Usefulness of Cone-Beam CT-Based Liver Perfusion Mapping for Evaluating the Response of Hepatocellular Carcinoma to Conventional Transarterial Chemoembolization
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection and Study Design
2.2. TACE Technique and Post-Procession of CBCT Data
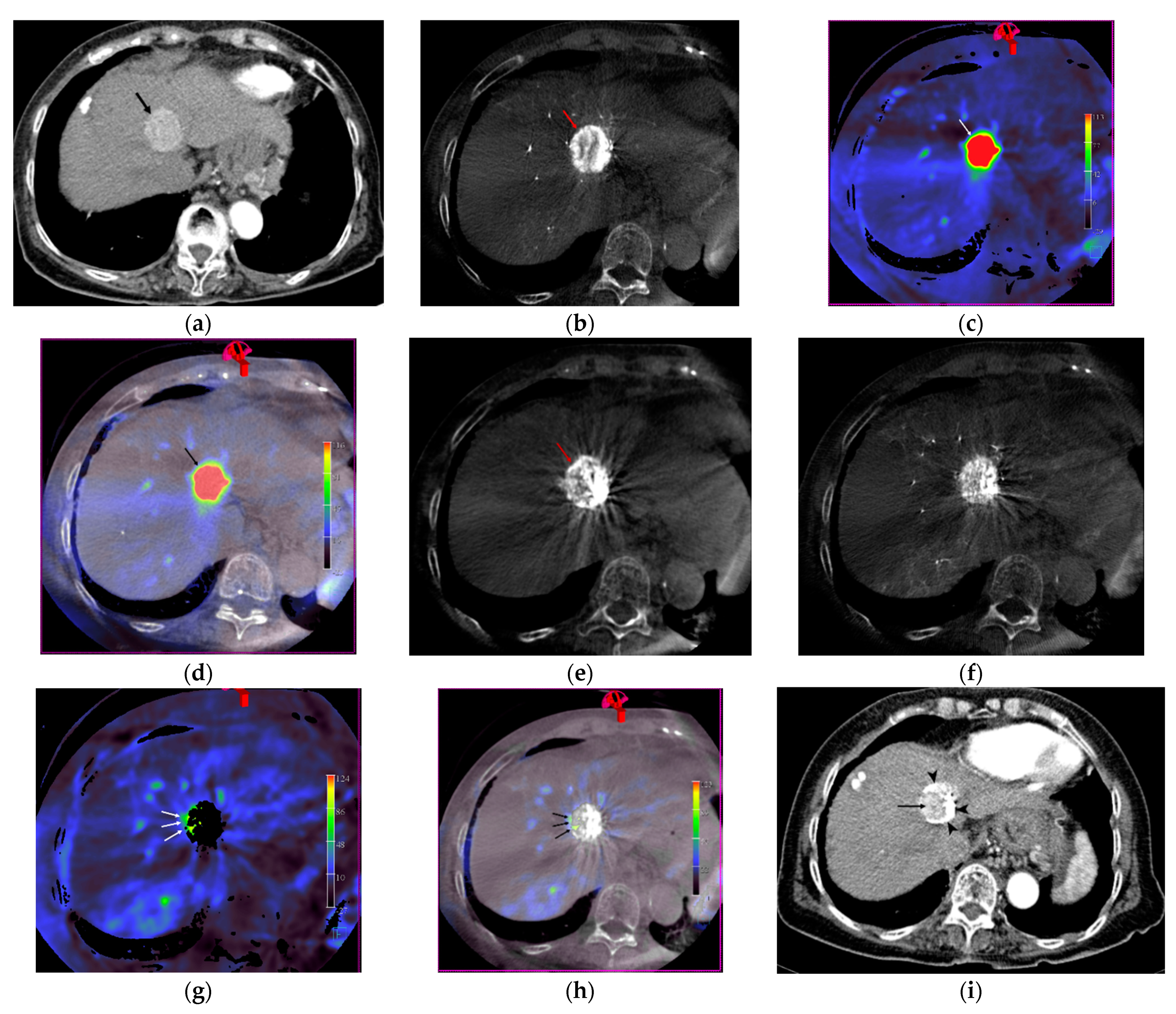
2.3. Image Analysis
2.4. Statistical Analysis
3. Results
3.1. Patient Demographics and Treatment Response
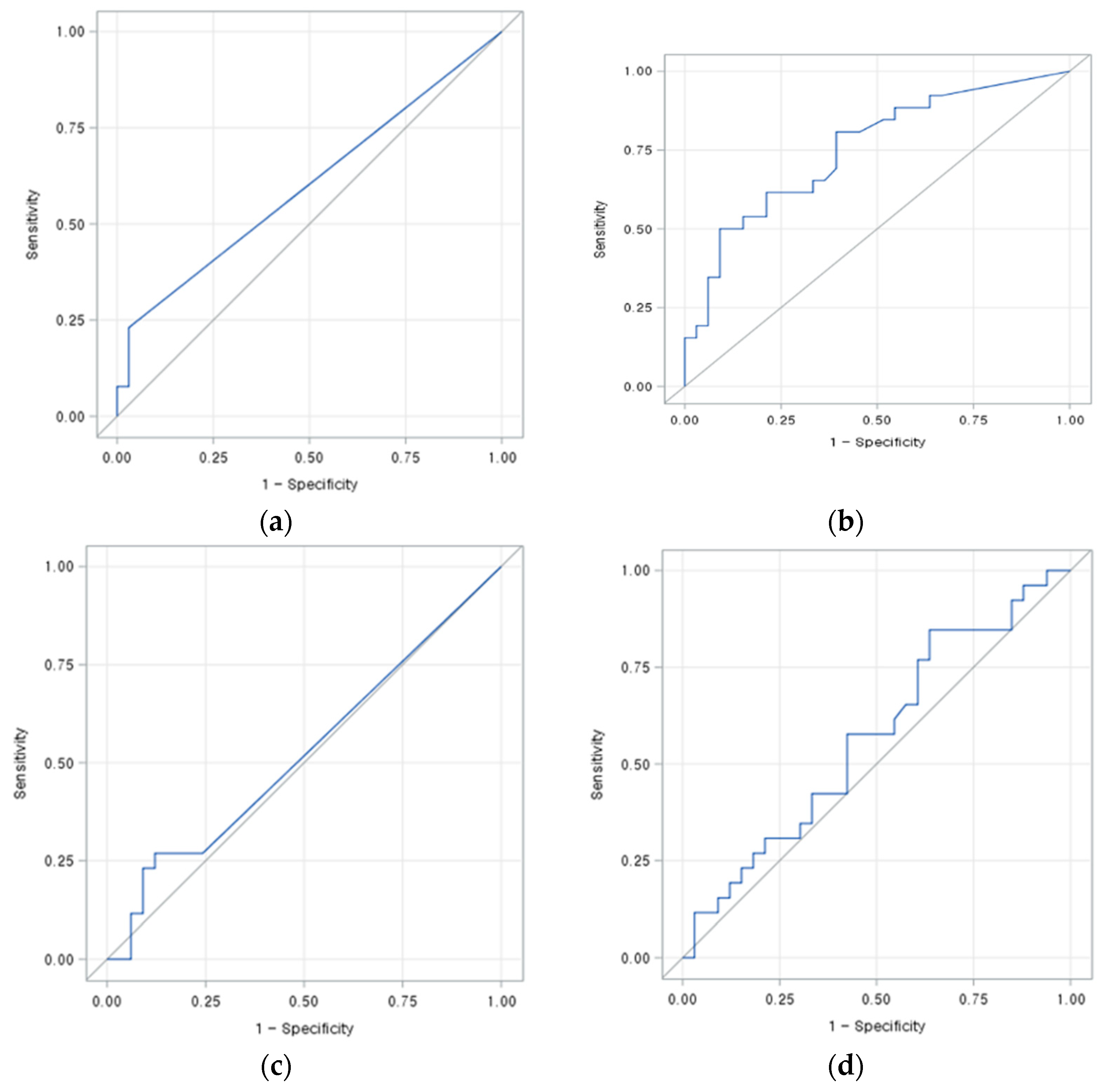
3.2. Diagnostic Performance of Treatment Response According to Imaging Type
3.3. Ability to Predict Treatment Response According to Imaging Type and Perfusion Parameters
3.4. Quantitative Analysis of Perfusion Parameters
3.5. Diagnostic Performance of the Operator’s Judgment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Miles, K.A. Functional computed tomography in oncology. Eur. J. Cancer 2002, 38, 2079–2084. [Google Scholar] [CrossRef]
- Miles, K.A. Perfusion CT for the assessment of tumour vascularity: Which protocol? Br. J. Radiol. 2003, 76, S36–S42. [Google Scholar] [CrossRef]
- Kim, S.H.; Kamaya, A.; Willmann, J.K. CT perfusion of the liver: Principles and applications in oncology. Radiology 2014, 272, 322–344. [Google Scholar] [CrossRef]
- Spira, D.; Schulze, M.; Sauter, A.; Brodoefel, H.; Brechtel, K.; Claussen, C.; Horger, M. Volume perfusion-CT of the liver: Insights and applications. Eur. J. Radiol. 2012, 81, 1471–1478. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.A.; Kartalis, N.; Grigoriadis, A.; Loizou, L.; Stal, P.; Leidner, B.; Aspelin, P.; Brismar, T.B. Perfusion computed tomography for detection of hepatocellular carcinoma in patients with liver cirrhosis. Eur. Radiol. 2015, 25, 3123–3132. [Google Scholar] [CrossRef] [PubMed]
- European Association For The Study Of The Liver; European Organisation For Research And Treatment Of Cancer. EASL-EORTC clinical practice guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2012, 56, 908–943. [Google Scholar] [CrossRef] [PubMed]
- Georgiades, C.; Geschwind, J.F.; Harrison, N.; Hines-Peralta, A.; Liapi, E.; Hong, K.; Wu, Z.; Kamel, I.; Frangakis, C. Lack of response after initial chemoembolization for hepatocellular carcinoma: Does it predict failure of subsequent treatment? Radiology 2012, 265, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.S.; Kim, J.H.; Yoon, E.L.; Lee, H.J.; Lee, S.J.; Suh, S.J.; Lee, B.J.; Seo, Y.S.; Yim, H.J.; Seo, T.S.; et al. Comparison of the methods for tumor response assessment in patients with hepatocellular carcinoma undergoing transarterial chemoembolization. J. Hepatol. 2013, 58, 1181–1187. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.K.; Kim, K.A.; Park, J.Y.; Ahn, S.H.; Chon, C.Y.; Han, K.H.; Kim, S.U.; Kim, M.J. Prospective comparison of prognostic values of modified Response Evaluation Criteria in Solid Tumours with European Association for the Study of the Liver criteria in hepatocellular carcinoma following chemoembolisation. Eur. J. Cancer 2013, 49, 826–834. [Google Scholar] [CrossRef]
- Kim, B.K.; Kim, S.U.; Kim, K.A.; Chung, Y.E.; Kim, M.J.; Park, M.S.; Park, J.Y.; Kim, D.Y.; Ahn, S.H.; Kim, M.D.; et al. Complete response at first chemoembolization is still the most robust predictor for favorable outcome in hepatocellular carcinoma. J. Hepatol. 2015, 62, 1304–1310. [Google Scholar] [CrossRef]
- Sieghart, W.; Hucke, F.; Peck-Radosavljevic, M. Transarterial chemoembolization: Modalities, indication, and patient selection. J. Hepatol. 2015, 62, 1187–1195. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Sherman, M.; American Association for the Study of Liver, D. Management of hepatocellular carcinoma: An update. Hepatology 2011, 53, 1020–1022. [Google Scholar] [CrossRef]
- Tacher, V.; Radaelli, A.; Lin, M.; Geschwind, J.F. How I do it: Cone-beam CT during transarterial chemoembolization for liver cancer. Radiology 2015, 274, 320–334. [Google Scholar] [CrossRef]
- Takayasu, K.; Arii, S.; Matsuo, N.; Yoshikawa, M.; Ryu, M.; Takasaki, K.; Sato, M.; Yamanaka, N.; Shimamura, Y.; Ohto, M. Comparison of CT findings with resected specimens after chemoembolization with iodized oil for hepatocellular carcinoma. AJR Am. J. Roentgenol. 2000, 175, 699–704. [Google Scholar] [CrossRef]
- Chiu, R.Y.; Yap, W.W.; Patel, R.; Liu, D.; Klass, D.; Harris, A.C. Hepatocellular Carcinoma Post Embolotherapy: Imaging Appearances and Pitfalls on Computed Tomography and Magnetic Resonance Imaging. Can. Assoc. Radiol. J. 2016, 67, 158–172. [Google Scholar] [CrossRef]
- Kim, K.A.; Choi, S.Y.; Kim, M.U.; Baek, S.Y.; Park, S.H.; Yoo, K.; Kim, T.H.; Kim, H.Y. The Efficacy of Cone-Beam CT-Based Liver Perfusion Mapping to Predict Initial Response of Hepatocellular Carcinoma to Transarterial Chemoembolization. J. Vasc. Interv. Radiol. 2019, 30, 358–369. [Google Scholar] [CrossRef] [PubMed]
- Datta, S.; Muller, K.; Moore, T.; Molvin, L.; Gehrisch, S.; Rosenberg, J.; Saenz, Y.; Manhart, M.; Deuerling-Zheng, Y.; Kothary, N.; et al. Dynamic Measurement of Arterial Liver Perfusion With an Interventional C-Arm System. Investig. Radiol. 2017, 52, 456–461. [Google Scholar] [CrossRef]
- Bley, T.; Strother, C.M.; Pulfer, K.; Royalty, K.; Zellerhoff, M.; Deuerling-Zheng, Y.; Bender, F.; Consigny, D.; Yasuda, R.; Niemann, D. C-arm CT measurement of cerebral blood volume in ischemic stroke: An experimental study in canines. AJNR Am. J. Neuroradiol. 2010, 31, 536–540. [Google Scholar] [CrossRef] [PubMed]
- Syha, R.; Grozinger, G.; Grosse, U.; Maurer, M.; Zender, L.; Horger, M.; Nikolaou, K.; Ketelsen, D. Parenchymal Blood Volume Assessed by C-Arm-Based Computed Tomography in Immediate Posttreatment Evaluation of Drug-Eluting Bead Transarterial Chemoembolization in Hepatocellular Carcinoma. Investig. Radiol. 2016, 51, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Niu, K.; Yang, P.; Wu, Y.; Struffert, T.; Doerfler, A.; Schafer, S.; Royalty, K.; Strother, C.; Chen, G.H. C-Arm Conebeam CT Perfusion Imaging in the Angiographic Suite: A Comparison with Multidetector CT Perfusion Imaging. AJNR Am. J. Neuroradiol. 2016, 37, 1303–1309. [Google Scholar] [CrossRef] [PubMed]
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Gaba, R.C.; Lewandowski, R.J.; Hickey, R.; Baerlocher, M.O.; Cohen, E.I.; Dariushnia, S.R.; Janne d’Othee, B.; Padia, S.A.; Salem, R.; Wang, D.S.; et al. Transcatheter Therapy for Hepatic Malignancy: Standardization of Terminology and Reporting Criteria. J. Vasc. Interv. Radiol. 2016, 27, 457–473. [Google Scholar] [CrossRef] [PubMed]
- Loffroy, R.; Lin, M.; Yenokyan, G.; Rao, P.P.; Bhagat, N.; Noordhoek, N.; Radaelli, A.; Blijd, J.; Liapi, E.; Geschwind, J.F. Intraprocedural C-arm dual-phase cone-beam CT: Can it be used to predict short-term response to TACE with drug-eluting beads in patients with hepatocellular carcinoma? Radiology 2013, 266, 636–648. [Google Scholar] [CrossRef] [PubMed]

| Clinical Characteristics | Value | |
|---|---|---|
| Sex | Man | 31 |
| Woman | 4 | |
| Age, median (range)(year) | 66 (45–85) | |
| Cause of liver cirrhosis | HBV | 20 |
| HCV | 4 | |
| Alcohol | 4 | |
| HBV + alcohol | 2 | |
| NBNC | 5 | |
| Child-turotte-pugh class | A | 29 |
| B | 6 | |
| ECOG performance status | 0 | 33 |
| 1 | 2 | |
| BCLC stage | 0 | 10 |
| A | 19 | |
| B | 6 | |
| Previous treatment of HCC | No treatment | 15 |
| RFA | 1 | |
| TACE | 18 | |
| TACE + RFA | 1 | |
| Tumor multiplicity | Single | 21 |
| Multiple, two nodules | 5 | |
| three nodules | 8 | |
| four nodules | 1 | |
| HCC diagnosis modality | CT | 17 |
| MRI | 18 | |
| Follow-up examination | CT | 23 |
| MRI | 10 | |
| Pathology | 1 | |
| Reviewer | Modality | TACE Result (Treatment Response) | * Response Outcome (n = 59) | |||
|---|---|---|---|---|---|---|
| CR | PR | SD | PD | |||
| R1 | L-CBCT | 0 (S) | 19 | 10 | 3 | 1 |
| 1 (S) | 10 | 6 | 2 | 0 | ||
| 2 (F) | 3 | 5 | 0 | 0 | ||
| 3 (F) | 0 | 0 | 0 | 0 | ||
| CE-CBCT | 0 (S) | 11 | 6 | 0 | 0 | |
| 1 (S) | 17 | 12 | 4 | 0 | ||
| 2 (F) | 4 | 3 | 1 | 1 | ||
| 3 (F) | 0 | 0 | 0 | 0 | ||
| CBCT-based-liver-perfusion-mapping | 0 (S) | 11 | 6 | 0 | 0 | |
| 1 (S) | 7 | 6 | 0 | 0 | ||
| 2 (F) | 9 | 4 | 5 | 0 | ||
| 3 (F) | 5 | 5 | 0 | 1 | ||
| R2 | L-CBCT | 0 (S) | 8 | 6 | 1 | 0 |
| 1 (S) | 21 | 13 | 4 | 1 | ||
| 2 (F) | 3 | 2 | 0 | 0 | ||
| 3 (F) | 0 | 0 | 0 | 0 | ||
| CE-CBCT | 0 (S) | 7 | 5 | 0 | 0 | |
| 1 (S) | 22 | 13 | 5 | 0 | ||
| 2 (F) | 3 | 3 | 0 | 1 | ||
| 3 (F) | 0 | 0 | 0 | 0 | ||
| CBCT-based-liver-perfusion-mapping | 0 (S) | 6 | 6 | 0 | 0 | |
| 1 (S) | 17 | 8 | 0 | 0 | ||
| 2 (F) | 4 | 2 | 5 | 0 | ||
| 3 (F) | 5 | 5 | 0 | 1 | ||
| R3 | L-CBCT | 0 (S) | 5 | 3 | 1 | 1 |
| 1 (S) | 13 | 8 | 3 | 0 | ||
| 2 (F) | 12 | 9 | 1 | 0 | ||
| 3 (F) | 2 | 1 | 0 | 0 | ||
| CE-CBCT | 0 (S) | 4 | 4 | 1 | 0 | |
| 1 (S) | 12 | 7 | 1 | 1 | ||
| 2 (F) | 13 | 7 | 3 | 0 | ||
| 3 (F) | 3 | 3 | 0 | 0 | ||
| CBCT-based-liver-perfusion-mapping | 0 (S) | 9 | 8 | 1 | 0 | |
| 1 (S) | 11 | 4 | 0 | 0 | ||
| 2 (F) | 4 | 4 | 3 | 0 | ||
| 3 (F) | 8 | 5 | 1 | 1 | ||
| Modality | Reviewer | Sensitivity (95%CI) | Specificity (95%CI) | PPV (95%CI) | NPV (95%CI) | Accuracy (95%CI) |
|---|---|---|---|---|---|---|
| L-CBCT | R1 | 25.93 (9.4–42.46) | 96.88 (90.86–100) | 87.5 (64.58–100) | 60.78 (47.38–74.18) | 64.41 (52.19–76.62) |
| R2 | 14.81 (1.41–28.21) | 96.88 (90.86–100) | 80 (44.94–100) | 57.41 (44.22–70.6) | 59.32 (46.79–71.86) | |
| R3 | 44.44 (25.7–63.19) | 59.38 (42.36–76.39) | 48 (28.42–67.58) | 55.88 (39.19–72.57) | 52.54 (39.8–65.28) | |
| CE-CBCT | R1 | 25.93 (9.4–42.46) | 93.75 (85.36–100) | 77.78 (50.62–100) | 60 (46.42–73.58) | 62.71 (50.37–75.05) |
| R2 | 25.93 (6.72–42.46) | 100 (100–100) | 100 (100–100) | 61.54 (48.32–74.76) | 66.1 (54.02–78.18) | |
| R3 | 55.56 (36.82–74.3) | 56.25 (39.06–73.44) | 51.72 (33.53–69.91) | 60 (42.47–77.53) | 55.93 (43.26–68.6) | |
| CBCT-based-liver-perfusion mapping | R1 | 96.3 (89.18–100) | 90.63 (80.53–100) | 89.66 (78.58–100) | 96.67 (90.25–100) | 93.22 (86.81–99.64) |
| R2 | 81.48 (47.43–96.13) | 100 (100–100) | 100 (100–100) | 86.49 (75.48–97.5) | 91.53 (84.42–98.63) | |
| R3 | 85.19 (71.79–98.59) | 90.63 (80.53–100) | 88.46 (76.18–100) | 87.88 (76.74–99.02) | 88.14 (79.88–96.39) |
| Modality | Reviewer | Sensitivity (95%CI) | Specificity (95%CI) | PPV (95%CI) | NPV (95%CI) | Accuracy (95%CI) |
|---|---|---|---|---|---|---|
| L-CBCT | R1 | 64.44 (48.78–78.13) | 62.5 (24.49–91.48) | 90.62 (74.98–98.02) | 23.81 (8.22–47.17) | 64.15 (49.8–76.86) |
| R2 | 60.42 (45.27–74.23) | 40 (5.27–85.34) | 90.62 (74.98–98.02) | 9.52 (1.17–30.38) | 58.49 (44.13–71.86) | |
| R3 | 62.07 (42.26–79.31) | 41.67 (22.11–63.36) | 56.25 (37.66–73.64) | 47.62 (25.71–70.22) | 52.83 (38.64–66.7) | |
| CE-CBCT | R1 | 58.7 (43.23–73) | 42.86 (9.9–81.59) | 87.1 (70.17–96.37) | 13.64 (2.91–34.91) | 56.6 (42.28–70.16) |
| R2 | 61.7 (46.38–75.49) | 50 (11.81–88.19) | 90.62 (74.98–98.02) | 14.29 (3.05–36.34) | 60.38 (46–73.55) | |
| R3 | 59.26 (38.8–77.61) | 38.46 (20.23–59.43) | 50 (31.89–68.11) | 47.62 (25.71–70.22) | 49.06 (35.06–63.16) | |
| CBCT-based liver-perfusion mapping | R1 | 60 (40.6–77.34) | 39.13 (19.71–61.46) | 56.25 (37.66–73.64) | 42.86 (21.82–65.98) | 50.94 (36.84–64.94) |
| R2 | 62.16 (44.76–77.54) | 43.75 (19.75–70.12) | 71.88 (53.25–86.25) | 33.33 (14.59–56.97) | 56.6 (42.28–70.16) | |
| R3 | 62.5 (43.69–78.9) | 42.86 (21.82–65.98) | 62.5 (43.69–78.9) | 42.86 (21.82–65.98) | 54.72 (40.45–68.44) |
| Model | Odds Ratio (95% CI) | p Value | C-Statistics | |
|---|---|---|---|---|
| 1 | Unenhanced cone beam CT | 1.221 (0.389–3.835) | 0.7323 | 0.726 |
| 2 | Contreast-enhanced cone-beam CT | 1.632 (0.516–5.161) | 0.4047 | 0.73 |
| 3 | Cone-beam CT-based-liver-perfusion-mapping | 72.661 (10.257–514.756) | <0.0001 | 0.954 |
| 4 | PBVmean of tumor | 1.032 (0.984–1.081) | 0.1964 | 0.7593 |
| 5 | PBVmax of tumor | 1.012 (1.000–1.023) | 0.0488 | 0.7844 |
| 6 | PBVmean of liver | 1.014 (0.971–1.060) | 0.5253 | 0.7372 |
| 7 | PBVmax of liver | 1.001 (0.997–1.005) | 0.6958 | 0.7401 |
| Region of Interest | Perfusion Factor | Before TACE (mL/L) | After TACE (mL/L) | p Value |
|---|---|---|---|---|
| Tumor | PBVmean | 113.09 ± 56.42 | 7.02 ± 26.5 | <0.0001 |
| PBVmax | 195.32 ± 84.77 | 57.27 ± 77.91 | <0.0001 | |
| Liver | PBVmean | 26.04 ± 21.82 | 6.67 ± 13.69 | <0.0001 |
| PBVmax | 273.92 ± 150.35 | 204.32 ± 140.1 | 0.0002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, S.Y.; Kim, K.A.; Choi, W.; Kwon, Y.; Cho, S.B. Usefulness of Cone-Beam CT-Based Liver Perfusion Mapping for Evaluating the Response of Hepatocellular Carcinoma to Conventional Transarterial Chemoembolization. J. Clin. Med. 2021, 10, 713. https://doi.org/10.3390/jcm10040713
Choi SY, Kim KA, Choi W, Kwon Y, Cho SB. Usefulness of Cone-Beam CT-Based Liver Perfusion Mapping for Evaluating the Response of Hepatocellular Carcinoma to Conventional Transarterial Chemoembolization. Journal of Clinical Medicine. 2021; 10(4):713. https://doi.org/10.3390/jcm10040713
Chicago/Turabian StyleChoi, Sun Young, Kyung Ah Kim, Woosun Choi, Yohan Kwon, and Soo Buem Cho. 2021. "Usefulness of Cone-Beam CT-Based Liver Perfusion Mapping for Evaluating the Response of Hepatocellular Carcinoma to Conventional Transarterial Chemoembolization" Journal of Clinical Medicine 10, no. 4: 713. https://doi.org/10.3390/jcm10040713
APA StyleChoi, S. Y., Kim, K. A., Choi, W., Kwon, Y., & Cho, S. B. (2021). Usefulness of Cone-Beam CT-Based Liver Perfusion Mapping for Evaluating the Response of Hepatocellular Carcinoma to Conventional Transarterial Chemoembolization. Journal of Clinical Medicine, 10(4), 713. https://doi.org/10.3390/jcm10040713






