COVID-19 in Autoinflammatory Diseases with Immunosuppressive Treatment
Abstract
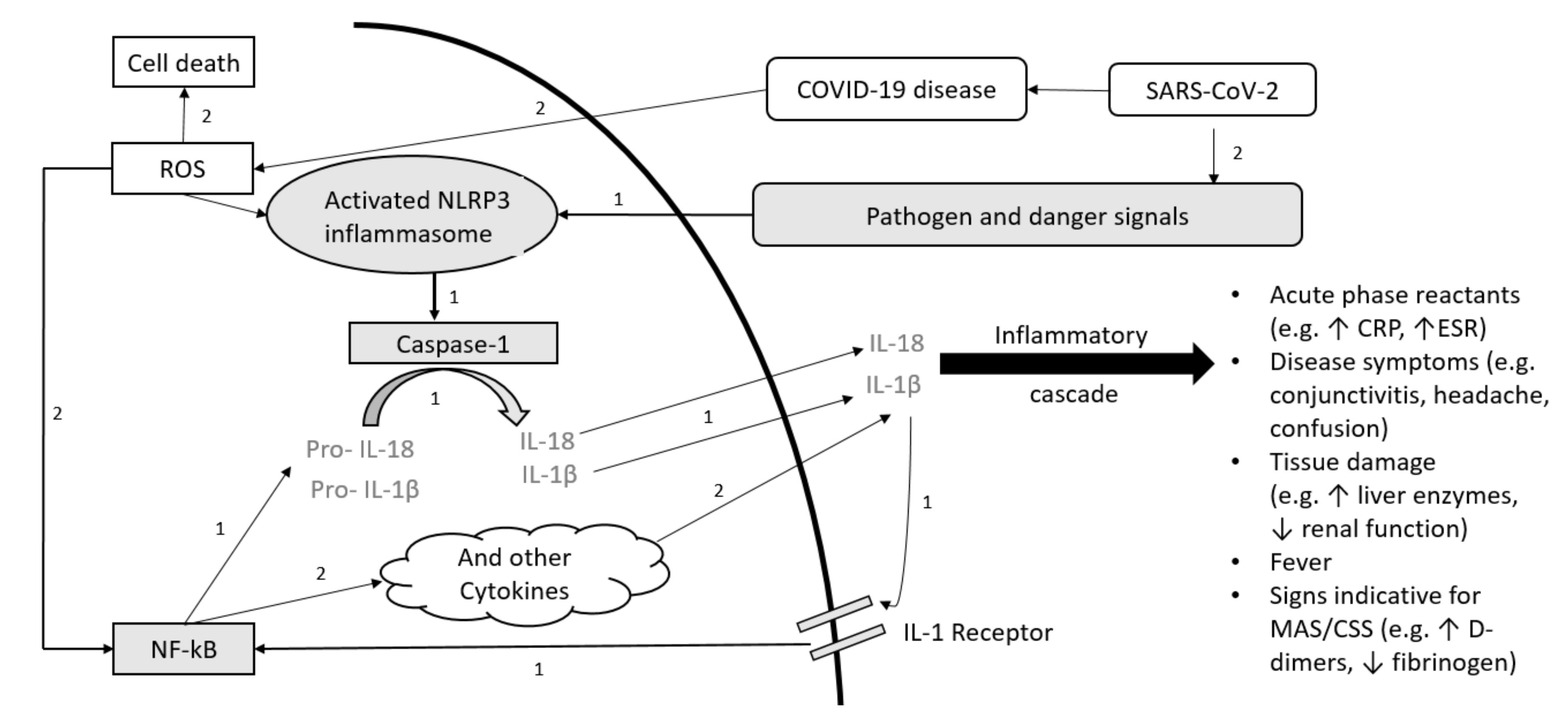
1. Introduction
2. Methods and Patients
2.1. Study Design
2.2. Patients
2.2.1. Patient 1
2.2.2. Patient 2
2.2.3. Patient 3
2.2.4. Patient 4
2.3. Monitoring and Follow-Up Visits
3. Results
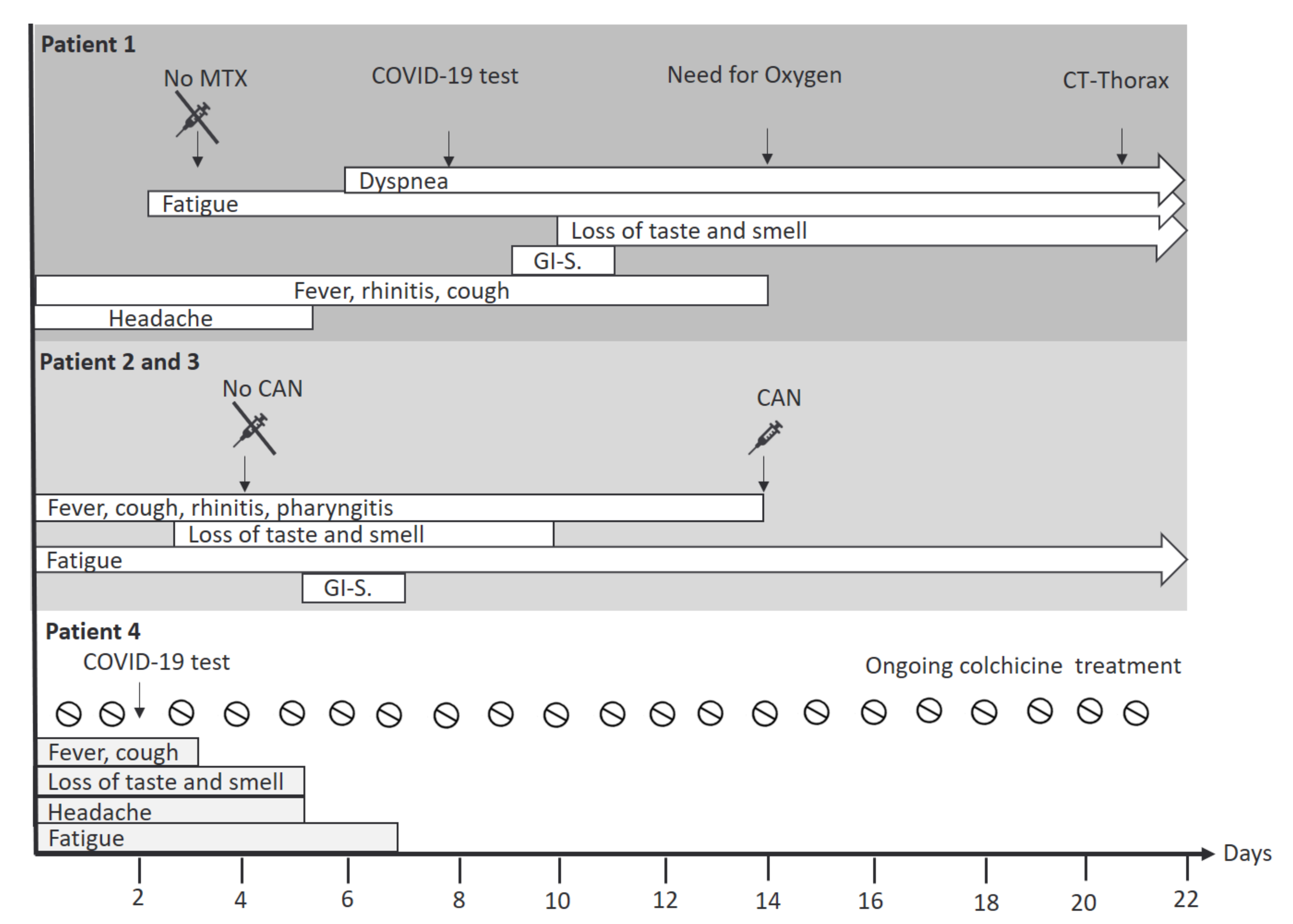
3.1. Acute COVID-19 Course
3.1.1. Patient 1
3.1.2. Patient 2 and 3
3.1.3. Patient 4
3.2. Follow-Up Visits
3.2.1. Patient 1
3.2.2. Patient 2
3.2.3. Patient 3
3.2.4. Patient 4
3.3. Nasopharyngeal COVID-19 Tests and Antibody Response to SARS-CoV-2
4. Discussion
4.1. COVID-19 Disease Course and AID
4.2. Antibody Response to SARS-CoV-2
4.3. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Broderick, L.; De Nardo, D.; Franklin, B.S.; Hoffman, H.M.; Latz, E. The inflammasomes and autoinflammatory syndromes. Annu. Rev. Pathol. 2015, 10, 395–424. [Google Scholar] [CrossRef]
- De Torre-Minguela, C.; Mesa Del Castillo, P.; Pelegrin, P. The NLRP3 and Pyrin Inflammasomes: Implications in the Pathophysiology of Autoinflammatory Diseases. Front. Immunol. 2017, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Ozen, S.; Demirkaya, E.; Erer, B.; Livneh, A.; Ben-Chetrit, E.; Giancane, G.; Ozdogan, H.; Abu, I.; Gattorno, M.; Hawkins, P.N.; et al. EULAR recommendations for the management of familial Mediterranean fever. Ann. Rheum. Dis. 2016, 75, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Ter Haar, N.M.; Oswald, M.; Jeyaratnam, J.; Anton, J.; Barron, K.S.; Brogan, P.A.; Cantarini, L.; Galeotti, C.; Grateau, G.; Hentgen, V.; et al. Recommendations for the management of autoinflammatory diseases. Ann. Rheum. Dis. 2015, 74, 1636–1644. [Google Scholar] [CrossRef] [PubMed]
- Henderson, L.A.; Canna, S.W.; Schulert, G.S.; Volpi, S.; Lee, P.Y.; Kernan, K.F.; Caricchio, R.; Mahmud, S.; Hazen, M.M.; Halyabar, O.; et al. On the alert for cytokine storm: Immunopathology in COVID-19. Arthritis Rheumatol. 2020, 72, 1059–1063. [Google Scholar] [CrossRef]
- Rigante, D.; Emmi, G.; Fastiggi, M.; Silvestri, E.; Cantarini, L. Macrophage activation syndrome in the course of monogenic autoinflammatory disorders. Clin. Rheumatol. 2015, 34, 1333–1339. [Google Scholar] [CrossRef]
- Horneff, G.; Rhouma, A.; Weber, C.; Lohse, P. Macrophage activation syndrome as the initial manifestation of tumour necrosis factor receptor 1-associated periodic syndrome (TRAPS). Clin. Exp. Rheumatol. 2013, 31, 99–102. [Google Scholar]
- Rigante, D.; Capoluongo, E.; Bertoni, B.; Ansuini, V.; Chiaretti, A.; Piastra, M.; Pulitano, S.; Genovese, O.; Compagnone, A.; Stabile, A. First report of macrophage activation syndrome in hyperimmunoglobulinemia D with periodic fever syndrome. Arthritis Rheumatol. 2007, 56, 658–661. [Google Scholar] [CrossRef]
- Lachmann, H.J. Periodic fever syndromes. Best Pract. Res. Clin. Rheumatol. 2017, 31, 596–609. [Google Scholar] [CrossRef]
- Conti, P.; Ronconi, G.; Caraffa, A.; Gallenga, C.E.; Ross, R.; Frydas, I.; Kritas, S.K. Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by Coronavirus-19 (COVI-19 or SARS-CoV-2): Anti-inflammatory strategies. J. Biol. Regul. Homeost. Agents 2020, 34, 327–331. [Google Scholar] [PubMed]
- Ouedraogo, D.D.; Tiendrebeogo, W.J.S.; Kabore, F.; Ntsiba, H. COVID-19, chronic inflammatory rheumatic disease and anti-rheumatic treatments. Clin. Rheumatol. 2020, 39, 2069–2075. [Google Scholar] [CrossRef]
- Zhang, C.; Wu, Z.; Li, J.W.; Zhao, H.; Wang, G.Q. Cytokine release syndrome in severe COVID-19: Interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int. J. Antimicrob. Agents 2020, 55, 105954. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, S.; Sinha, A.; Banach, M.; Mittoo, S.; Weissert, R.; Kass, J.S.; Rajagopal, S.; Pai, A.R.; Kutty, S. Cytokine Storm in COVID-19-Immunopathological Mechanisms, Clinical Considerations, and Therapeutic Approaches: The REPROGRAM Consortium Position Paper. Front. Immunol. 2020, 11, 1648. [Google Scholar] [CrossRef]
- Qin, C.; Zhou, L.; Hu, Z.; Zhang, S.; Yang, S.; Tao, Y.; Xie, C.; Ma, K.; Shang, K.; Wang, W.; et al. Dysregulation of Immune Response in Patients With Coronavirus 2019 (COVID-19) in Wuhan, China. Clin. Infect. Dis. 2020, 71, 762–768. [Google Scholar] [CrossRef]
- Fajgenbaum, D.C.; June, C.H. Cytokine Storm. New Engl. J. Med. 2020, 383, 2255–2273. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, S.; Yu, M.; Wang, K.; Tao, Y.; Zhou, Y.; Shi, J.; Zhou, M.; Wu, B.; Yang, Z.; et al. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J. Allergy Clin. Immunol. 2020, 146, 110–118. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; Northwell, C.-R.C.; Barnaby, D.P.; Becker, L.B.; Chelico, J.D.; et al. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef]
- Zheng, Z.; Peng, F.; Xu, B.; Zhao, J.; Liu, H.; Peng, J.; Li, Q.; Jiang, C.; Zhou, Y.; Liu, S.; et al. Risk factors of critical & mortal COVID-19 cases: A systematic literature review and meta-analysis. J. Infect. 2020, 81, e16–e25. [Google Scholar]
- Lu, X.; Zhang, L.; Du, H.; Zhang, J.; Li, Y.Y.; Qu, J.; Zhang, W.; Wang, Y.; Bao, S.; Li, Y.; et al. SARS-CoV-2 Infection in Children. New Engl. J. Med. 2020, 382, 1663–1665. [Google Scholar] [CrossRef]
- Dong, Y.; Mo, X.; Hu, Y.; Qi, X.; Jiang, F.; Jiang, Z.; Tong, S. Epidemiology of COVID-19 Among Children in China. Pediatrics 2020, 145, e20200702. [Google Scholar] [CrossRef] [PubMed]
- Belhadjer, Z.; Meot, M.; Bajolle, F.; Khraiche, D.; Legendre, A.; Abakka, S.; Auriau, J.; Grimaud, M.; Oualha, M.; Beghetti, M.; et al. Acute Heart Failure in Multisystem Inflammatory Syndrome in Children in the Context of Global SARS-CoV-2 Pandemic. Circulation 2020, 142, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, E.; Bamford, A.; Kenny, J.; Kaforou, M.; Jones, C.E.; Shah, P.; Ramnarayan, P.; Fraisse, A.; Miller, O.; Davies, P.; et al. Clinical Characteristics of 58 Children With a Pediatric Inflammatory Multisystem Syndrome Temporally Associated With SARS-CoV-2. JAMA 2020, 324, 259–269. [Google Scholar] [CrossRef]
- Riphagen, S.; Gomez, X.; Gonzalez-Martinez, C.; Wilkinson, N.; Theocharis, P. Hyperinflammatory shock in children during COVID-19 pandemic. Lancet 2020, 395, 1607–1608. [Google Scholar] [CrossRef]
- Haslak, F.; Yildiz, M.; Adrovic, A.; Sahin, S.; Koker, O.; Aliyeva, A.; Barut, K.; Kasapcopur, O. Management of childhood-onset autoinflammatory diseases during the COVID-19 pandemic. Rheumatol. Int. 2020, 40, 1423–1431. [Google Scholar] [CrossRef] [PubMed]
- Filocamo, G.; Minoia, F.; Carbogno, S.; Costi, S.; Romano, M.; Cimaz, R. Pediatric Rheumatology Group of the Milan A: Absence of severe complications from SARS-CoV-2 infection in children with rheumatic diseases treated with biologic drugs. J. Rheumatol. 2020. [Google Scholar] [CrossRef]
- Koker, O.; Demirkan, F.G.; Kayaalp, G.; Cakmak, F.; Tanatar, A.; Karadag, S.G.; Sonmez, H.E.; Omeroglu, R.; Aktay Ayaz, N. Does immunosuppressive treatment entail an additional risk for children with rheumatic diseases? A survey-based study in the era of COVID-19. Rheumatol. Int. 2020, 40, 1613–1623. [Google Scholar] [CrossRef] [PubMed]
- Favalli, E.G.; Monti, S.; Ingegnoli, F.; Balduzzi, S.; Caporali, R.; Montecucco, C. Incidence of COVID-19 in Patients With Rheumatic Diseases Treated With Targeted Immunosuppressive Drugs: What Can We Learn From Observational Data? Arthritis Rheumatol. 2020, 72, 1600–1606. [Google Scholar] [CrossRef]
- Hansmann, S.; Lainka, E.; Horneff, G.; Holzinger, D.; Rieber, N.; Jansson, A.F.; Rosen-Wolff, A.; Erbis, G.; Prelog, M.; Brunner, J.; et al. Consensus protocols for the diagnosis and management of the hereditary autoinflammatory syndromes CAPS, TRAPS and MKD/HIDS: A German PRO-KIND initiative. Pediatr. Rheumatol. Online J. 2020, 18, 17. [Google Scholar] [CrossRef]
- Kuemmerle-Deschner, J.; Ihle, J.; Orlikowski, T.; Dannecker, G.E. ARDIS-Arthritis and Rheumatism Database and Information System. Arthritis Rheum. 1999, 42, S327. [Google Scholar]
- Kuemmerle-Deschner, J.B.; Verma, D.; Endres, T.; Broderick, L.; de Jesus, A.A.; Hofer, F.; Blank, N.; Krause, K.; Rietschel, C.; Horneff, G.; et al. Clinical and Molecular Phenotypes of Low-Penetrance Variants of NLRP3: Diagnostic and Therapeutic Challenges. Arthritis Rheumatol. 2017, 69, 2233–2240. [Google Scholar] [CrossRef] [PubMed]
- Piram, M.; Frenkel, J.; Gattorno, M.; Ozen, S.; Lachmann, H.J.; Goldbach-Mansky, R.; Hentgen, V.; Neven, B.; Stojanovic, K.S.; Simon, A.; et al. A preliminary score for the assessment of disease activity in hereditary recurrent fevers: Results from the AIDAI (Auto-Inflammatory Diseases Activity Index) Consensus Conference. Ann. Rheum. Dis. 2011, 70, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Glaeser, L.; Henes, J.; Kotter, I.; Vogel, W.; Kanz, L.; Klein, R. Molecular recognition patterns of anti-topoisomerase I-antibodies in patients with systemic sclerosis before and after autologous stem cell transplantation. Clin. Exp. Rheumatol. 2018, 36, 28–35. [Google Scholar]
- Paderno, A.; Schreiber, A.; Grammatica, A.; Raffetti, E.; Tomasoni, M.; Gualtieri, T.; Taboni, S.; Zorzi, S.; Lombardi, D.; Deganello, A.; et al. Smell and taste alterations in COVID-19: A cross-sectional analysis of different cohorts. Int. Forum Allergy Rhinol. 2020, 10, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Baj, J.; Karakula-Juchnowicz, H.; Teresinski, G.; Buszewicz, G.; Ciesielka, M.; Sitarz, E.; Forma, A.; Karakula, K.; Flieger, W.; Portincasa, P.; et al. COVID-19: Specific and Non-Specific Clinical Manifestations and Symptoms: The Current State of Knowledge. J. Clin. Med. 2020, 9, 1753. [Google Scholar] [CrossRef]
- Yang, W.; Cao, Q.; Qin, L.; Wang, X.; Cheng, Z.; Pan, A.; Dai, J.; Sun, Q.; Zhao, F.; Qu, J.; et al. Clinical characteristics and imaging manifestations of the 2019 novel coronavirus disease (COVID-19): A multi-center study in Wenzhou city, Zhejiang, China. J. Infect. 2020, 80, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Zhao, Z.; Zhang, T.; Guo, W.; Guo, W.; Zheng, J.; Zhang, J.; Dong, C.; Na, R.; Zheng, L.; et al. A systematic review and meta-analysis of children with coronavirus disease 2019 (COVID-19). J. Med. Virol. 2021, 93, 1057–1069. [Google Scholar] [CrossRef]
- Zhang, C.; Gu, J.; Chen, Q.; Deng, N.; Li, J.; Huang, L.; Zhou, X. Clinical and epidemiological characteristics of pediatric SARS-CoV-2 infections in China: A multicenter case series. PLoS Med. 2020, 17, e1003130. [Google Scholar] [CrossRef]
- Perez-Martinez, A.; Guerra-Garcia, P.; Melgosa, M.; Frauca, E.; Fernandez-Camblor, C.; Remesal, A.; Calvo, C. Clinical outcome of SARS-CoV-2 infection in immunosuppressed children in Spain. Eur. J. Pediatr. 2020, 12, 1–5. [Google Scholar] [CrossRef]
- Gerussi, A.; Rigamonti, C.; Elia, C.; Cazzagon, N.; Floreani, A.; Pozzi, R.; Pozzoni, P.; Claar, E.; Pasulo, L.; Fagiuoli, S.; et al. Coronavirus Disease 2019 (COVID-19) in autoimmune hepatitis: A lesson from immunosuppressed patients. Hepatol. Commun. 2020, 4, 1257–1262. [Google Scholar] [CrossRef]
- Kainth, M.K.; Goenka, P.K.; Williamson, K.A.; Fishbein, J.S.; Subramony, A.; Barone, S.; Belfer, J.A.; Feld, L.M.; Krief, W.I.; Palumbo, N.; et al. Early Experience of COVID-19 in a US Children’s Hospital. Pediatrics 2020, 146, e2020003186. [Google Scholar] [CrossRef]
- Moutsopoulos, H.M. Anti-inflammatory therapy may ameliorate the clinical picture of COVID-19. Ann. Rheum. Dis. 2020, 79, 1253–1254. [Google Scholar] [CrossRef]
- Freeman, T.L.; Swartz, T.H. Targeting the NLRP3 Inflammasome in Severe COVID-19. Front. Immunol. 2020, 11, 1518. [Google Scholar] [CrossRef] [PubMed]
- Dalrymple, J.M.; Stamp, L.K.; O’Donnell, J.L.; Chapman, P.T.; Zhang, M.; Barclay, M.L. Pharmacokinetics of oral methotrexate in patients with rheumatoid arthritis. Arthritis Rheum. 2008, 58, 3299–3308. [Google Scholar] [CrossRef] [PubMed]
- Murchu, E.O.; Byrne, P.; Walsh, K.A.; Carty, P.G.; Connolly, M.; De Gascun, C.; Jordan, K.; Keoghan, M.; O’Brien, K.K.; O’Neill, M.; et al. Immune response following infection with SARS-CoV-2 and other coronaviruses: A rapid review. Rev. Med. Virol. 2020, e2162. [Google Scholar] [CrossRef]
- Kellam, P.; Barclay, W. The dynamics of humoral immune responses following SARS-CoV-2 infection and the potential for reinfection. J. Gen. Virol. 2020, 101, 791–797. [Google Scholar] [CrossRef] [PubMed]
- Long, Q.X.; Liu, B.Z.; Deng, H.J.; Wu, G.C.; Deng, K.; Chen, Y.K.; Liao, P.; Qiu, J.F.; Lin, Y.; Cai, X.F.; et al. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat. Med. 2020, 26, 845–848. [Google Scholar] [CrossRef]
- Callow, K.A.; Parry, H.F.; Sergeant, M.; Tyrrell, D.A. The time course of the immune response to experimental coronavirus infection of man. Epidemiol. Infect. 1990, 105, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Seow, J.; Graham, C.; Merrick, B.; Acors, S.; Pickering, S.; Steel, K.J.A.; Hemmings, O.; O’Byrne, A.; Kouphou, N.; Galao, R.P.; et al. Longitudinal observation and decline of neutralizing antibody responses in the three months following SARS-CoV-2 infection in humans. Nat. Microbiol. 2020, 5, 1598–1607. [Google Scholar] [CrossRef]
- To, K.K.; Hung, I.F.; Ip, J.D.; Chu, A.W.; Chan, W.M.; Tam, A.R.; Fong, C.H.; Yuan, S.; Tsoi, H.W.; Ng, A.C.; et al. COVID-19 re-infection by a phylogenetically distinct SARS-coronavirus-2 strain confirmed by whole genome sequencing. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Tillett, R.L.; Sevinsky, J.R.; Hartley, P.D.; Kerwin, H.; Crawford, N.; Gorzalski, A.; Laverdure, C.; Verma, S.C.; Rossetto, C.C.; Jackson, D.; et al. Genomic evidence for reinfection with SARS-CoV-2: A case study. Lancet Infect. Dis. 2021, 21, 52–58. [Google Scholar] [CrossRef]
- Bentivegna, E.; Sentimentale, A.; Luciani, M.; Speranza, M.L.; Guerritore, L.; Martelletti, P. New IgM seroconversion and positive RT-PCR test after exposure to the virus in recovered COVID-19 patient. J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Torres, D.A.; Ribeiro, L.; Riello, A.; Horovitz, D.D.G.; Pinto, L.F.R.; Croda, J. Reinfection of COVID-19 after 3 months with a distinct and more aggressive clinical presentation: Case report. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Freeman, M.C.; Rapsinski, G.J.; Zilla, M.L.; Wheeler, S.E. Immunocompromised Seroprevalence and Course of Illness of SARS-CoV-2 in One Pediatric Quaternary Care Center. J. Pediatric Infect. Dis. Soc. 2020. [Google Scholar] [CrossRef] [PubMed]
| PGA (0–10 cm) | PPGA (0–10 cm) | Flares/3 Months | Fever | Rash | Fatigue | Dyspnea | Arthralgia | |
|---|---|---|---|---|---|---|---|---|
| Patient 1 | ||||||||
| Before COVID-19 | 2 | 2 | 1 | - | - | + | - | + |
| ~Day 40 * | 2 | n.a. | n.a. | - | - | +++ | ++ | + |
| ~Day 100 * | 2 | n.a | n.a. | - | - | ++ | + | + |
| Patient 2 | ||||||||
| Before COVID-19 | ≤2 | ≤2 | 0 | - | - | - | - | - |
| Day 40 * | 2 | 5 | n.a | - | + | +/++ | - | - |
| Day 100 * | 2 | 2 | 1 | - | + | + | - | - |
| Patient 3 | ||||||||
| Before COVID-19 | 1–3 | 2 | 0 | - | - | + | - | - |
| Day 40 * | 4 | 7 | n.a. | - | ++ | +++ | - | + |
| Day 100 *,1 | 5 | 7 | 3 | +/++ | - | +++ | - | ++ |
| Patient 4 | ||||||||
| Before COVID-19 | ≤2 | 0 | 0 | - | - | - | - | - |
| Day 40 * | 2 | n.a. | n.a. | - | - | - | - | + |
| Day 100 * | 1 | 0 | 0 | - | - | - | - | - |
| Hb (g/dl) | Leuc (10S9/l) | Plt (10S9/l) | ESR (mm/h) | CRP (mg/dl) | IL-6 (ng/L) | S100A8/A9 (μg/mL) | sl-ILR2 (U/mL) | SAA (mg/L) | |
|---|---|---|---|---|---|---|---|---|---|
| Patient 1 | |||||||||
| Before COVID-19 | 11.8 | 5.52 | 197 | 8 | 0.34 | <2.0 | 2.9 | n.a. | 2 |
| ~Day 40 * | 12.4 | 5.30 | 256 | 22 | 0.72 | 3.0 | 16.4 | 267 | 4 |
| ~Day 100 * | 11.6 | 5.92 | 231 | 13 | 1.54 | n.a. | 6.2 | n.a. | 6 |
| Patient 2 | |||||||||
| Before COVID-19 | 13.5 | 4.30 | 303 | 5 | 0.01 | <2.0 | 11.3 | n.a. | 1 |
| Day 40 * | 14.3 | 5.09 | 239 | n.a. | 0.01 | 2.7 | 10.4 | 215 | 2 |
| Day 100 * | 13.9 | 5.38 | 301 | 2 | 0.01 | n.a. | 6.3 | n.a. | 1 |
| Patient 3 | |||||||||
| Before COVID-19 | 14.3 | 5.37 | 319 | 2 | 0.01 | <2.0 | 3.2 | n.a. | 1 |
| Day 40 * | 15.0 | 5.63 | 305 | 4 | 0.02 | 2.7 | 6.9 | 164 | 2 |
| Day 100 *,1 | 14.5 | 5.78 | 300 | 2 | 0.01 | 2.8 | 5.7 | n.a. | 2 |
| Patient 4 | |||||||||
| Before COVID-19 | 13.2 | 5.54 | 191 | 13 | 0.14 | 2.9 | 64.4 | n.a. | 19 |
| Day 40 * | 13.3 | 4.61 | 180 | 2 | 0.05 | n.a. | n.a. | n.a. | 10 |
| Day 100 * | 12.5 | 4.06 | 174 | 7 | 0.07 | 3.7 | 40.9 | n.a | 15 |
| COVID-19 Test (Nasopharyngeal) | SARS-CoV-2 Antibodies § (Serum) | |
|---|---|---|
| Patient 1 | ||
| Day 8 * | Positive | n.a. |
| ~Day 40 * | n.a. | n.a. |
| ~Day 100 * | n.a. | Negative |
| Patient 2 | ||
| Day -2 (before COVID-19 symptom onset) | Negative | |
| Day 40 * | n.a. | Positive IgG (19.4 U/mL) and IgA (15.2 U/mL) SARS-CoV-2-nucleocapside, Positive IgG SARS-CoV-2-spike (18.3 U/mL), Positive IgG SARS-CoV-2-RBD (19.9 U/mL) |
| Day 100 * | n.a. | Negative |
| Patient 3 | ||
| Day -2 (before COVID-19 symptom onset) | Negative | n.a |
| Day 40 * | n.a. | n.a. |
| Day 100*,1 | n.a. | Positive IgG (59.3 U/mL) and IgA (28.6 U/mL) SARS-CoV-2-nucleocapside |
| Patient 4 | ||
| Day 2* | Positive | n.a. |
| Day 40* | n.a. | n.a. |
| Day 100* | n.a. | Negative |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Welzel, T.; Samba, S.D.; Klein, R.; van den Anker, J.N.; Kuemmerle-Deschner, J.B. COVID-19 in Autoinflammatory Diseases with Immunosuppressive Treatment. J. Clin. Med. 2021, 10, 605. https://doi.org/10.3390/jcm10040605
Welzel T, Samba SD, Klein R, van den Anker JN, Kuemmerle-Deschner JB. COVID-19 in Autoinflammatory Diseases with Immunosuppressive Treatment. Journal of Clinical Medicine. 2021; 10(4):605. https://doi.org/10.3390/jcm10040605
Chicago/Turabian StyleWelzel, Tatjana, Samuel Dembi Samba, Reinhild Klein, Johannes N. van den Anker, and Jasmin B. Kuemmerle-Deschner. 2021. "COVID-19 in Autoinflammatory Diseases with Immunosuppressive Treatment" Journal of Clinical Medicine 10, no. 4: 605. https://doi.org/10.3390/jcm10040605
APA StyleWelzel, T., Samba, S. D., Klein, R., van den Anker, J. N., & Kuemmerle-Deschner, J. B. (2021). COVID-19 in Autoinflammatory Diseases with Immunosuppressive Treatment. Journal of Clinical Medicine, 10(4), 605. https://doi.org/10.3390/jcm10040605








