SARS-CoV-2 Infection in Hereditary Hemorrhagic Telangiectasia Patients Suggests Less Clinical Impact Than in the General Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Population under Study
2.2. ELISA (Enzyme-Linked Immune Adsorbent Assay) for Detection of Inflammatory Cytokines in Macrophages of HHT Patients
2.3. Analysis of ACE2 Expression by RT-qPCR
2.4. Statistical Analysis
3. Results
3.1. SARS-CoV-2 Infection Data among a Cohort of 138 HHT Patients
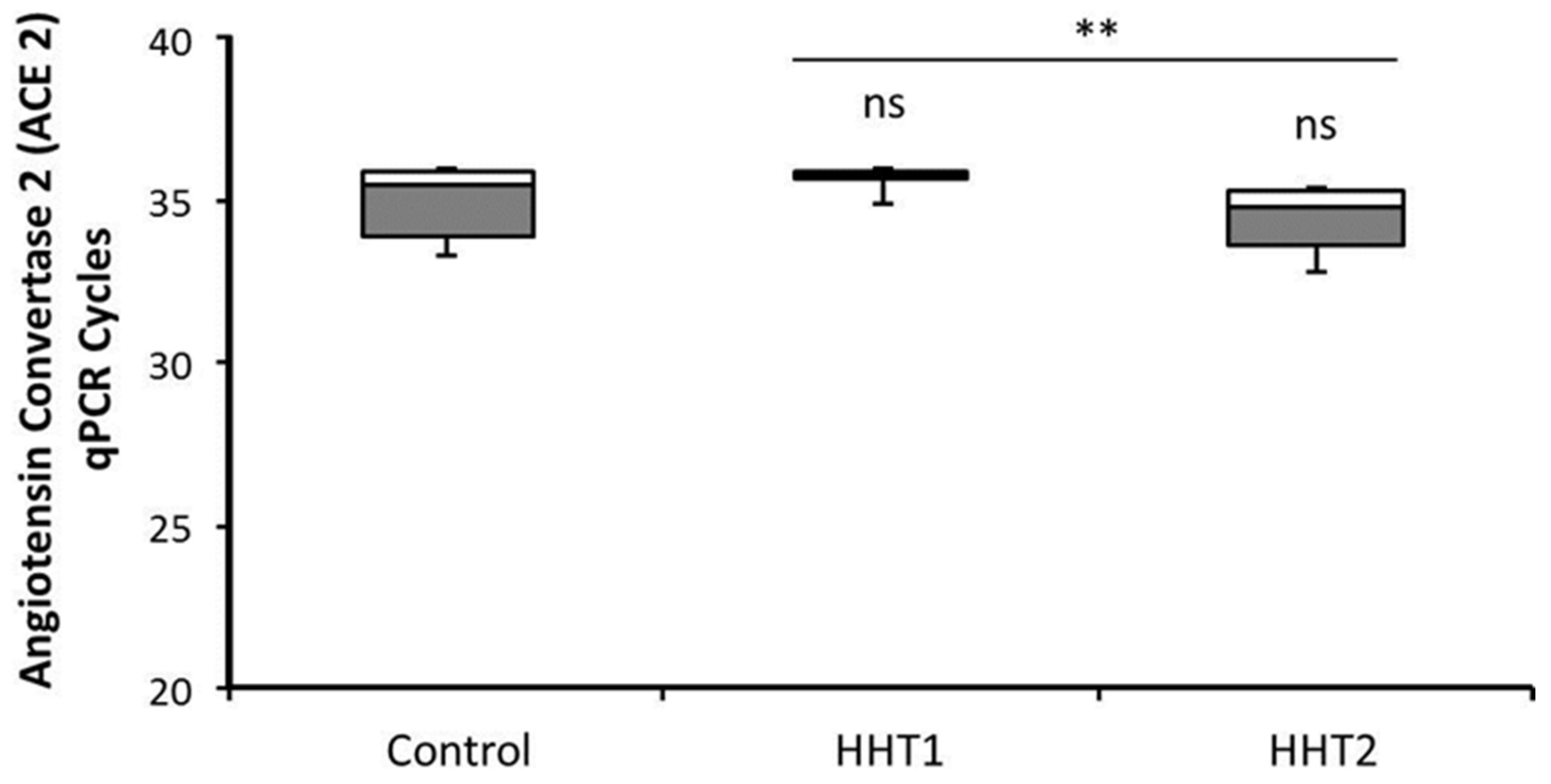
3.2. ACE2 Expression in HHT and Control Macrophages
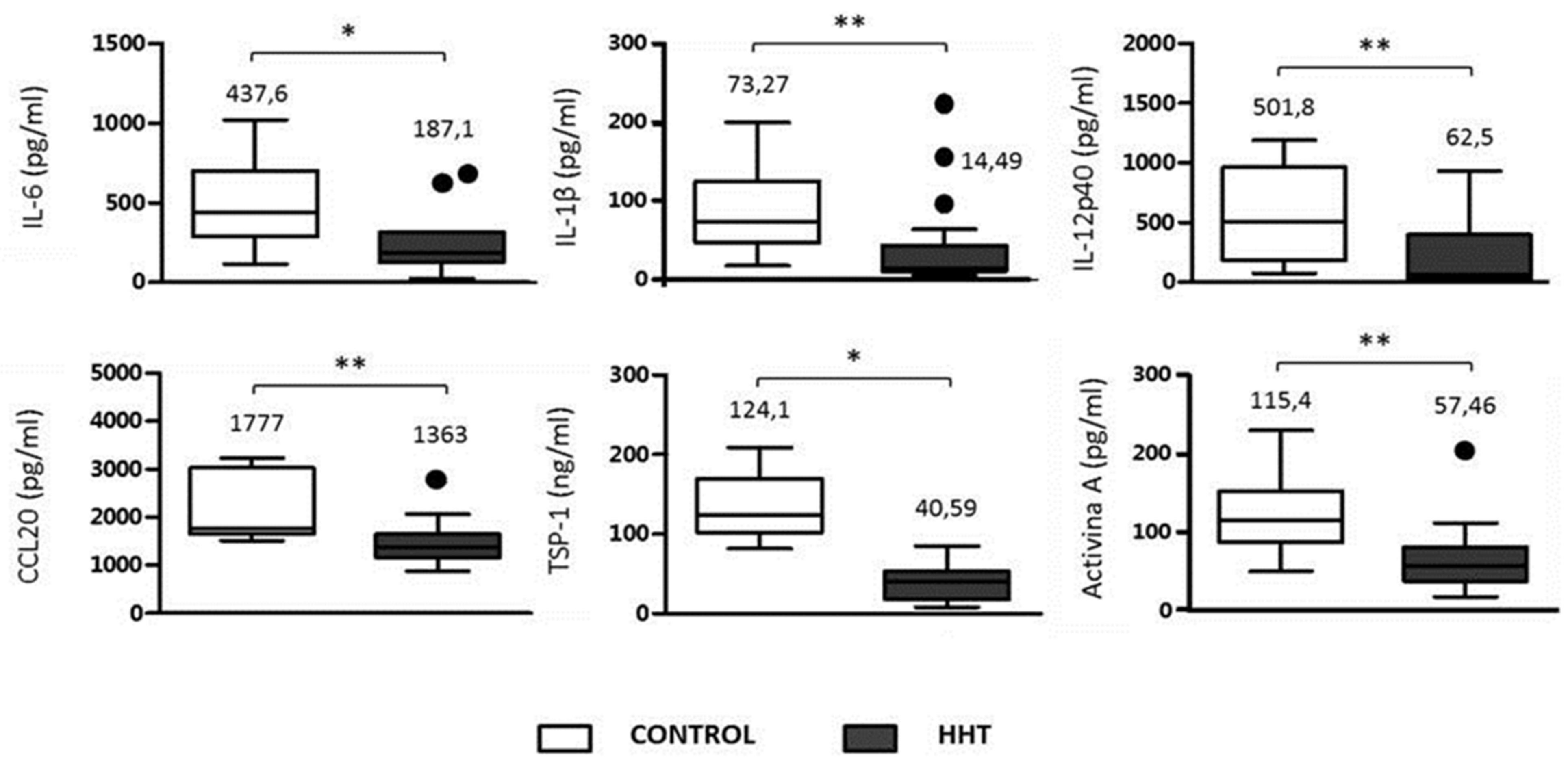
3.3. Inflammatory Cytokines in HHT and Control Macrophages
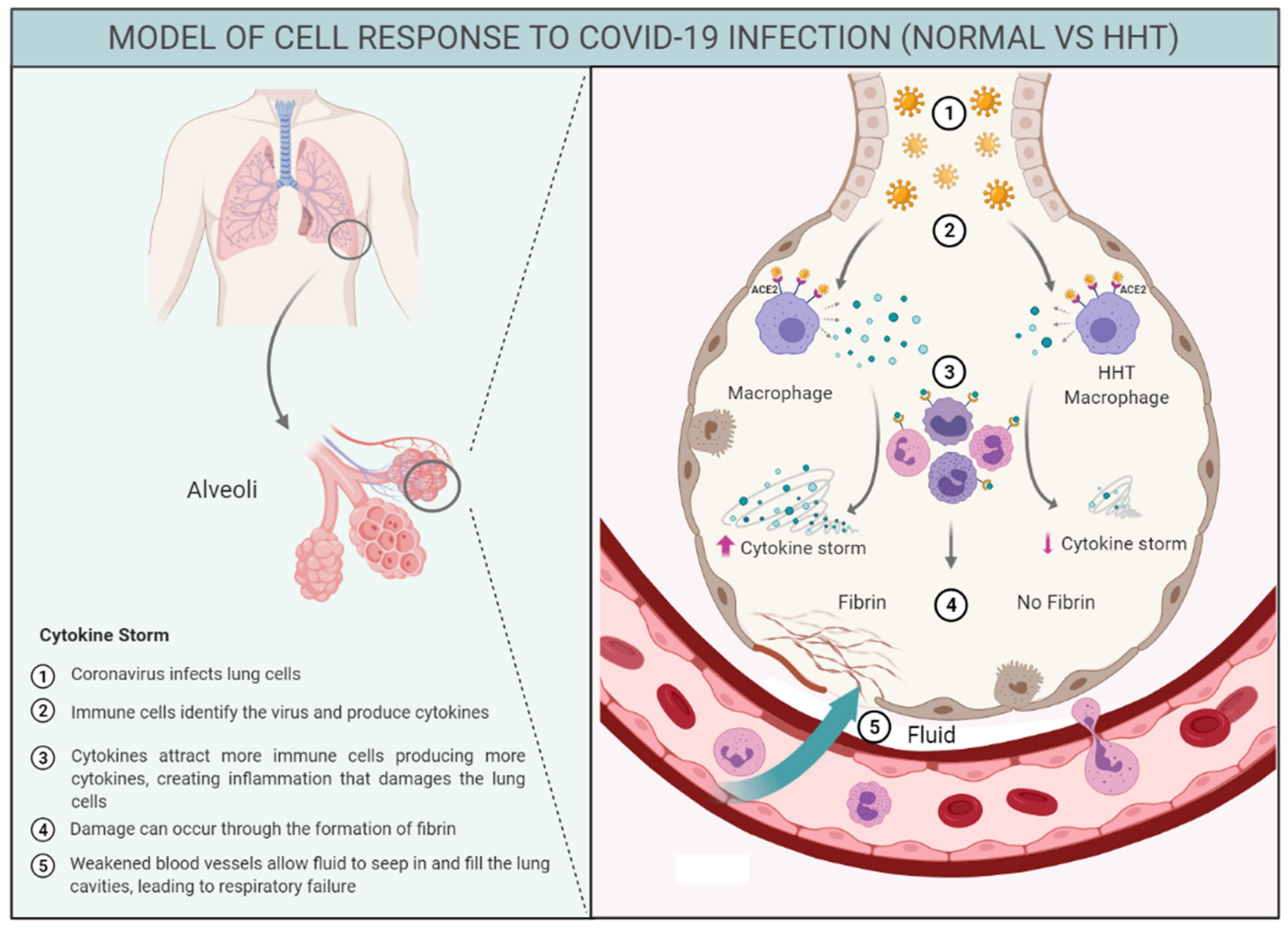
4. Discussion
Limitations and Interest of the Study
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ministerio de Sanidad, Consumo y Bienestar Social. Available online: https://www.mscbs.gob.es/profesionales/saludPublica/ccayes/alertasActual/nCov/situacionActual.htm (accessed on 23 March 2021).
- Hamming, I.; Timens, W.; Bulthuis, M.L.; Lely, A.T.; Navis, G.; van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004, 203, 6317. [Google Scholar] [CrossRef]
- Ayres, J.S. A metabolic handbook for the COVID-19 pandemic. Nat. Metab. 2020, 2, 572–585. [Google Scholar] [CrossRef]
- Teuwen, L.A.; Geldhof, V.; Pasut, A.; Carmeliet, P. COVID-19: The vasculature unleashed. Nat. Rev. Immunol. 2020, 20, 389–391. [Google Scholar] [CrossRef] [PubMed]
- Shovlin, C.L.; Guttmacher, A.E.; Buscarini, E.; Faughnan, M.E.; Hyland, R.H.; Westermann, C.J.J.; Kjeldsen, A.D.; Plauchu, H. Diagnostic criteria for Hereditary Hemorrhagic Telangiectasia (Rendu-Osler-Weber Syndrome). Am. J. Med. Genet. 2000, 91, 66–67. [Google Scholar] [CrossRef]
- Shovlin, C.L. Hereditary haemorrhagic telangiectasia: Pathophysiology, diagnosis and treatment. Blood Rev. 2010, 24, 203–219. [Google Scholar] [CrossRef] [PubMed]
- Shovlin, C.L. Pulmonary arteriovenous malformations. Am. J. Respir. Crit. Care Med. 2014, 190, 1217–1228. [Google Scholar] [CrossRef]
- Kjeldsen, A.D.; Vase, P.; Green, A. Hereditary haemorrhagic telangiectasia: A population-based study of prevalence and mortality in Danish patients. J. Intern. Med. 1999, 245, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Jessurun, G.A.J.; Kamphuis, D.J.; van der Zande, F.H.R.; Nossent, J.C. Cerebral arteriovenous malformations in the Netherlands Antilles. High prevalence of hereditary hemorrhagic telangiectasia-related single and multiple cerebral arteriovenous malformations. Clin. Neurol. Neurosurg. 1993, 95, 193–198. [Google Scholar] [CrossRef]
- McAllister, K.A.; Grogg, K.M.; Johnson, D.W.; Gallione, C.J.; Baldwin, M.A.; Jackson, C.E.; Helmbold, E.A.; Markel, D.S.; McKinnon, W.C.; Murrel, J.; et al. Endoglin, a TGF-β binding protein of endothelial cells, is the gene for hereditary haemorrhagic telangiectasia type 1. Nat. Genet. 1994, 8, 345–351. [Google Scholar] [CrossRef]
- Johnson, D.W.; Berg, J.N.; Baldwin, M.A.; Gallione, C.J.; Marondel, I.; Yoon, S.J.; Stenzel, T.T.; Speer, M.; Pericak-Vance, M.A.; Diamond, A.; et al. Mutations in the activin receptor-like kinase 1 gene in hereditary haemorrhagic telangiectasia type. Nat. Genet. 1996, 13, 189–195. [Google Scholar] [CrossRef]
- Morales-Angulo, C.; Del Valle-Zapico, A. Hereditary hemorrhagic telangiectasia. Otolaryngol. Head Neck Surg. 1998, 119, 293. [Google Scholar]
- Assar, O.S.; Friedman, C.M.; White, R.I. The Natural History of Epistaxis in Hereditary Hemorrhagic Telangiectasia. Laryngoscope 1991, 101, 977–980. [Google Scholar] [CrossRef]
- Geisthoff, U.W.; Schneider, G.; Fischinger, J.; Plinkert, P.K. Hereditary hemorrhagic telangiectasia (Osler’s disease). An interdisciplinary challenge. HNO 2002, 50, 114–128. [Google Scholar] [CrossRef]
- Guttmacher, A.E.; Marchuk, D.A.; White, R.I. Hereditary hemorrhagic telangiectasia. N. Engl. J. Med. 1995, 333, 918–924. [Google Scholar] [CrossRef]
- Esteban-Casado, S.; Martín de Rosales Cabrera, A.M.; Usarral de Pérez, A.; Martínez Simón, J.J.; Zhan Zhou, E.; Marcos Salazar, M.S.; Pérez Encinas, M.; Botella Cubells, L. Sclerotherapy and Topical Nasal Propranolol: An Effective and Safe Therapy for HHT-Epistaxis. Laryngoscope 2019, 129, 2216–2223. [Google Scholar] [CrossRef]
- Sánchez-Martínez, R.; Iriarte, A.; Mora-Luján, J.M.; Patier, J.L.; López-Wolf, D.; Ojeda, A.; Torralba, M.A.; Juyol, M.C.; Gil, R.; Añón, S.; et al. RiHHTa Investigators of the Rare Diseases Working Group from the Spanish Society of Internal Medicine. Current HHT genetic overview in Spain and its phenotypic correlation: Data from RiHHTa registry. Orphanet J. Rare Dis. 2020, 15, 138. [Google Scholar] [CrossRef]
- Fernandez-L, A.; Sanz-Rodriguez, F.; Zarrabeitia, R.; Perez-Molino, A.; Morales, C.; Restrepo, C.M.; Ramirez, J.R.; Coto, E.; Lenato, G.M.; Bernabeu, C.; et al. Mutation study of spanish patients with hereditary hemorrhagic telangiectasia and expression analysis of endoglin and ALK1. Hum. Mutat. 2006, 27, 295. [Google Scholar] [CrossRef]
- Health National Institute Carlos III. Report No. 70, COVID-19 Situation in Spain. Available online: https://www.isciii.es/QueHacemos/Servicios/VigilanciaSaludPublicaRENAVE/EnfermedadesTransmisibles/Documents/INFORMES/Informes%20COVID-19/INFORMES%20COVID-19%202021/Informe%20COVID-19.%20N%C2%BA%2070_%2017%20de%20marzo%20de%202021.pdf (accessed on 17 March 2021).
- Sierra-Filardi, E.; Puig-Kröger, A.; Blanco, F.J.; Nieto, C.; Bragado, R.; Palomero, M.I.; Bernabéu, C.; Vega, M.A.; Corbí, A.L. Activin A skews macrophage polarization by promoting a proinflammatory phenotype and inhibiting the acquisition of anti-inflammatory macrophage markers. Blood 2011, 117, 5092–5101. [Google Scholar] [CrossRef]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, in press. [Google Scholar] [CrossRef]
- Riera-Mestre, A.; Iriarte, A.; Moreno, M.; del Castillo, R.; López-Wolf, D. Angiogenesis, hereditary hemorrhagic telangiectasia and COVID-19. Angiogenesis 2021, 24, 13–15. [Google Scholar] [CrossRef]
- Zhou, P.; Yang, X.L.; Wang, X.G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.R.; Zhu, Y.; Li, B.; Huang, C.L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef]
- Alijotas-Reig, J.; Esteve-Valverde, E.; Belizna, C.; Selva-O’Callaghan, A.; Pardos-Gea, J.; Quintana, A.; Mekinian, A.; Anunciacion-Llunell, A.; Miró-Mur, F. Immunomodulatory therapy for the management of severe COVID-19. Beyond the anti-viral therapy: A comprehensive review. Autoimmun. Rev. 2020, 19, 102569. [Google Scholar] [CrossRef]
- WHO Solidarity Trial Consortium; Pan, H.; Peto, R.; Henao-Restrepo, A.-M.; Preziosi, M.-P.; Sathiyamoorthy, V.; Abdool Karim, Q.; Alejandria, M.M.; Hernández García, C.; Kieny, M.-P.; et al. Repurposed Antiviral Drugs for COVID-19—Interim WHO Solidarity Trial Results. N. Engl. J. Med. 2021, 384, 497–511. [Google Scholar]
- Moffat, J.G.; Vincent, F.; Lee, J.A.; Eder, J.; Prunotto, M. Opportunities and challenges in phenotypic drug discovery: An industry perspective. Nat. Rev. Drug Discov. 2017, 16, 531–543. [Google Scholar] [CrossRef]
- RECOVERY Collaborative Group; Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; et al. Dexamethasone in Hospitalized Patients with COVID-19. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Ojeda-Fernández, L.; Recio-Poveda, L.; Aristorena, M.; Lastres, P.; Blanco, F.J.; Sanz-Rodríguez, F.; Gallardo-Vara, E.; de las Casas-Engel, M.; Corbí, Á.; Arthur, H.M.; et al. Mice Lacking Endoglin in Macrophages Show an Impaired Immune Response. PLoS Genet. 2016, 12, e1005935. [Google Scholar] [CrossRef]
- Pérez-Gómez, E.; Del Castillo, G.; Juan Francisco, S.; López-Novoa, J.M.; Bernabéu, C.; Quintanilla, M. The role of the TGF-β coreceptor endoglin in cancer. Sci. World J. 2010, 10, 2367–2384. [Google Scholar] [CrossRef]
- Hosman, A.E.; Devlin, H.L.; Silva, B.M.; Shovlin, C.L. Specific cancer rates may differ in patients with hereditary haemorrhagic telangiectasia compared to controls. Orphanet J. Rare Dis. 2013, 8, 195. [Google Scholar] [CrossRef]
- Toi, H.; Tsujie, M.; Haruta, Y.; Fujita, K.; Duzen, J.; Seon, B.K. Facilitation of endoglin-targeting cancer therapy by development/utilization of a novel genetically engineered mouse model expressing humanized endoglin (CD105). Int. J. Cancer 2015, 136, 452–461. [Google Scholar] [CrossRef]
| Patient | Gender | Gene | Type | AVMS | HHT-ESS | Age | Symptoms | Hospital |
|---|---|---|---|---|---|---|---|---|
| #1 | ♀ | ENG | HHT1 | HAVM | 0.91 | 37 | asymptomatic | - |
| #2 | ♂ | ENG | HHT1 | PAVM, HAVM | 1.41 α | 74 | asymptomatic | - |
| #3 | ♀ | ALK1 | HHT2 | HAVM | 1.01 | 57 | headache, diarrhea | - |
| #4 | ♂ | ALK1 | HHT2 | HAVM | 1.91 | 64 | headache, diarrhea, myalgias | - |
| #5 | ♂ | ENG | HHT1 | PAVM, HAVM | 0.51 | 70 | cough, shortness of breath | - |
| #6 | ♂ | ALK1 | HHT2 | HAVM | 7.46 ε | 63 | suspected diarrhea, serologic detection months later | - |
| #7 | ♀ | ALK1 | HHT2 | HAVM | 3.51 | 63 | myalgias, headache, nosebleed, diarrhea | - |
| #8 | ♂ | ALK1 | HHT2 | - | 0.0 | 29 | asymptomatic | - |
| #9 | ♀ | ENG | HHT1 | PAVM, CAVM, HAVM | 1.41 | 32 | asymptomatic | - |
| #10 | ♀ | ALK1 | HHT2 | PAVM, HAVM | 2.43 | 47 | pneumonia, cough, dyspnea | YES |
| #11 | ♀ | ALK1 | HHT2 | HAVM | 1.41 | 56 | asymptomatic | - |
| #12 | ♀ | ENG | HHT1 | PAVM, HAVM | 1.41 | 62 | asymptomatic | - |
| #13 | ♀ | ALK1 | HHT2 | HAVM | 3.33 | 70 | asymptomatic | - |
| #14 | ♀ | ALK1 | HHT2 | HAVM | 5.18 γ | 87 | pneumonia, anemia | YES |
| #15 | ♂ | ALK1 | HHT2 | HAVM | 0.0 | 55 | headache, diarrhea | - |
| #16 | ♀ | ALK1 | HHT2 | - | 0.0 | 25 | anosmia, headache | - |
| #17 | ♀ | ALK1 | HHT2 | PAVM, HAVM | 3.33 | 50 | anosmia, headache, diarrhea | - |
| #18 | ♀ | ENG | HHT1 | PAVM, HAVM | 3.33 | 52 | pneumonia, no dyspnea | YES |
| #19 | ♀ | ENG | HHT1 | PAVM, SpAVM | 0.0 | 16 | asymptomatic | - |
| #20 | ♀ | ENG | HHT1 | HAVM | 2.43 Δ | 49 | anosmia, ageusia, moderate fever and slight muscular pain, diarrhea | - |
| #21 | ♂ | ALK1 | HHT2 | - | 0.0 | 41 | anosmia, ageusia, moderate fever and slight muscular pain | - |
| #22 | ♂ | ALK1 | HHT2 | - | 0.0 | 18 | infected twice, rhinitis | - |
| #23 | ♀ | ALK1 | HHT2 | HAVM | 0.51 | 50 | tonsil and ear infections, cough, fever, pain in the chest, and low oxygen saturation (91%) | - |
| #24 | ♀ | ALK1 | HHT2 | - | 0.0 | 16 | fever, vomit, tiredness and breathless | - |
| #25 | ♂ | ALK1 | HHT2 | HAVM | 0.0 | 63 | anosmia, headache | - |
| HHT1 n (%) | HHT2 n (%) | |
|---|---|---|
| Sex | ||
| Male Female | 2 (25) 6 (75) | 7 (41.1) 10 (58.8) |
| Age | 49 (±19.9) * | 49.3 (±19.6) * |
| Symptoms | ||
| No | 5 (62.5) | 3 (18.8) |
| Yes | 3 (37.5) | 14 (81.2) |
| Hospitalization | ||
| No | 7 (87.5) | 15 (87.5) |
| Yes | 1 (12.5) | 2 (12.5) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcos, S.; Albiñana, V.; Recio-Poveda, L.; Tarazona, B.; Verde-González, M.P.; Ojeda-Fernández, L.; Botella, L.-M. SARS-CoV-2 Infection in Hereditary Hemorrhagic Telangiectasia Patients Suggests Less Clinical Impact Than in the General Population. J. Clin. Med. 2021, 10, 1884. https://doi.org/10.3390/jcm10091884
Marcos S, Albiñana V, Recio-Poveda L, Tarazona B, Verde-González MP, Ojeda-Fernández L, Botella L-M. SARS-CoV-2 Infection in Hereditary Hemorrhagic Telangiectasia Patients Suggests Less Clinical Impact Than in the General Population. Journal of Clinical Medicine. 2021; 10(9):1884. https://doi.org/10.3390/jcm10091884
Chicago/Turabian StyleMarcos, Sol, Virginia Albiñana, Lucia Recio-Poveda, Belisa Tarazona, María Patrocinio Verde-González, Luisa Ojeda-Fernández, and Luisa-María Botella. 2021. "SARS-CoV-2 Infection in Hereditary Hemorrhagic Telangiectasia Patients Suggests Less Clinical Impact Than in the General Population" Journal of Clinical Medicine 10, no. 9: 1884. https://doi.org/10.3390/jcm10091884
APA StyleMarcos, S., Albiñana, V., Recio-Poveda, L., Tarazona, B., Verde-González, M. P., Ojeda-Fernández, L., & Botella, L.-M. (2021). SARS-CoV-2 Infection in Hereditary Hemorrhagic Telangiectasia Patients Suggests Less Clinical Impact Than in the General Population. Journal of Clinical Medicine, 10(9), 1884. https://doi.org/10.3390/jcm10091884







