Meta-Analysis on the Effect of Contingency Management for Patients with Both Psychotic Disorders and Substance Use Disorders
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Selection Criteria
2.2. Outcome Measures
2.3. Data Analysis
3. Results
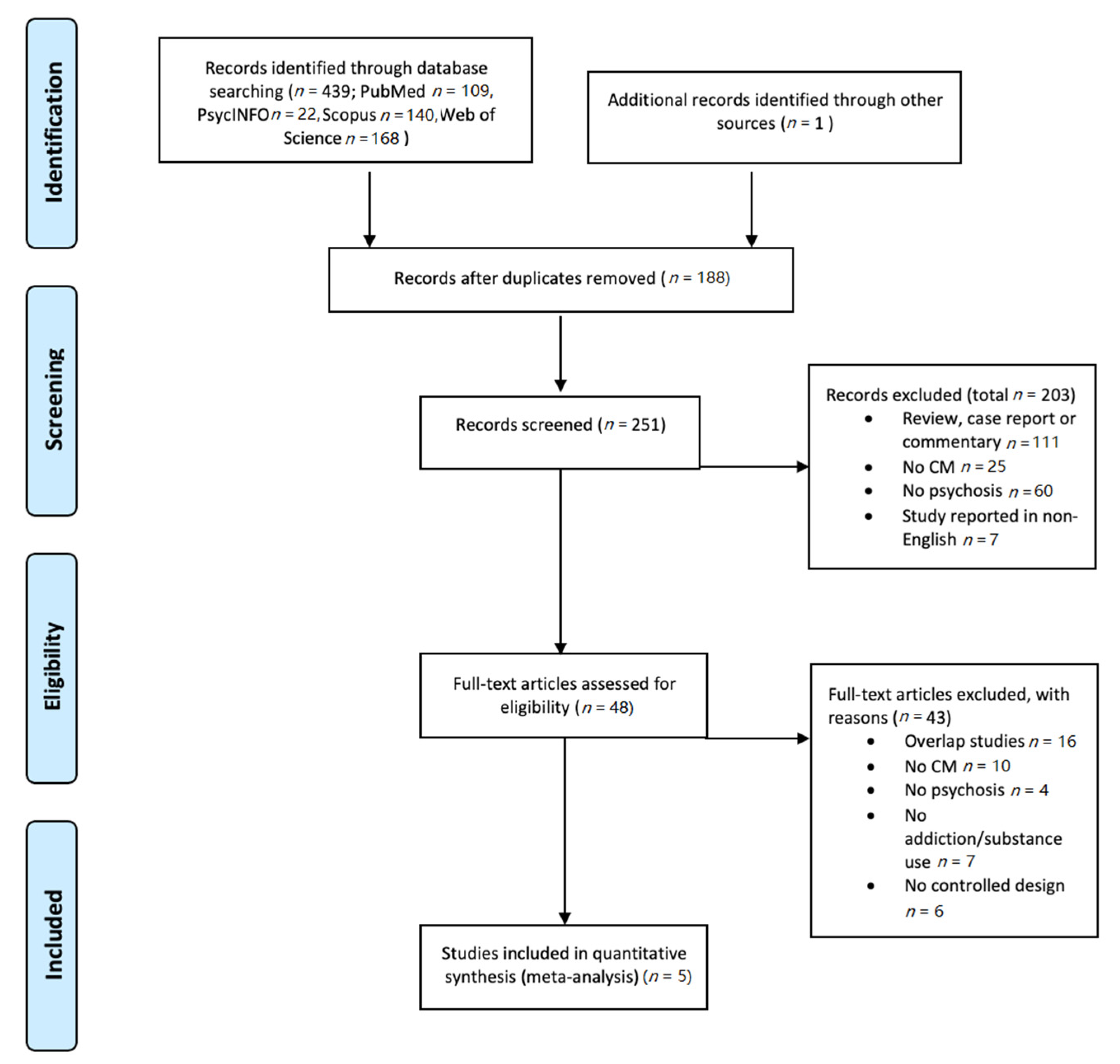
3.1. Study Selection
3.2. Demographic
3.3. Effectivity of CM
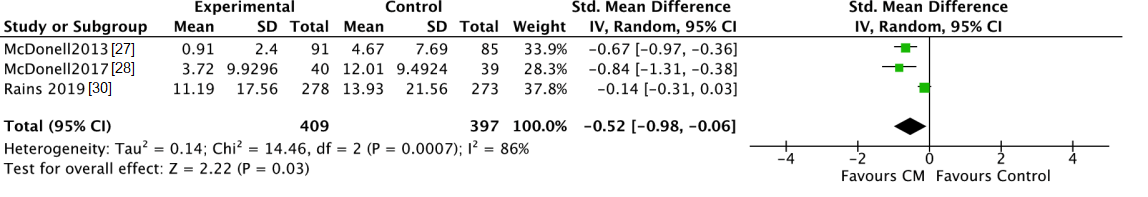
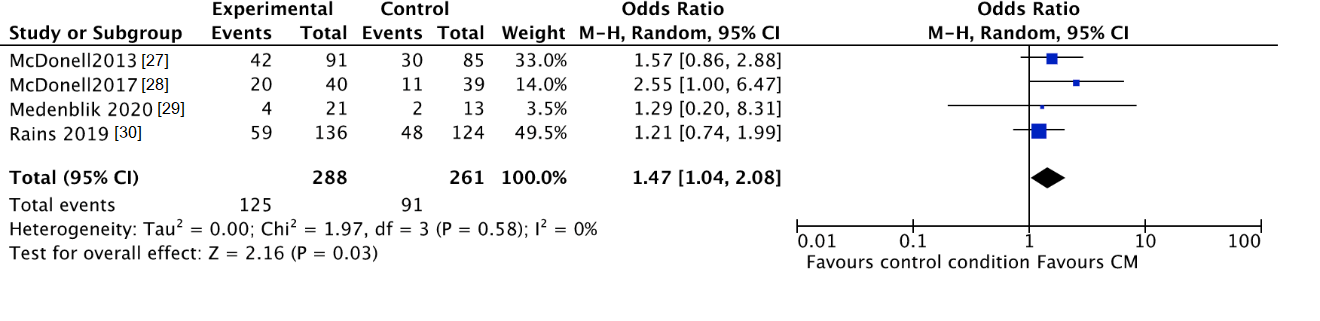
3.3.1. Self-Reported Substance Use
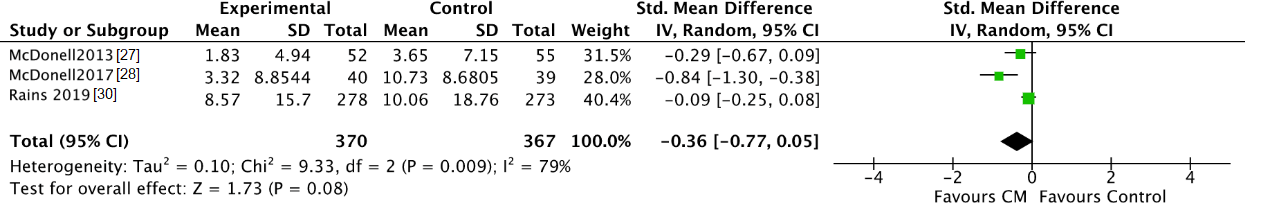
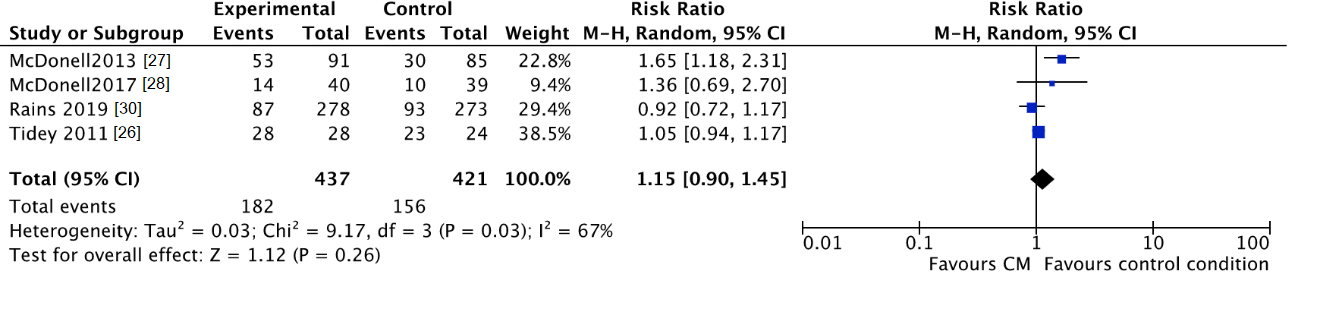
3.3.2. Biochemically Verified Abstinence
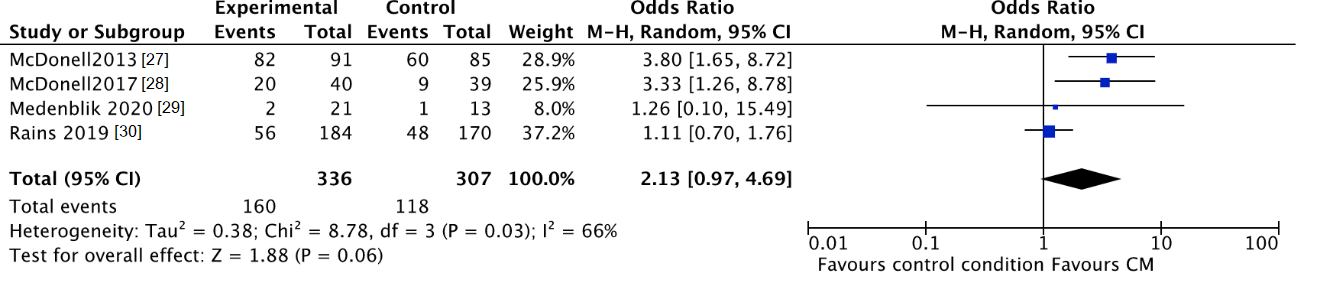
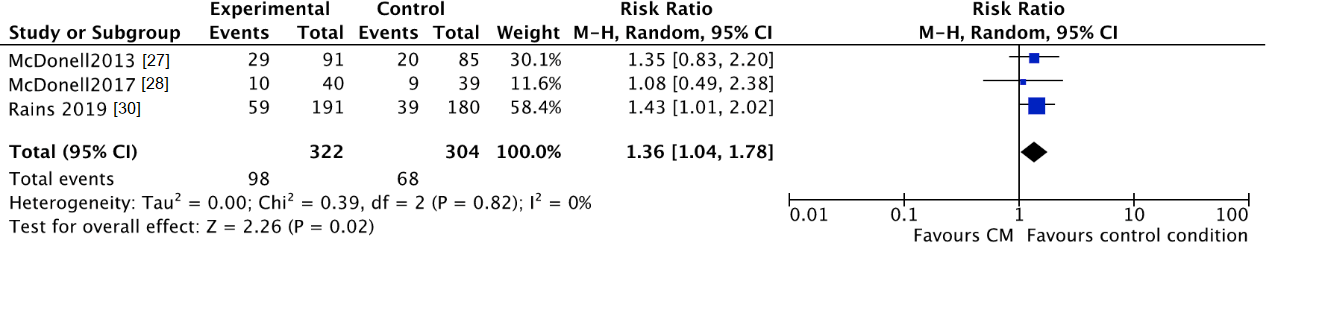
3.3.3. Lost to Treatment and Lost to Follow-Up
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Bennett, M.E.; Bellack, A.S.; Brown, C.H.; DiClemente, C. Substance dependence and remission in schizophrenia: A comparison of schizophrenia and affective disorders. Addict. Behav. 2009, 34, 806–814. [Google Scholar] [CrossRef][Green Version]
- Margolese, H.C.; Malchy, L.; Negrete, J.C.; Tempier, R.; Gill, K. Drug and alcohol use among patients with schizophrenia and related psychoses: Levels and consequences. Schizophr. Res. 2004, 67, 157–166. [Google Scholar] [CrossRef]
- Xie, H.; McHugo, G.J.; Fox, M.B.; Drake, R.E. Substance abuse relapse in a ten-year prospective follow-up of clients with mental and substance use disorders. Psychiatr. Serv. 2005, 56, 1282–1287. [Google Scholar] [CrossRef]
- Schmidt, L.M.; Hesse, M.; Lykke, J. The impact of substance use disorders on the course of schizophrenia--a 15-year follow-up study: Dual diagnosis over 15 years. Schizophr. Res. 2011, 130, 228–233. [Google Scholar] [CrossRef]
- Fazel, S.; Grann, M.; Carlström, E.; Lichtenstein, P.; Långström, N. Risk factors for violent crime in Schizophrenia: A national cohort study of 13,806 patients. J. Clin. Psychiatry 2009, 70, 362–369. [Google Scholar] [CrossRef]
- Soyka, M. Substance misuse, psychiatric disorder and violent and disturbed behaviour. Br. J. Psychiatry 2000, 176, 345–350. [Google Scholar] [CrossRef]
- Suokas, J.T.; Perälä, J.; Suominen, K.; Saarni, S.; Lönnqvist, J.; Suvisaari, J.M. Epidemiology of suicide attempts among persons with psychotic disorder in the general population. Schizophr. Res. 2010, 124, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Hunt, G.E.; Large, M.M.; Cleary, M.; Lai, H.M.X.; Saunders, J.B. Prevalence of comorbid substance use in schizophrenia spectrum disorders in community and clinical settings, 1990–2017: Systematic review and meta-analysis. Drug Alcohol Depend. 2018, 191, 234–258. [Google Scholar] [CrossRef] [PubMed]
- Willet, S.; Alderson, P.; Sparrow, K.; Moreea, O. Surveillance report 2016–Coexisting severe mental illness (psychosis) and substance misuse: Assessment and management in healthcare settings (2011) NICE guideline CG120 [Internet], National Institute for Health and Care Excellence, London, UK.
- Higgins, S.T.; Kurti, A.N.; Davis, D.R. Voucher-based contingency management is efficacious but underutilized in treating addictions. Perspect. Behav. Sci. 2019, 42, 501–524. [Google Scholar] [CrossRef] [PubMed]
- Brown, H.D.; DeFulio, A. Contingency management for the treatment of methamphetamine use disorder: A systematic review. Drug Alcohol Depend. 2020, 216, 108307. [Google Scholar] [CrossRef]
- Ellis, J.D.; Struble, C.A.; Fodor, M.C.; Cairncross, M.; Lundahl, L.H.; Ledgerwood, D.M. Contingency management for individuals with chronic health conditions: A systematic review and meta-analysis of randomized controlled trials. Behav. Res. Ther. 2020, 136, 103781. [Google Scholar] [CrossRef] [PubMed]
- Petry, N.M. Contingency Management for Substance Abuse Treatment: A Guide to Implementing This Evidence-Based Practice; Routledge/Taylor & Francis Group: Abingdon, UK, 2012. [Google Scholar]
- Benishek, L.A.; Dugosh, K.L.; Kirby, K.C.; Matejkowski, J.; Clements, N.T.; Seymour, B.L.; Festinger, D.S. Prize-based contingency management for the treatment of substance abusers: A meta-analysis. Addiction 2014, 109, 1426–1436. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.L.; Powers, M.B.; Handelsman, P.; Medina, J.L.; Zvolensky, M.; Smits, J.A. Behavioral therapies for treatment-seeking cannabis users: A meta-analysis of randomized controlled trials. Eval. Health Prof. 2015, 38, 94–114. [Google Scholar] [CrossRef]
- De Crescenzo, F.; Ciabattini, M.; D’Alò, G.L.; De Giorgi, R.; Del Giovane, C.; Cassar, C.; Janiri, L.; Clark, N.; Ostacher, M.J.; Cipriani, A. Comparative efficacy and acceptability of psychosocial interventions for individuals with cocaine and amphetamine addiction: A systematic review and network meta-analysis. PLoS Med. 2018, 15, e1002715. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Cao, J.; Guo, T.; Xiao, Y. Association between alcoholic interventions and abstinence rates for alcohol use disorders: A meta-analysis. Medicine 2018, 97, e13566. [Google Scholar] [CrossRef] [PubMed]
- Prendergast, M.; Podus, D.; Finney, J.; Greenwell, L.; Roll, J. Contingency management for treatment of substance use disorders: A meta-analysis. Addiction 2006, 101, 1546–1560. [Google Scholar] [CrossRef]
- Dutra, L.; Stathopoulou, G.; Basden, S.L.; Leyro, T.M.; Powers, M.B.; Otto, M.W. A meta-analytic review of psychosocial interventions for substance use disorders. Am. J. Psychiatry 2008, 165, 179–187. [Google Scholar] [CrossRef]
- Petry, N.M.; Alessi, S.M.; Olmstead, T.A.; Rash, C.J.; Zajac, K. Contingency management treatment for substance use disorders: How far has it come, and where does it need to go? Psychol. Addict. Behav. 2017, 31, 897–906. [Google Scholar] [CrossRef]
- Hunt, G.E.; Siegfried, N.; Morley, K.; Brooke-Sumner, C.; Cleary, M. Psychosocial interventions for people with both severe mental illness and substance misuse. Cochrane Database Syst. Rev. 2019, 12, CD001088. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Wang, W.; Liu, J.; Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 2014, 14, 135. [Google Scholar] [CrossRef] [PubMed]
- Riley, R.D.; Higgins, J.P.; Deeks, J.J. Interpretation of random effects meta-analyses. BMJ 2011, 342, d549. [Google Scholar] [CrossRef]
- Tidey, J.W.; Rohsenow, D.J.; Kaplan, G.B.; Swift, R.M.; Reid, N. Effects of contingency management and bupropion on cigarette smoking in smokers with schizophrenia. Psychopharmacology 2011, 217, 279–287. [Google Scholar] [CrossRef][Green Version]
- McDonell, M.G.; Srebnik, D.; Angelo, F.; McPherson, S.; Lowe, J.M.; Sugar, A.; Short, R.A.; Roll, J.M.; Ries, R.K. Randomized controlled trial of contingency management for stimulant use in community mental health patients with serious mental illness. Am. J. Psychiatry 2013, 170, 94–101. [Google Scholar] [CrossRef]
- McDonell, M.G.; Leickly, E.; McPherson, S.; Skalisky, J.; Srebnik, D.; Angelo, F.; Vilardaga, R.; Nepom, J.R.; Roll, J.M.; Ries, R.K. A Randomized Controlled Trial of Ethyl Glucuronide-Based Contingency Management for Outpatients with Co-Occurring Alcohol Use Disorders and Serious Mental Illness. Am. J. Psychiatry 2017, 174, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Medenblik, A.M.; Mann, A.M.; Beaver, T.A.; Dedert, E.A.; Wilson, S.M.; Calhoun, P.S.; Beckham, J.C. Treatment Outcomes of a Multi-Component Mobile Health Smoking Cessation Pilot Intervention for People with Schizophrenia. J. Dual Diagn. 2020, 16, 420–428. [Google Scholar] [CrossRef]
- Rains, L.S.; Marston, L.; Hinton, M.; Marwaha, S.; Craig, T.; Fowler, D.; King, M.; Omar, R.Z.; McCrone, P.; Spencer, J.; et al. Clinical and cost-effectiveness of contingency management for cannabis use in early psychosis: The CIRCLE randomised clinical trial. BMC Med. 2019, 17, 161. [Google Scholar]
- Jongsma, H.E.; Turner, C.; Kirkbride, J.B.; Jones, P.B. International incidence of psychotic disorders, 2002–2017: A systematic review and meta-analysis. Lancet Public Health 2019, 4, e229–e244. [Google Scholar] [CrossRef]
- Davis, D.R.; Kurti, A.N.; Skelly, J.M.; Redner, R.; White, T.J.; Higgins, S.T. A review of the literature on contingency management in the treatment of substance use disorders, 2009–2014. Prev. Med. 2016, 92, 36–46. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). Clinical Guideline on Drug Misuse in over 16’s: Psychosocial Interventions (CG51). National Institute for Health and Care Excellence (NICE): London, UK, 2007.
- Forster, S.E.; DePhillippis, D.; Forman, S.D. “I’s” on the prize: A systematic review of individual differences in Contingency Management treatment response. J. Subst. Abus. Treat. 2019, 100, 64–83. [Google Scholar] [CrossRef]
- Weinstock, J.; Alessi, S.M.; Petry, N.M. Regardless of psychiatric severity the addition of contingency management to standard treatment improves retention and drug use outcomes. Drug Alcohol Depend. 2007, 87, 288–296. [Google Scholar] [CrossRef]
- Lussier, J.P.; Heil, S.H.; Mongeon, J.A.; Badger, G.J.; Higgins, S.T. A meta-analysis of voucher-based reinforcement therapy for substance use disorders. Addiction 2006, 101, 192–203. [Google Scholar] [CrossRef]
- Rash, C.J.; DePhilippis, D. Considerations for Implementing Contingency Management in Substance Abuse Treatment Clinics: The Veterans Affairs Initiative as a Model. Perspect. Behav. Sci. 2019, 42, 479–499. [Google Scholar] [CrossRef]
- McPherson, S.M.; Burduli, E.; Smith, C.L.; Herron, J.; Oluwoye, O.; Hirchak, K.; Orr, M.F.; McDonell, M.G.; Roll, J.M. A review of contingency management for the treatment of substance-use disorders: Adaptation for underserved populations, use of experimental technologies, and personalized optimization strategies. Subst. Abus. Rehabilatation 2018, 9, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Horsfall, J.; Cleary, M.; Hunt, G.E.; Walter, G. Psychosocial treatments for people with co-occurring severe mental illnesses and substance use disorders (dual diagnosis): A review of empirical evidence. Harv. Rev. Psychiatry 2009, 17, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Brown, H.E.; Hart, K.L.; Snapper, L.A.; Roffman, J.L.; Perlis, R.H. Impairment in delay discounting in schizophrenia and schizoaffective disorder but not primary mood disorders. NPJ Schizophr. 2018, 4, 9. [Google Scholar] [CrossRef] [PubMed]
- Dom, G.; D’haene, P.; Hulstijn, W.; Sabbe, B. Impulsivity in abstinent early- and late-onset alcoholics: Differences in self-report measures and a discounting task. Addiction 2006, 101, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Kluwe-Schiavon, B.; Viola, T.W.; Sanvicente-Vieira, B.; Lumertz, F.S.; Salum, G.A.; Grassi-Oliveira, R.; Quednow, B.B. Substance related disorders are associated with impaired valuation of delayed gratification and feedback processing: A multilevel meta-analysis and meta-regression. Neurosci. Biobehav. Rev. 2020, 108, 295–307. [Google Scholar] [CrossRef]
- Regier, P.S.; Redish, A.D. Contingency Management and Deliberative Decision-Making Processes. Front. Psychiatry 2015, 6, 76. [Google Scholar] [CrossRef]
- Destoop, M.; Morrens, M.; Coppens, V.; Dom, G.; Addiction, Anhedonia, and Comorbid Mood Disorder. A Narrative Review. Front. Psychiatry 2019, 22, 311. [Google Scholar] [CrossRef]
- Frost, K.H.; Strauss, G.P. A Review of Anticipatory Pleasure in Schizophrenia. Curr. Behav. Neurosci. Rep. 2016, 3, 232–247. [Google Scholar] [CrossRef]
- Noordraven, E.L.; Audier, C.H.; Staring, A.B.; Wierdsma, A.I.; Blanken, P.; van der Hoorn, B.E.; Roijen, L.H.; Mulder, C.L. Money for medication: A randomized controlled study on the effectiveness of financial incentives to improve medication adherence in patients with psychotic disorders. BMC Psychiatry 2014, 14, 343. [Google Scholar] [CrossRef] [PubMed]
- Petry, N.M.; Alessi, S.M.; Rash, C.J.; Barry, D.; Carroll, K.M. A randomized trial of contingency management reinforcing attendance at treatment: Do duration and timing of reinforcement matter? J. Consult. Clin. Psychol. 2018, 86, 799–809. [Google Scholar] [CrossRef]
- Cooper, K.; Chatters, R.; Kaltenthaler, E.; Wong, R. Psychological and psychosocial interventions for cannabis cessation in adults: A systematic review short report. Health Technol. Assess. 2015, 19, 1–130. [Google Scholar] [CrossRef]
- Rains, S.L.; Steare, T.; Mason, O.; Johnson, S. Improving substance misuse outcomes in contingency management treatment with adjunctive formal psychotherapy: A systematic review and meta-analysis. BMJ Open 2020, 10, e034735. [Google Scholar] [CrossRef] [PubMed]
- González-Roz, A.; Weidberg, S.; García-Pérez, Á.; Martínez-Loredo, V.; Secades-Villa, R. One-year efficacy and incremental cost-effectiveness of contingency management for cigarette smokers with depression. Nicotine Tob. Res. 2021, 23, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Petry, N.M.; Alessi, S.M.; Carroll, K.M.; Hanson, T.; MacKinnon, S.; Rounsaville, B.; Sierra, S. Contingency management treatments: Reinforcing abstinence versus adherence with goal-related activities. J. Consult. Clin. Psychol. 2006, 74, 592–601. [Google Scholar] [CrossRef] [PubMed]
- Petry, N.M.; Weinstock, J.; Alessi, S.M.; Lewis, M.W.; Dieckhaus, K. Group-based randomized trial of contingencies for health and abstinence in HIV patients. J. Consult. Clin. Psychol. 2010, 78, 89–97. [Google Scholar] [CrossRef]
- Gagnon, M.; Guta, A.; Payne, A. “Setting people up for success and then failure"-health care and service providers’ experiences of using prize-based contingency management. Subst. Abus. Treat. Prev. Policy 2020, 15, 71. [Google Scholar] [CrossRef]
| Author | Randomisation | Intervention Bias | Missing Outcome Data | Measurement | Reporting Bias |
|---|---|---|---|---|---|
| McDonell 2013 [27] | Low risk | Low risk | Low risk | Low risk | Low risk |
| McDonell 2017 [28] | Low risk | Low risk | Low risk | Low risk | Low risk |
| Medenblik 2020 [29] | Some concerns | Low risk | Some concerns | Low risk | Some concerns |
| Rains 2019 [30] | Low risk | High risk | High risk | Low risk | Low risk |
| Tidey 2011 [26] | Low risk | Low risk | Low risk | Low risk | Low risk |
| Authors, Design | Sample Inclusion Criteria | Sample Exclusion Criteria | CM Intervention | Reward | Control Intervention | Follow-Up | Results |
|---|---|---|---|---|---|---|---|
| McDonell, 2013, RCT [27] | Have used stimulants during past 30 d, methamphetamine/amphetamine/cocaine dependence and schizophrenia/schizoaffective disorder/bipolar I or II/recurrent major depressive disorder | Organic brain disorder, dementia or medical disorders or psychiatric symptoms severe enough to compromise safe participation | CM: 3 m 3 x/w breath and urine samples, 1 opportunity for each negative sample and 1 additional for each week of continuous stimulant abstinence + TAU n = 91 | Prize draws from container of tokens respresenting magnitudes of reinforcement (50% “good job” and 50% tangible prize | 3 m Noncontingent reinforcement for each urine sample regardless result + TAU: psychoeducation + mental health care by case manager + psychiatric medication management + group treatment + housing and vocational services n = 85 | 3 m | CM group less likely to complete treatment period, more likely to submit a stimulant-negative urine test. CM group lower levels of alcohol use, injection drug use, fewer psychiatric symptoms, less likely to be admitted for psychiatric hospitalization. |
| McDonell, 2017, RCT [28] | Alcohol dependence and schizophrenia/schizoaffective disorder/bipolar I or II/recurrent major depressive disorder, alcohol use on >4d of the last 30d, enrollment in outpatient addiction group treatment | Comorbid drug dependence, medical or psychiatric severity that would compromise safe participation | CM: 12 w 3 x/w urine samples, 3 prize draws for each EtG-negative urine sample and 1 additional prize draw for each week of continuous alcohol abstinence + TAU n = 40 | Prize draws from container of tokens respresenting magnitudes of reinforcement (50% “good job” and 50% tangible prize | 12 w Noncontingent reinforcement for each urine sample regardless of EtG result + TAU: psychoeducation + mental health care by case manager + psychiatric medication management + group treatment + housing and vocational services n = 39 | 3 m | CM group had longest duration of alcohol abstinence, fewer days of drinking, fewer heavy drinking episodes and lower EtG levels. They were more likely to submit stimulant-negative urine and smoking-negative breath samples. |
| Medenblik, 2020, RCT pilot [29] | Smoked for at least 1 year, 18–70 y, at least 10 cig/d, sufficient English, willing to make a smoking cessation attempt, criteria for schizophrenia/schizoaffective disorder/psychotic disorder | History of AMI last 6 m, contraindication for NRT, unwilling to quit other forms of nicotine, pregnancy, criteria for current mania, current incarceration of inpatient hospitalization | iCOMMIT: 30 d behavioral therapy in the form of mCM + pharmacotherapy for smoking cessation + five sessions of cognitive-behavioral smoking cessation couseling n = 21 | Money checks for bioverification of abstinence through using phone app | ITC: 30 d pharmacotherapy for smoking cessation + five sessions of cognitive-behavioral smoking cessation couseling n = 13 | 6 m | No statistically significant difference between iCOMMIT and ITC groups. |
| Rains, 2019, RCT [30] | On an EIP service caseload, THC once in 12/24 w, 18–36 y, living in stable accommodation, sufficient English | Compulsory treatment or court | 12 w, weekly CM sessions with immediate reward with vouchers if negative urinalaysis for THC n = 278 | Variable reward schedule with £ 5 for every 2 clean samples | 12 w optimised TAU psychoeducational intervention recommended in EIP practice n = 273 | 18 m | No statistically significant difference in cannabis use, in engagement in work of education and positive psychotic symptoms. CM significant improvement in time to acute psychiatric admission |
| Tidey, 2011, RCT [26] | Schizophrenia/schizoaffective disorder, >18y, >20 cig/d, >6 FTND, stable on medication for >2 m, interested in quitting smoking | Pregnancy, positive breath alcohol level or urine drug toxicity test, medical condition contraindicating bupropion, very high psychiatric symptom severity | CM + BUP: 3w 3 x/w urine and CO samples + bupropion 150mg 3d and bupropion 2 × 150 mg 4–22 d n = 12 CM + PLA: 3 w 3 x/w urine and CO samples + placebo n = 16 | USD 25 for attending study sessions and increased by USD 5 for each abstinent sample | NRT + BUP: 3 w 25 dollar for attending study sessions and providing urine samples at each visit + bupropion 150 mg 3 d and bupropion 2 × 150 mg 4–22 d n = 11 NRT+ PLA: 3w USD 25 for attending study sessions and providing urine samples at each visit + placebo n = 13 | No | Significantly decreased cotinine and CO levels in CM group. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Destoop, M.; Docx, L.; Morrens, M.; Dom, G. Meta-Analysis on the Effect of Contingency Management for Patients with Both Psychotic Disorders and Substance Use Disorders. J. Clin. Med. 2021, 10, 616. https://doi.org/10.3390/jcm10040616
Destoop M, Docx L, Morrens M, Dom G. Meta-Analysis on the Effect of Contingency Management for Patients with Both Psychotic Disorders and Substance Use Disorders. Journal of Clinical Medicine. 2021; 10(4):616. https://doi.org/10.3390/jcm10040616
Chicago/Turabian StyleDestoop, Marianne, Lise Docx, Manuel Morrens, and Geert Dom. 2021. "Meta-Analysis on the Effect of Contingency Management for Patients with Both Psychotic Disorders and Substance Use Disorders" Journal of Clinical Medicine 10, no. 4: 616. https://doi.org/10.3390/jcm10040616
APA StyleDestoop, M., Docx, L., Morrens, M., & Dom, G. (2021). Meta-Analysis on the Effect of Contingency Management for Patients with Both Psychotic Disorders and Substance Use Disorders. Journal of Clinical Medicine, 10(4), 616. https://doi.org/10.3390/jcm10040616







