid meningioma | YES | 5 | Motor |
| 6 | M | 6 | 3 | Motor | L5-S2, posterior | Epidermal cyst | 3 | ||
| 7 | M | 1 | 2 | - | C1-C5, posterolateral | Lipoma | 1 | ||
| 8 | F | 14 | 4 | Sensory, motor | T12, L4, posterior | Clear cell meningioma | 3 | ||
| 9 | M | 18 | 4 | Sensory, motor | T12-L2, posterolateral | Myxopapillary ependymoma | 4 | ||
| 10 | F | 15 | 1 | - | L1-L4, posterior | Ependymoma | 1 | ||
| 11 | M | 8 | 5 | Motor, urinary incontinence | C2-C4, posterior | NGGCT (CSF seeding) | 4 | ||
| 12 | M | 7 | 3 | Sensory, motor | C5-T4, posterolateral | Anaplastic ependymoma | YES | 4 | Motor |
| 13 | F | 7 | 5 | Sensory, motor, urinary incontinence | T12-L4, posterior | Dermoid cyst | 5 | ||
| 14 | F | 16 | 1 | - | L3-L5, lateral | Clear cell meningioma | 1 | ||
| 15 | F | 7 | 1 | - | L4, posterior | Myxopapillary ependymoma | 1 | ||
| 16 | M | 14 | 3 | Motor | C1-C2, anterolateral | Endodermal cyst | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antkowiak, L.; Putz, M.; Sordyl, R.; Pokora, S.; Mandera, M. Predictive Value of Motor Evoked Potentials in the Resection of Intradural Extramedullary Spinal Tumors in Children. J. Clin. Med. 2023, 12, 41. https://doi.org/10.3390/jcm12010041
Antkowiak L, Putz M, Sordyl R, Pokora S, Mandera M. Predictive Value of Motor Evoked Potentials in the Resection of Intradural Extramedullary Spinal Tumors in Children. Journal of Clinical Medicine. 2023; 12(1):41. https://doi.org/10.3390/jcm12010041
Chicago/Turabian StyleAntkowiak, Lukasz, Monika Putz, Ryszard Sordyl, Szymon Pokora, and Marek Mandera. 2023. "Predictive Value of Motor Evoked Potentials in the Resection of Intradural Extramedullary Spinal Tumors in Children" Journal of Clinical Medicine 12, no. 1: 41. https://doi.org/10.3390/jcm12010041
APA StyleAntkowiak, L., Putz, M., Sordyl, R., Pokora, S., & Mandera, M. (2023). Predictive Value of Motor Evoked Potentials in the Resection of Intradural Extramedullary Spinal Tumors in Children. Journal of Clinical Medicine, 12(1), 41. https://doi.org/10.3390/jcm12010041






