The Effect of Transoesophageal Echocardiography on Treatment Change in a High-Volume Stroke Unit
Abstract
1. Introduction
2. Methods
2.1. Study Design and Population
2.2. Cardiovascular Diagnostic Work-up
2.3. Transesophageal Echocardiography (TEE)
2.4. Statistical Analysis
3. Results
3.1. Patients Characteristics
3.2. Transesophageal Echocardiography (TEE) with Abnormal Findings and TEE with Subsequent Change in Medical Management
3.3. Predictors of Abnormal Findings in TEE and Subsequent Change in Medical Management following TEE
4. Discussion
4.1. Frequent Abnormal Findings and Few Change in Medical Management
4.2. Transesophageal Echocardiography (TEE) in Hemorrhagic Stroke: Towards an Extended Indication and New Horizon for TEE?
4.3. Futility, Benefit and Applicability of Transesophageal Echocardiography in the Real-World Practice
5. Study Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations and Acronyms
| AHS | Acute hemorrhagic stroke |
| AIS | Acute ischemic stroke |
| AF | Atrial Fibrillation |
| ASA | Atrial septal aneurysm |
| AHS | Acute hemorrhagic stroke |
| AIS | Acute ischemic stroke |
| CAD | Coronary artery disease |
| LA | Left atrial |
| LAA | left atrial appendage |
| PAD | Peripheral artery disease |
| PFO | patent foramen ovale |
| NIHSS Score | National Institutes of Health Stroke Scale score |
| TIA | Transient ischemic Attack |
| TTE | Transthoracic echocardiography |
| TEE | Transesophageal echocardiography |
| SEC | Spontaneous echo contrast |
| SU | Stroke unit |
References
- Adams, H.P., Jr.; Bendixen, B.H.; Kappelle, L.J.; Biller, J.; Love, B.B.; Gordon, D.L.; Marsh, E.E., III. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 1993, 24, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brown, M.; Demaerschalk, B.M.; Hoh, B.; et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 2019, 50, e344–e418. [Google Scholar] [CrossRef] [PubMed]
- Ferro, J.M. Cardioembolic stroke: An update. Lancet Neurol. 2003, 2, 177–188. [Google Scholar] [CrossRef]
- Pepi, M.; Evangelista, A.; Nihoyannopoulos, P.; Flachskampf, F.A.; Athanassopoulos, G.; Colonna, P.; Habib, G.; Ringelstein, E.B.; Sicari, R.; Zamorano, J.L. Recommendations for echocardiography use in the diagnosis and management of cardiac sources of embolism: European Association of Echocardiography (EAE) (a registered branch of the ESC). Eur. J. Echocardiogr. 2010, 11, 461–476. [Google Scholar] [CrossRef] [PubMed]
- Saric, M.; Armour, A.C.; Arnaout, M.S.; Chaudhry, F.A.; Grimm, R.A.; Kronzon, I.; Landeck, B.F.; Maganti, K.; Michelena, H.I.; Tolstrup, K. Guidelines for the Use of Echocardiography in the Evaluation of a Cardiac Source of Embolism. J. Am. Soc. Echocardiogr. 2016, 29, 1–42. [Google Scholar] [CrossRef] [PubMed]
- Heidrich, J.; Heuschmann, P.U.; Kolominsky-Rabas, P.; Rudd, A.G.; Wolfe, C.D.; European BIOMED II Study of Stroke Care Group. Variations in the use of diagnostic procedures after acute stroke in Europe: Results from the BIOMED II study of stroke care. Eur. J. Neurol. 2007, 14, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Rosol, Z.; Kopecky, K.; Minehart, B.; Tecson, K.; Vasudevan, A.; Schussler, J. The overuse of transesophageal echocardiography in patients with confirmed ischemic stroke. J. Am. Coll. Cardiol. 2019, 73, 1593. [Google Scholar] [CrossRef]
- Charbonnel, C.; Jégo, C.; Jourda, F.; Vinsonneau, U.; Garçon, P.; Turlotte, G.; Rivière, J.F.; Maurin, M.; Lubret, R.; Meimoun, P.; et al. ADAM-C score: New risk score for predicting diagnostic yield of transesophageal echocardiography after cerebral ischemia. Echocardiogr. 2018, 35, 1171–1182. [Google Scholar] [CrossRef] [PubMed]
- Hahn, R.T.; Abraham, T.; Adams, M.S.; Bruce, C.J.; Glas, K.E.; Lang, R.M.; Reeves, S.T.; Shanewise, J.S.; Siu, S.C.; Stewart, W.; et al. Guidelines for Performing a Comprehensive Transesophageal Echocardiographic Examination: Recommendations from the American Society of Echocardiography and the Society of Cardiovascular Anesthesiologists. J. Am. Soc. Echocardiogr. 2013, 26, 921–964. [Google Scholar] [CrossRef] [PubMed]
- Van Woerkom, R.C.; Lester, S.J.; Demaerschalk, B.M.; LaBanc, A.J.; Davis, M.L.; Coakley, M.F.; Cha, S.S.; Wilansky, S. Comparison of the Utility of Transesophageal Echocardiography in Patients With Acute Ischemic Stroke and Transient Ischemic Attack Stratified by Age Group (<60, 60 to 80,≥80 Years). Am. J. Cardiol. 2018, 122, 2142–2146. [Google Scholar] [PubMed]
- Khariton, Y.; House, J.A.; Comer, L.; Coggins, T.R.; Magalski, A.; Skolnick, D.G.; Good, T.H.; Main, M.L. Impact of Transesophageal Echocardiography on Management in Patients With Suspected Cardioembolic Stroke. Am. J. Cardiol. 2014, 114, 1912–1916. [Google Scholar] [CrossRef] [PubMed]
- Boersma, L.V.; Ince, H.; Kische, S.; Pokushalov, E.; Schmitz, T.; Schmidt, B.; Gori, T.; Meincke, F.; Protopopov, A.V.; Betts, T.; et al. Evaluating real-world clinical outcomes in atrial fibrillation patients receiving the WATCHMAN left atrial appendage closure technology: Final 2-year outcome data of the EWOLUTION trial focusing on history of stroke and hemorrhage. Circ. Arrhythmia Electrophysiol. 2019, 12, e006841. [Google Scholar] [CrossRef] [PubMed]
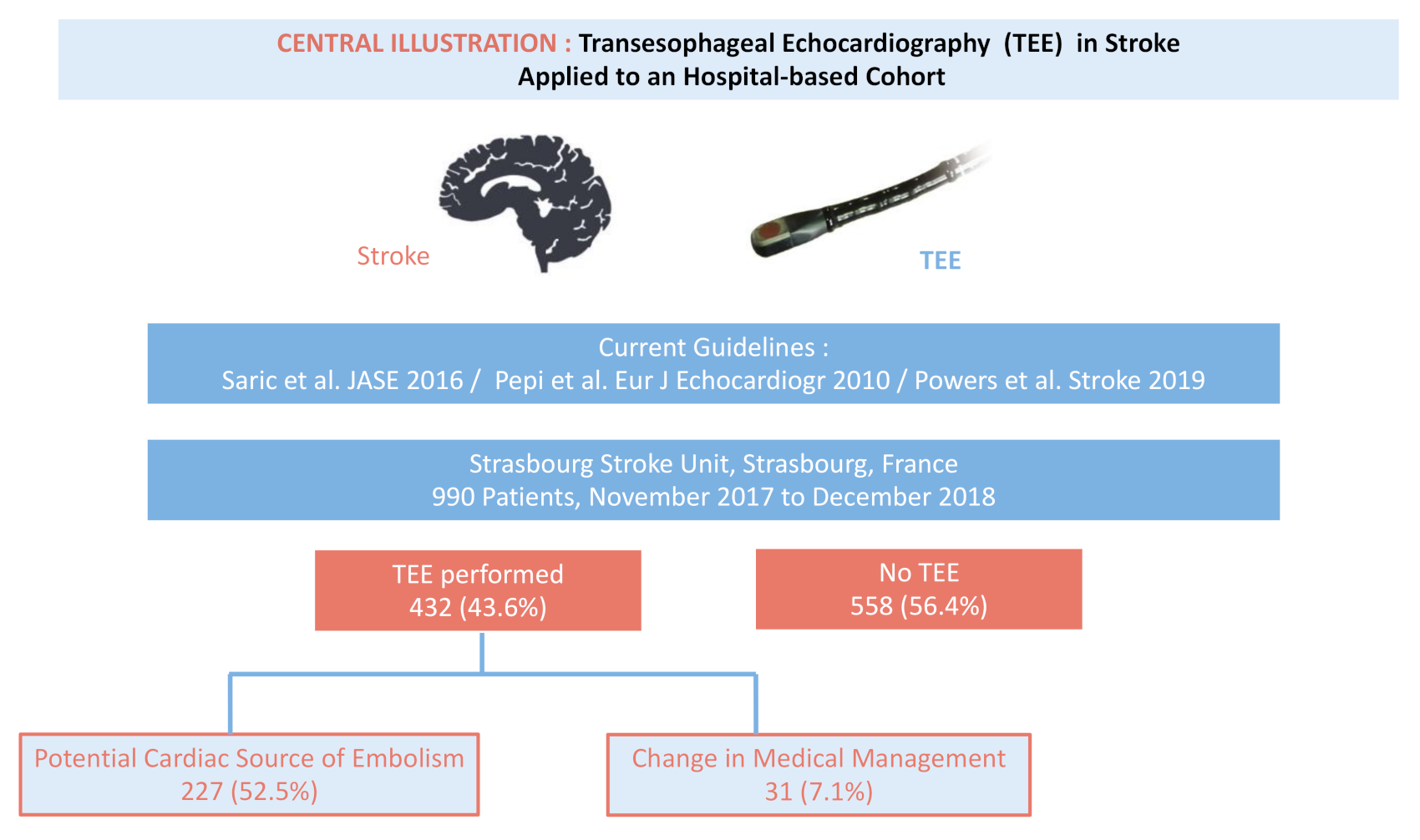
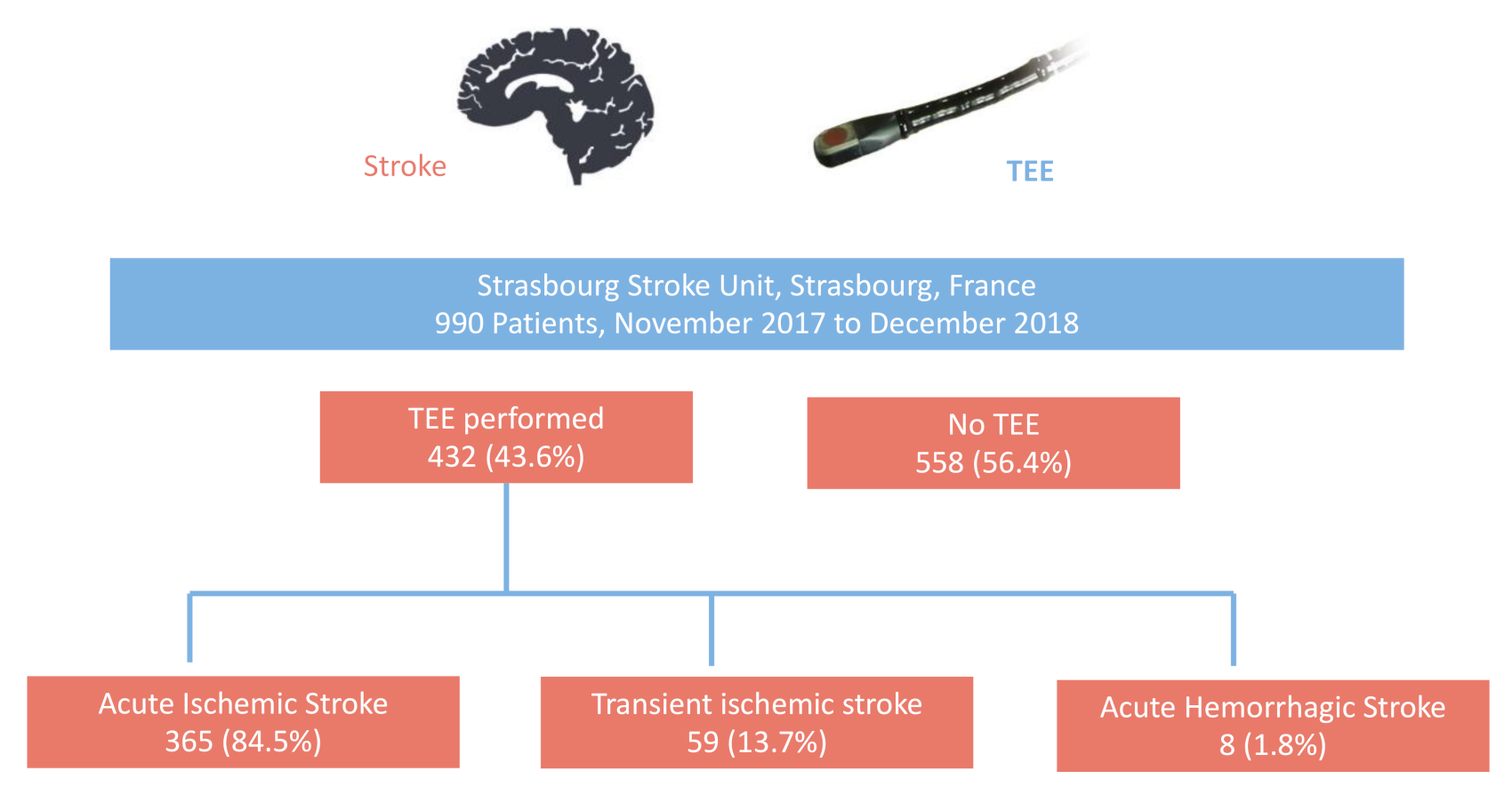
| Global Population | TEE + | No TEE − | p | |
|---|---|---|---|---|
| (n = 990) | (n = 432) | (n = 558) | ||
| Types of Stroke | ||||
| Hemorrhagic Stroke—n (%) | 67 (6.8%) | 8 (1.9%) | 59 (10.6%) | <0.001 |
| Transient Ischemic Attack—n (%) | 121 (12.2%) | 59 (13.7%) | 62 (11.1%) | 0.241 |
| Ischemic Stroke—n (%) | 802(81.0%) | 365 (84.5%) | 437(78.3%) | 0.014 |
| Large-artery atherosclerosis | 171 | 65 | 106 | 0.025 |
| Cardioembolism | 239 | 58 | 181 | <0.001 |
| Small-vessel occlusion | 7 | 3 | 4 | 1 |
| Stroke of other determined etiology | 7 | 0 | 7 | 0.018 |
| Stroke of undetermined etiology | 378 | 239 | 139 | <0.001 |
| Vascular territory of ischemic stroke | ||||
| Deep middle cerebral artery—n (%) | 91 (8.2%) | 31 (7.2%) | 50 (9.0%) | 0.350 |
| Superficial middle cerebral artery—n (%) | 354 (35.8%) | 130 (31.9%) | 216 (38.7%) | 0.032 |
| Borderzone territory—n (%) | 30 (3.0%) | 6 (1.4%) | 24 (4.3%) | 0.008 |
| Complete middle cerebral artery—n (%) | 51 (5.2%) | 17 (3.9%) | 34 (6.1%) | 0.148 |
| Anterior cerebral artery—n (%) | 24 (2.4%) | 9 (2.1%) | 15 (2.7%) | 0.678 |
| Anterior choroidal artery—n (%) | 61 (6.2%) | 23 (5.3%) | 38 (6.8%) | 0.354 |
| Posterior cerebral artery—n (%) | 95 (9.6%) | 42 (9.7%) | 53 (9.5%) | 0.914 |
| Vertebro-basilar—n (%) | 86 (8.7%) | 48 (11.1%) | 38 (6.8%) | 0.022 |
| Cerebellar—n (%) | 42 (4.2%) | 29 (6.7%) | 13 (2.3%) | 0.001 |
| Multiple—n (%) | 82 (8.3%) | 32 (7.4%) | 50(8.9%) | 0.060 |
| Global Population | TEE + | No TEE - | p | ||
|---|---|---|---|---|---|
| (n = 990) | (n = 432) | (n = 558) | |||
| Age (years) | 69.03 ± 14.6 | 62.88 ± 14.8 | 73.8 ± 12.5 | <0.001 | |
| Sex (Male)—n (%) | 553 (55.9%) | 263 (60.9%) | 290 (52.0%) | 0.006 | |
| BMI (Kg.m2) | 26.7 ± 5.4 | 26.9 ± 5.7 | 26.4 ± 5.1 | 0.149 | |
| CV Risk Factors | |||||
| Hypertension—n (%) | 631 (63.7%) | 237 (54.9%) | 394 (70.6) | <0.001 | |
| Dyslipidemia—n (%) | 254 (25.7%) | 103 (23.8%) | 151 (27.1%) | 0.271 | |
| Past or current smoker—n (%) | 365 (36.9%) | 178 (41.2%) | 187 (33.5%) | 0.014 | |
| Diabetes mellitus—n (%) | 219 (22.1%) | 78 (18.1%) | 141 (25.3%) | 0.007 | |
| Familial history of CAD—n (%) | 5 (0.5%) | 5 (1.2%) | 0 (0.0%) | 0.016 | |
| Medical History | |||||
| Hemorrhagic stroke—n (%) | 4 (0.4%) | 1 (0.2%) | 3 (0.5%) | 0.636 | |
| Ischemic stroke—n (%) | 124 (12.5%) | 40 (9.3%) | 84 (15.1%) | 0.007 | |
| TIA—n (%) | 41 (4.1%) | 20 (4.6%) | 21 (3.8%) | 0.523 | |
| PAD—n (%) | 67 (6.8%) | 20 (4.6%) | 47 (8.4%) | 0.021 | |
| Carotid endarterectomy—n (%) | 11 (1.1%) | 2 (0.5%) | 9 (1.6%) | 0.126 | |
| AF—n (%) | 172 (17.4%) | 26 (6.0%) | 146 (26.2%) | <0.001 | |
| Mechanical valve—n (%) | 17 (1.7%) | 11 (2.5%) | 6 (1.1%) | 0.088 | |
| Bioprosthetic valve—n (%) | 4 (0.4%) | 1 (0.2%) | 3 (0.5%) | 0.165 | |
| CAD—n (%) | 118 (11.9%) | 50 (11.6%) | 68 (12.2%) | 0.843 | |
| Coronary Angioplasty—n (%) | 78 (7.9%) | 36 (8.3%) | 42 (7.5%) | 0.637 | |
| STEMI—n (%) | 47 (4.7%) | 22 (5.1%) | 25 (4.5%) | 0.655 | |
| CABG—n (%) | 12 (1.2%) | 5 (1.2%) | 7 (1.3%) | 1.000 | |
| CKD eGFR <60—n (%) | 68 (6.9%) | 21 (4.9%) | 47 (8.4%) | 0.031 | |
| Neoplasia—n (%) | 121 (12.2%) | 40 (9.3%) | 81 (14.5%) | 0.014 | |
| Thromboembolic disease- n (%) | 55 (5.6%) | 11 (2.5%) | 44 (7.9%) | <0.001 | |
| Baseline biological parameters | |||||
| Cr eGFR (mL/min/1.73m²) ± DS | 77.6 ± 21.7 | 81.2± 21.6 | 74.8 ± 21.4 | <0.001 | |
| Cr level (µmol.L) ± DS | 84.7 ± 39.9 | 88.6 ± 37.4 | 82.8 ± 40.9 | 0.058 | |
| Total cholesterol (g/L) ± DS | 1.9 ± 0.36 | 1.7 ± 0.45 | 2.08 ± 0.38 | 0.447 | |
| LDL-C (g/L) ± DS | 1.06 ± 0.37 | 1.10 ± 0.36 | 1.03 ± 0.38 | 0.003 | |
| HDL-C (g/L) ± DS | 0.50 ± 0.32 | 0.48 ± 0.15 | 0.50 ± 0.41 | 0.232 | |
| TG (g/L) ± DS | 1.23 ± 0.88 | 1.22 ± 0.68 | 1.24 ± 1.02 | 0.759 | |
| Glycated Hb(%) ± DS | 5.9 ± 1.29 | 5.9 ± 1.22 | 5.9 ± 1.34 | 0.139 | |
| Scoring systems and scales | |||||
| CHA2DS2-VASc score * | 5.3 ± 1.2 | 5.1 ± 1.5 | 5.3 ± 1.2 | 0.359 | |
| HAS BLED Score * | 3.4± 1.1 | 3.3 ± 1.3 | 3.5± 1.08 | 0.291 | |
| NIHSS (IQR) | 2 (0–10) | 2(0–4) | 3(0–10) | <0.01 | |
| Modified RANKIN Scale (IQR) | 1 (0–3) | 0 (0–1) | 1 (0–3) | <0.01 | |
| Average Lengh of Stay (days) | 12.2 ± 13.0 | 9.9 ± 10.6 | 13.9 ±14.4 | <0.001 | |
| Baseline/Pre hospital medication | |||||
| Antiplatelet Agents—n(%) | 309 (31.2%) | 119 (27.5%) | 190 (34.1%) | 0.032 | |
| ASA—n(%) | 275 (27.8%) | 110 (25.5%) | 165 (29.6) | 0.174 | |
| Clopidogrel—n(%) | 83 (8.4%) | 33 (7.6%) | 50 (9.0%) | 0.263 | |
| DAPT—n(%) | 34 (3.4%) | 14 (3.2%) | 20 (3.6%) | 0.456 | |
| VKA—n(%) | 93 (9.4%) | 18 (4.2%) | 75 (13.4%) | <0.001 | |
| DOAC—n(%) | 59 (6.0%) | 13 (3.0%) | 46 (8.3%) | <0.001 | |
| Beta blockers—n(%) | 338 (34.1%) | 125 (28.9%) | 213 (38.2%) | 0.001 | |
| ACE inhibitors/ARBs—n (%) | 412 (41.6%) | 155 (35.9%) | 257 (46.1%) | 0.001 | |
| Aldosterone-receptor antagonists—n (%) | 40 (4.0%) | 15 (3.5%) | 25 (4.5%) | 0.264 | |
| Statin—n (%) | 320 (32.3%) | 139 (32.2%) | 181 (32.4%) | 0.493 | |
| Furosemide—n (%) | 93 (9.4%) | 21 (4.9%) | 72 (12.9%) | <0.001 | |
| Population with TEE Performed | Abnormal TEE Findings | |
|---|---|---|
| (n = 432) | (n = 227) | |
| Infective Endocarditis—n (%) | 0 (0.0%) | 0 (0.0%) |
| LA Spontaneous Contrast—n (%) | 14 (3.2%) | 14 (6.2%) |
| LAA Spontaneous Contrast—n (%) | 8 (1.9%) | 8 (3.5%) |
| LA/LV Thrombus—n (%) | 3 (0.7%) | 3 (1.3%) |
| Left Atrial Appendage Thrombus—n (%) | 6 (1.4%) | 6 (2.6%) |
| Prosthetic Thrombus—n (%) | 4 (0.9%) | 4 (1.8%) |
| Patent Foramen Ovale—n (%) * | 134 (31.0%) | 134 (59.0%) |
| Grade 1: < 5 bubbles | 82 (61.2%) | |
| Grade 2: 5 to 25 bubbles | 29 (21.6%) | |
| Grade 3: > 25 bubbles | 13 (9.7%) | |
| Grade 4: opacification of chamber | 10 (7.5) | |
| Atrial Septal Aneurysm—n (%) | 48 (11.1%) | 48 (21.1%) |
| Complex aortic plaque—n (%) | 118 (27.3%) | 118 (52.0%) |
| TEE | Abnormal TEE Findings | Normal TEE | p | |
|---|---|---|---|---|
| (n = 432) | (n = 227) | (n = 205) | ||
| Age (years) | 62.9 ± 14.8 | 63.0 ±15.3 | 62.7 ± 14.2 | 0.859 |
| Sex (Male)—n (%) | 263 (60.9%) | 140 (61.7%) | 123 (60.0%) | 0.767 |
| BMI (Kg/m2) | 26.9 ± 5.6 | 26.9 ± 6.1 | 26.9 ± 5.1 | 0.924 |
| CV Risk Factors | ||||
| Hypertension—n (%) | 237 (55.0%) | 130 (57.3%) | 107 (52.2%) | 0.333 |
| Dyslipidemia—n (%) | 103 (23.8%) | 56 (24.7%) | 47 (22.9%) | 0.735 |
| Past or current smoker—n (%) | 178 (41.2%) | 95(41.9%) | 83 (40.5%) | 0.845 |
| Diabetes mellitus—n (%) | 78 (18.1%) | 45 (19.8%) | 33 (16.1%) | 0.320 |
| Familial history of CAD—n (%) | 5 (1.2%) | 5(2.2%) | 0 (0.0%) | 0.063 |
| Medical History | ||||
| Hemorrhagic stroke—n (%) | 1 (0.2%) | 1 (0.4%) | 0 (0.0%) | 1.000 |
| Ischemic stroke—n (%) | 40 (9.3%) | 18 (7.9%) | 22 (10.7%) | 0.324 |
| TIA—n (%) | 20 (4.6%) | 14 (6.2%) | 6 (2.9%) | 0.168 |
| PAD—n (%) | 20(4.6%) | 17 (7.5%) | 3 (1.5%) | 0.003 |
| AF—n (%) | 26 (6.0%) | 13 (5.7%) | 13 (6.3%) | 0.841 |
| Mechanical valve—n (%) | 3 (0.7%) | 1 (0.4%) | 2 (1.0%) | 0.606 |
| Bioprosthetic valve—n (%) | 1 (0.2%) | 0 (0.0%) | 1 (0.5%) | 0.475 |
| CAD—n (%) | 50 (11.6%) | 29 (12.8%) | 21 (10.2%) | 0.453 |
| CKD eGFR <60—n (%) | 21 (4.9%) | 9 (4.0%) | 12 (5.9%) | 0.380 |
| Neoplasia—n(%) | 40 (9.3%) | 21 (9.3%) | 19 (9.3%) | 1.000 |
| Thromboembolic disease—n (%) | 11(2.5%) | 4 (1.8%) | 7 (3.4%) | 0.363 |
| Baseline biological parameters | ||||
| Cr eGFR (mL/min/1.73m²) ± DS | 81.2 ± 21.6 | 80.8± 21.6 | 81.7 ± 21.5 | 0.694 |
| Cr level (µmol.L) ± DS | 88.6± 37.4 | 89.8 ± 39.7 | 87.3± 35.1 | 0.583 |
| LDL-C (g/L) ± DS | 1.10 ± 0.36 | 1.11 ± 0.39 | 1.09 ± 0.33 | 0.504 |
| TG (g/l) ± DS | 1.22 ± 0.68 | 1.23 ± 0.70 | 1.21 ± 0.67 | 0.688 |
| Glycated Hb (%) ± DS | 5.9 ± 1.22 | 5.9 ± 1.15 | 5.9 ± 1.29 | 0.876 |
| Scoring systems and scales | ||||
| CHA2DS2-VASc score | 5.1 ± 1.5 | 5.3± 1.7 | 5.0 ± 1.4 | 0.608 |
| HAS BLED Score | 3.3± 1.3 | 3.6 ± 1.7 | 3.0± 0.9 | 0.167 |
| NIHSS (IQR) | 2 (0–4) | 2 (0–4) | 1.5 (0–4) | 0.854 |
| Modified RANKIN Scale (IQR) | 1 (0–1) | 0 (0–1) | 1 (0–1) | 0.441 |
| Baseline/Pre hospital medication | ||||
| Antiplatelet Agents—n(%) | 119 (27.5%) | 66 (29.1%) | 53 (25.9%) | 0.518 |
| VKA—n(%) | 18(4.2%) | 10 (4.4%) | 8 (3.9%) | 0.815 |
| DOAC—n(%) | 125 (28.9%) | 69 (30.4%) | 56 (27.3%) | 0.524 |
| TEE with Change in Medical Management | |
|---|---|
| (n = 31) | |
| TEE Findings | |
| LA/LV Thrombus—n(%) | 3 (9.6%) |
| Left Atrial Appendage Thrombus—n(%) | 2 (6.4%) |
| Prosthetic Thrombus—n(%) | 3 (9.6%) |
| Left Atrial Appendage Closure—n(%) | 6 (19.4%) |
| PFO Closure—n(%) | 16 (51.6%) |
| Ulcerated complex aortic plaque with anticoagulation—n(%) | 1 (3.2%) |
| Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|
| HR | 95%CI | p | HR | 95%CI | p | |
| Abnormal Findings in TEE | ||||||
| PAD | 5.424 | 1.566–18.790 | 0.008 | 3.144 | 0.812–12.164 | 0.097 |
| CAD | 3.222 | 1.167–8.898 | 0.024 | 1.921 | 0.611–6.040 | 0.264 |
| LA volume (mL/m2) | 1.014 | 1.000–1.029 | 0.045 | 1.013 | 0.999–1.027 | 0.070 |
| Change In Medical Management following TEE | ||||||
| Age (years) | 0.967 | 0.946–0.989 | 0.003 | 0.948 | 0.923–0.974 | <0.001 |
| Past ischemic stroke | 3.297 | 1.322–8.224 | 0.011 | 3.542 | 1.290–9.722 | 0.014 |
| Past TIA | 5.013 | 1.690–14.868 | 0.004 | 7.830 | 2.214–27.689 | 0.001 |
| AF History | 4.400 | 1.629–11.882 | 0.003 | 3.981 | 0.738–21.468 | 0.108 |
| Mechanical valve | 4.243 | 1.104–16.300 | 0.035 | 1.424 | 0.095–21.299 | 0.798 |
| VKA at admission | 3. 390 | 1.066–10.781 | 0.039 | 1.366 | 0.115–16.261 | 0.805 |
| Superficial middle cerebral artery stroke territory | 2.083 | 0.999–4.345 | 0.050 | 2.774 | 1.168–6.589 | 0.021 |
| Cerebellar stroke territory | 3.061 | 1.080–8.679 | 0.035 | 2.687 | 0.727–9.924 | 0.138 |
| Baseline Hb (g/dL) | 0.827 | 0.685–0.998 | 0.047 | 0.906 | 0.729–1.126 | 0.373 |
| De novo AF | 3.656 | 1.456–9.176 | 0.006 | 2.582 | 0.601–11.100 | 0.202 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siegel, C.; Marchandot, B.; Matsushita, K.; Trimaille, A.; Mirea, C.; Peillex, M.; Sauer, F.; How-Choong, C.; Zeyons, F.; Rouyer, O.; et al. The Effect of Transoesophageal Echocardiography on Treatment Change in a High-Volume Stroke Unit. J. Clin. Med. 2021, 10, 805. https://doi.org/10.3390/jcm10040805
Siegel C, Marchandot B, Matsushita K, Trimaille A, Mirea C, Peillex M, Sauer F, How-Choong C, Zeyons F, Rouyer O, et al. The Effect of Transoesophageal Echocardiography on Treatment Change in a High-Volume Stroke Unit. Journal of Clinical Medicine. 2021; 10(4):805. https://doi.org/10.3390/jcm10040805
Chicago/Turabian StyleSiegel, Camille, Benjamin Marchandot, Kensuke Matsushita, Antonin Trimaille, Corina Mirea, Marilou Peillex, François Sauer, Cecile How-Choong, Floriane Zeyons, Olivier Rouyer, and et al. 2021. "The Effect of Transoesophageal Echocardiography on Treatment Change in a High-Volume Stroke Unit" Journal of Clinical Medicine 10, no. 4: 805. https://doi.org/10.3390/jcm10040805
APA StyleSiegel, C., Marchandot, B., Matsushita, K., Trimaille, A., Mirea, C., Peillex, M., Sauer, F., How-Choong, C., Zeyons, F., Rouyer, O., Trinh, A., Petit-Eisenmann, H., Jesel, L., Ohlmann, P., Wolff, V., & Morel, O. (2021). The Effect of Transoesophageal Echocardiography on Treatment Change in a High-Volume Stroke Unit. Journal of Clinical Medicine, 10(4), 805. https://doi.org/10.3390/jcm10040805






