Pulmonary Arterial Hypertension and Adverse Outcomes after Kidney Transplantation: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Sources and Search Strategy
2.2. Eligibility Criteria and Outcomes
2.3. Data Collection and Synthesis
2.4. Quality Assessment
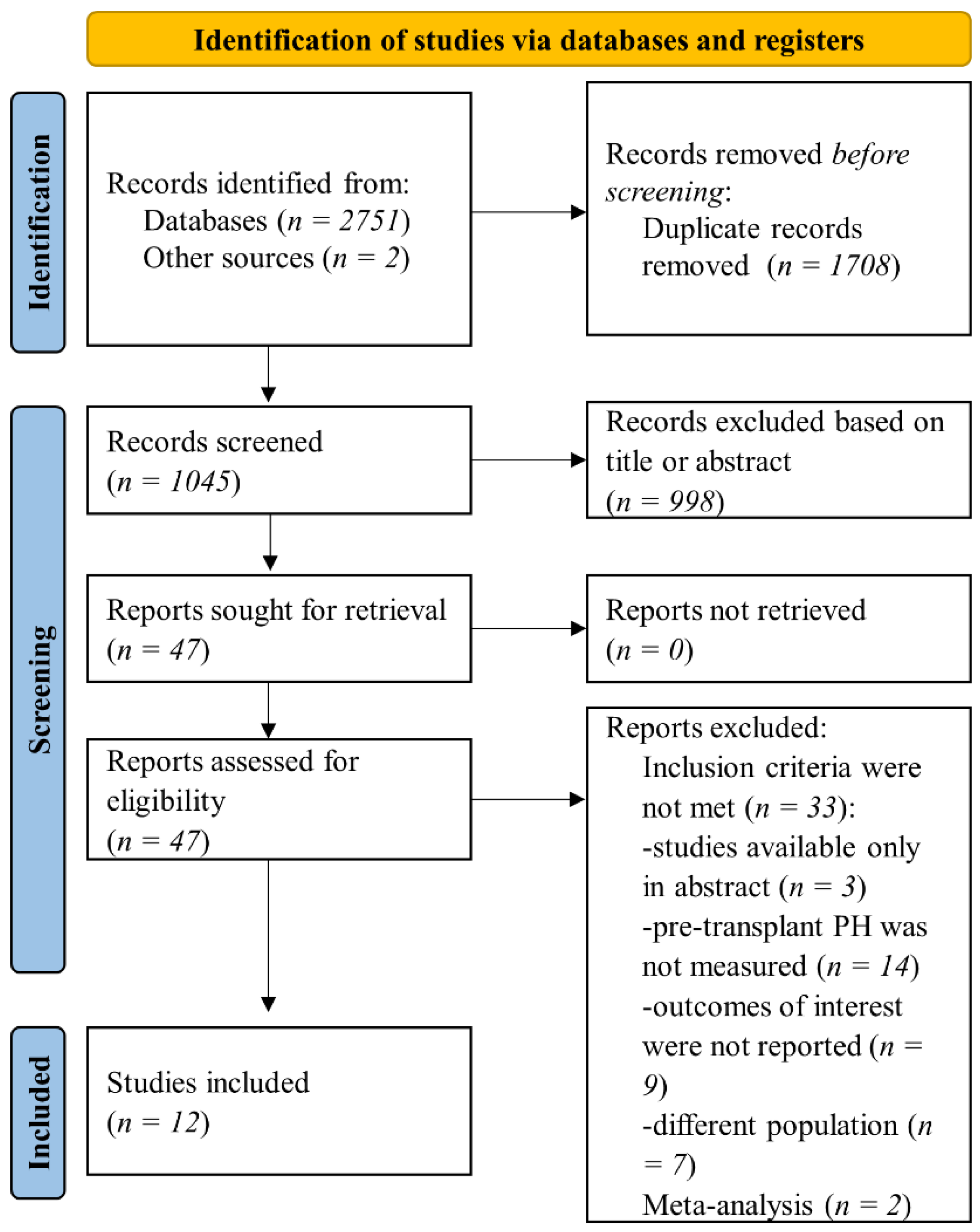
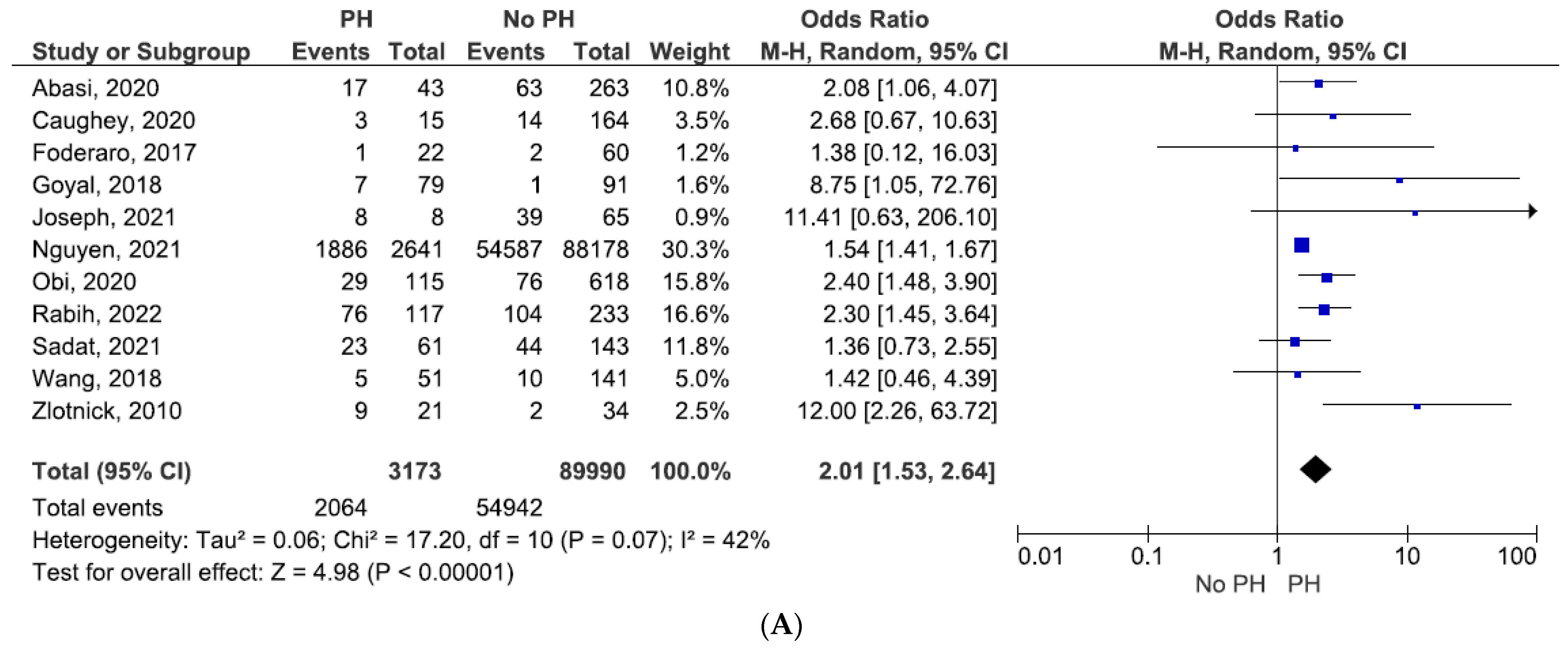
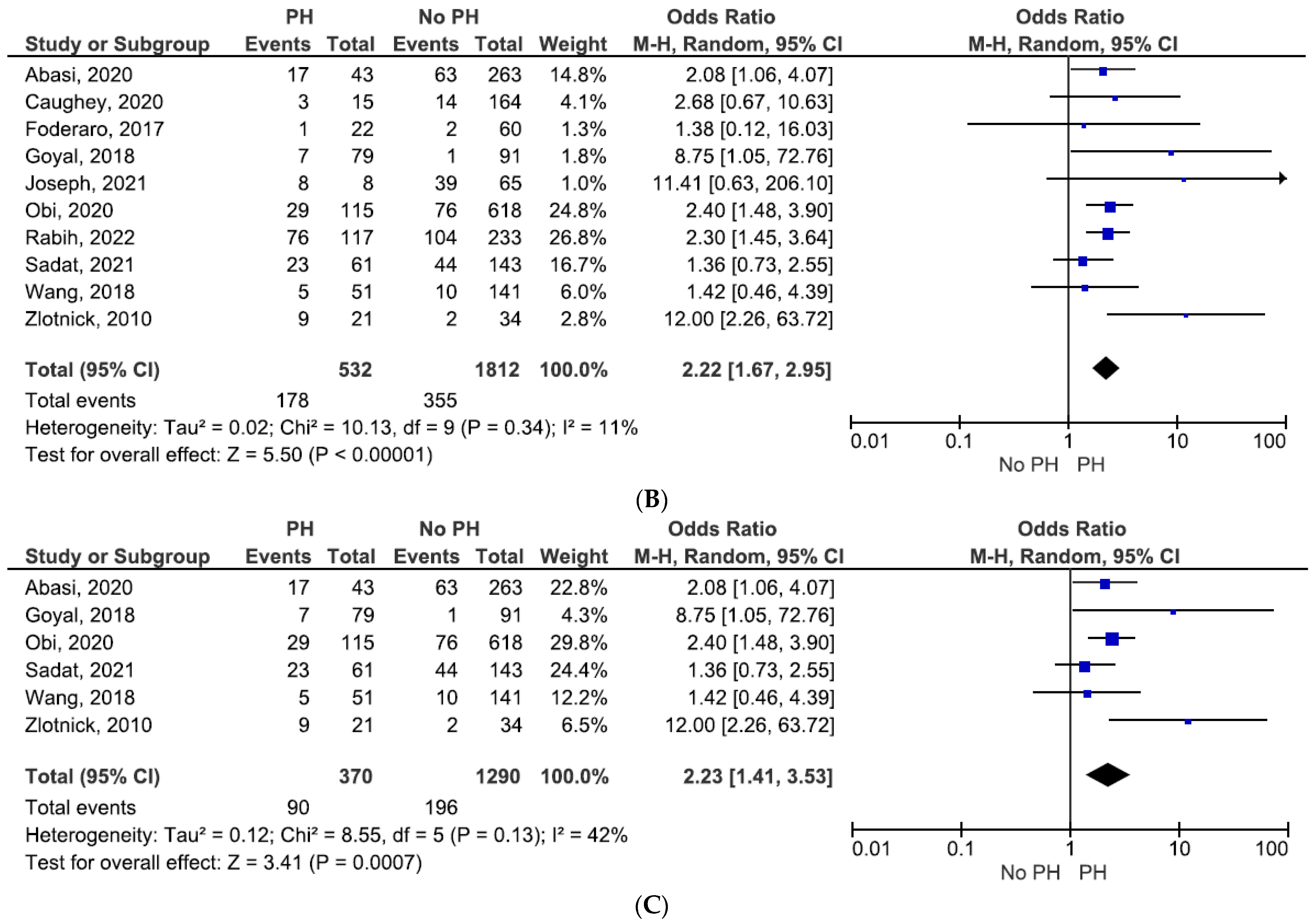
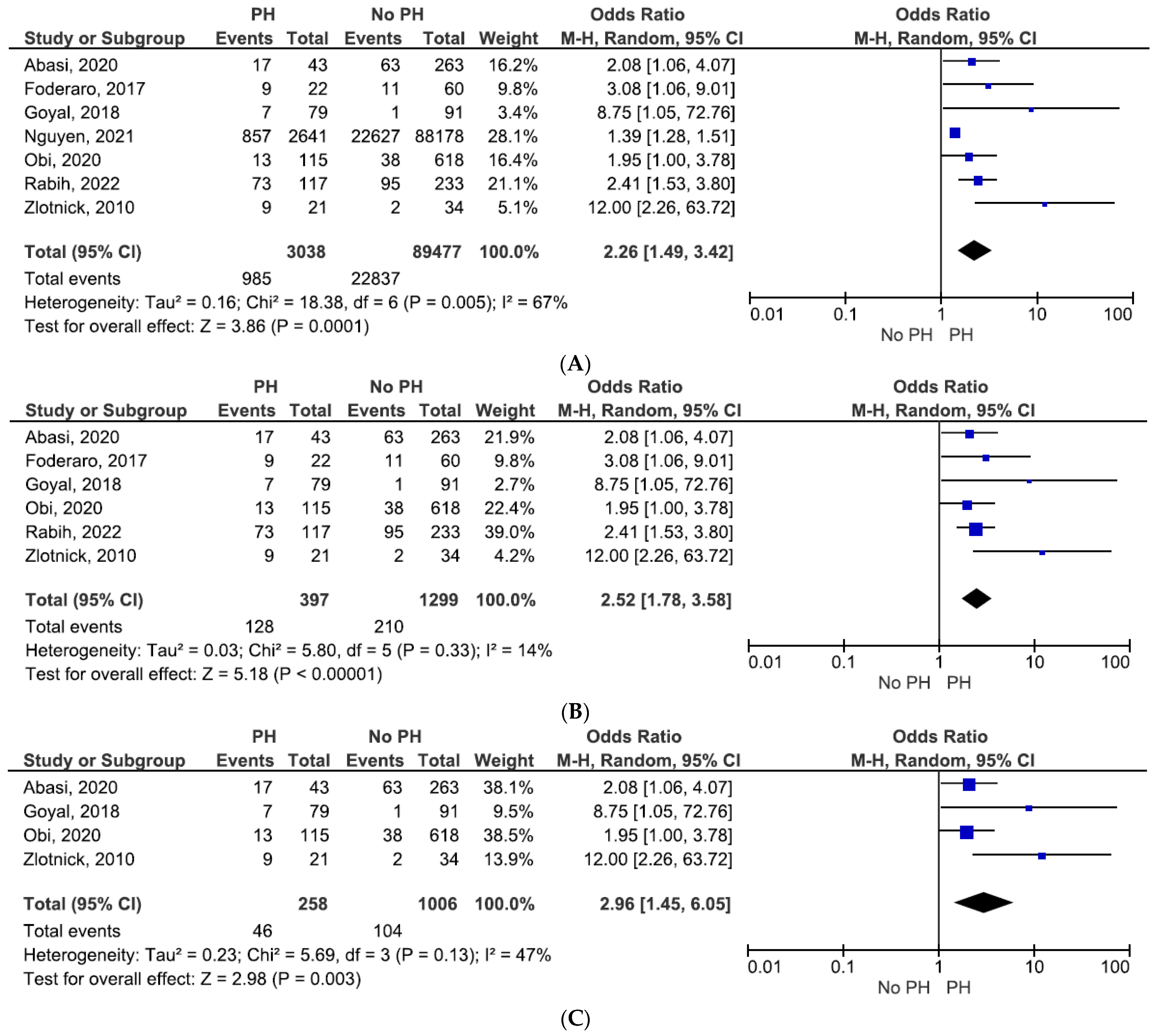
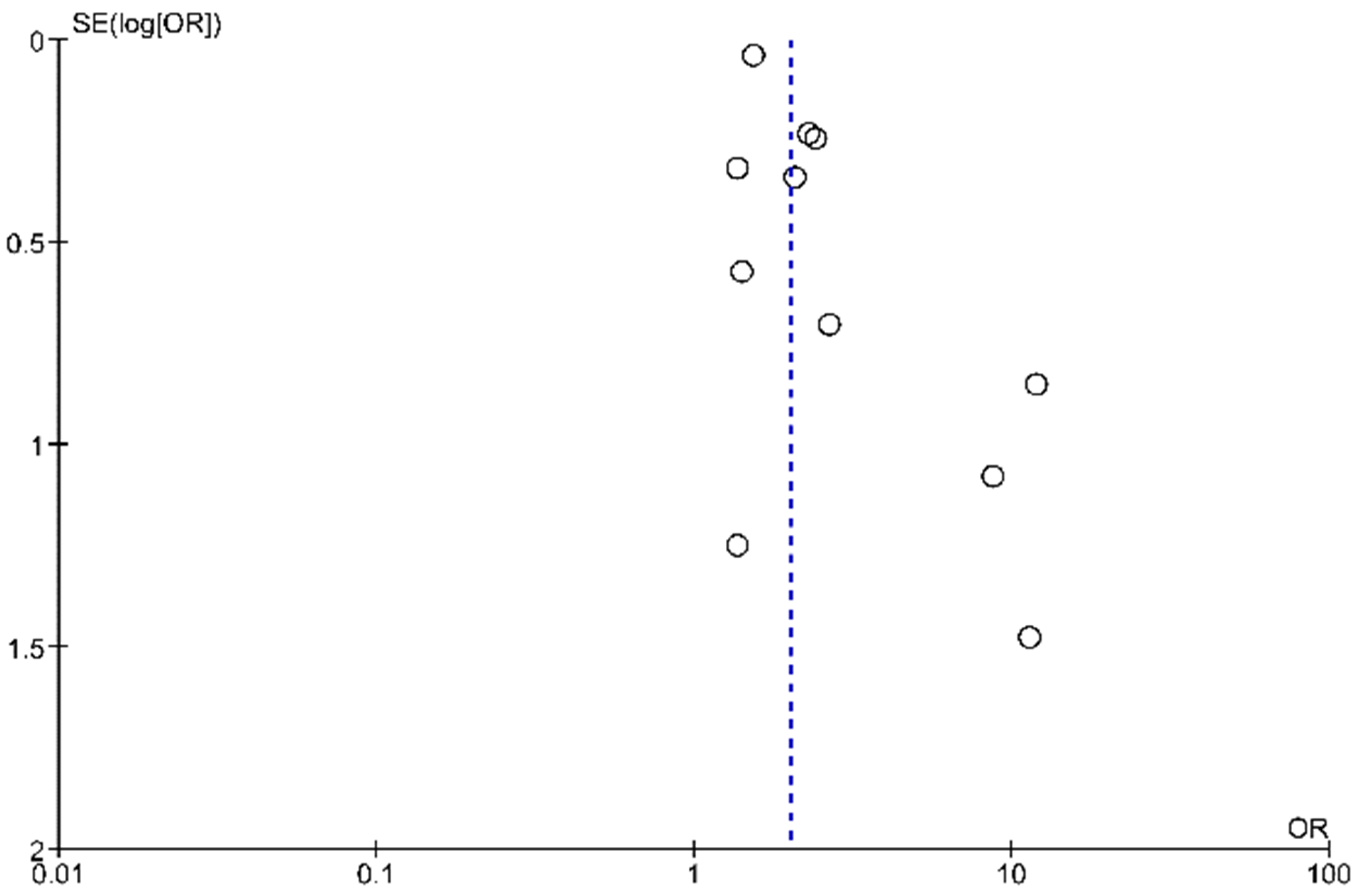
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, Regional, and National Burden of Chronic Kidney Disease, 1990-2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef] [Green Version]
- Baek, S.D.; Baek, C.H.; Kim, J.S.; Kim, S.M.; Kim, J.H.; Kim, S.B. Does Stage Iii Chronic Kidney Disease Always Progress to End-Stage Renal Disease? A Ten-Year Follow-up Study. Scand. J. Urol. Nephrol. 2012, 46, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Abecassis, M.; Bartlett, S.T.; Collins, A.J.; Davis, C.L.; Delmonico, F.L.; Friedewald, J.J.; Hays, R.; Howard, A.; Jones, E.; Leichtman, A.B.; et al. Kidney Transplantation as Primary Therapy for End-Stage Renal Disease: A National Kidney Foundation/Kidney Disease Outcomes Quality Initiative (Nkf/Kdoqi™) Conference. Clin. J. Am. Soc. Nephrol. 2008, 3, 471–480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fu, R.; Sekercioglu, N.; Berta, W.; Coyte, P.C. Cost-effectiveness of Deceased-donor Renal Transplant Versus Dialysis to Treat End-stage Renal Disease: A Systematic Review. Transplant. Direct 2020, 6, e522. [Google Scholar] [CrossRef] [Green Version]
- Tonelli, M.; Wiebe, N.; Knoll, G.; Bello, A.; Browne, S.; Jadhav, D.; Klarenbach, S.; Gill, J. Systematic Review: Kidney Transplantation Compared with Dialysis in Clinically Relevant Outcomes. Am. J. Transplant. 2011, 11, 2093–2109. [Google Scholar] [CrossRef]
- Jansz, T.T.; Bonenkamp, A.A.; Boereboom, F.T.J.; Van Reekum, F.E.; Verhaar, M.; Van Jaarsveld, B.C. Health-related quality of life compared between kidney transplantation and nocturnal hemodialysis. PLoS ONE 2018, 13, e0204405. [Google Scholar] [CrossRef] [Green Version]
- Kostro, J.Z.; Hellmann, A.; Kobiela, J.; Skóra, I.; Lichodziejewska-Niemierko, M.; Dębska-Ślizień, A.; Śledziński, Z. Quality of Life after Kidney Transplantation: A Prospective Study. Transplant. Proc. 2016, 48, 50–54. [Google Scholar] [CrossRef]
- Sarhan, A.L.; Jarareh, R.H.; Shraim, M. Quality of Life for Kidney Transplant Recipients and Hemodialysis Patients in Palestine: A Cross-Sectional Study. BMC Nephrol. 2021, 22, 210. [Google Scholar] [CrossRef]
- Kaballo, A.M.; Canney, M.; O’Kelly, P.; Williams, Y.; O’Seaghdha, C.M.; Conlon, P.J. A comparative analysis of survival of patients on dialysis and after kidney transplantation. Clin. Kidney J. 2017, 11, 389–393. [Google Scholar] [CrossRef]
- Bolignano, D.; Pisano, A.; Coppolino, G.; Tripepi, G.L.; D’Arrigo, G. Pulmonary Hypertension Predicts Adverse Outcomes in Renal Patients: A Systematic Review and Meta-Analysis. Ther. Apher. Dial. 2019, 23, 369–384. [Google Scholar] [CrossRef]
- Zhang, Y.; Ding, X.-H.; Rao, R.; Wang, Y.; Pang, F.; Tang, S.; Nie, L.; Bian, S.-Z. The Prevalence of Pulmonary Hypertension Among Maintenance Dialysis Patients With ESRD and Its Associated Factors: A Retrospective Study. Front. Med. 2020, 7, 884. [Google Scholar] [CrossRef] [PubMed]
- Navaneethan, S.D.; Walther, C.P.; Gregg, L.P.; Bansal, S.; Winkelmayer, W.C.; Nambi, V.; Niu, J. Mortality, Kidney Failure, and Hospitalization Among Medicare Beneficiaries With CKD and Pulmonary Hypertension. Am. J. Kidney Dis. 2021, 78, 700–708.e1. [Google Scholar] [CrossRef] [PubMed]
- Augustine, D.X.; Coates-Bradshaw, L.D.; Willis, J.; Harkness, A.; Ring, L.; Grapsa, J.; Coghlan, G.; Kaye, N.; Oxborough, D.; Robinson, S.; et al. Echocardiographic assessment of pulmonary hypertension: A guideline protocol from the British Society of Echocardiography. Echo Res. Pr. 2018, 5, G11–G24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moza, A.; Khan, A.R.; Parashar, R.; Khan, S.; Khouri, S.J.; Ortiz, J.; Malhotra, D.K.; Rees, A.M.; Moukarbel, G.V. Pulmonary hypertension in renal transplant candidates: A systematic review and meta-analysis of the available evidence and a proposed algorithm for pre-transplant management. Trends Transplant. 2017, 10. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, A.R.; Knorst, M.M. Pulmonary Hypertension Identified by Echocardiography with Color Doppler in Renal Transplant Candidates: Prevalence and Outcome. Am. J. Respir. Crit. Care Med. 2013, 187, A4690. [Google Scholar]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, 105906. [Google Scholar]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef] [Green Version]
- Issa, N.; Krowka, M.J.; Griffin, M.D.; Hickson, L.J.; Stegall, M.D.; Cosio, F.G. Pulmonary Hypertension Is Associated With Reduced Patient Survival After Kidney Transplantation. Transplantation 2008, 86, 1384–1388. [Google Scholar] [CrossRef]
- Nguyen, M.C.; Chiang, T.P.-Y.; Massie, A.B.; Bae, S.; Motter, J.D.; Brennan, D.C.; Desai, N.M.; Segev, D.L.; Garonzik-Wang, J.M. Kidney Transplantation Confers Survival Benefit for Candidates With Pulmonary Hypertension. Transplant. Direct 2021, 7, e738. [Google Scholar] [CrossRef]
- Obi, C.; Frost, A.E.; Graviss, E.A.; Nguyen, D.T.; Gaber, A.O.; Suki, W.N. The Association of Pretransplant Pulmonary Hypertension With Patient and Graft Survival After Kidney Transplantation: A Retrospective Cohort Study. Transplant. Proc. 2020, 52, 3023–3032. [Google Scholar] [CrossRef]
- Rabih, F.; Holden, R.L.; Vasanth, P.; Pastan, S.O.; Fisher, M.R.; Trammell, A.W. Effect of Pulmonary Hypertension on 5-Year Outcome of Kidney Transplantation. Pulm. Circ. 2022, 12, e12010. [Google Scholar] [CrossRef]
- Sadat, B.; Tirunagari, D.; Karthikeyan, V.; Patel, A.; Van Harn, M.; Saleem, M.M.; Ananthasubramaniam, K. Clinical impact of pre-kidney transplant pulmonary hypertension on post-transplant outcomes. Int. J. Cardiovasc. Imaging 2021, 37, 1979–1986. [Google Scholar] [CrossRef] [PubMed]
- Goyal, V.K.; Solanki, S.L.; Baj, B. Pulmonary Hypertension and Post-Operative Outcome in Renal Transplant: A Retrospective Analysis of 170 Patients. Indian J. Anaesth. 2018, 62, 131–135. [Google Scholar] [PubMed]
- Wang, S.C.; Garcia, R.; Torosoff, M.; Fein, S.A.; Ashar, A.; Chandolias, N.; Conti, D.; Lyubarova, R. Influence of mildly and moderately elevated pulmonary artery systolic pressure on post-renal transplantation survival and graft function. Echocardiography 2018, 36, 22–27. [Google Scholar] [CrossRef] [Green Version]
- Zlotnick, D.M.; Axelrod, D.A.; Chobanian, M.C.; Friedman, S.; Brown, J.; Catherwood, E.; Costa, S.P. Non-Invasive Detection of Pulmonary Hypertension Prior to Renal Transplantation Is a Predictor of Increased Risk for Early Graft Dysfunction. Nephrol. Dial Transplant. 2010, 25, 3090–3096. [Google Scholar] [CrossRef] [Green Version]
- Caughey, M.C.; Detwiler, R.K.; Sivak, J.A.; Rose-Jones, L.J.; Kshirsagar, A.V.; Hinderliter, A.L. Five-Year Outcomes of Pulmonary Hypertension with and without Elevated Left Atrial Pressure in Patients Evaluated for Kidney Transplantation. Transplantation 2020, 104, 2113–2119. [Google Scholar] [CrossRef]
- Abasi, K.; Lotfi, Z.; Ahmadi, M.; Majd, H.M.; Hasanzamani, B. Relationship between Pulmonary Hypertension before Kidney Transplantation and Early Graft Dysfunction. Iran J. Kidney Dis. 2020, 14, 399–404. [Google Scholar]
- Foderaro, A.; Baird, G.; Bazargan-Lari, A.; Morrissey, P.; Gohh, R.; Poppas, A.; Klinger, J.; Ventetuolo, C. Echocardiographic Pulmonary Hypertension Predicts Post-transplantation Renal Allograft Failure. Transplant. Proc. 2017, 49, 1256–1261. [Google Scholar] [CrossRef]
- Joseph, M.S.; Tinney, F.; Naik, A.; Parasuraman, R.; Samaniego-Picota, M.; Bhave, N.M. Right Ventricular Dysfunction and Adverse Outcomes after Renal Transplantation. Cardiorenal. Med. 2021, 11, 109–118. [Google Scholar] [CrossRef]
- Weitsman, T.; Weisz, G.; Farkash, R.; Klutstein, M.; Butnaru, A.; Rosenmann, D.; Hasin, T. Pulmonary Hypertension with Left Heart Disease: Prevalence, Temporal Shifts in Etiologies And outcome. Am. J. Med. 2017, 130, 1272–1279. [Google Scholar] [CrossRef]

| Author, Year | Design | Patients, No. | Age, Median/Mean ± SD | Setting | Parameters Evaluated | Methods | Outcomes | Follow-Up Period |
|---|---|---|---|---|---|---|---|---|
| Issa et al., 2008 [18] | Observational, single-center, retrospective | 215 | 55 ± 11 | Adult KT recipients between January 2004 and June 2007 who had a pre-transplant TTE | RVSP LVEF LVH | PH was estimated by RVSP, using TTE: <35 mmHg (normal range), 35–50 mmHg (mild to moderate PH) and >50 mmHg (severe PH) | Primary endpoint: patient death with a functioning kidney graft | 22.8 ± 11.8 months |
| Nguyen et al., 2021 [19] | Observational, multicenter (centers for Medicare and Medicaid Services), retrospective | 90,819 | 52.5 ± 13.5 (without PH) | First-time adult KT recipients (between 2000 and 2016) reported by the US Renal Data System | PH | PH defined by a 2-component algorithm, including right heart catheterization | (a) Delayed graft function (dialysis within 7 days after transplant) (b) Death-censored graft failure (c) Mortality | 4.3 years (with PH) |
| 55.7 ± 12.1 (with PH) | 6.7 years (without PH) | |||||||
| Obi et al., 2020 [20] | Observational, single-center, retrospective | 733 | 49.0 (without PH) | Adult KT patients between 2010 and 2015 who had a pre-transplant TTE | PASP LVEF Right atrial pressure | PH was estimated by using TTE: PASP < 35 mmHg (patients without PH) or PASP ≥ 35 mmHg (patients with PH) | (a) Mortality (b) Graft failure (c) Composite outcome of mortality or graft failure | 46.9 months (without PH) |
| 56.0 (with PH) | 36.9 months (with PH) | |||||||
| Rabih et al., 2022 [21] | Observational, single-center, retrospective | 350 | 51.0 (without PH) | Adult KT recipients at Emory Transplant Center between 2010 and 2011 who had a pre-transplant TTE | RVSP TRJV LV systolic or diastolic dysfunction | PH was defined as RVSP ≥ 35 mmHg and/or maximum TRJV ≥ 2.9 m/s, as measured by TTE | (a) All-cause mortality (b) Graft dysfunction (stable creatinine ≥ 1.4 mg/dL) (c) Graft failure (requiring dialysis or retransplant) | 5 years |
| 52 (with PH) | ||||||||
| Sadat et al., 2021 [22] | Observational, single-center, retrospective | 204 | – | Adult KT patients from 2010 to 2016 who had a pre-transplant TTE | PASP LVEF | PH was estimated by using TTE: PASP ≥ 40 mmHg (patients with PH) or PASP < 40 mmHg (patients without PH) | (a) Mortality (b) Graft function | 77.9 ± 36.12 months |
| Goyal et al., 2018 [23] | Observational, single-center, retrospective | 170 | 36.2 ± 11.2 (without PH) | Adult KT recipients who underwent a pre-transplant TTE examination | PASP LVEF | PH was estimated by using TTE: PASP ≥ 35 mmHg (patients with PH) or PASP < 35 mmHg (patients without PH) | (a) Primary outcome: delayed graft function (dialysis within 7 days after transplant) (b) Secondary outcomes: perioperative complications (hypotension, arrhythmias, need of post-operative mechanical ventilation, atelectasis, pulmonary edema) | – |
| 35.7 ± 9.8 (with PH) | ||||||||
| Wang et al., 2018 [24] | Observational, single-center, retrospective | 192 | 50.3 ± 12.9 (without PH) | Consecutive adult KT recipients between 2008 and 2015 who had a pre-transplant TTE | PASP LVEF Right ventricular systolic function | PH was estimated by using TTE: PASP ≥ 37 mmHg (patients with PH) or PASP < 37 mmHg (patients without PH) | (a) Length of hospital stay after transplant (b) Renal allograft function by 1 and 2 years (creatinine and eGFR measurement at 1 and 2 years) (c) Recipient mortality (d) Rate of graft loss | 4.0 ± 1.9 years |
| 52.7 ± 10.8 (with PH) | ||||||||
| Zlotnick et al., 2010 [25] | Observational, single-center, retrospective | 55 | 52.4 ± 9.9 (without PH) | Adult KT recipients over a period of 3 years who had a pre-transplant TTE | PASP | PH was estimated by using TTE: PASP ≥ 35 mmHg (patients with PH) or PASP < 35 mmHg (patients without PH) | Early graft dysfunction: delayed graft function (dialysis within 7 days after transplant) or slow graft function (creatinine ≥ 3 mg/dL on day 5 after transplant, without dialysis) | – |
| 54.6 ± 13.2 (with PH) | ||||||||
| Caughey et al., 2020 [26] | Observational, retrospective | 778 (179 KT recipients) | 56.0 ± 10 (without PH) | Adult patients with advanced CKD included in the University of North Carolina Cardiorenal Registry | TRJV LVEF LVH Left atrial pressure | PH was estimated by using TRJV: ≥2.9 m/s ± other signs (interventricular septal flattening, dilated inferior vena cava) | Mortality | 4.4 years |
| 57 ± 12 (with PH) | ||||||||
| Abasi et al., 2020 [27] | Observational, single-center, retrospective | 306 | 37.33 ± 10.92 (without PH) | Adult KT recipient over a period of 4 years who had a pre-transplant TTE | PASP | PH was estimated by using TTE: PASP ≥ 35 mmHg (patients with PH) or PASP < 35 mmHg (patients without PH) | Delayed graft function (dialysis within the first week after transplant or creatinine ≥ 3 mg/dL on day 5 after transplant) | – |
| 35.26 ± 10.3 (with PH) | ||||||||
| Foderaro et al., 2017 [28] | Observational, single-center, retrospective | 82 | 48.0 (without PH) | First-time adult KT recipients between 2003 and 2009 who had a pre-transplant TTE | RVSP LVEF | PH was estimated by using TTE: RVSP ≥ 40 mmHg (patients with PH) | (a) Death-censored allograft failure (b) Mortality | 3 years |
| 50.0 (with PH) | ||||||||
| Joseph et al., 2021 [29] | Observational, single-center, retrospective | 80 (RV function assessed in 73 patients) | 51.3 ± 14.2 | Adult KT recipients between 2008 and 2010 who had a TTE within 1 year prior to surgery | RV dilation and dysfunction LVEF | RVS dilation and function was established using TTE and standardized cutoff values | Primary outcome: composite of delayed graft function, graft failure, and all-cause mortality | 9.4 ± 0.8 years |
| Author, Year | Parameters | Outcomes | Results | |
|---|---|---|---|---|
| Issa et al., 2008 [18] | RVSP > 50 mmHg | Reduced recipient survival | HR 3.75 (95% CI, 1.17–11.97) | p = 0.016 |
| Nguyen et al., 2021 [19] | PH | Delayed graft function | OR 1.23 (95% CI, 1.10–1.36) | p < 0.001 |
| Mortality | HR 1.56 (95% CI, 1.44–1.69) | p < 0.001 | ||
| Death-censored graft failure | HR 1.23 (95% CI, 1.11–1.38) | p < 0.001 | ||
| Obi et al., 2020 [20] | PASP ≥ 35 mmHg | Univariate analysis: | ||
| Mortality (1 year) | HR 1.16 (95% CI, 0.33–4.04) | p = 0.82 | ||
| Mortality (3 years) | HR 1.71 (95% CI, 0.84–3.47) | p = 0.14 | ||
| Mortality (5 years) | HR 1.98 (95% CI, 1.11–3.56) | p = 0.02 | ||
| Composite of death or graft loss (5 years) | HR 1.69 (95% CI, 1.03–2.78) | p = 0.04 | ||
| Multivariate analysis: | ||||
| Mortality (5 years) | HR 1.26 (95% CI, 0.66–2.41) | p = 0.49 | ||
| Graft failure (5 years) | HR 0.77 (95% CI, 0.31–1.91) | p = 0.57 | ||
| Rabih et al., 2022 [21] | RVSP ≥ 35 mmHg and/or TRJV ≥ 2.9 m/s | Death, graft dysfunction, or failure | RR 1.432 (95% CI, 1.189–1.724) | p < 0.001 |
| LV systolic dysfunction | Death, graft dysfunction, or failure | RR 0.672 (95% CI, 0.347–1.302) | p = 0.239 | |
| LV diastolic dysfunction | Death, graft dysfunction, or failure | RR 1.073 (95% CI, 0.824–1.399) | p = 0.600 | |
| Sadat et al., 2021 [22] | PASP ≥ 40 mmHg | Mortality | 30.7% in patients without PH vs. 37.7% in patients with PH | p = 0.334 |
| Goyal et al., 2018 [23] | PASP ≥ 35 mmHg | Delayed graft function | OR 8.75 (95% CI, 1.05–72.75)—univariate analysis | p = 0.017 |
| On multivariate analysis PH was not associated with delayed graft function | ||||
| Wang et al., 2018 [24] | PASP ≥ 37 mmHg | Death or graft loss (>2 years) | 7.090% in patients without PH vs. 9.800% in patients with PH | p = 0.536 |
| Mean eGFR (2 years) | 60.28 mL/min ± 20.94 in patients without PH vs. 51.04 ± 15.07 in patients with PH | p = 0.006 | ||
| Zlotnick et al., 2010 [25] | PASP ≥ 35 mmHg | Early graft dysfunction | OR 15.0 (95% CI, 1.2–188.9)—adjusted for multiple variables | p = 0.03 |
| AUROC 0.74 (95% CI, 0.58–0.91) | ||||
| Caughey et al., 2020 [26] | TRJV: ≥2.9 m/s ± other signs | Mortality | 8% in patients without PH and normal left atrial pressure vs. 17% in patients with PH with normal left atrial pressure | |
| Abasi et al., 2020 [27] | PASP ≥ 35 mmHg | Delayed graft function | 39.5% in patients with PH vs. 24% in patients without PH | p < 0.05 |
| Foderaro et al., 2017 [28] | RVSP ≥ 40 mmHg | Death-censored allograft failure | Three-fold higher risk in PH group (95% CI, 1.20–7.32) | p = 0.02 |
| Mortality | 5% in patients with PH vs. 3% in patients without PH | p = 0.80 | ||
| Joseph et al., 2021 [29] | RV dilation and dysfunction | Composite of delayed graft function, graft failure and all-cause mortality | 100% in patients with RV dysfunction vs. 60% in patients without RV dysfunction | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brinza, C.; Covic, A.; Stefan, A.E.; Floria, M.; Popa, I.V.; Scripcariu, D.-V.; Burlacu, A. Pulmonary Arterial Hypertension and Adverse Outcomes after Kidney Transplantation: A Systematic Review and Meta-Analysis. J. Clin. Med. 2022, 11, 1944. https://doi.org/10.3390/jcm11071944
Brinza C, Covic A, Stefan AE, Floria M, Popa IV, Scripcariu D-V, Burlacu A. Pulmonary Arterial Hypertension and Adverse Outcomes after Kidney Transplantation: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2022; 11(7):1944. https://doi.org/10.3390/jcm11071944
Chicago/Turabian StyleBrinza, Crischentian, Adrian Covic, Anca Elena Stefan, Mariana Floria, Iolanda Valentina Popa, Dragos-Viorel Scripcariu, and Alexandru Burlacu. 2022. "Pulmonary Arterial Hypertension and Adverse Outcomes after Kidney Transplantation: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 11, no. 7: 1944. https://doi.org/10.3390/jcm11071944
APA StyleBrinza, C., Covic, A., Stefan, A. E., Floria, M., Popa, I. V., Scripcariu, D.-V., & Burlacu, A. (2022). Pulmonary Arterial Hypertension and Adverse Outcomes after Kidney Transplantation: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 11(7), 1944. https://doi.org/10.3390/jcm11071944








