Incidence, Severity and Clinical Factors Associated with Hypotension in Patients Admitted to an Intensive Care Unit: A Prospective Observational Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Measurements
2.4. Analyses
2.5. Signal Quality and Arterial Waveform Analyses
2.6. Data Acquisition
2.7. Statistical Analyses
3. Results
3.1. Patient Characteristics
3.2. Incidence and Severity of Hypotension
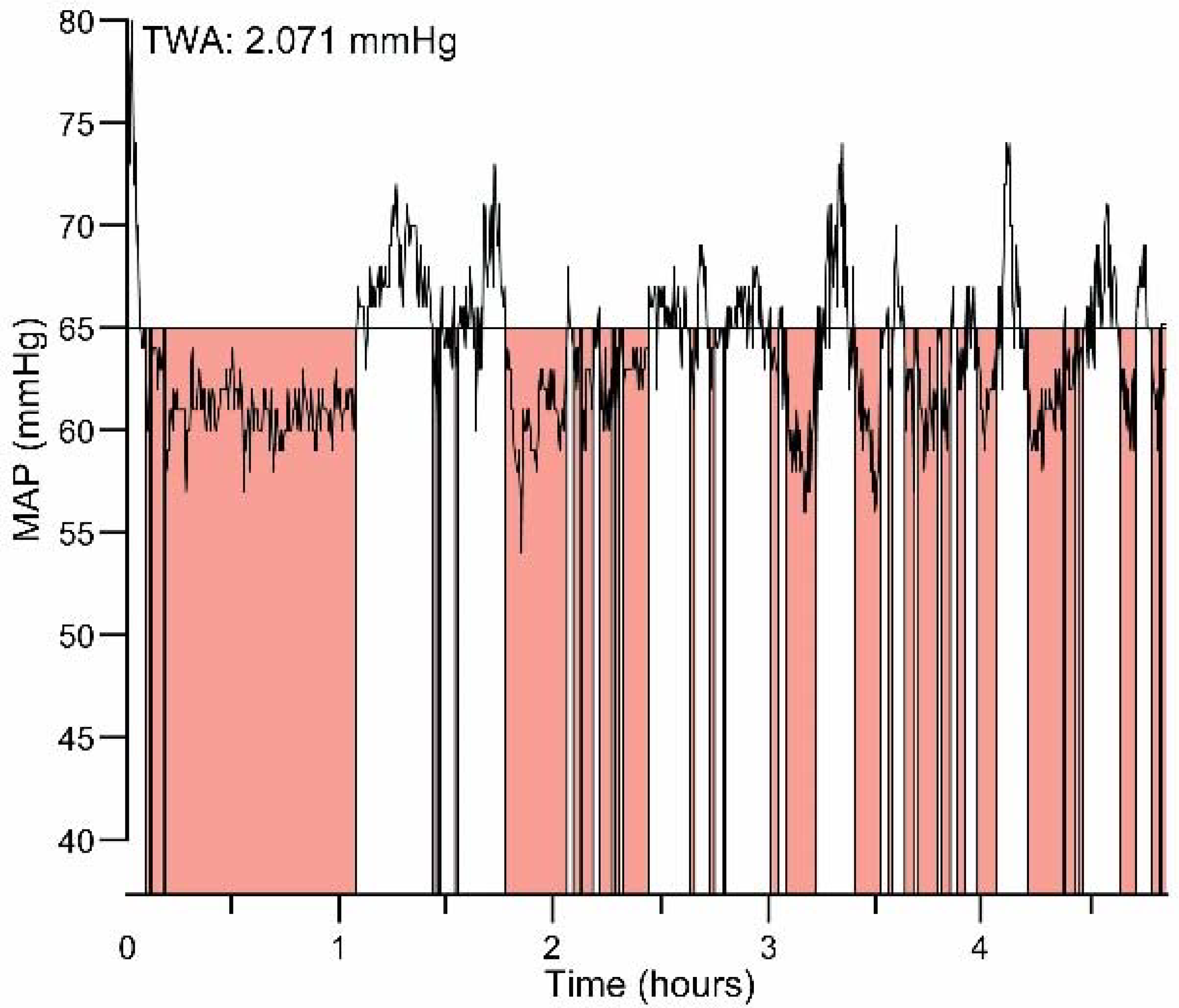
3.3. Severity Expressed in TWA
3.4. Clinical Factors Associated with Hypotension
4. Discussion
4.1. Incidence
4.2. Severity
4.3. Clinical Factors Associated with Hypotension
4.4. Limitations
4.5. Strengths
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Study Acronym
Editor’s Key Points
Abbreviations
| AKI | Acute kidney injury |
| AUT | Area under the threshold |
| BP | Blood pressure |
| BMI | Body mass index |
| CO | Cardiac output |
| CI | Cardiac index |
| HR | Heart rate |
| ICU | Intensive care unit |
| MAP | Mean arterial pressure |
| MI | Myocardial injury |
| PPV | Pulse pressure variation |
| SOFA | Sequential organ failure assessment |
| SV | Stroke volume |
| SVI | Stroke volume index |
| SVV | Stroke volume variation |
| TWA | Time weighted average |
| WMO | Dutch Medical Research Involving Human Subjects Act |
References
- Barrett, M.L.; Smith, M.W.; Elixhauser, A.; Honigman, L.S.; Pines, J.M. Utilization of Intensive Care Services, 2011. In Healthcare Cost and Utilization Project (HCUP) Statistical Briefs; Statistical Brief #185; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2006. [Google Scholar]
- Rhodes, A.; Evans, L.E.; Alhazzani, W.; Levy, M.M.; Antonelli, M.; Ferrer, R.; Kumar, A.; Sevransky, J.E.; Sprung, C.L.; Nunnally, M.E.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Crit. Care Med. 2017, 45, 486–552. [Google Scholar] [CrossRef] [PubMed]
- Maheshwari, K.; Nathanson, B.H.; Munson, S.H.; Khangulov, V.; Stevens, M.; Badani, H.; Khanna, A.K.; Sessler, D.I. The relationship between ICU hypotension and in-hospital mortality and morbidity in septic patients. Intensive Care Med. 2018, 44, 857–867. [Google Scholar] [CrossRef] [PubMed]
- Poukkanen, M.; Wilkman, E.; Vaara, S.T.; Pettilä, V.; Kaukonen, K.-M.; Korhonen, A.-M.; Uusaro, A.; Hovilehto, S.; Inkinen, O.; Laru-Sompa, R.; et al. Hemodynamic variables and progression of acute kidney injury in critically ill patients with severe sepsis: Data from the prospective observational FINNAKI study. Crit. Care 2013, 17, R295. [Google Scholar] [CrossRef] [PubMed]
- Varpula, M.; Tallgren, M.; Saukkonen, K.; Voipio-Pulkki, L.M.; Pettila, V. Hemodynamic variables related to outcome in septic shock. Intensive Care Med. 2005, 31, 1066–1071. [Google Scholar] [CrossRef]
- Leone, M.; Asfar, P.; Radermacher, P.; Vincent, J.L.; Martin, C. Optimizing mean arterial pressure in septic shock: A critical reappraisal of the literature. Crit. Care 2015, 19, 101. [Google Scholar] [CrossRef]
- Vincent, J.-L.; Nielsen, N.D.; Shapiro, N.I.; Gerbasi, M.E.; Grossman, A.; Doroff, R.; Zeng, F.; Young, P.J.; Russell, J.A. Mean arterial pressure and mortality in patients with distributive shock: A retrospective analysis of the MIMIC-III database. Ann. Intensive Care 2018, 8, 107. [Google Scholar] [CrossRef]
- Sessler, D.I.; Bloomstone, J.A.; Aronson, S.; Berry, C.; Gan, T.J.; Kellum, J.A.; Plumb, J.; Mythen, M.G.; Grocott, M.P.; Edwards, M.R.; et al. Perioperative Quality Initiative consensus statement on intraoperative blood pressure, risk and outcomes for elective surgery. Br. J. Anaesth. 2019, 122, 563–574. [Google Scholar] [CrossRef]
- Khanna, A.K.; Maheshwari, K.; Mao, G.; Liu, L.; Perez-Protto, S.E.; Chodavarapu, P.; Schacham, Y.N.; Sessler, D.I. Association Between Mean Arterial Pressure and Acute Kidney Injury and a Composite of Myocardial Injury and Mortality in Postoperative Critically Ill Patients: A Retrospective Cohort Analysis. Crit. Care Med. 2019, 47, 910–917. [Google Scholar] [CrossRef]
- Coopersmith, C.M.; De Backer, D.; Deutschman, C.S.; Ferrer, R.; Lat, I.; Machado, F.R.; Martin, G.S.; Martin-Loeches, I.; Nunnally, M.E.; Antonelli, M.; et al. Surviving sepsis campaign: Research priorities for sepsis and septic shock. Intensive Care Med. 2018, 44, 1400–1426. [Google Scholar] [CrossRef]
- Wesseling, K.H. Physiocal, Calibrating Finger Vascular Physiology for Finapress; TNO Biomedical Instrumentation: Amsterdam, The Netherlands, 1995; pp. 1–16. [Google Scholar]
- Maheshwari, K.; Khanna, S.; Bajracharya, G.R.; Makarova, N.; Riter, Q.; Raza, S.; Cywinski, J.B.; Argalious, M.; Kurz, A.; Sessler, D.I. A Randomized Trial of Continuous Noninvasive Blood Pressure Monitoring During Noncardiac Surgery. Anesth. Analg. 2018, 127, 424–431. [Google Scholar] [CrossRef]
- Zong, W.; Heldt, T.; Moody, G.; Mark, R. An open-source algorithm to detect onset of arterial blood pressure pulses. Comput. Cardiol. 2003, 30, 259–262. [Google Scholar]
- Sun, J.; Reisner, A.; Mark, R. A signal abnormality index for arterial blood pressure waveforms. Comput. Cardiol. 2006, 2006, 13–16. [Google Scholar]
- Bighamian, R.; Rubbo, C.; Thorsen, J.E.; Jin-Oh, H.; Reisner, A.T. Is there opportunity for automated decision-support and closed-loop control in ICU patients receiving vasopressor infusion? In Proceedings of the 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2014; pp. 1949–1952. [Google Scholar] [CrossRef]
- Yapps, B. Hypotension in ICU patients receiving vasopressor therapy. Sci. Rep. 2017, 7, 8551. [Google Scholar] [CrossRef] [PubMed]
- Lamontagne, F.; for the Canadian Critical Care Trials Group; Meade, M.O.; Hébert, P.C.; Asfar, P.; Lauzier, F.; Seely, A.J.; Day, A.G.; Mehta, S.; Muscedere, J.; et al. Higher versus lower blood pressure targets for vasopressor therapy in shock: A multicentre pilot randomized controlled trial. Intensive Care Med. 2016, 42, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Asfar, P.; Meziani, F.; Hamel, J.-F.; Grelon, F.; Megarbane, B.; Anguel, N.; Mira, J.-P.; Dequin, P.-F.; Gergaud, S.; Weiss, N.; et al. High versus low blood-pressure target in patients with septic shock. N. Engl. J. Med. 2014, 370, 1583–1593. [Google Scholar] [CrossRef]
- Russell, J.A.; Walley, K.R.; Singer, J.; Gordon, A.C.; Hébert, P.C.; Cooper, D.J.; Holmes, C.L.; Mehta, S.; Granton, J.T.; Storms, M.M.; et al. Vasopressin versus norepinephrine infusion in patients with septic shock. N. Engl. J. Med. 2008, 358, 877–887. [Google Scholar] [CrossRef]
- van Doorn, K.J. The duration of hypotension determines the evolution of bacteremia-induced acute kidney injury in the intensive care unit. PLoS ONE 2014, 9, e114312. [Google Scholar]
- Schmidt, H.; Hoyer, D.; Wilhelm, J.; Söffker, G.; Heinroth, K.; Hottenrott, K.; Said, S.M.; Buerke, M.; Müller-Werdan, U.; Werdan, K. The alteration of autonomic function in multiple organ dysfunction syndrome. Crit. Care Clin. 2008, 24, 149–163. [Google Scholar] [CrossRef]
- Abhishekh, H.A.; Nisarga, P.; Kisan, R.; Meghana, A.; Chandran, S.; Raju, T.; Sathyaprabha, T.N. Influence of age and gender on autonomic regulation of heart. J. Clin. Monit. Comput. 2013, 27, 259–264. [Google Scholar] [CrossRef]
- Schmidt, H.B.; Werdan, K.; Müller-Werdan, U. Autonomic dysfunction in the ICU patient. Curr. Opin. Crit. Care 2001, 7, 314–322. [Google Scholar] [CrossRef]
- Wijnberge, M.; Geerts, B.F.; Hol, L.; Lemmers, N.; Mulder, M.P.; Berge, P.; Schenk, J.; Terwindt, L.E.; Hollmann, M.W.; Vlaar, A.P.; et al. Effect of a Machine Learning-Derived Early Warning System for Intraoperative Hypotension vs Standard Care on Depth and Duration of Intraoperative Hypotension During Elective Noncardiac Surgery: The HYPE Randomized Clinical Trial. JAMA 2020, 323, 1052–1060. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, G. Artificial Intelligence in the Intensive Care Unit. Crit. Care 2020, 24, 101. [Google Scholar] [CrossRef] [PubMed]
| Baseline Parameters | All Patients n = 499 |
|---|---|
| Sex, male, n (%) | 327 (66) |
| Age, years, mean (sd) | 61 (14) |
| Number of patients older than 65 years, n (%) | 221 (44) |
| Weight (kg), mean (sd) | 82.97 (19.5) |
| Height (cm), mean (sd) | 174 (9.9) |
| BMI, mean (sd) | 27 (6) |
| SOFA score, mean (sd) | 10 (3) |
| Vasoactive medication during measurements, n (%) | 302 (61) |
| Mechanical ventilation, n (%) | 358 (72) |
| Measurement details | |
| Blood pressure monitoring time per patient (minutes), median [Q1–Q3] | 441 [411–962] |
| Signal quality percentage per patient, median [Q1–Q3] | 98.0 [94.6–99.0] |
| Number of daytime measurements, n (%) | 305 (61) |
| Number of night-time measurements, n (%) | 194 (39) |
| Reason of ICU admission | |
| Respiratory failure, n (%) | 57 (11) |
| Neurological disease, n (%) | 82 (16) |
| Sepsis, n (%) | 38 (8) |
| Cardiac shock/other cardiac, n (%) | 19 (4) |
| Postoperative after surgery, n (%) | 216 (43) |
| Assigned shock groups | |
| Cardiogenic shock, n (%) | 66 (13) |
| Distributive shock, n (%) | 94 (19) |
| Hypovolemic shock, n (%) | 12 (2) |
| Obstructive shock, n (%) | 2 (0.4) |
| Combination type of shock, n (%) | 32 (6) |
| Non-shock classification, n (%) | 293 (59) |
| Hypotension Classification | |||
|---|---|---|---|
| Baseline Parameters | Absent MAP ≥ 65 mmHg n = 123 (25%) | Present MAP < 65 mmHg n = 376 (75%) | p-Value |
| Sex, male, n (%) | 71 (58) | 256 (68) | 0.062 |
| Age, years, mean (sd) | 58 (14) | 62 (14) | 0.003 |
| BMI, mean (sd) | 28 (6) | 27 (6) | 0.049 |
| Day-time measurements, n (%) | 87 (71) | 218 (58) | 0.008 |
| Medical history | |||
| Myocardial infarction, n (%) | 11 (9) | 59 (16) | 0.251 |
| Hypertension, n (%) | 37 (30) | 109 (29) | 0.819 |
| Pulmonary disease, n (%) | 1 (1) | 12 (3) | 0.314 |
| Diabetes mellitus type II, n (%) | 15 (12) | 66 (18) | 0.204 |
| Cerebral vascular accident, n (%) | 6 (5) | 18 (5) | 1.000 |
| Gastrointestinal disease, n (%) | 19 (16) | 57 (15) | 0.427 |
| Renal insufficiencies, n (%) | 6 (5) | 29 (8) | 0.664 |
| Oncological disease, n (%) | 7 (6) | 16 (4) | 0.293 |
| Reason of ICU admission | |||
| OHCA, n (%) | 4 (3) | 27 (7) | 0.136 |
| Post cardiac surgery, n (%) | 32 (26) | 167 (44) | <0.001 |
| Intracranial bleeding (SAB), n (%) | 27 (22) | 24 (6) | <0.001 |
| Sepsis, n (%) | 12 (10) | 26 (7) | 0.326 |
| Assigned shock groups | |||
| Cardiogenic shock, n (%) | 4 (3) | 62 (16) | <0.001 |
| Distributive shock, n (%) | 20 (16) | 74 (20) | 0.506 |
| Hypovolemic shock, n (%) | 1 (1) | 11 (3) | 0.309 |
| Obstructive shock, n (%) | 1 (1) | 1 (0.3) | 0.430 |
| Non-shock classification, n (%) | 89 (72) | 204 (54) | <0.001 |
| Clinical data | |||
| Length of stay ICU (days), median [Q1–Q3] * | 3 [1–10] | 2 [1–7] | 0.407 |
| Length of stay in hospital (days), median [Q1–Q3] * | 10 [5–23] | 10 [6–20] | 0.877 |
| Lactate (mmol/L), median [Q1–Q3] | 1.5 [1.2–2.0] | 1.4 [1.1–2.0] | 0.985 |
| Diuresis (ml/kg/h), median [Q1–Q3] | 0.93 [0.61–1.48] | 0.87 [0.58–1.32] | 0.377 |
| Haemoglobin (mmol/L), median [Q1–Q3] | 6.8 [5.9–7.8] | 6.5 [5.7–7.4] | 0.118 |
| Saturation (%), median [Q1–Q3] | 94 [91–96] | 93 [71–96] | 0.002 |
| No treatment with Norepinephrine, n (%) | 55 (45) | 221 (59) | 0.012 |
| Propofol sedation, n (%) | 39 (32) | 142 (38) | 0.280 |
| Mechanical ventilation, n (%) | 83 (68) | 274 (73) | 0.356 |
| Days of mechanical ventilation during admission ICU, median [Q1–Q3] * | 12 [2–74] | 8 [2–47] | 0.299 |
| Apache score, median [Q1–Q3] * | 34 [32–60] | 49 [36–63] | 0.031 |
| Died during ICU admission, n (%) * | 12 (9.8) | 54 (14.4) | 0.220 |
| Died during hospital admission, n (%) * | 18 (14.6) | 70 (18.6) | 0.340 |
| Haemodynamic data | |||
| Systolic BP (mmHg), median [Q1–Q3] | 133 [114–163] | 121 [108–143] | 0.005 |
| Diastolic BP (mmHg), median [Q1–Q3] | 60 [52–69] | 55 [50–62] | <0.001 |
| Number of events per patients, median [Q1–Q3] | - | 6 [2–13] | |
| Total duration of events per patient (min), median [Q1–Q3] | - | 52 [5–170] | |
| Total percentage duration of measurement in hypotension (%), median [Q1–Q3] | - | 9.3 [0.7–29.1] | |
| TWA per patient (mmHg), 10 s median [Q1–Q3] | - | 0.3 [0.03–1.0] | |
| Mild TWA Group (≤0.0233 mmHg) n = 166 (33%) | Moderate TWA Group (0.0234–0.5719 mmHg) n = 166 (33%) | Severe TWA Group (≥0.5720 mmHg) n = 167 (34%) | p-Value | |
|---|---|---|---|---|
| Baseline parameters | ||||
| Sex, male, n (%) | 96 (58) | 117 (71) | 114 (68) | 0.035a |
| Age, years, mean (sd) | 57 (14) ¥® | 63 (12) ¥ | 62 (15) ® | <0.001 |
| BMI, mean (sd) | 28 (7) | 27 (6) | 27 (6) | 0.422 |
| SOFA score, median [Q1–Q3] | 9 [6–11] ¥® | 11 [9–12] ¥ | 10 [9–12] ® | <0.001 |
| Temperature °C, mean (sd) | 37.0 (0.8) ¥® | 36.7 (0.8) ¥ | 36.7 (0.8) ® | 0.006 |
| Medical history | ||||
| Myocardial infarction, n (%) | 17 (10) | 25 (15) | 28 (17) | 0.731 |
| Hypertension, n (%) | 52 (31) | 43 (26) | 51 (31) | 0.502 |
| Diabetes mellitus type 2, n (%) | 27 (16) | 23 (14) | 31 (19) | 0.507 |
| Cerebral vascular accident, n (%) | 8 (5) | 7 (4) | 9 (5) | 0.883 |
| Renal disease and insufficiencies, n (%) | 14 (8) | 7 (4) | 14 (8) | 0.097 |
| Reason of ICU admission | ||||
| OHCA, n (%) | 8 (5) | 9 (5) | 14 (8) | 0.353 |
| IHCA, n (%) | 2 (1) | - | 7 (4) | 0.013 |
| Post cardiac surgery, n (%) | 38 (23) | 77 (46) | 84 (50) | <0.001 |
| Pneumonia, n (%) | 2 (1) | 11 (7) | 5 (3) | 0.026 |
| Intracranial bleeding (SAB), n (%) | 42 (25) | 5 (3) | 4 (2) | <0.001 |
| Neurological other, n (%) | 19 (11) | 6 (4) | 3 (2) | <0.001 |
| Sepsis, n (%) | 9 (5) | 13 (8) | 16 (10) | 0.356 |
| Assigned shock groups | ||||
| Cardiogenic shock, n (%) | 7 (4) | 23 (14) | 36 (22) | <0.001 |
| Distributive shock, n (%) | 13 (8) | 39 (24) | 42 (25) | <0.001 |
| Hypovolemic shock, n (%) | 2 (1) | 5 (3) | 5 (3) | 0.466 |
| Obstructive shock, n (%) | - | 1 (1) | 1 (1) | 0.603 |
| Combination type of shock, n (%) | 2 (1) | 13 (8) | 17 (10) | 0.002 |
| Non-shock classification, n (%) | 141 (85) | 85 (51) | 67 (40) | <0.001 |
| Clinical data | ||||
| Lactate (mmol/L), mean (sd) | 1.6 (1.2) | 1.7 (1.5) | 2.07 (1.9) | 0.088 |
| Diuresis (ml/kg/h), mean (sd) | 1.2 (1.0) ® | 1.0(0.79) | 0.9 (0.8) ® | 0.002 |
| Saturation (%), median [Q1–Q3] | 93 [89–95] ¥® | 92 [70–94] ¥ | 92 [68–94] ® | <0.001 |
| Haemoglobin (mmol/L), mean (sd) | 6.8 (1.4) ¥® | 6.5 (1.2) ¥ | 6.3 (1.2) ® | <0.001 |
| Maximum need for norepinephrine dose (mcg/kg/min) during measurement), mean (sd) | 0.12 (0.11) ® | 0.16 (0.13) ‡ | 0.22 (0.20) ®‡ | <0.001 |
| Minimum need for norepinephrine dose (mcg/kg/min) during measurement, mean (sd) | 0.07 (0.13) | 0.05 (0.08) ‡ | 0.11 (0.15) ‡ | 0.002 |
| Mechanical ventilation, n (%) | 108 (65) | 124 (75) | 125 (75) | 0.077 |
| Haemodynamic data * | ||||
| Cardiac output (L/min), median [Q1–Q3] | 6.0 [4.9–7.5] ¥ | 5.6 [4.5–6.9] ¥ | 5.6 [4.7–6.7] | 0.027 |
| Systolic BP (mmHg), median [Q1–Q3] | 140 [124–158] ¥® | 117 [107–128] ¥ | 109 [99–120] ® | <0.001 |
| Diastolic BP (mmHg), median [Q1–Q3] | 67 [61–72] ¥® | 57 [54–62] ¥‡ | 51 [48–55] ‡® | <0.001 |
| MAP (mmHg), median [Q1–Q3] | 89 [82–101] ® | 75 [73–80] ‡ | 69 [66–71] ®‡ | <0.001 |
| Hypotension MAP < 65 mmHg | Severe TWA Group TWA > 0.5720 mmHg | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Covariate | Odds ratio | 95% C.I. | p-Value | Covariate | Odds ratio | 95% C.I. | p-Value | ||
| Lower | Upper | Lower | Upper | ||||||
| Age | 1.03 | 1.01 | 1.06 | 0.005 | Reason of ICU admission: post cardiac surgery | 2.33 | 1.30 | 4.19 | 0.005 |
| Sex (Male) | 2.59 | 1.38 | 4.85 | 0.003 | Reason of ICU admission: IHCA | 5.99 | 0.63 | 56.74 | 0.119 |
| BMI | 0.95 | 0.90 | 1.00 | 0.046 | Reason of ICU admission: Intracranial bleeding (SAB) | 0.14 | 0.01 | 1.45 | 0.100 |
| Cardiogenic shock classification | 3.70 | 1.17 | 11.66 | 0.026 | Non-shock classification | 0.59 | 0.31 | 1.12 | 0.104 |
| Non-shock classification | 0.55 | 0.27 | 1.14 | 0.106 | Haemoglobin (mmol/L) during measurement | 0.84 | 0.68 | 1.04 | 0.108 |
| Minimum need for norepinephrine dose (mcg/kg/min) during measurement | 0.12 | 0.01 | 1.23 | 0.074 | Maximum need for norepinephrine dose (mcg/kg/min) during measurement | 7.14 | 0.83 | 61.42 | 0.073 |
| Minimum need for norepinephrine dose (mcg/kg/min) during measurement | 31.80 | 0.95 | 1061.59 | 0.053 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terwindt, L.E.; Schuurmans, J.; van der Ster, B.J.P.; Wensing, C.A.G.C.L.; Mulder, M.P.; Wijnberge, M.; Cherpanath, T.G.V.; Lagrand, W.K.; Karlas, A.A.; Verlinde, M.H.; et al. Incidence, Severity and Clinical Factors Associated with Hypotension in Patients Admitted to an Intensive Care Unit: A Prospective Observational Study. J. Clin. Med. 2022, 11, 6832. https://doi.org/10.3390/jcm11226832
Terwindt LE, Schuurmans J, van der Ster BJP, Wensing CAGCL, Mulder MP, Wijnberge M, Cherpanath TGV, Lagrand WK, Karlas AA, Verlinde MH, et al. Incidence, Severity and Clinical Factors Associated with Hypotension in Patients Admitted to an Intensive Care Unit: A Prospective Observational Study. Journal of Clinical Medicine. 2022; 11(22):6832. https://doi.org/10.3390/jcm11226832
Chicago/Turabian StyleTerwindt, Lotte E., Jaap Schuurmans, Björn J. P. van der Ster, Carin A. G. C. L. Wensing, Marijn P. Mulder, Marije Wijnberge, Thomas G. V. Cherpanath, Wim K. Lagrand, Alain A. Karlas, Mark H. Verlinde, and et al. 2022. "Incidence, Severity and Clinical Factors Associated with Hypotension in Patients Admitted to an Intensive Care Unit: A Prospective Observational Study" Journal of Clinical Medicine 11, no. 22: 6832. https://doi.org/10.3390/jcm11226832
APA StyleTerwindt, L. E., Schuurmans, J., van der Ster, B. J. P., Wensing, C. A. G. C. L., Mulder, M. P., Wijnberge, M., Cherpanath, T. G. V., Lagrand, W. K., Karlas, A. A., Verlinde, M. H., Hollmann, M. W., Geerts, B. F., Veelo, D. P., & Vlaar, A. P. J. (2022). Incidence, Severity and Clinical Factors Associated with Hypotension in Patients Admitted to an Intensive Care Unit: A Prospective Observational Study. Journal of Clinical Medicine, 11(22), 6832. https://doi.org/10.3390/jcm11226832





