Current Knowledge and Future Challenges in Takotsubo Syndrome: Part 2—Treatment and Prognosis
Abstract
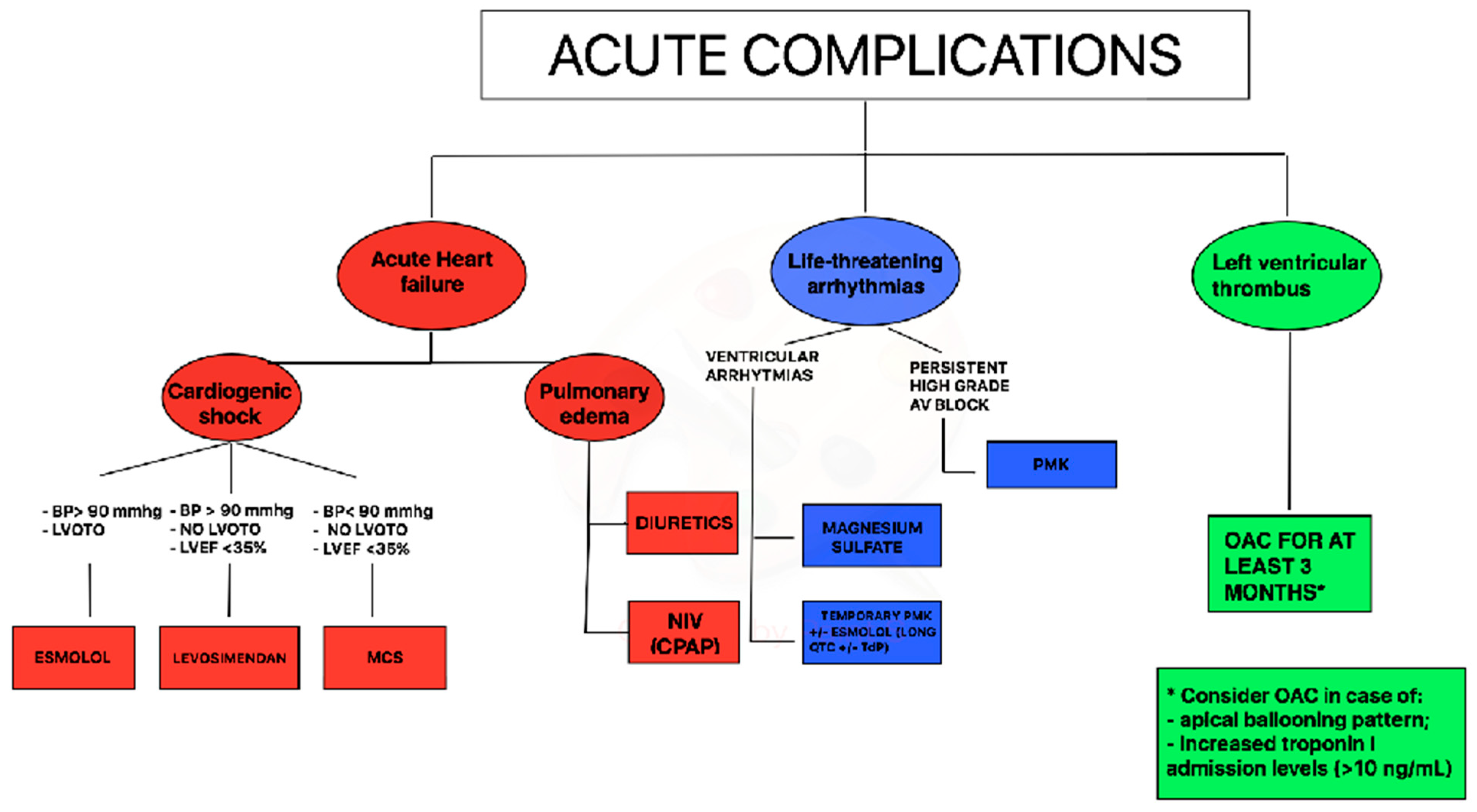
1. Acute Treatment in Takotsubo Syndrome (TTS)
1.1. Treatment of Acute Heart Failure in TTS
1.2. Treatment of Arrhythmias in TTS
1.3. Treatment of Thromboembolic Events in TTS
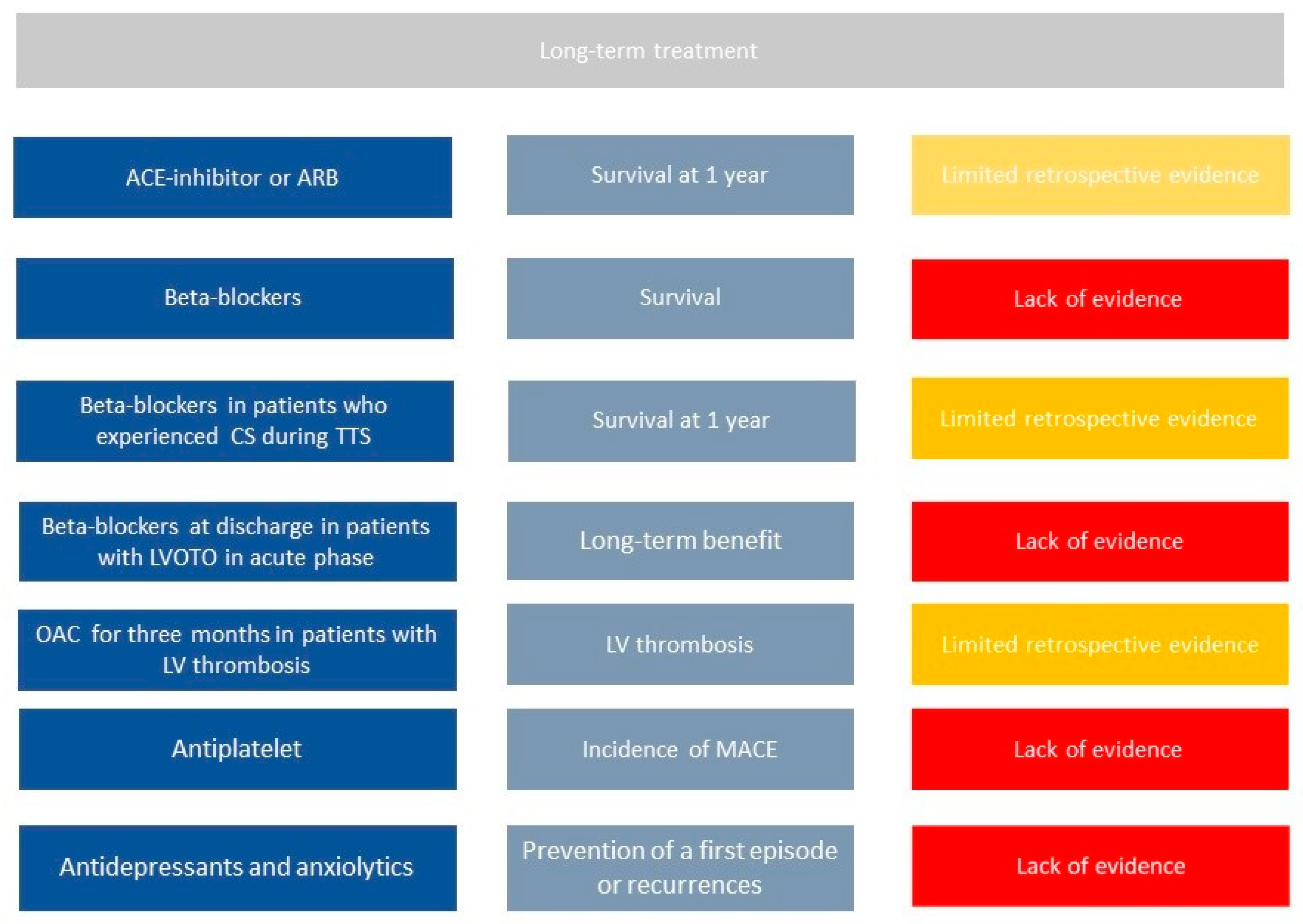
2. Chronic Treatment in TTS
3. Short Term and Long-Term Prognosis
3.1. Short Term Prognosis
3.2. Long Term Prognosis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Almendro-Delia, M.; Núñez-Gil, I.J.; Lobo, M.; Andrés, M.; Vedia, O.; Sionis, A.; Martin-García, A.; Cruz Aguilera, M.; Pereyra, E.; Martín de Miguel, I.; et al. Short- and Long-Term Prognostic Relevance of Cardiogenic Shock in Takotsubo Syndrome: Results from the RETAKO Registry. JACC Heart Fail. 2018, 6, 928–936. [Google Scholar] [CrossRef]
- Santoro, F.; Ferraretti, A.; Ieva, R.; Musaico, F.; Fanelli, M.; Tarantino, N.; Scarcia, M.; Caldarola, P.; Di Biase, M.; Brunetti, N.D. Renal impairment and outcome in patients with takotsubo cardiomyopathy. Am. J. Emerg. Med. 2016, 34, 548–552. [Google Scholar] [CrossRef]
- Templin, C.; Ghadri, J.R.; Diekmann, J.; Napp, L.C.; Bataiosu, D.R.; Jaguszewski, M.; Lüscher, T.F. Clinical features and outcomes of Takotsubo (stress)cardiomyopathy. N. Engl. J. Med. 2015, 373, 929–938. [Google Scholar] [CrossRef]
- Santoro, F.; Gil, I.J.N.; Stiermaier, T.; El-Battrawy, I.; Guerra, F.; Novo, G.; Guastafierro, F.; Tarantino, N.; Novo, S.; Mariano, E.; et al. Assessment of the German and Italian Stress Cardiomyopathy Score for Risk Stratification for In-hospital Complications in Patients with Takotsubo Syndrome. JAMA Cardiol. 2019, 4, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, N.D.; Santoro, F.; De Gennaro, L.; Correale, M.; Kentaro, H.; Gaglione, A.; Di Biase, M. Therapy of stress cardiomyopathy: Present shortcoming and future perspectives. Future Cardiol. 2016, 12, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Santoro, F.; Ieva, R.; Musaico, F.; Ferraretti, A.; Triggiani, G.; Tarantino, N.; Di Biase, M.; Brunetti, N.D. Lack of efficacy of drug therapy in preventing Takotsubo cardiomyopathy recurrence: A meta-analysis. Clin. Cardiol. 2014, 37, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Isogai, T.; Matsui, H.; Tanaka, H.; Fushimi, K.; Yasunaga, H. Early β-blocker use and in-hospital mortality in patients with Takotsubo cardiomyopathy. Heart 2016, 102, 1029–1035. [Google Scholar] [CrossRef] [PubMed]
- Paur, H.; Wright, P.T.; Sikkel, M.B.; Tranter, M.H.; Mansfield, C.; O’Gara, P.; Stuckey, D.J.; Nikolaev, V.O.; Diakonov, I.; Pannell, L.; et al. High levels of circulating epinephrine trigger apical cardiodepression in a β2-adrenergic receptor/Gi-dependent manner: A new model of Takotsubo cardiomyopathy. Circulation 2012, 126, 697–706. [Google Scholar] [CrossRef]
- Santoro, F.; Ieva, R.; Ferraretti, A.; Fanelli, M.; Musaico, F.; Tarantino, N.; Di Martino, L.; De Gennaro, L.; Caldarola, P.; Di Biase, M.; et al. Hemodynamic effects, safety, and feasibility of intravenous esmolol infusion during Takotsubo cardiomyopathy with left ventricular outflow tract obstruction: Results from a multicenter registry. Cardiovasc. Ther. 2016, 34, 161–166. [Google Scholar] [CrossRef]
- Akashi, Y.J.; Nef, H.M.; Lyon, A.R. Epidemiology and pathophysiology of Takotsubo syndrome. Nat. Rev. Cardiol. 2015, 12, 387–397. [Google Scholar] [CrossRef]
- Sharkey, S.W.; Windenburg, D.C.; Lesser, J.R.; Maron, M.S.; Hauser, R.G.; Lesser, J.N.; Haas, T.S.; Hodges, J.S.; Maron, B.J. Natural history and expansive clinical profile of stress (Takotsubo) cardiomyopathy. J. Am. Coll. Cardiol. 2010, 55, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Schneider, B.; Athanasiadis, A.; Schwab, J.; Pistner, W.; Gottwald, U.; Schoeller, R.; Toepel, W.; Winter, K.-D.; Stellbrink, C.; Müller-Honold, T.; et al. Complications in the clinical course of Takotsubo cardiomyopathy. Int. J. Cardiol. 2014, 176, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Redmond, M.; Knapp, C.; Salim, M.; Shanbhag, S.; Jaumdally, R. Use of vasopressors in Takotsubo cardiomyopathy: A cautionary tale. Br. J. Anaesth. 2013, 110, 487–488. [Google Scholar] [CrossRef] [PubMed]
- Ansari, U.; El-Battrawy, I.; Fastner, C.; Behnes, M.; Sattler, K.; Huseynov, A.; Baumann, S.; Tülümen, E.; Borggrefe, M.; Akin, I. Clinical outcomes associated with catecholamine use in patients diagnosed with Takotsubo cardiomyopathy. BMC Cardiovasc. Disord. 2018, 18, 54. [Google Scholar] [CrossRef] [PubMed]
- Lyon, A.R.; Bossone, E.; Schneider, B.; Sechtem, U.; Citro, R.; Underwood, S.R.; Sheppard, M.N.; Figtree, G.A.; Parodi, G.; Akashi, Y.J.; et al. Current state of knowledge on takotsubo syndrome: A position statement from the task force on Takotsubo syndrome of the Heart Failure Association of the European Society of Cardiology. Eur. Heart J. Heart Fail. 2016, 18, 8–27. [Google Scholar] [CrossRef]
- Yaman, M.; Arslan, U.; Kaya, A.; Akyol, A.; Ozturk, F.; Okudan, Y.E.; Bayramoglu, A.; Bektas, O. Levosimendan accelerates recovery in patients with takotsubo cardiomyopathy. Cardiol. J. 2016, 23, 610–615. [Google Scholar] [CrossRef]
- Santoro, F.; Ieva, R.; Ferraretti, A.; Ienco, V.; Carpagnano, G.; Lodispoto, M.; Di Biase, L.; Di Biase, M.; Brunetti, N.D. Safety and feasibility of levosimendan administration in takotsubo cardiomyopathy: A case series. Cardiovasc. Ther. 2013, 31, e133–e137. [Google Scholar] [CrossRef]
- Flevari, P.; Parissis, J.T.; Leftheriotis, D.; Panou, F.; Kourea, K.; Kremastinos, D.T. Effect of levosimendan on ventricular arrhythmias and prognostic autonomic indexes in patients with decompensated advanced heart failure secondary to ischemic or dilated cardiomyopathy. Am. J. Cardiol. 2006, 98, 1641–1645. [Google Scholar] [CrossRef]
- Dias, A.; Núñez Gil, I.J.; Santoro, F.; Madias, J.E.; Pelliccia, F.; Brunetti, N.D.; Salmoirago-Blotcher, E.; Sharkey, S.W.; Eitel, I.; Akashi, Y.J.; et al. Takotsubo syndrome: State-of-the-art review by an expert panel—Part 2. Cardiovasc. Revasc. Med. 2019, 20, 153–166. [Google Scholar] [CrossRef]
- Santoro, F.; Nunez-Gil, I.-J.; Stiermaier, T.; El-Battrawy, I.; Guerra, F.; Arcari, L.; Musumeci, B.; Novo, G.; Cannone, M.; Almendro, M.; et al. Intra-aortic balloon counterpulsation in takotsubo syndrome complicated by cardiogenic shock: Short- and long-term results from a cohort of 2250 patients of the German-Italian-Spanish registry. Eur. Heart J. 2019, 40, ehz745.0545. [Google Scholar] [CrossRef]
- Mariani, S.; Richter, J.; Pappalardo, F.; Bělohlávek, J.; Lorusso, R.; Schmitto, J.D.; Bauersachs, J.; Napp, L.C. Mechanical circulatory support for Takotsubo syndrome: A systematic review and meta-analysis. Int. J. Cardiol. 2020, 316, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Madias, C.; Fitzgibbons, T.P.; Alsheikh-Ali, A.A.; Bouchard, J.L.; Kalsmith, B.; Garlitski, A.C.; Tighe, D.A.; Estes, N.M.; Aurigemma, G.P.; Link, M.S. Acquired long QT syndrome from stress cardiomyopathy is associated with ventricular arrhythmias and torsades de pointes. Heart Rhythm. 2011, 8, 555–561. [Google Scholar] [CrossRef] [PubMed]
- El-Battrawy, I.; Santoro, F.; Stiermaier, T.; Möller, C.; Guastafierro, F.; Novo, G.; Novo, S.; Santangelo, A.; Mariano, E.; Romeo, F.; et al. Prevalence, management, and out- come of adverse rhythm disorders in takotsubo syndrome: Insights from the international multicenter GEIST registry. Heart Fail Rev. 2020, 25, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Gili, S.; Cammann, V.L.; Schlossbauer, S.A.; Kato, K.; D’Ascenzo, F.; Di Vece, D.; Jurisic, S.; Micek, J.; Obeid, S.; Bacchi, B.; et al. Cardiac arrest in takotsubo syndrome: Results from the InterTAK Registry. Eur. Heart J. 2019, 40, 2142–2151. [Google Scholar] [CrossRef] [PubMed]
- Santoro, F.; Brunetti, N.D.; Tarantino, N.; Romero, J.; Guastafierro, F.; Ferraretti, A.; Di Martino, L.F.M.; Ieva, R.; Pellegrino, P.L.; Di Biase, M.; et al. Dynamic changes of QTc interval and prognostic significance in takotsubo (stress) cardiomyopathy. Clin. Cardiol. 2017, 40, 1116–1122. [Google Scholar] [CrossRef] [PubMed]
- Stiermaier, T.; Rommel, K.P.; Eitel, C.; Möller, C.; Graf, T.; Desch, S.; Thiele, H.; Eitel, I. Management of arrhythmias in patients with Takotsubo cardiomyopathy: Is the implantation of permanent devices necessary? Heart Rhythm. 2016, 13, 1979–1986. [Google Scholar] [CrossRef]
- El-Battrawy, I.; Erath, J.W.; Lang, S.; Ansari, U.; Behnes, M.; Gietzen, T.; Zhou, X.; Borggrefe, M.; Akin, I. Takotsubo syndrome and cardiac implantable electronic device therapy. Sci. Rep. 2019, 9, 16559. [Google Scholar] [CrossRef]
- Santoro, F.; Stiermaier, T.; Tarantino, N.; De Gennaro, L.; Moeller, C.; Guastafierro, F.; Marchetti, M.F.; Montisci, R.; Carapelle, E.; Graf, T.; et al. Left ventricular thrombi in Takotsubo syndrome: Incidence, predictors, and management: Results from the GEIST (German Italian Stress Cardiomyopathy) Registry. J. Am. Heart Assoc. 2017, 6, e006990. [Google Scholar] [CrossRef]
- El-Battrawy, I.; Gietzen, T.; Lang, S.; Ansari, U.; Behnes, M.; Zhou, X.; Borggrefe, M.; Akin, I. Short- and Long-Term Incidence of Thromboembolic Events in Takotsubo Syndrome as Compared With Acute Coronary Syndrome. Angiology 2019, 70, 838–843. [Google Scholar] [CrossRef]
- Ding, K.J.; Cammann, V.L.; Szawan, K.A.; Stähli, B.E.; Wischnewsky, M.; Di Vece, D.; Citro, R.; Jaguszewski, M.; Seifert, B.; Sarcon, A.; et al. Intraventricular Thrombus Formation and Embolism in Takotsubo Syndrome: Insights From the International Takotsubo Registry. Arter. Thromb. Vasc. Biol. 2020, 40, 279–287. [Google Scholar] [CrossRef]
- D’Ascenzo, F.; Gili, S.; Bertaina, M.; Iannaccone, M.; Cammann, V.L.; Di Vece, D.; Kato, K.; Saglietto, A.; Szawan, K.A.; Frangieh, A.H.; et al. Impact of aspirin on takotsubo syndrome: A propensity score-based analysis of the InterTAK Registry. Eur. J. Heart Fail. 2020, 22, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Eitel, I.; Stiermaier, T.; Graf, T.; Möller, C.; Rommel, K.P.; Eitel, C.; Schuler, G.; Thiele, H.; Desch, S. Optical Coherence Tomography to Evaluate Plaque Burden and Morphology in Patients With Takotsubo Syndrome. J. Am. Heart Assoc. 2016, 5, e004474. [Google Scholar] [CrossRef] [PubMed]
- Lüscher, T.F.; Templin, C. Is takotsubo syndrome a microvascular acute coronary syndrome? Towards of a new definition. Eur. Heart J. 2016, 37, 2816–2820. [Google Scholar] [CrossRef] [PubMed]
- El-Battrawy, I.; Santoro, F.; Stiermaier, T.; Möller, C.; Guastafierro, F.; Novo, G.; Novo, S.; Mariano, E.; Romeo, F.; Romeo, F.; et al. Incidence and Clinical Impact of Recurrent Takotsubo Syndrome: Results From the GEIST Registry. J. Am. Heart Assoc. 2019, 8, e010753. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, N.D.; Santoro, F.; De Gennaro, L.; Correale, M.; Gaglione, A.; Di Biase, M. Drug treatment rates with beta-blockers and ACE-inhibitors/angiotensin receptor blockers and recurrences in takotsubo cardiomyopathy: A meta-regression analysis. Int. J. Cardiol. 2016, 214, 340–342. [Google Scholar] [CrossRef]
- Brunetti, N.D.; Santoro, F.; De Gennaro, L.; Correale, M.; Gaglione, A.; Di Biase, M.; Madias, J.E. Combined therapy with beta-blockers and ACE-inhibitors/angiotensin receptor blockers and recurrence of Takotsubo (stress) cardiomyopathy: A meta-regression study. Int. J. Cardiol. 2017, 230, 281–283. [Google Scholar] [CrossRef]
- Waldenborg, M.; Soholat, M.; Kähäri, A.; Emilsson, K.; Fröbert, O. Multidisciplinary assessment of tako tsubo cardiomyopathy: A prospective case study. BMC Cardiovasc. Disord. 2011, 11, 14. [Google Scholar] [CrossRef]
- Hovgaard, H.L.; Zaremba, T.; Aaroe, J. Relapsing Classical Takotsubo Syndrome in a Postmenopausal Woman Successfully Managed with Psychology Consultations. Cureus 2019, 11, e5361. [Google Scholar] [CrossRef]
- Stiermaier, T.; Santoro, F.; Eitel, C.; Graf, T.; Möller, C.; Tarantino, N.; Guastafierro, F.; Di Biase, M.; Thiele, H.; Brunetti, N.D.; et al. Prevalence and prognostic relevance of atrial fibrillation in patients with Takotsubo syndrome. Int. J. Cardiol. 2017, 245, 156–161. [Google Scholar] [CrossRef]
- Santoro, F.; Stiermaier, T.; Tarantino, N.; Guastafierro, F.; Graf, T.; Möller, C.; Di Martino, L.F.M.; Thiele, H.; Di Biase, M.; Eitel, I.; et al. Impact of persistent ST elevation on outcome in patients with Takotsubo syndrome. Results from the GErman Italian STress Cardiomyopathy (GEIST) registry. Int. J. Cardiol. 2018, 255, 140–144. [Google Scholar] [CrossRef]
- Stiermaier, T.; Santoro, F.; Graf, T.; Guastafierro, F.; Tarantino, N.; De Gennaro, L.; Caldarola, P.; Di Biase, M.; Thiele, H.; Brunetti, N.D.; et al. Prognostic value of N-Terminal Pro-B-Type Natriuretic Peptide in Takotsubo syndrome. Clin. Res. Cardiol. 2018, 107, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Stiermaier, T.; Santoro, F.; El-Battrawy, I.; Möller, C.; Graf, T.; Novo, G.; Santangelo, A.; Mariano, E.; Romeo, F.; Caldarola, P.; et al. Prevalence and Prognostic Impact of Diabetes in Takotsubo Syndrome: Insights From the International, Multicenter GEIST Registry. Diabetes Care 2018, 41, 1084–1088. [Google Scholar] [CrossRef] [PubMed]
- Citro, R.; Rigo, F.; D’Andrea, A.; Ciampi, Q.; Parodi, G.; Provenza, G.; Piccolo, R.; Mirra, M.; Zito, C.; Giudice, R.; et al. Echocardiographic correlates of acute heart failure, cardiogenic shock, and in-hospital mortality in tako-tsubo cardiomyopathy. JACC Cardiovasc. Imaging 2014, 7, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Kagiyama, N.; Okura, H.; Tamada, T.; Imai, K.; Yamada, R.; Kume, T.; Hayashida, A.; Neishi, Y.; Kawamoto, T.; Yoshida, K. Impact of right ventricular involvement on the prognosis of takotsubo cardiomyopathy. Eur. Heart J. Cardiovasc. Imaging 2016, 17, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Stiermaier, T.; Moeller, C.; Oehler, K.; Desch, S.; Graf, T.; Eitel, C.; Vonthein, R.; Schuler, G.; Thiele, H.; Eitel, I. Long-term excess mortality in takotsubo cardiomyopathy: Predictors, causes and clinical consequences. Eur. J. Heart Fail. 2016, 18, 650–656. [Google Scholar] [CrossRef]
- Ghadri, J.R.; Kato, K.; Cammann, V.L.; Gili, S.; Jurisic, S.; di Vece, D.; Candreva, A.; Ding, K.J.; Micek, J.; Szawan, K.A.; et al. Long-Term Prognosis of Patients with Takotsubo Syndrome. J. Am. Coll. Cardiol. 2018, 72, 874–882. [Google Scholar] [CrossRef]
- Uribarri, A.; Núñez-Gil, I.J.; Conty, D.A.; Vedia, O.; Almendro-Delia, M.; Cambra, A.D.; Martin-Garcia, A.C.; Barrionuevo-Sánchez, M.; Martínez-Sellés, M.; Raposeiras-Roubín, S.; et al. Short- and Long-Term Prognosis of Patients with Takotsubo Syndrome Based on Different Triggers: Importance of the Physical Nature. J. Am. Heart Assoc. 2019, 8, e013701. [Google Scholar] [CrossRef]
- Kato, K.; Di Vece, D.; Cammann, V.L.; Micek, J.; Szawan, K.A.; Bacchi, B.; Lüscher, T.F.; Ruschitzka, F.; Ghadri, J.R.; Templin, C. InterTAK Collaborators. Takotsubo Recurrence: Morphological Types and Triggers and Identification of Risk Factors. J. Am. Coll. Cardiol. 2019, 73, 982–984. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santoro, F.; Mallardi, A.; Leopizzi, A.; Vitale, E.; Rawish, E.; Stiermaier, T.; Eitel, I.; Brunetti, N.D. Current Knowledge and Future Challenges in Takotsubo Syndrome: Part 2—Treatment and Prognosis. J. Clin. Med. 2021, 10, 468. https://doi.org/10.3390/jcm10030468
Santoro F, Mallardi A, Leopizzi A, Vitale E, Rawish E, Stiermaier T, Eitel I, Brunetti ND. Current Knowledge and Future Challenges in Takotsubo Syndrome: Part 2—Treatment and Prognosis. Journal of Clinical Medicine. 2021; 10(3):468. https://doi.org/10.3390/jcm10030468
Chicago/Turabian StyleSantoro, Francesco, Adriana Mallardi, Alessandra Leopizzi, Enrica Vitale, Elias Rawish, Thomas Stiermaier, Ingo Eitel, and Natale D. Brunetti. 2021. "Current Knowledge and Future Challenges in Takotsubo Syndrome: Part 2—Treatment and Prognosis" Journal of Clinical Medicine 10, no. 3: 468. https://doi.org/10.3390/jcm10030468
APA StyleSantoro, F., Mallardi, A., Leopizzi, A., Vitale, E., Rawish, E., Stiermaier, T., Eitel, I., & Brunetti, N. D. (2021). Current Knowledge and Future Challenges in Takotsubo Syndrome: Part 2—Treatment and Prognosis. Journal of Clinical Medicine, 10(3), 468. https://doi.org/10.3390/jcm10030468






