Impaired Kidney Function Associated with Increased Risk of Side Effects in Patients with Small Vessel Vasculitis Treated with Rituximab as an Induction Therapy
Abstract
1. Introduction
2. Methods
- complete remission: 0 points in the BVAS/WG score after 6 months;
- partial remission: drop by 50% in the BVAS/WG score after 6 months;
- inadequate response: <50% reduction of disease activity in the BVAS/WG score after 6 months.
Statistical Analysis
3. Results
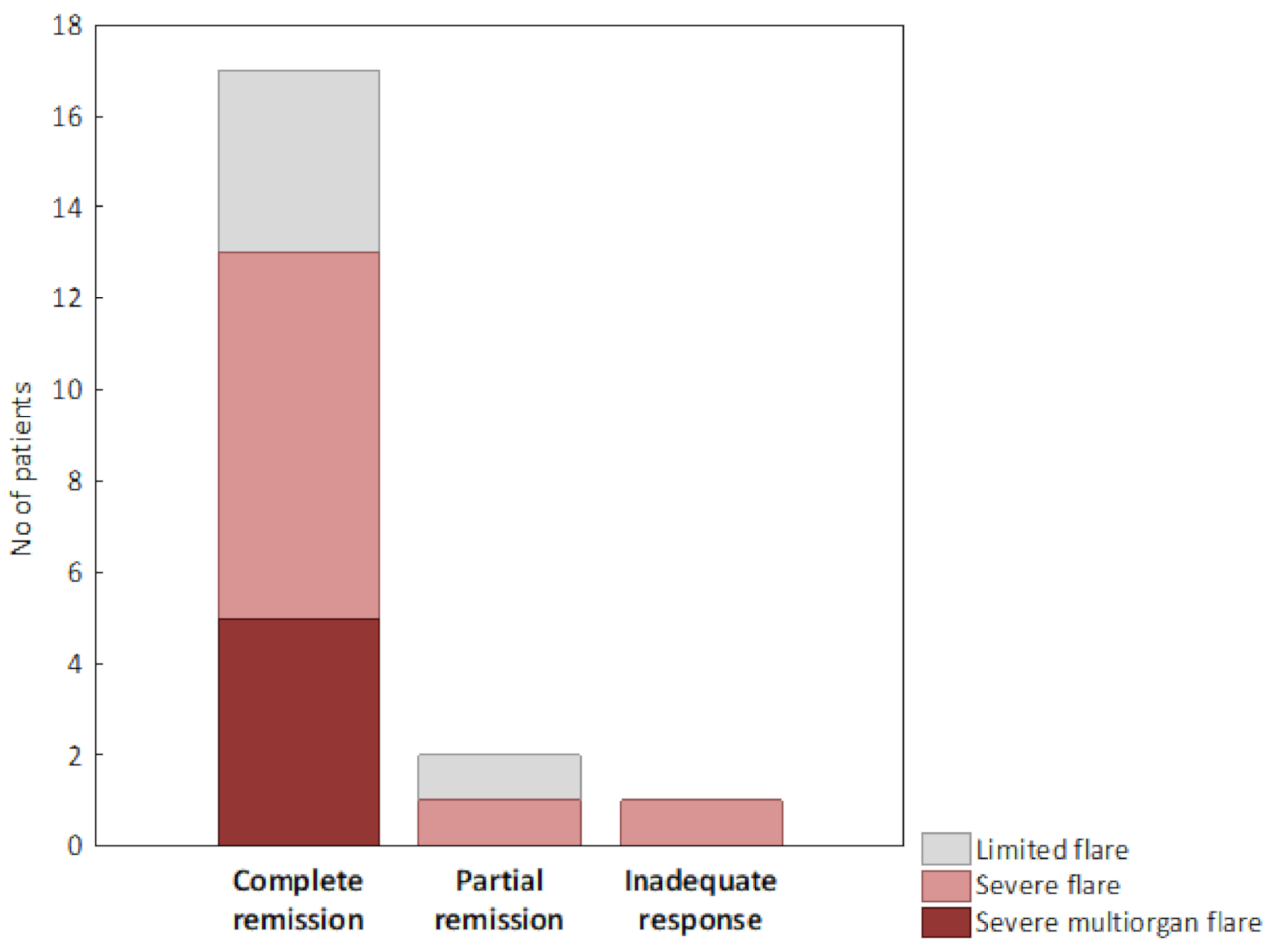
3.1. Efficacy
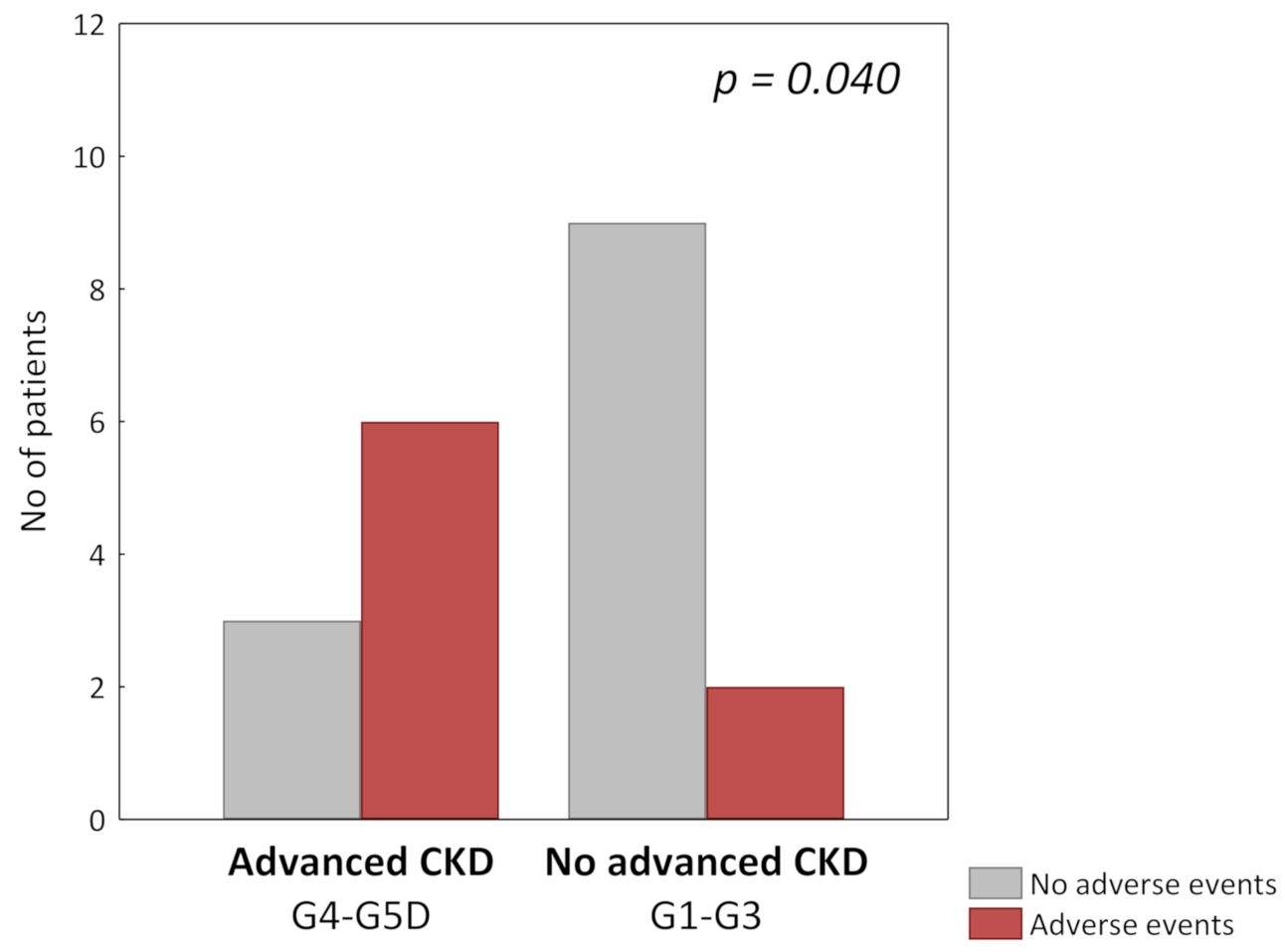
3.2. Safety
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jennette, J.C.; Falk, R.J.; Bacon, P.A.; Basu, N.; Cid, M.C.; Ferrario, F.; Flores-Suarez, L.F.; Gross, W.L.; Guillevin, L.; Hagen, E.C.; et al. 2012 Revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013, 65, 1–11. [Google Scholar] [CrossRef]
- Geetha, D.; Jefferson, J.A. ANCA-Associated Vasculitis: Core Curriculum 2020. Am. J. Kidney Dis. 2020, 75, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Lyons, P.A.; Rayner, T.F.; Trivedi, S.; Holle, J.U.; Watts, R.A.; Jayne, D.R.; Baslund, B.; Brenchley, P.; Bruchfeld, A.; Chaudhry, A.N.; et al. Genetically distinct subsets within ANCA-associated vasculitis. N. Engl. J. Med. 2012, 367, 214–223. [Google Scholar] [CrossRef] [PubMed]
- De Jonge, M.E.; Huitema, A.D.R.; Rodenhuis, S.; Beijnen, J.H. Clinical Pharmacokinetics of Cyclophosphamide. Clin. Pharmacokinet. 2005, 44, 1135–1164. [Google Scholar] [CrossRef]
- Harper, L.; Morgan, M.D.; Walsh, M.; Hoglund, P.; Westman, K.; Flossmann, O.; Tesar, V.; Vanhille, P.; De Groot, K.; Luqmani, R.; et al. Pulse versus daily oral cyclophosphamide for induction of remission in ANCA-associated vasculitis: Long-term follow-up. Ann. Rheum. Dis. 2012, 71, 955–960. [Google Scholar] [CrossRef]
- Stasi, R. Rituximab in Autoimmune Hematologic Diseases: Not Just a Matter of B Cells. Semin. Hematol. 2010, 47, 170–179. [Google Scholar] [CrossRef]
- Yates, M.; Watts, R.A.; Bajema, I.M.; Cid, M.C.; Crestani, B.; Hauser, T.; Hellmich, B.; Holle, J.U.; Laudien, M.; Little, M.A.; et al. EULAR/ERA-EDTA recommendations for the management of ANCA-associated vasculitis. Ann. Rheum. Dis. 2016, 75, 1583–1594. [Google Scholar] [CrossRef] [PubMed]
- Unizony, S.; Villarreal, M.; Miloslavsky, E.M.; Lu, N.; Merkel, P.A.; Spiera, R.; Seo, P.; Langford, C.A.; Hoffman, G.S.; Kallenberg, C.G.M.; et al. Clinical outcomes of treatment of anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis based on ANCA type. Ann. Rheum. Dis. 2016, 75, 1166–1169. [Google Scholar] [CrossRef]
- Guillevin, L.; Pagnoux, C.; Karras, A.; Khouatra, C.; Aumaître, O.; Cohen, P.; Maurier, F.; Decaux, O.; Ninet, J.; Gobert, P.; et al. Rituximab versus Azathioprine for Maintenance in ANCA-Associated Vasculitis. N. Engl. J. Med. 2014, 371, 1771–1780. [Google Scholar] [CrossRef] [PubMed]
- Trivin, C.; Tran, A.; Moulin, B.; Choukroun, G.; Gatault, P.; Courivaud, C.; Augusto, J.-F.; Ficheux, M.; Vigneau, C.; Thervet, E.; et al. Infectious complications of a rituximab-based immunosuppressive regimen in patients with glomerular disease. Clin. Kidney J. 2016, 10, 461–469. [Google Scholar] [CrossRef]
- Gottenberg, J.-E.; Ravaud, P.; Bardin, T.; Cacoub, P.; Cantagrel, A.; Combe, B.; Dougados, M.; Flipo, R.M.; Godeau, B.; Guillevin, L.; et al. Risk factors for severe infections in patients with rheumatoid arthritis treated with rituximab in the autoimmunity and rituximab registry. Arthritis Rheum. 2010, 62, 2625–2632. [Google Scholar] [CrossRef] [PubMed]
- Terrier, B.; Amoura, Z.; Ravaud, P.; Hachulla, E.; Jouenne, R.; Combe, B.; Bonnet, C.; Cacoub, P.; Cantagrel, A.; De Bandt, M.; et al. Safety and efficacy of rituximab in systemic lupus erythematosus: Results from 136 patients from the French autoimmunity and rituximab registry. Arthritis Rheum. 2010, 62, 2458–2466. [Google Scholar] [CrossRef] [PubMed]
- Thery-Casari, C.; Euvrard, R.; Mainbourg, S.; Durupt, S.; Reynaud, Q.; Durieu, I.; Belot, A.; Lobbes, H.; Cabrera, N.; Lega, J.-C. Severe infections in patients with anti-neutrophil cytoplasmic antibody-associated vasculitides receiving rituximab: A meta-analysis. Autoimmun. Rev. 2020, 19, 102505. [Google Scholar] [CrossRef]
- Stone, J.H.; Hoffman, G.S.; Merkel, P.A.; Min, Y.I.; Uhlfelder, M.L.; Hellmann, D.B.; Specks, U.; Allen, N.B.; Davis, J.C.; Spiera, R.F.; et al. International Network for the Study of the Systemic Vasculitides (INSSYS). A disease-specific activ-ity index for Wegener’s granulomatosis: Modification of the Birmingham Vasculitis Activity Score. International Network for the Study of the Systemic Vasculitides (INSSYS). Arthritis Rheum. 2011, 44, 912–920. [Google Scholar] [CrossRef]
- Lionaki, S.; Blyth, E.R.; Hogan, S.L.; Hu, Y.; Senior, J.B.A.; Jennette, C.E.; Nachman, P.H.; Jennette, J.C.; Falk, R.J. Classification of antineutrophil cytoplasmic autoantibody vasculitides: The role of antineutrophil cytoplasmic autoantibody specificity for myeloperoxidase or proteinase 3 in disease recognition and prognosis. Arthritis Rheum. 2012, 64, 3452–3462. [Google Scholar] [CrossRef]
- Mahr, A.; Katsahian, S.; Varet, H.; Fichet, J.; Hagen, E.C.; Höglund, P.; Merkel, P.A.; Pagnoux, C.; Rasmussen, N.; Westman, K.; et al. Revisiting the classification of clinical phenotypes of anti-neutrophil cytoplasmic antibody-associated vasculitis: A cluster analysis. Ann. Rheum. Dis. 2012, 72, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, J.H.; Wright, M.N.; Herrmann, K.; Laudien, M.; Nölle, B.; Reinhold-Keller, E.; Bremer, J.P.; Moosig, F.; Holle, J.U. Myeloperoxidase-Antineutrophil Cytoplasmic Antibody (ANCA)-Positive Granulomatosis With Polyangiitis (Wegener’s) Is a Clinically Distinct Subset of ANCA-Associated Vasculitis: A Retrospective Analysis of 315 Patients From a German Vasculitis Referral Cent. Arthritis Rheumatol. 2016, 68, 2953–2963. [Google Scholar] [CrossRef]
- Walsh, M.; Flossmann, O.; Berden, A.; Westman, K.; Höglund, P.; Stegeman, C.; Jayne, D.; European Vasculitis Study Group. Risk factors for relapse of antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheum. 2012, 64, 542–548. [Google Scholar] [CrossRef]
- Kado, R.; Sanders, G.; McCune, W.J. Suppression of normal immune responses after treatment with rituximab. Curr. Opin. Rheumatol. 2016, 28, 251–258. [Google Scholar] [CrossRef]
- Cortazar, F.B.; Iii, W.F.P.; Wenger, J.; Owens, C.T.; Laliberte, K.; Niles, J.L. Effect of Continuous B Cell Depletion with Rituximab on Pathogenic Autoantibodies and Total IgG Levels in Antineutrophil Cytoplasmic Antibody–Associated Vasculitis. Arthritis Rheumatol. 2017, 69, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Roberts, D.M.; Jones, R.B.; Smith, R.M.; Alberici, F.; Kumaratne, D.S.; Burns, S.; Jayne, D.R. Rituximab-associated hypogammaglobulinemia: Incidence, predictors and outcomes in patients with multi-system autoimmune disease. J. Autoimmun. 2015, 57, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Hruskova, Z.; Segelmark, M.; Morgan, M.D.; Hogan, J.; Lee, S.K.; Dale, J.; Harper, L.; Tesar, V.; Jayne, D.R.; et al. Treatment of Severe Renal Disease in ANCA Positive and Negative Small Vessel Vasculitis with Rituximab. Am. J. Nephrol. 2015, 41, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Scemla, A.; Loupy, A.; Candon, S.; Mamzer, M.-F.; Martinez, F.; Zuber, J.; Sberro, R.; Legendre, C.; Thervet, E. Incidence of Infectious Complications in Highly Sensitized Renal Transplant Recipients Treated by Rituximab: A Case-Controlled Study. Transplantation 2010, 90, 1180–1184. [Google Scholar] [CrossRef] [PubMed]
- Jillella, A.P.; Dainer, P.M.; Kallab, A.M.; Ustun, C. Treatment of a patient with end-stage renal disease with Rituximab: Pharmacokinetic evaluation suggests Rituximab is not eliminated by hemodialysis. Am. J. Hematol. 2002, 71, 219–222. [Google Scholar] [CrossRef]
- Dalrymple, L.S.; Go, A.S. Epidemiology of Acute Infections among Patients with Chronic Kidney Disease: Figure 1. Clin. J. Am. Soc. Nephrol. 2008, 3, 1487–1493. [Google Scholar] [CrossRef]
- Kausz, A.T.; Gilbertson, D.T. Overview of Vaccination in Chronic Kidney Disease. Adv. Chronic Kidney Dis. 2006, 13, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Chmielewski, M.; Honda, H.; Pecoits-Filho, R.; Matsuo, S.; Yuzawa, Y.; Tranaeus, A.; Stenvinkel, P.; Lindholm, B. Aspects of Immune Dysfunction in End-stage Renal Disease. Clin. J. Am. Soc. Nephrol. 2008, 3, 1526–1533. [Google Scholar] [CrossRef]
| Parameter | RTX Induction n = 20 | |
|---|---|---|
| Age (years) | mean ± SD min–max | 49 ± 13 24–70 |
| Sex: | ||
| Male | n (%) | 10 (50%) |
| Female | n (%) | 10 (50%) |
| ANCA type: | ||
| PR3-ANCA | n (%) | 18 (90%) |
| MPO-ANCA | n (%) | 1 (5%) |
| ANCA negative | n (%) | 1 (5%) |
| Duration of SVV (years) | median (IQR) min–max | 3 (1–5.5) 0–19 |
| Treatment of SVV | ||
| Indication for RTX induction: | ||
| —Failure of CYC | n (%) | 8 (40%) |
| —High cumulative dose of CYC | n (%) | 11 (55%) |
| —Adverse effects related to CYC | n (%) | 1 (5%) |
| CYC cumulative dose (g) | median (IQR) min–max | 11 (7.5–23) 0.7–60 |
| CYC cumulative dose: | ||
| >36 g | n (%) | 3 (15%) |
| ≤36 g | n (%) | 17 (85%) |
| CYC cumulative dose (mg/kg) | median (IQR) min–max | 152 (96–303) 13–759 |
| CYC cumulative dose: | ||
| >120 mg/kg | n (%) | 13 (65%) |
| ≤120 mg/kg | n (%) | 7 (35%) |
| Parameter | RTX Induction n = 20 | |
|---|---|---|
| Clinical picture of SVV | ||
| Renal involvement: | ||
| —no 1 | n (%) | 3 (15%) |
| —yes, CKD G1-G3 | n (%) | 8 (40%) |
| —yes, CKD G4-G5D 2 | n (%) | 9 (45%) |
| Disease activity by BVAS/WG: | ||
| —general symptoms | n (%) | 9 (45%) |
| —cutaneous | n (%) | 5 (25%) |
| —mucosal membranes/eyes | n (%) | 1 (5%) |
| —ear–nose–throat (ENT) | n (%) | 15 (75%) |
| —cardiovascular | n (%) | 0 |
| —gastrointestinal | n (%) | 1 (5%) |
| —pulmonary | n (%) | 15 (75%) |
| —renal | n (%) | 12 (60%) |
| —nervous system | n (%) | 4 (20%) |
| —other | n (%) | 8 (40%) |
| SVV course: | ||
| —limited flare | n (%) | 5 (25%) |
| —severe flare | n (%) | 10 (50%) |
| —severe multiorgan flare | n (%) | 5 (25%) |
| Dominant organ activity (presence of major symptoms): | ||
| —eye | ||
| —ENT | n (%) | 1 (5%) |
| —pulmonary | n (%) | 4 (20%) |
| —gastrointestinal | n (%) | 2 (10%) |
| —renal | n (%) | 1 (5%) |
| —nervous system | n (%) | 4 (20%) |
| —other | n (%) | 3 (15%) |
| Dominant organ activity (presence of major symptoms): | n (%) | 4 (20%) * |
| Parameter | Baseline | Month 6 | p-Value | |
|---|---|---|---|---|
| BVAS/WG score | median (IQR) min–max | 6 (5–9) 3–12 | 0 (0–0) 0–4 | 0.0001 * |
| ANCA titre (IU/mL) | median (IQR) min–max | 30 (10–124) 0–177 | 6.2 (0–24) 0–174 | 0.0003 * |
| IgA (mg/dL) | mean ± SD min–max | 196.5 ± 66.7 109–330 | 141.9 ± 53.2 58–247 | 0.0001 |
| IgM (mg/dL) | median (IQR) min–max | 65 (34–117) 26–164 | 39.5 (28–65) 10–127 | 0.0003 * |
| IgG (mg/dL) | mean ± SD min–max | 969.5 ± 320.7 369–1740 | 726.3 ± 243.3 325–1230 | 0.002 |
| IgG (mg/dL) <400 400–699 ≥700 | n (%) n (%) n (%) | 1 (5%) 3 (15%) 16 (80%) | 1 (5%) 9 (45%) 10 (50%) | 0.112 # |
| Serum creatinine (mg/dL) | median (IQR) min–max | 1.6 (0.9–2.5) 0.8–4.5 | 1.4 (1.0–2.1) 0.8–5.7 | 1.000 |
| eGFR ^ (mL/min/1.73 m2) | median (IQR) min–max | 54 (25–79) 15–108 | 55 (29–75) 11–90 | 0.691 * |
| Hematuria ^ | 0.039 | |||
| —yes, massive | n (%) | 5 (31.25%) | 0 | |
| —yes, non-massive | n (%) | 5 (31.25%) | 5 (31.25%) | |
| —no | n (%) | 6 (37.5%) | 11 (68.75%) | |
| Proteinuria (mg/dL) | median (IQR) | 29 (10–46) | 21 (10–43) | 0.722 |
| Parameter | No Adverse Events n = 12 | Adverse Events n = 8 | p-Value | |
|---|---|---|---|---|
| Age (years) | mean ± SD | 42 ± 11 | 57 ± 12 | 0.014 * |
| Sex: Male Female | n (%) n (%) | 5 (41.7%) 7 (58.3%) | 5 (62.5%) 3 (37.5%) | 0.650 |
| Duration of AAV (years) | median (IQR) | 3 (3–10.5) | 1.5 (1–4) | 0.107 |
| CYC cumulative dose (g) | median (IQR) | 18 (9.63–35.5) | 8.75 (6.5–10.38) | 0.082 |
| CYC cumulative dose: >36 g ≤36 g | n (%) n (%) | 3 (25%) 9 (75%) | 0 8 (100%) | 0.242 |
| CYC cumulative dose (mg/kg) | median (IQR) | 185 (133–465) | 105 (82–209) | 0.153 |
| CYC cumulative dose: >120 mg/kg ≤120 mg/kg | n (%) n (%) | 10 (83.3%) 16.7%) | 3 (37.5%) 5 (62.5%) | 0.062 |
| RTX single dose (mg) | mean ± SD | 721 ± 96 | 700 ± 122 | 0.682 |
| Baseline BVAS/WG score | median (IQR) | 5 (4.5–8) | 6 (6–10) | 0.111 |
| Baseline ANCA titre (IU/mL) | median (IQR) | 37 (4–127) | 26 (8–73) | 0.847 |
| Serum creatinine (mg/dL) | median (IQR) | 1.3 (0.85–2.05) | 3.35 (2.2–4.9) | 0.044 |
| eGFR (mL/min/1.73 m2) G1-G3: ≥30 G4-G5D: <30 | n (%) n (%) | 9 (75%) 3 (25%) | 2 (25%) 6 (75%) | 0.040 # |
| eGFR (mL/min/1.73 m2) | mean ± SD | 57 ± 32 | 29 ± 26 | 0.049 |
| IgA (mg/dL) | mean ± SD | 207.2 ± 65.5 | 175.9 ± 71.4 | 0.337 * |
| IgM (mg/dL) | median (IQR) | 67 (53.5–128.5) | 52.5 (31.5–85) | 0.203 |
| IgG (mg/dL) | mean ± SD | 995.7 ± 317.3 | 921.9 ± 345.8 | 0.636 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rymarz, A.; Matyjek, A.; Sułek-Jakóbczyk, M.; Mosakowska, M.; Niemczyk, S. Impaired Kidney Function Associated with Increased Risk of Side Effects in Patients with Small Vessel Vasculitis Treated with Rituximab as an Induction Therapy. J. Clin. Med. 2021, 10, 786. https://doi.org/10.3390/jcm10040786
Rymarz A, Matyjek A, Sułek-Jakóbczyk M, Mosakowska M, Niemczyk S. Impaired Kidney Function Associated with Increased Risk of Side Effects in Patients with Small Vessel Vasculitis Treated with Rituximab as an Induction Therapy. Journal of Clinical Medicine. 2021; 10(4):786. https://doi.org/10.3390/jcm10040786
Chicago/Turabian StyleRymarz, Aleksandra, Anna Matyjek, Magdalena Sułek-Jakóbczyk, Magdalena Mosakowska, and Stanisław Niemczyk. 2021. "Impaired Kidney Function Associated with Increased Risk of Side Effects in Patients with Small Vessel Vasculitis Treated with Rituximab as an Induction Therapy" Journal of Clinical Medicine 10, no. 4: 786. https://doi.org/10.3390/jcm10040786
APA StyleRymarz, A., Matyjek, A., Sułek-Jakóbczyk, M., Mosakowska, M., & Niemczyk, S. (2021). Impaired Kidney Function Associated with Increased Risk of Side Effects in Patients with Small Vessel Vasculitis Treated with Rituximab as an Induction Therapy. Journal of Clinical Medicine, 10(4), 786. https://doi.org/10.3390/jcm10040786








