Can Use of Digital Technologies by People with Dementia Improve Self-Management and Social Participation? A Systematic Review of Effect Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources and Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Study Selection and Data Extraction
2.4. Methodological Quality and Weight of Evidence
2.5. Synthesis
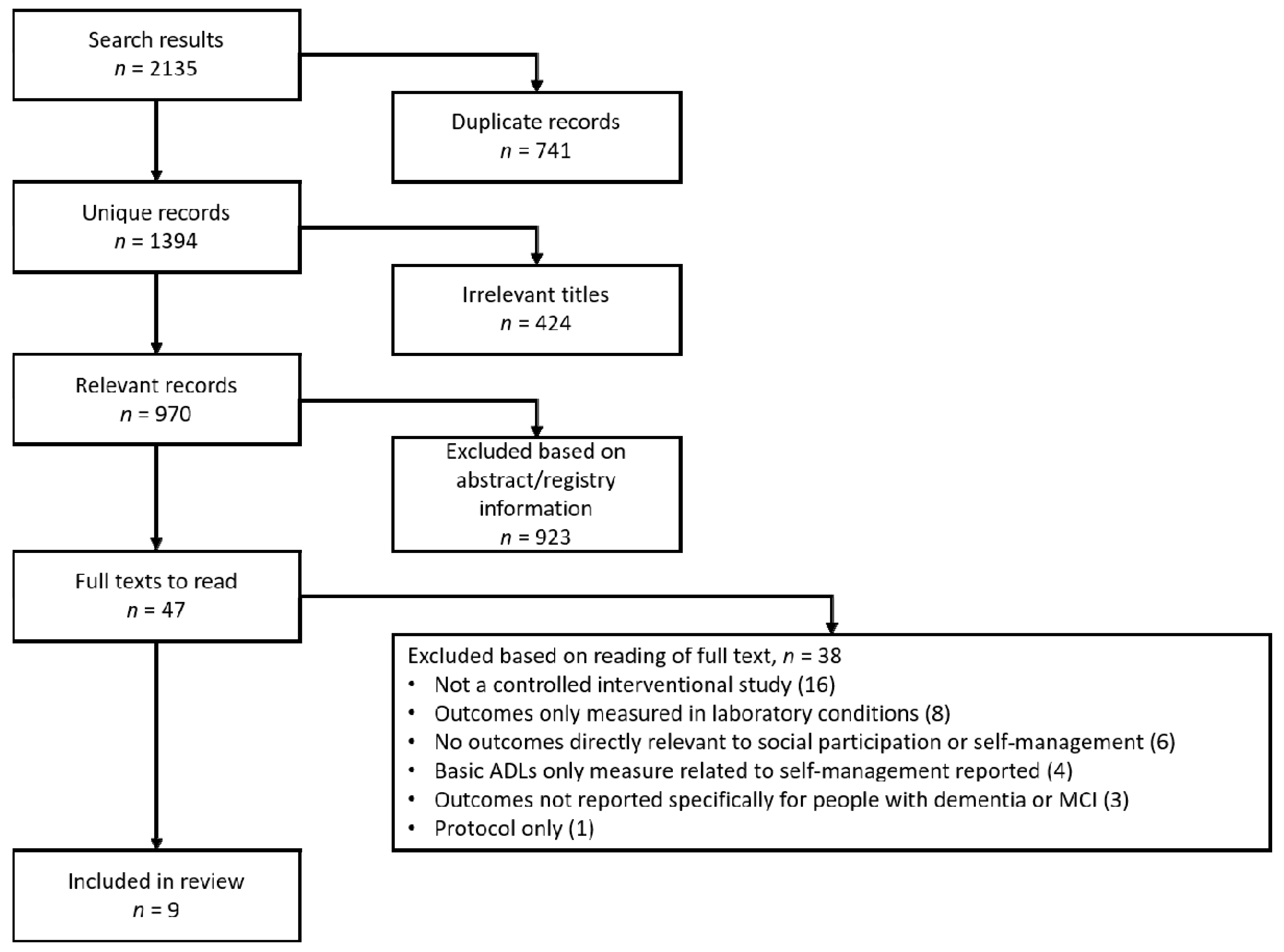
3. Results
3.1. Characteristics of Included Studies
- Virtual reality (VR)-based interventions
- -
- Combined physical and cognitive VR-based training [31]: The primary aims of the intervention were to improve the user’s cognitive function and reduce neuronal oxidative stress. A VR-based program combining aerobic exercise and cognitive training was administered over 6 weeks, in three sessions per week (18 sessions total). Each session was of 40–45 min duration, in which participants interacted with three virtual environments (15–20 min cycle in a virtual park, 5 min crossing at virtual crossroads, 20 min shopping in a virtual supermarket).
- -
- VR-based physical and cognitive training [29]: The primary aims of the intervention were to improve the user’s cognitive function and performance of IADL. The Microsoft Kinect system was used to engage participants in Tai Chi, resistance and aerobic exercises, and simulated functional tasks, such as window cleaning and stair climbing. The HTC VIVE system (VR glasses) was used to engage participants in VR games based on IADL (such as shopping and food preparation). The intervention was delivered in sessions (three sessions per week for 12 weeks, 36 sessions in total). Each session lasted one hour (40 min physical activity, 20 min cognitive training).
- -
- VR-based non-specific computer training [33]: The aims of the intervention were to improve users’ cognitive function and quality of life. Users were trained to use the Nintendo Wii to engage in virtual reality sports activities, including table tennis, fencing, and archery. The intervention was delivered by occupational therapists, during three sessions per week for ten weeks, of 10 min duration.
- Other wearable technologies
- -
- Social Support Aid, a web-based mobile app with a smartwatch [30]: The primary aim of the intervention was to improve the users’ social engagement. Social Support Aid is a mobile phone-based app, which connects to a smart watch. Facial recognition software is used to assist people with dementia in the identification (names and relationships) of people that they interact with. Participants were given the technology to use for 6 months and were free to use it as much or as little as they chose.
- -
- SenseCam, a wearable camera [35]: The primary aims of the intervention were to improve the cognitive function (specifically episodic and autobiographical memory), and the wellbeing and quality of life of the user. The SenseCam wearable camera was worn by participants around the neck while performing routine activities. The images captured were reviewed and discussed with a psychologist at interval appointments (face-to-face, twice per week). Participants were given SenseCam to wear for six weeks and were encouraged to wear the camera every day for the longest time possible.
- Software applications
- -
- Tablet-based cognitive training and rehabilitation [34]: The primary aims of the intervention were to improve cognitive function and functional capabilities of the user, and to unburden the user’s informal caregiver. A tablet computer was used for guided and independent cognitive training. Participants also engaged in a wellbeing program via the tablet, with videocalls for peer support. The telerehabilitation program was administered over 13 weeks—guided for the first 9 weeks, then completed independently for the remaining 4 weeks. Participants engaged in seven face-to-face sessions (four sessions of 4–6 h each for patients and family members together, and three peer support sessions of 2 h each separately for patients and family members).
- -
- Electric calendar [32]: The primary aims of the intervention were to improve the user’s cognitive function and reduce behavioral disturbances by means of a software application for an Android tablet with page-a-day calendar, clock, and alarm function for scheduled “events”. The intention was that the person with dementia or MCI would regularly view the calendar and interact with alarms. The calendar was set-up and kept up to date by a caregiver. The calendar was continuously present in the participant’s home for 12 weeks.
- -
- WESIHAT 2.0©, a web-based health education tool [36]: The primary aim of the intervention was to improve cognitive function of the user. Secondary aims were to improve users’ functional capabilities, social support, mood, and quality of life, and to reduce loneliness. A web-based application with four components: (1) a screening tool for risk of memory impairment; (2) lifestyle advice for promoting memory and health; (3) health diary; (4) healthy food menu, with meal preparation tips, shopping guidelines, and a nutrition-related quiz. Participants were exposed to the application in sessions: four sessions per week for six months (at least 100 sessions total). Sessions lasted at least 30 min.
- -
- Computerized errorless learning program (CELP) for memory training [28]: The primary aim of the intervention was to improve the cognitive function of the user. Improving mood and functional capabilities were secondary aims. The memory training included several components: basic training on various memory types; the use of mnemonics and learning principles, as well as name–face association; and advanced memory training and strategies for applying memory techniques to ADLs. Participants received the intervention in sessions: approximately two sessions per week up to a total of twelve sessions. Each session lasted around 30 min.
3.2. Quality and Weight-of-Evidence Assessment of Included Studies
3.3. Results of Included Studies
3.4. Facilitators and Barriers to Implementation
4. Discussion
4.1. Strengths and Limitations of This Review
4.2. Recommendations for Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huber, M.; Knottnerus, J.A.; Green, L.; van der Horst, H.; Jadad, A.R.; Kromhout, D.; Leonard, B.; Lorig, K.; Loureiro, M.I.; van der Meer, J.W.; et al. How should we define health? BMJ 2011, 343. [Google Scholar] [CrossRef]
- Alzheimer Nederland. Handreiking (dag)activiteiten Bij Dementie. 2014. Available online: https://www.alzheimer-nederland.nl/sites/default/files/directupload/activiteiten-bij-dementie.pdf (accessed on 17 December 2020).
- The Alzheimer’s Society. Dementia 2013: The Hidden Voice of Loneliness; Alzheimer’s Society: London, UK, 2013; p. 78. [Google Scholar]
- Dröes, R.-M.; Boelens-Van Der Knoop, E.C.; Bos, J.; Meihuizen, L.; Ettema, T.P.; Gerritsen, D.L.; Hogeveen, F.; De Lange, J.; Schölzel-Dorenbos, C.J.M. Quality of life in dementia in perspective: An explorative study of variations in opinions among people with dementia and their professional caregivers, and in literature. Dementia 2006, 5, 533–558. [Google Scholar]
- Van der Roest, H.G.; Meiland, F.J.M.; Comijs, H.C.; Derksen, E.; Jansen, A.P.D.; Van Hout, H.P.J.; Jonker, C.; Dröes, R.-M. What do community-dwelling people with dementia need? A survey of those who are known to care and welfare services. Int. Psychogeriatr. 2009, 21, 949–965. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.; Turner, A.; Wallace, L.M.; Choudhry, K.; Bradbury, N. Perceived barriers to self-management for people with dementia in the early stages. Dementia 2013, 12, 481–493. Available online: http://www.ncbi.nlm.nih.gov/pubmed/24336956 (accessed on 17 December 2020). [CrossRef] [PubMed]
- World Health Organization. Dementia as a Public Health Priority; WHO: Geneva, Switzerland, 2012; ISBN 978 92 4 156445 8. [Google Scholar]
- Meiland, F.J.M.; Innes, A.; Mountain, G.; Robinson, L.; van der Roest, H.; García-Casal, J.A.; Gove, D.; Thyrian, J.R.; Evans, S.; Dröes, R.-M.; et al. Technologies to Support Community-Dwelling Persons With Dementia: A Position Paper on Issues Regarding Development, Usability, Effectiveness and Cost-Effectiveness, Deployment, and Ethics. JMIR Rehabil. Assist. Technol. 2017, 4, e1. [Google Scholar] [CrossRef]
- Dutch National Dementia Strategy. Deltaplan Dementie. Available online: https://www.vilans.nl/vilans/media/documents/producten/zorgstandaard-dementie.pdf (accessed on 17 December 2020).
- Dröes, R.-M.; Vermeer, Y.; Libert, S.; Gaber, S.; Wallcook, S.; Rai, H.; Barroso, A.C.; Van Santen, J.; Mangiaracina, F.; Beentjes, K.; et al. Best Practice Guidance Human Interaction with Technology in Dementia. Recommendations based on the research conducted in the Marie Sklodowska Curie International Training Network INDUCT. Available online: https://www.dementiainduct.eu/guidance/recommendation/3.2.2.3/ (accessed on 17 December 2020).
- Manca, R.; De Marco, M.; Venneri, A. The Impact of COVID-19 Infection and Enforced Prolonged Social Isolation on Neuropsychiatric Symptoms in Older Adults with and Without Dementia: A Review. Front. Psychiatry 2020, 11, 585540. [Google Scholar] [CrossRef] [PubMed]
- Rijksdienst voor Ondernemend Nederland. Available online: https://www.rvo.nl/subsidie-en-financieringswijzer/set-covid-19-2-zorg-en-ondersteuning-op-afstand (accessed on 17 December 2020).
- Lauriks, S.; Reinersmann, A.; van der Roest, H.; Meiland, F.J.M.; Davies, R.J.; Moelaert, F.; Mulvenna, M.D.; Nugent, C.D.; Dröes, R.-M. Review of ICT-based services for identified unmet needs in people with dementia. Ageing Res. Rev. 2007, 6, 223–246. [Google Scholar] [CrossRef]
- Gates, N.J.; Vernooij, R.W.M.; Di Nisio, M.; Karim, S.; March, E.; Martinez, G.; Rtujes, A. Computerised cognitive training for preventing dementia in people with mild cognitive impairment. Cochrane Database Syst. Rev. 2019, CD012279. [Google Scholar] [CrossRef]
- García-Casal, J.A.; Loizeau, A.; Csipke, E.; Franco-Martín, M.; Perea-Bartolomé, M.V.; Orrell, M. Computer-based cognitive interventions for people living with dementia: A systematic literature review and meta-analysis. Aging Ment. Health 2017, 21, 454–467. [Google Scholar] [CrossRef]
- Moore, G.E. Cramming more components onto integrated circuits. Electronics 1965, 38, 8. [Google Scholar] [CrossRef]
- Waldrop, M.M. The chips are down for Moore’s law. Nature 2016, 530, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Dröes, R.-M.; Chattat, R.; Diaz, A.; Gove, D.; Graff, M.; Murphy, K.; Verbeek, H.; Vernooij-Dassen, M.; Clare, L.; Johannessen, A.; et al. Social health and dementia: A European consensus on the operationalization of the concept and directions for research and practice. Aging Ment. Health 2017, 21, 4–17. [Google Scholar] [CrossRef]
- Neal, D.P.; Kerkhof, Y.J.F.; Ettema, T.P.; Muller, M.; Bosmans, J.; Finnema, E.; Graff, M.; Dijkstra, K.; Stek, M.; Dröes, R.-M. Evaluation of FindMyApps: Protocol for a Randomized Controlled Trial of the effectiveness and cost-effectiveness of a tablet-based intervention to improve self-management and social participation of community-dwelling people with mild dementia, compared to usual tablet use. BMC Geriatr. 2021, in press. [Google Scholar] [CrossRef]
- Lorenz, K.; Freddolino, P.P.; Comas-Herrera, A.; Knapp, M.; Damant, J. Technology-based tools and services for people with dementia and carers: Mapping technology onto the dementia care pathway. Dementia 2019, 18, 725–741. [Google Scholar] [CrossRef]
- Craig, P.; Dieppe, P.; Macintyre, S.; Mitchie, S.; Nazareth, I.; Petticrew, M. Developing and evaluating complex interventions: The new Medical Research Council guidance. BMJ 2008, 337, 979–983. [Google Scholar] [CrossRef]
- Moore, G.F.; Audrey, S.; Barker, M.; Bond, L.; Bonell, C.; Hardeman, W.; Moore, L.; O’Cathain, A.; Tinati, T.; Wight, D.; et al. Process evaluation of complex interventions: Medical Research Council guidance. BMJ 2015, 350. [Google Scholar] [CrossRef]
- Centre for Reviews and Dissemination. CRD’s Guidance for Undertaking Reviews in Health Care; CRD, University of York: York, UK, 2009; ISBN 978-1-900640-47-3. [Google Scholar]
- Mitchell, A.J.; Shiri-Feshki, M. Rate of progression of mild cognitive impairment to dementia--meta-analysis of 41 robust inception cohort studies. Acta Psychiatr. Scand. 2009, 119, 252–265. [Google Scholar] [CrossRef]
- Gough, D. Weight of evidence: A framework for the appraisal of the quality and relevance of evidence. Res. Pap. Educ. 2007, 22, 213–228. [Google Scholar] [CrossRef]
- National Institutes of Health. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 17 December 2020).
- Mays, N.; Pope, C.; Popay, J. Details of Approaches to Synthesis. A Methodological Appendix to the Paper: Systematically Reviewing Qualitative and Quantitative Evidence to Inform Management and Policy Making in the Health Field. Available online: http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.113.2530&rep=rep1&type=pdf (accessed on 27 December 2020).
- Lee, G.Y.; Yip, C.C.K.; Yu, E.C.S.; Man, D.W.K. Evaluation of a computer-assisted errorless learning-based memory training program for patients with early Alzheimer’s disease in Hong Kong: A pilot study. Clin. Interv. Aging. 2013, 8, 623–633. [Google Scholar]
- Liao, Y.Y.; Tseng, H.-Y.; Lin, Y.-J.; Wang, C.-J.; Hsu, W.-C. Using virtual reality-based training to improve cognitive function, instrumental activities of daily living and neural efficiency in older adults with mild cognitive impairment. Eur. J. Phys. Rehabil. Med. 2020, 56, 47–57. [Google Scholar] [CrossRef]
- McCarron, H.R.; Zmora, R.; Gaugler, J.E. A Web-Based Mobile App with a Smartwatch to Support Social Engagement in Persons With Memory Loss: Pilot Randomized Controlled Trial. JMIR Aging. 2019, 2, e13378. [Google Scholar] [CrossRef]
- Mrakic-Sposta, S.; Di Santo, S.G.; Franchini, F.; Arlati, S.; Zangiacomi, A.; Greci, L.; Moretti, S.; Jesuthasan, N.; Marzorati, M.; Rizzo, G.; et al. Effects of Combined Physical and Cognitive Virtual Reality-Based Training on Cognitive Impairment and Oxidative Stress in MCI Patients: A Pilot Study. Front. Aging Neurosci. 2018, 10, 282. [Google Scholar] [CrossRef]
- Nishiura, Y.; Nihei, M.; Nakamura-Thomas, H.; Inoue, T. Effectiveness of using assistive technology for time orientation and memory, in older adults with or without dementia. Disabil. Rehabil. Assist. Technol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-H.; Park, J.-H. Does cognition-specific computer training have better clinical outcomes than non-specific computer training? A single-blind, randomized controlled trial. Clin. Rehabil. 2018, 32, 213–222. [Google Scholar] [CrossRef]
- Pietilä, P.; Poutiainen, E.; Nukari, J.; Launiainen, H.; Arifullen-Hämäläinen, U.; Sarajuuri, J.; Koskinen, S. Multi-methodological rehabilitation with emphasis on cognitive training alleviates Alzheimer patients’ mood symptoms and supports coping among family members. Psykologia 2017, 52, 356–375. [Google Scholar]
- Silva, A.R.; Pinho, M.S.; Macedo, L.; Moulin, C.; Caldeira, S.; Firmino, H. It is not only memory: Effects of sensecam on improving well-being in patients with mild alzheimer disease. Int. Psychogeriatr. 2017, 29, 741–754. [Google Scholar] [CrossRef]
- Vanoh, D.; Shahar, S.; Razali, R.; Ali, N.M.; Manaf, Z.A.; Mohd Noah, S.A.; Nur, A.M. The effectiveness of a Web-Based Health Education Tool, WESIHAT 2.0, among Older Adults: A Randomized Controlled Trial. J. Alzheimer’s Dis. 2019, 70, S255–S270. [Google Scholar] [CrossRef]
- Bejan, A.; Murko, P.; Müller, N.; König, P.; Kunze, C. Using Surface Table Computers to Promote the Well-Being of People with Dementia. Stud. Health Technol. Inform. 2017, 242, 23–26. [Google Scholar]
- Casey, D.; Whelan, S.; Barrett, E.; Murphy, K.; Santorelli, A.; Burke, M.; Kovacic, T. The Impact of a Companion Robot in Combatting Loneliness in People with Dementia Living in Residential Care. The MARIO Project. Age Ageing 2018, 47, v13–v60. [Google Scholar] [CrossRef]
- Cherniack, E.P. Not just fun and games: Applications of virtual reality in the identification and rehabilitation of cognitive disorders of the elderly. Disabil. Rehabil. Assist. Technol. 2011, 6, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Maia, J.C.; Coutinho, J.F.V.; Sousa, C.R.; Barbosa, R.G.B.; Mota, F.R.N.; Marques, M.B.; Silva, R.R.L.; Lima, R.B.S. Assistive technologies for demented elderly: A systematic review. Acta Paul. Enferm. 2018, 31, 651–658. [Google Scholar] [CrossRef]
- Cutler, C.; Hicks, B.; Innes, A. Does Digital Gaming Enable Healthy Aging for Community-Dwelling People with Dementia? Games Cult. 2016, 11, 104–129. [Google Scholar] [CrossRef]
- D’Cunha, N.M.; Nguyen, D.; Naumovski, N.; McKune, A.J.; Kellett, J.; Georgousopoulou, E.N.; Frost, J.; Isbel, S. Mini-Review of Virtual Reality-Based Interventions to Promote Well-Being for People Living with Dementia and Mild Cognitive Impairment. Gerontology 2019, 65, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Djabelkhir, L.; Wu, Y.H.; Vidal, J.S.; Cristancho-Lacroix, V.; Marlats, F.; Lenoir, H.; Carno, A.; Rigaud, A.S. Computerized cognitive stimulation and engagement programs in older adults with mild cognitive impairment: Comparing feasibility, acceptability, and cognitive and psychosocial effects. Clin. Interv. Aging 2017, 12, 1967–1975. [Google Scholar] [CrossRef] [PubMed]
- Djabelkhir-Jemmi, L.; Wu, Y.H.; Boubaya, M.; Marlats, F.; Lewis, M.; Vidal, J.S.; Lenoir, H.; Charlieux, B.; Isabet, B.; Rigaud, A.S. Differential effects of a computerized cognitive stimulation program on older adults with mild cognitive impairment according to the severity of white matter hyperintensities. Clin. Interv. Aging 2018, 13, 1543–1554. [Google Scholar]
- Dossey, L. FOMO, Digital Dementia, and Our Dangerous Experiment. Explore J. Sci. Heal. 2014, 10, 69–73. [Google Scholar] [CrossRef]
- Moyle, W.; Jones, C.; Murfield, J.; Thalib, L.; Beattie, E.; Shum, D.; Draper, B. Using a therapeutic companion robot for dementia symptoms in long-term care: Reflections from a cluster-RCT. Aging Ment. Health 2019, 23, 329–336. [Google Scholar] [CrossRef]
- Sánchez-Rico, A.; Garel, P.; Notarangelo, I.; Quintana, M.; Hernández, G.; Asteriadis, S.; Popa, M.; Vretos, N.; Solachidis, V.; Burgos, M.; et al. ICT Services for Life Improvement for the Elderly. Stud. Health Technol. Inform. 2017, 242, 600–605. [Google Scholar]
- Sung, H.C.; Chang, S.M.; Chin, M.Y.; Lee, W.L. Robot-assisted therapy for improving social interactions and activity participation among institutionalized older adults: A pilot study. Asia-Pac. Psychiatry 2015, 7, 1–6. [Google Scholar] [CrossRef]
- Tobiasson, H.; Sundblad, Y.; Walldius, Å.; Hedman, A. Designing for Active Life: Moving and Being Moved Together with Dementia Patients. Int. J. Des. 2015, 9, 47–62. [Google Scholar]
- Tyack, C.; Camic, P.M.; Heron, M.J.; Hulbert, S. Viewing Art on a Tablet Computer: A Well-Being Intervention for People with Dementia and Their Caregivers. J. Appl. Gerontol. 2017, 36, 864–894. [Google Scholar] [CrossRef] [PubMed]
- Weakley, A.; Weakley, A.T.; Schmitter-Edgecombe, M. Compensatory strategy use improves real-world functional performance in community dwelling older adults. Neuropsychology 2019, 33, 1121–1135. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Foloppe, D.A.; Richard, P.; Richard, E.; Allain, P. A Dual-Modal Virtual Reality Kitchen for (Re) Learning of Everyday Cooking Activities in Alzheimer’s Disease. Presence Teleoperators Virtual Environ. 2012, 21, 43–57. [Google Scholar] [CrossRef]
- Astell, A.J.; Joddrell, P.; Groenewoud, H.; de Lange, J.; Goumans, M.; Cordia, A.; Schikhof, Y. Does familiarity affect the enjoyment of touchscreen games for people with dementia? Int. J. Med. Inform. 2016, 91, e1–e8. [Google Scholar] [CrossRef] [PubMed]
- In Soon, K.; Hee Sun, K. Effects of Intervention Using PARO on the Cognition, Emotion, Problem Behavior, and Social Interaction of Elderly People with Dementia. J. Korean Acad. Community Health Nurs. Jiyeog Sahoe Ganho Hakoeji 2018, 29, 300–309. [Google Scholar]
- Leng, F.Y.; Yeo, D.; George, S.; Barr, C. Comparison of iPad applications with traditional activities using person-centred care approach: Impact on well-being for persons with dementia. Dementia 2014, 13, 265–273. [Google Scholar] [CrossRef]
- Liang, A.; Piroth, I.; Robinson, H.; MacDonald, B.; Fisher, M.; Nater, U.M.; Skoluda, N.; Broadbent, E. A Pilot Randomized Trial of a Companion Robot for People with Dementia Living in the Community. J. Am. Med. Dir. Assoc. 2017, 18, 871–878. [Google Scholar] [CrossRef]
- O’Neill, B.; Moran, K.; Gillespie, A. Scaffolding rehabilitation behaviour using a voice-mediated assistive technology for cognition. Neuropsychol. Rehabil. 2010, 20, 509–527. [Google Scholar] [CrossRef]
- Palac, D.; Bullard, T.; Cohen, J.D.; Nguyen, L.T.; Mudar, R.A.; Mullen, S.P. Effects of Traditional vs. iPad-Enhanced Aerobic Exercise on Wayfinding Efficacy and Cognition: A Pilot Randomized Controlled Trial. Int. J. Environ. Res. Public. Health 2019, 16, 3495. [Google Scholar] [CrossRef] [PubMed]
- Rouaix, N.; Retru-Chavastel, L.; Rigaud, A.S.; Monnet, C.; Lenoir, H.; Pino, M. Affective and Engagement Issues in the Conception and Assessment of a Robot-Assisted Psychomotor Therapy for Persons with Dementia. Front. Psychol. 2017, 8, 950. [Google Scholar] [CrossRef] [PubMed]
- Song, J.H. Effects of a robot pet-assisted program for elderly people with dementia. J. Korean Acad. Nurs. 2009, 39, 562–573. [Google Scholar] [CrossRef] [PubMed]
- Bahar-Fuchs, A.; Webb, S.; Bartsch, L.; Clare, L.; Rebok, G.; Cherbuin, N.; Anstey, K.J. Tailored and Adaptive Computerized Cognitive Training in Older Adults at Risk for Dementia: A Randomized Controlled Trial. J. Alzheimer’s Dis. 2017, 60, 889–911. [Google Scholar] [CrossRef] [PubMed]
- Ben-Sadoun, G.; Sacco, G.; Manera, V.; Bourgeois, J.; König, A.; Foulon, P.; Fosty, B.; Bremond, F.; d’Arripe-Longueville, F.; Robert, P. Physical and Cognitive Stimulation Using an Exergame in Subjects with Normal Aging, Mild and Moderate Cognitive Impairment. J. Alzheimer’s Dis. 2016, 53, 1299–1314. [Google Scholar] [CrossRef]
- Poptsi, E.; Lazarou, I.; Markou, N.; Vassiloglou, M.; Nikolaidou, E.; Diamantidou, A.; Siatra, V.; Karathanassi, E.; Karakostas, A.; Zafeiropoulou, F.K.; et al. A Comparative Single-Blind Randomized Controlled Trial with Language Training in People with Mild Cognitive Impairment. Am. J. Alzheimer’s Dis. Other Dement. 2019, 34, 176–187. [Google Scholar] [CrossRef]
- Rostill, H.; Nilforooshan, R.; Barnaghi, P.; Morgan, A. Technology-integrated dementia care: Trial results. Nurs. Resid. Care 2019, 21, 489–494. [Google Scholar] [CrossRef]
- Rouse, H.J.; Small, D.J.; Faust, M.E. Assessment of Cognitive Training & Social Interaction in People with Mild to Moderate Dementia: A Pilot Study. Clin. Gerontol. 2019, 42, 421–434. [Google Scholar]
- Tsai, P.F.; Kitch, S.; Beck, C.; Jakobs, T.; Rettiganti, M.; Jordan, K.; Jakobs, E.; Adair, S. Using an Interactive Video Simulator to Improve Certified Nursing Assistants’ Dressing Assistance and Nursing Home Residents’ Dressing Performance: A Pilot Study. CIN Comput. Inform. Nurs. 2018, 36, 183–192. [Google Scholar] [CrossRef]
- Lee, G.J.; Bang, H.J.; Lee, K.M.; Kong, H.H.; Seo, H.S.; Oh, M.; Bang, M. A comparison of the effects between 2 computerized cognitive training programs, Bettercog and COMCOG, on elderly patients with MCI and mild dementia A single-blind randomized controlled study. Medicine 2018, 97. [Google Scholar] [CrossRef]
- Oh, J.H.; Yi, Y.J.; Shin, C.J.; Park, C.; Kang, S.; Kim, J.; Kim, I.S. Effects of Silver-Care-Robot Program on Cognitive Function, Depression, and Activities of Daily Living for Institutionalized Elderly People. J. Korean Acad. Nurs. 2015, 45, 388–396. [Google Scholar] [CrossRef]
- Rohrbach, N.; Gulde, P.; Armstrong, A.R.; Hartig, L.; Abdelrazeq, A.; Schröder, S.; Neuse, J.; Grimmer, T.; Diehl-Schmid, J.; Hermsdörfer, J. An augmented reality approach for ADL support in Alzheimer’s disease: A crossover trial. J. Neuroeng. Rehabil. 2019, 16, 66. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Lim, K.-C. Effects of a Computerized Cognitive Training on Cognitive Function, Depression, Self-esteem, and Activities of Daily Living among Older Adults with Mild Cognitive Impairment. Korean J. Adult Nurs. 2016, 28, 691–700. [Google Scholar] [CrossRef]
- Appel, L.; Appel, E.; Bogler, O.; Wiseman, M.; Cohen, L.; Ein, N.; Abrams, H.B.; Campos, J.L. Older Adults With Cognitive and/or Physical Impairments Can Benefit From Immersive Virtual Reality Experiences: A Feasibility Study. Front. Med. 2019, 6, 329. [Google Scholar] [CrossRef]
- Archer, N.; Keshavjee, K.; Demers, C.; Lee, R. Online self-management interventions for chronically ill patients: Cognitive impairment and technology issues. Int. J. Med. Inform. 2014, 83, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Dewar, B.K.; Kapur, N.; Kopelman, M. Do memory aids help everyday memory? A controlled trial of a Memory Aids Service. Neuropsychol. Rehabil. 2018, 28, 614–632. [Google Scholar] [CrossRef]
- Andreassen, M.; Boman, I.L.; Danielsson, H.; Hemmingsson, H. Digital Support for Persons with Cognitive Impairment. Stud. Health Technol. Inform. 2017, 242, 5–8. [Google Scholar] [PubMed]
- Richardson, J.T.E. Eta squared and partial eta squared as measures of effect size in educational research. Educ. Res. Rev. 2011, 6, 135–147. [Google Scholar] [CrossRef]
- Abraha, I.; Cozzolino, F.; Orso, M.; Marchesi, M.; Germani, A.; Lombardo, G.; Eusebi, P.; De Florio, R.; Luchetta, M.L.; Iorio, A.; et al. A systematic review found that deviations from intention-to-treat are common in randomized trials and systematic reviews. J. Clin. Epidemiol. 2017, 84, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Al-Durra, M.; Nolan, R.P.; Seto, E.; Cafazzo, J.A.; Eysenbach, G. Nonpublication Rates and Characteristics of Registered Randomized Clinical Trials in Digital Health: Cross-Sectional Analysis. J. Med. Internet Res. 2018, 20, e11924. [Google Scholar] [CrossRef]
- Mohr, D.C.; Riper, H.; Schueller, S.M. A Solution-Focused Research Approach to Achieve an Implementable Revolution in Digital Mental Health. JAMA Psychiatry 2018, 75, 113–114. [Google Scholar] [CrossRef]
- Torous, J.; Firth, J. The digital placebo effect: Mobile mental health meets clinical psychiatry. Lancet Psychiatry 2016, 3, 100–102. [Google Scholar] [CrossRef]
| Authors, Year, and Language of Publication | Setting and Reported Funding Source | Study Design and Sample Size | Experimental Intervention | Control or Comparison Intervention | Sample Characteristics | Outcome Measures Relevant to Self-Management | Outcome Measures Relevant to Social Participation | Outcome Measures Related to Caregivers |
|---|---|---|---|---|---|---|---|---|
| Lee et al. 2013 (English). [28] | Hong Kong. Both outpatient and inpatient; urban setting. Source of funding not reported. | Single-center, single-blinded, three-arm, randomized controlled trial (RCT). Measurements at baseline, 6 weeks, and 3 months; n = 24 (7 experimental; 6 offline training; 6 control). | Computerized errorless memory training program (CELP). | (Therapist-led errorless learning programme) TELP arm: Therapist-led memory training Control arm: waiting list. | Persons with ‘early dementia’ (Clinical Dementia Rating Scale [CDR] = 1); Mean age: 77.7 ± 6.07; 6 male, 13 female. Mean Mini Mental-State Examination (MMSE) CELP group 15.3 ± 2.7; mean MMSE TELP group 17.0 ± 3.5; mean MMSE control 17.6 ± 4.7 | Hong Kong Lawton Instrumental Activities of Daily Living Scale (HKLIADL) | None | None |
| Liao et al. 2020 (English). [29] | Taiwan. Community-dwelling, recruited through community and day care centers. Funding from Taiwan Ministry of Science and Technology. | Single-center, single-blinded, two-arm RCT. Measurements at baseline and after 12 weeks; n = 42 (21 experimental; 21 control). | Microsoft Kinect VR system, for Tai Chi, resistance, and aerobic exercises. | Combined physical and cognitive training | Older adults with MCI (MMSE ≥ 24, Montreal Cognitive Assessment [MoCA] < 26); VR group: mean age 75.5 ± 5.2, 11 female, 7 male; control group: mean age 73.1 ± 6.8; 12 female, 4 male. | Lawton Instrumental Activities of Daily Living scale (LIADL) | None | None |
| McCarron et al.2019 (English). [30] | United States of America. Described as community setting. Funding from United States National Institute on Aging to Advanced Medical Electronics. | Pilot single-center, non-blinded, two-arm RCT. Measurements at baseline, 3 months, and 6 months; n = 48 (20 experimental; 28 control). | Social Support Aid (SSA) mobile phone-based facial recognition application. | Usual care | Persons diagnosed with dementia (n = 29), persons with self-reported memory loss or concerns (n = 19). Mean age 74.90 ± 6.98; 23 male, 25 female. | None | Self-reported satisfaction with social contacts. Pleasant Events Schedule—Alzheimer’s Disease (PES-AD); Dementia Quality of Life (DQoL). | None |
| Mrakic-Sposta et al. 2018 (English). [31] | Italy. Not further described. Funding from the Italian Ministry of Education, Universities, and Research (MIUR). | Pilot single-center, non-blinded, two-arm RCT. Measurements at baseline and after 6 weeks; n = 10 (5 experimental; 5 control). | VR-based program combining aerobic exercise and cognitive training. | No treatment | Persons described as having MCI (n = 6), described as mild dementia (n = 4). Mean age: 73.3 ± 5.7 years; MMSE 23 ± 3.4; 4 male, 6 female. | Functional Activities Questionnaire (FAQ) | None | None |
| Nishihura et al. 2019 (English). [32] | Japan. Community dwelling participants recruited via day care center. Funding from the Japan Society for the Promotion of Science. | Single-centre, non-blinded, randomized cross-over study. Measurements at baseline, after 12 weeks, and after 24 weeks; n = 27 (15 experimental; 12 control). | Electric calendar software application for Android tablet. | Waiting list | Alzheimer’s disease (n = 12), cerebrovascular dementia (n = 5), senile dementia (n = 2), alcoholic dementia (n = 1), healthy older persons (n = 7). Mean age: 81.5 ± 6.9 years; mean MMSE 22 ± 4.0; 9 male, 18 female. | Semi-structured interviews with participants and their caregivers. | None | None |
| Park and Park 2018 (English). [33] | Korea. Community dwelling participants recruited via community welfare centers. No external funding received. | Single-blinded RCT. Measurements at baseline and after 10-week intervention; n = 78 (39 experimental; 39 control) | Non-specific computer training (NCT) with Nintendo Wii sports games. | Cognition-specific computer training (CCT) using the CoTras program. | MCI (MMSE NCT 26.41 ± 1.94, CCT group 26.67 ± 1.68); NCT group age 66.95 ± 4.10, CCT age 67.64 ± 4.55. 42 male, 36 female (NCT 20 male, 19 female; CCT 22 male, 17 female) | 36-Item Short-Form Health Survey (SF-36) 1 | SF-36 2 | |
| Pietilä et al. 2017 (Finnish). [34] | Finland. Community dwelling participants. Funding from the Miina Sillanpää Foundation. | Single-center, single-blinded, two-arm, RCT. Measurements at baseline, 13 weeks, and 6 months; n = 53 (28 experimental; 25 control) | FORAMEN rehab program, delivered by tablet computer. | Waiting list | Persons with early stage “mild” Alzheimer’s disease. Mean age 69.0 ± 5.0; 28 male, 27 female. Mean MMSE experimental group: 23.1 ± 3.8. Mean MMSE control group: 20.9 ± 3.8 | Alzheimer’s Disease Cooperative Study Activities of Daily Living Scale (ADCS-ADL) | None | Beck Depression Inventory (BDI-II); World Health Organization Quality of Life BREF (WHOQOL-BREF); Care of Older People in Europe (COPE-index) |
| Silva et al. 2017 (English). [35] | United Kingdom. Community dwelling, recruited through day care centers. Funding from Region of Bourgogne (FABER); Fondation Médéric; and the Portuguese Foundation for Science and Technology. | Single-center, single-blinded, three-arm RCT. Measurements at baseline, after 6 weeks, and 6 months; n = 51 (17 experimental; 17 offline memory support; 17 control). | SenseCam wearable camera. | Memo+ arm: a pencil and paper cognitive training program. Control arm: daily activities diary | Diagnosed probable Alzheimer’s disease. Sensecam group mean age: 75.41 ± 5.26; mean MMSE 21.88 ± 3.33 Memo + group mean age: 71.71 ± 5.15; mean MMSE 21.53 ± 3.01 Control group: mean age 73.82 ± 5.74; mean MMSE 22.82 ± 1.85 | Adults and Older Adults Functional Assessment Inventory (IAFAI). | World Health Organization Quality of Life OLD (WHOQOL-OLD) 3 | None |
| Vanoh et al. 2019 (English). [36] | Malaysia. Community dwelling participants. Funding from the Ministry of Higher Education Malaysia. | Single-center, single-blinded, two-arm, RCT. Measurements at baseline and 6 months; n = 60 (30 experimental; 30 control). | WESIHAT 2.0© web-based wellness application. | Dietary counselling based on the “Healthy Eating” pamphlet produced by Ministry of Health Malaysia | Older adults with MMSE 16–28. Mean MMSE WESIHAT group 28.3 ± 1.78; mean MMSE control group 27.0 ± 2.63; mean age: 67.84 ± 5.65; 21 male, 29 female | World Health Organization Disability Assessment Scale (WHODAS 2.0) 12-item version 4 | Medical Outcome Social Support Survey (MOSS); WHODAS 2.0 12-item version 5; three-item loneliness scale. | None |
| Authors | WoE A *, Intrinsic Methodological Quality (Good, Fair, Poor) | WoE B, Appropriateness of Method in Context of this Review (High, Fair, Low) | WoE C, Relevance of Study to this Review Question (High, Fair, Low) | WoE D, Overall Assessment (High, Fair, Low) |
|---|---|---|---|---|
| Lee et al. [28] | Poor | Fair | Low | Low |
| Liao et al. [29] | Fair | Fair | Low | Fair |
| McCarron et al. [30] | Fair | Fair | Low | Fair |
| Mrakic-Sposta et al. [31] | Fair | Fair | Low | Fair |
| Nishihura et al. [32] | Poor | Low | Low | Low |
| Park and Park. [33] | Good | Low | Low | Fair |
| Pietilä et al. [34] | Fair | Fair | Low | Fair |
| Silva et al. [35] | Fair | High | Low | Fair |
| Vanoh et al. [36] | Fair | Fair | Low | Fair |
| Authors | Outcome Measure | Results |
|---|---|---|
| Lee et al. [28] | HK-LIADL Scale (self-management) | Effect of intervention not statistically significant |
| Liao et al. [29] | LIADL (self-management) | Significant group and group by time interaction effects, in favor of the experimental group (group, p < 0.001, ƞ2p = 0.87; interaction, p < 0.01, ƞ2p = 0.217). |
| McCarron et al. [30] | PES-AD (social participation) | Effect of intervention not statistically significant |
| DQoL (social participation) | Effect of intervention not statistically significant | |
| Mrakic-Sposta et al. [31] | FAQ (self-management) | Effect of intervention not statistically significant |
| Nishihura et al. [32] | Semi-structured interviews (self-management) | SSI results not fully reported. Summary statement that positive self-management behavior changes were observed by caregivers in intervention group |
| Park and Park. [33] | SF-36 (self-management and social participation) | Role-emotional subscale: Significant group-by-time effect in favor of NCT (4.18 (95%CI 3.72 to 4.63), ƞ2p = 0.821 Role-physical subscale: Effect of intervention not statistically significant Social functioning subscale: Effect of intervention not statistically significant |
| Pietilä et al. [34] | ADCS-ADL (self-management) | Effect of intervention not statistically significant |
| BDI-II (caregiver) | Effect of intervention not statistically significant | |
| WHOQOL-BREF (caregiver) | Effect of intervention not statistically significant | |
| COPE index (caregiver) | Subscales analyzed separately. Significant improvement in intervention group vs. control with respect to positive attitudes (p = 0.023). Effect size not reported. No statistically significant differences on other subscales. | |
| Silva et al. [35] | IAFAI (self-management) | Global scores: statistically significant visit effect (F[2,43] = 16.26, p < 0.01, ƞ2p = 0.28) and group by visit interaction (F[2,43] = 8.71, p < 0.01, ƞ2p = 0.29). IADL familiar subscale: statistically significant visit effect (F[2,43] = 5.31, p < 0.01, ƞ2p = 0.11) and group by visit interaction (F[2,43] = 5.40, p < 0.01, ƞ2p = 0.21). IADL advanced subscale: statistically significant visit effect (F[2,43] = 11.74, p < 0.01, ƞ2p = 0.22) and group by visit interaction (F[2,43] = 4.83, p < 0.01, ƞ2p = 0.19). All reported significant effects demonstrated improvements in intervention group vs control |
| WHOQOL-OLD * (social participation) | No subscale analyses for social participation reported. Overall, effect of intervention not statistically significant | |
| Vanoh et al. [36] | WHODAS 2.0 (self-management and social participation) | No subscale analyses for social participation and self-management reported. Significant group, time, and interaction effects, in favor of the intervention group (group, p < 0.01, ƞ2p = 0.341; Time, p < 0.05, ƞ2p = 0.128; interaction p < 0.01, ƞ2p = 0.191) |
| MOSS (social participation) | The four subscales were analyzed separately. Significant group by time interaction effects for informational support (p < 0.05, ƞ2p = 0.123) and tangible support (p < 0.01, ƞ2p = 0.186). No statistically significant group, time, or interaction effects with respect to positive social interaction or affective support | |
| Three-item loneliness scale (social participation) | Significant effect of group (p < 0.05, ƞ2p = 0.184) but no statistically significant time or interaction effects |
| Authors | Statements Concerning Facilitators in Implementing Intervention | Classification (Implementation, Mechanism of Impact, Context) | Statements Concerning Barriers in Implementing Intervention | Classification (Implementation, Mechanism of Impact, Context) |
|---|---|---|---|---|
| Lee et al. [28] | Qualitative feedback from participants showed that they enjoyed and liked the memory training program. They considered that learning to use a computer was not difficult. They would recommend the training program to others. | Mechanism of impact | More regular stimulating positive feedback could be integrated into the training program. It is suggested to increase the number of training sessions from 12 to 15. | Implementation |
| Liao et al. [29] | The enjoyment and attractiveness of VR characteristics may increase motivation and lead to extensive training effects, resulting in cognitive improvement. | Mechanism of impact | None identified | Not applicable (N.A.) |
| McCarron et al. [30] | None identified | N.A. | The majority of participants did not find the technology useful. Reasons: (1) complexity of the SSA, (2) enrollment process, (3) impracticality, (4) stigma, and (5) functionality of the SSA. | Mechanism of impact |
| Mrakic-Sposta et al. [31] | None identified | N.A. | None identified | N.A. |
| Nishihura et al. [32] | Eleven of the fourteen participants received sufficient support from their main caregiver. For instance, the caregiver visited them more than once a week to maintain the electric calendar. | Context | Some negative comments about the lack of durability of the battery and difficulties inputting the schedule. Three participants mentioned they did not need it in their daily lives. | Mechanism of impact |
| Feedback interviews revealed that most participants had positive impressions of using the electric calendar. | Mechanism of impact | |||
| Park and Park [33] | NCT can facilitate the training in a fun manner. Thus, NCT might motivate subjects more efficiently as compared to CCT, which may decrease issues with intervention compliance. | Mechanism of Impact | CCT had to be provided at a healthcare facility. Both training groups only received training individually, without opportunity for social interaction. | Implementation |
| Pietilä et al. [34] | None identified | N.A. | None identified | N.A. |
| Silva et al. [35] | SenseCam does not provoke negative evaluations, but suggests a beneficial action on mood. Authors refer to research suggesting that wearable cameras are less demanding than other external aids and are also less demanding and more motivating than paper and pencil cognitive exercises. | Mechanism of impact | None identified | N.A. |
| Vanoh et al. [36] | Group discussion was conducted to explain the proper ways to use the website, to describe the content in WESIHAT 2.0, and to identify the problems faced by subjects. | Implementation | Difficulties were reported by the intervention group in dealing with unknown people due to the fear of being deceived. | Implementation |
| The counselling session was made interesting by having a quiz related to the content of WESIHAT 2.0 and playing free online brain games. | Mechanism of impact | The use of the compute requires complex motor functioning, language processing, and focus. | Mechanism of impact |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neal, D.; van den Berg, F.; Planting, C.; Ettema, T.; Dijkstra, K.; Finnema, E.; Dröes, R.-M. Can Use of Digital Technologies by People with Dementia Improve Self-Management and Social Participation? A Systematic Review of Effect Studies. J. Clin. Med. 2021, 10, 604. https://doi.org/10.3390/jcm10040604
Neal D, van den Berg F, Planting C, Ettema T, Dijkstra K, Finnema E, Dröes R-M. Can Use of Digital Technologies by People with Dementia Improve Self-Management and Social Participation? A Systematic Review of Effect Studies. Journal of Clinical Medicine. 2021; 10(4):604. https://doi.org/10.3390/jcm10040604
Chicago/Turabian StyleNeal, David, Floor van den Berg, Caroline Planting, Teake Ettema, Karin Dijkstra, Evelyn Finnema, and Rose-Marie Dröes. 2021. "Can Use of Digital Technologies by People with Dementia Improve Self-Management and Social Participation? A Systematic Review of Effect Studies" Journal of Clinical Medicine 10, no. 4: 604. https://doi.org/10.3390/jcm10040604
APA StyleNeal, D., van den Berg, F., Planting, C., Ettema, T., Dijkstra, K., Finnema, E., & Dröes, R.-M. (2021). Can Use of Digital Technologies by People with Dementia Improve Self-Management and Social Participation? A Systematic Review of Effect Studies. Journal of Clinical Medicine, 10(4), 604. https://doi.org/10.3390/jcm10040604








