Cold Ablation Robot-Guided Laser Osteotome (CARLO®): From Bench to Bedside
Abstract
1. Introduction
2. Methods and Results
2.1. Ethics
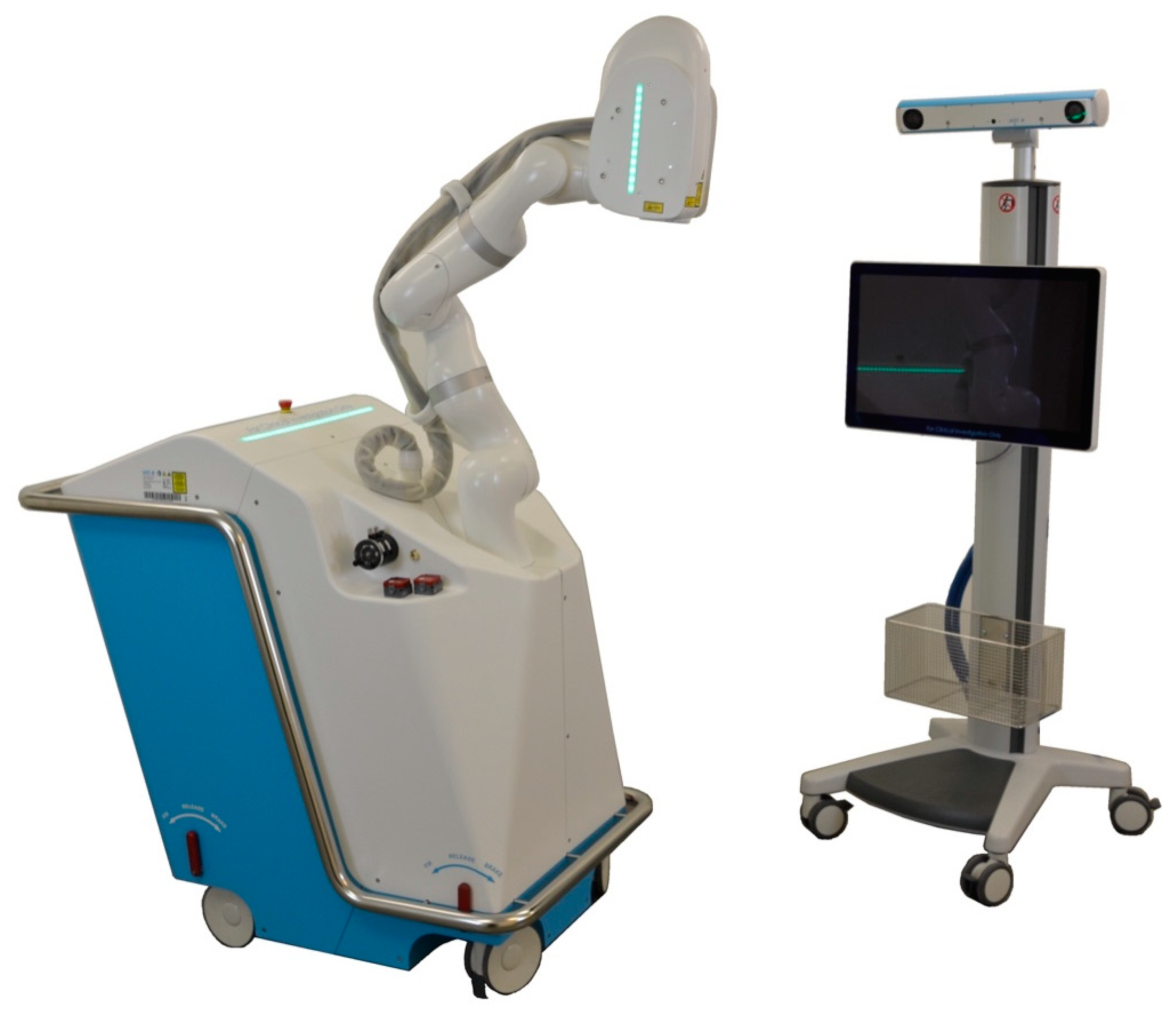
2.2. CARLO® Device
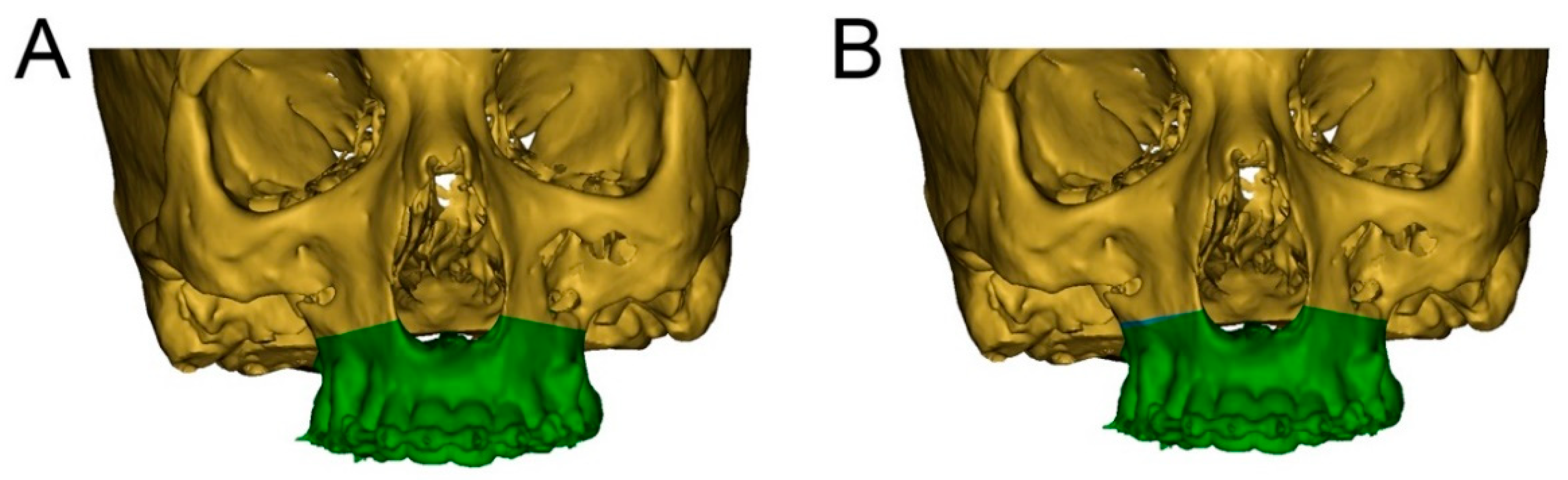
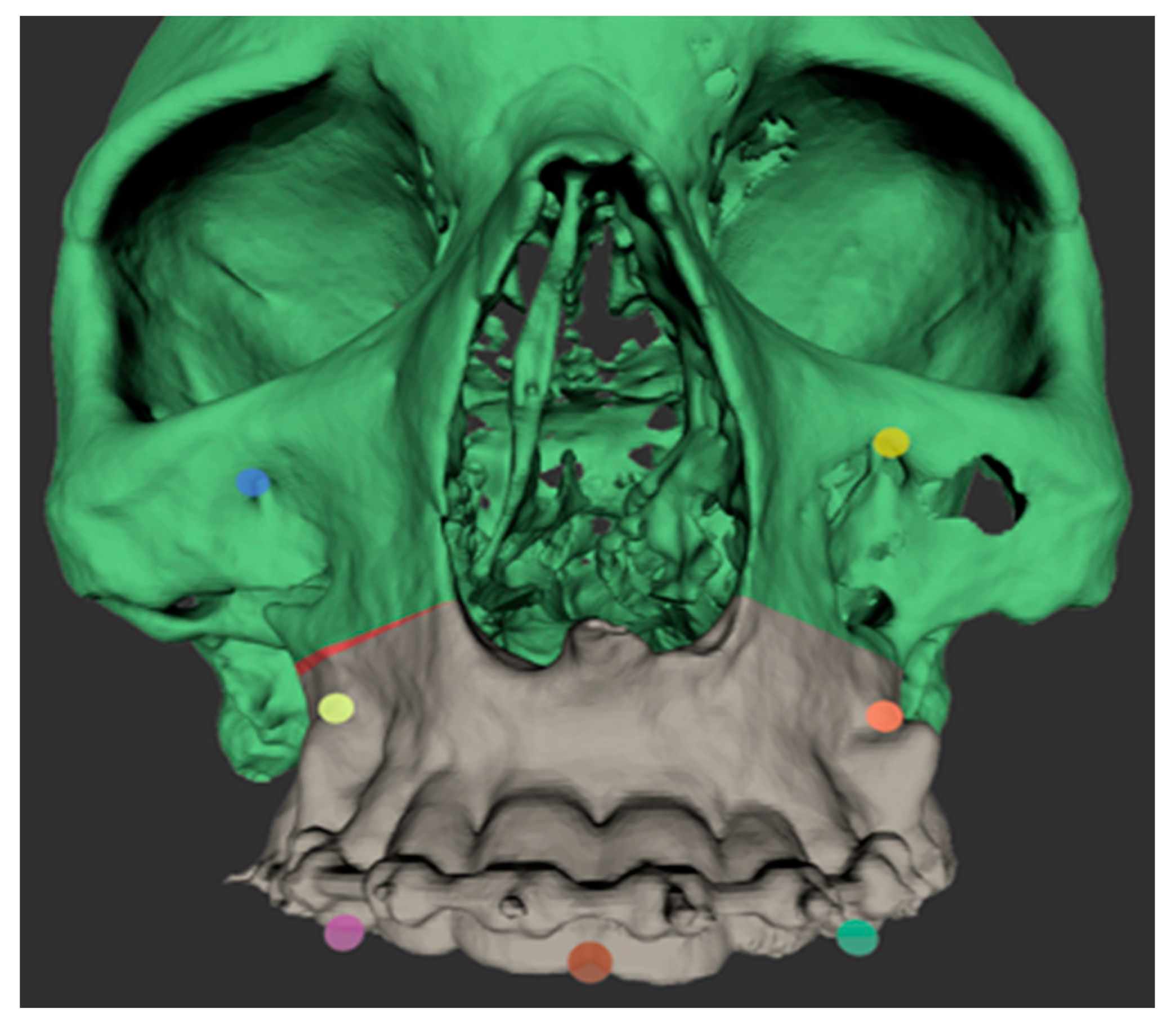
2.3. Digital Workflow
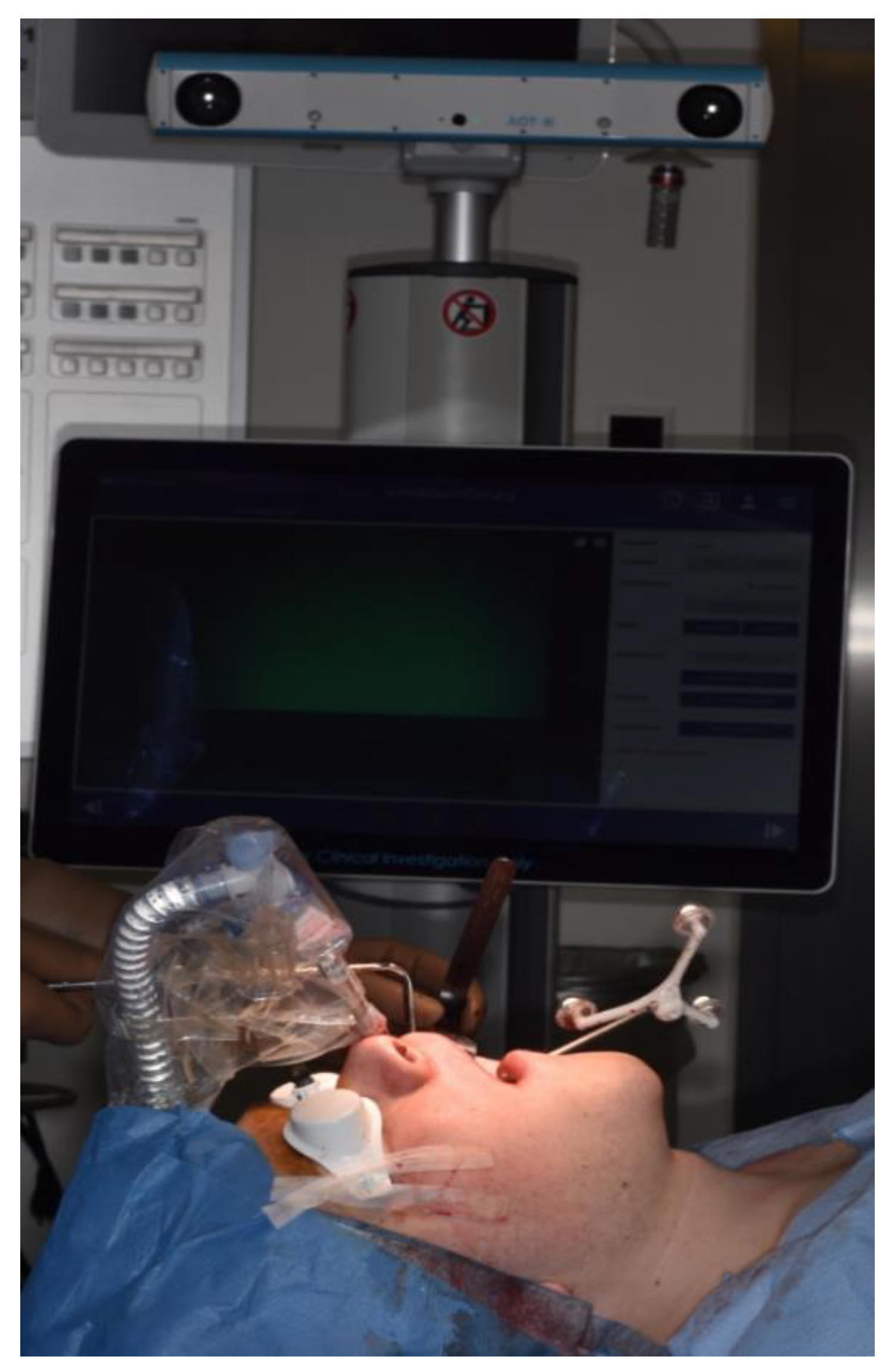
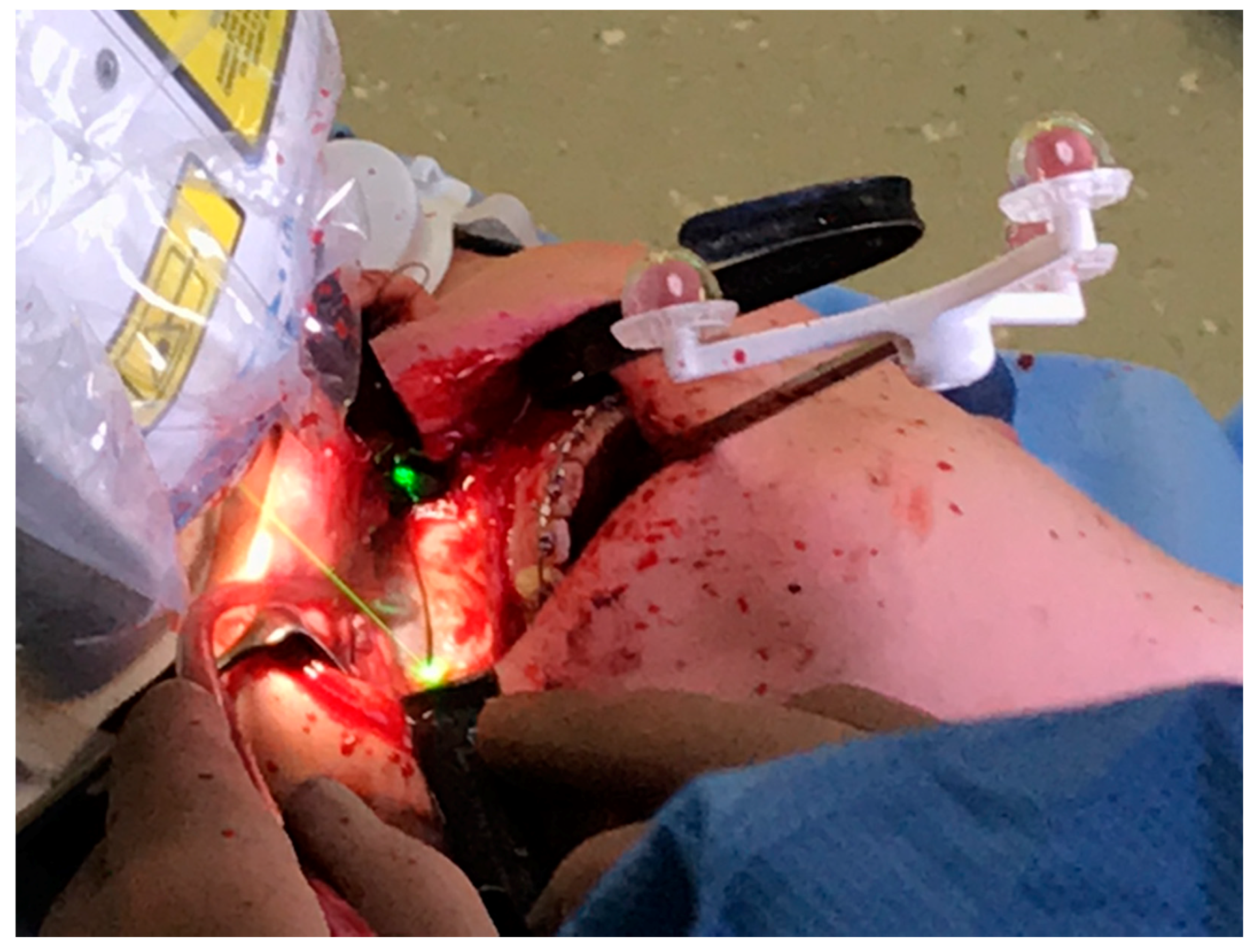
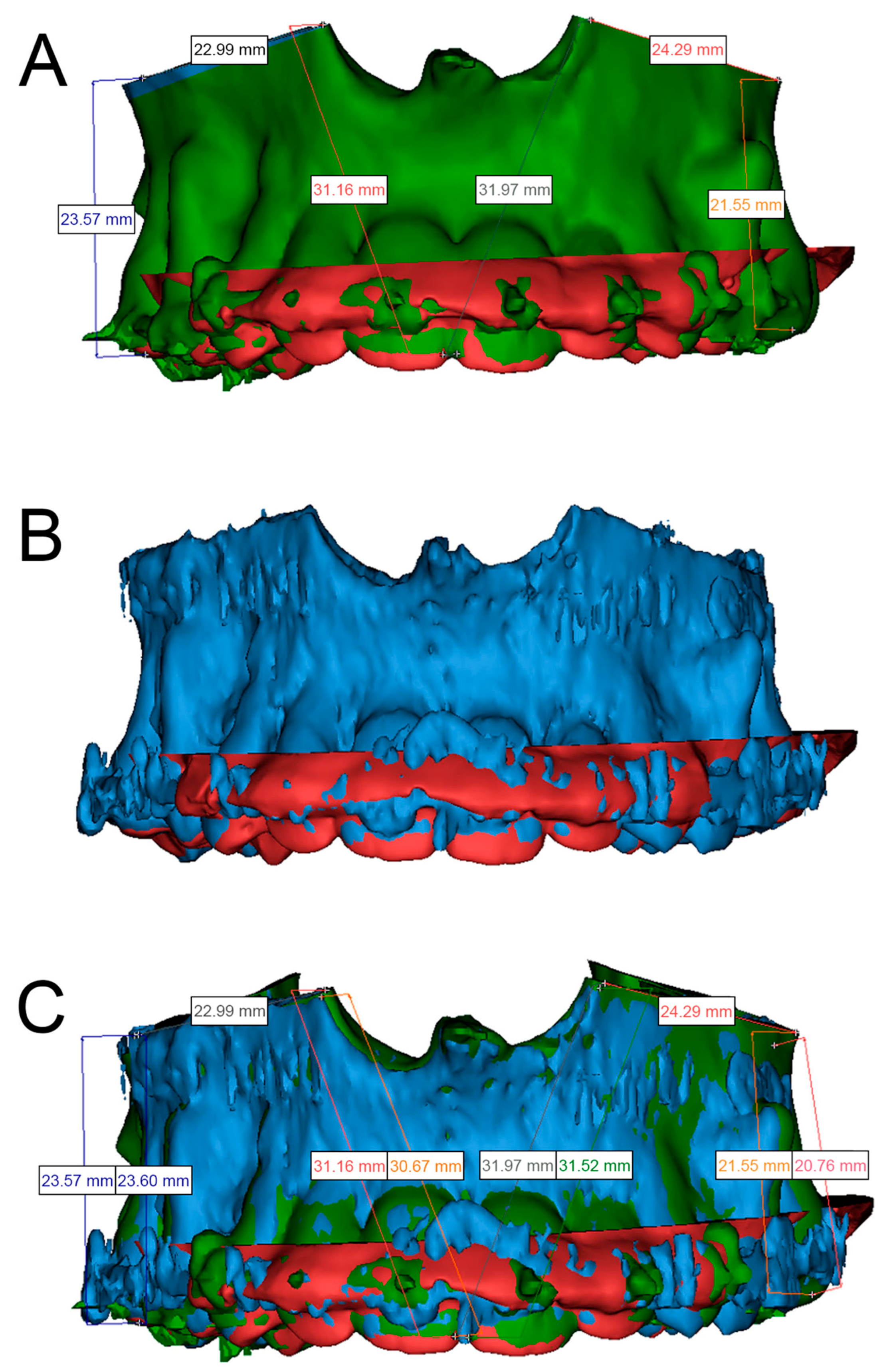
2.4. Surgical Procedure
2.5. Postoperative Procedure
3. Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Louvrier, A.; Marty, P.; Barrabé, A.; Euvrard, E.; Chatelain, B.; Weber, E.; Meyer, C. How useful is 3D printing in maxillofacial surgery? J. Stomatol. Oral Maxillofac. Surg. 2017, 118, 206–212. [Google Scholar] [CrossRef]
- Huang, M.F.; Alfi, D.; Alfi, J.; Huang, A.T. The Use of Patient-Specific Implants in Oral and Maxillofacial Surgery. Oral Maxillofac. Surg. Clin. N. Am. 2019, 31, 593–600. [Google Scholar] [CrossRef]
- Hua, J.; Aziz, S.; Shum, J.W. Virtual Surgical Planning in Oral and Maxillofacial Surgery. Oral Maxillofac. Surg. Clin. N. Am. 2019, 31, 519–530. [Google Scholar] [CrossRef]
- Jacobs, C.A.; Lin, A.Y. A New Classification of Three-Dimensional Printing Technologies. Plast. Reconstr. Surg. 2017, 139, 1211–1220. [Google Scholar] [CrossRef] [PubMed]
- Martelli, N.; Serrano, C.; Brink, H.V.D.; Pineau, J.; Prognon, P.; Borget, I.; El Batti, S. Advantages and disadvantages of 3-dimensional printing in surgery: A systematic review. Surgery 2016, 159, 1485–1500. [Google Scholar] [CrossRef] [PubMed]
- Gunaratne, R.; Khan, R.; Fick, D.; Robertson, B.; Dahotre, N.; Ironside, C. A review of the physiological and histological effects of laser osteotomy. J. Med Eng. Technol. 2016, 41, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Stübinger, S.; Ghanaati, S.; Saldamli, B.; Kirkpatrick, C.; Sader, R. Er:YAG Laser Osteotomy: Preliminary Clinical and Histological Results of a New Technique for Contact-Free Bone Surgery. Eur. Surg. Res. 2009, 42, 150–156. [Google Scholar] [CrossRef]
- Eriksson, A.R.; Albrektsson, T.; Albrektsson, B. Heat caused by drilling cortical bone: Temperature measuredin vivoin patients and animals. Acta Orthop. Scand. 1984, 55, 629–631. [Google Scholar] [CrossRef]
- Lagunas, J.G. Is the piezoelectric device the new standard for facial osteotomies? J. Stomatol. Oral Maxillofac. Surg. 2017, 118, 255–258. [Google Scholar] [CrossRef] [PubMed]
- Gilles, R.; Couvreur, T.; Dammous, S. Ultrasonic orthognathic surgery: Enhancements to established osteotomies. Int. J. Oral Maxillofac. Surg. 2013, 42, 981–987. [Google Scholar] [CrossRef]
- Jundt, J.S.; Marchena, J.M.; Hanna, I.; Dhanda, J.; Breit, M.J.; Perry, A.P. Evolving Technologies for Tissue Cutting. Oral Maxillofac. Surg. Clin. N. Am. 2019, 31, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Vercellotti, T.; Nevins, M.L.; Kim, D.M.; Nevins, M.; Wada, K.; Schenk, R.K.; Fiorellini, J.P. Osseous response following resective therapy with piezosurgery. Int. J. Periodontics Restor. Dent. 2005, 25, 543–549. [Google Scholar]
- Labanca, M.; Azzola, F.; Vinci, R.; Rodella, L.F. Piezoelectric surgery: Twenty years of use. Br. J. Oral Maxillofac. Surg. 2008, 46, 265–269. [Google Scholar] [CrossRef]
- Augello, M.; Baetscher, C.; Segesser, M.; Zeilhofer, H.-F.; Cattin, P.; Juergens, P. Performing partial mandibular resection, fibula free flap reconstruction and midfacial osteotomies with a cold ablation and robot-guided Er:YAG laser osteotome (CARLO®)—A study on applicability and effectiveness in human cadavers. J. Cranio-Maxillofac. Surg. 2018, 46, 1850–1855. [Google Scholar] [CrossRef] [PubMed]
- Berg, B.-I.; Peyer, M.; Kuske, L.; Augello, M.; Schötzau, A.; Steineck, M.; Deibel, W.; Mathys, D.; Jürgens, P.; Kunz, C.; et al. Comparison of an Er: YAG laser osteotome versus a conventional drill for the use in osteo- odonto-keratoprosthesis (OOKP). Lasers Surg. Med. 2019, 51, 531–537. [Google Scholar] [CrossRef]
- Sotsuka, Y.; Nishimoto, S.; Tsumano, T.; Kawai, K.; Ishise, H.; Kakibuchi, M.; Shimokita, R.; Yamauchi, T.; Okihara, S.-I. The dawn of computer-assisted robotic osteotomy with ytterbium-doped fiber laser. Lasers Med. Sci. 2013, 29, 1125–1129. [Google Scholar] [CrossRef]
- Romeo, U.; Del Vecchio, A.; Palata, G.; Tenore, G.; Visca, P.; Maggiore, C. Bone damage induced by different cutting instruments: An in vitro study. Braz. Dent. J. 2009, 20, 162–168. [Google Scholar] [CrossRef]
- Hibst, R. Mechanical effects of erbium:YAG laser bone ablation. Lasers Surg. Med. 1992, 12, 125–130. [Google Scholar] [CrossRef]
- Visuri, S.R.; Walsh, J.T.; Wigdor, H.A. Erbium Laser Ablation of Dental Hard Tissue: Effect of Water Cooling. Lasers Surg. Med. 1996, 18, 294–300. [Google Scholar] [CrossRef]
- Zeitouni, J.; Clough, B.; Zeitouni, S.; Saleem, M.; Al Aisami, K.; Gregory, C. The effects of the Er:YAG laser on trabecular bone micro-architecture: Comparison with conventional dental drilling by micro-computed tomographic and histological techniques. F1000Research 2017, 6, 1133. [Google Scholar] [CrossRef]
- Pantawane, M.V.; Chipper, R.T.; Robertson, W.B.; Khan, R.J.; Fick, D.P.; Dahotre, N.B. Evolution of surface morphology of Er:YAG laser-machined human bone. Lasers Med. Sci. 2020, 35, 1477–1485. [Google Scholar] [CrossRef] [PubMed]
- Lewandrowski, K.U.; Lorente, C.; Schomacker, K.T.; Flotte, T.J.; Wilkes, J.W.; Deutsch, T.F. Use of the Er:YAG Laser for Improved Plating in Maxillofacial Surgery: Comparison of Bone Healing in Laser and Drill Osteotomies. Lasers Surg. Med. 1996, 19, 40–45. [Google Scholar] [CrossRef]
- De Mello, E.D.A.; Pagnoncelli, R.M.; Munin, E.; Filho, M.S.; De Mello, G.P.S.; Arisawa, E.A.L.; De Oliveira, M. Comparative histological analysis of bone healing of standardized bone defects performed with the Er:YAG laser and steel burs. Lasers Med. Sci. 2007, 23, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Pourzarandian, A.; Watanabe, H.; Aoki, A.; Ichinose, S.; Sasaki, K.M.; Nitta, H.; Ishikawa, I. Histological and TEM Examination of Early Stages of Bone Healing after Er:YAG Laser Irradiation. Photomed. Laser Surg. 2004, 22, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Augello, M.; Deibel, W.; Nuss, K.; Cattin, P.; Jürgens, P. Comparative microstructural analysis of bone osteotomies after cutting by computer-assisted robot-guided laser osteotome and piezoelectric osteotome: An in vivo animal study. Lasers Med. Sci. 2018, 33, 1471–1478. [Google Scholar] [CrossRef]
- Stübinger, S.; Nuss, K.; Pongratz, M.; Price, J.; Sader, R.A.; Zeilhofer, H.-F.; Von Rechenberg, B. Comparison of Er:YAG laser and piezoelectric osteotomy: An animal study in sheep. Lasers Surg. Med. 2010, 42, 743–751. [Google Scholar] [CrossRef]
- Cloutier, M.; Girard, B.; Peel, S.A.; Wilson, D.; Sándor, G.K.; Clokie, C.M.; Miller, D. Calvarial bone wound healing: A comparison between carbide and diamond drills, Er:YAG and Femtosecond lasers with or without BMP-7. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 2010, 110, 720–728. [Google Scholar] [CrossRef]
- Baek, K.-W.; Deibel, W.; Marinov, D.; Griessen, M.; Bruno, A.; Zeilhofer, H.-F.; Cattin, P.; Juergens, P. Clinical applicability of robot-guided contact-free laser osteotomy in cranio-maxillo-facial surgery: In-vitro simulation and in-vivo surgery in minipig mandibles. Br. J. Oral Maxillofac. Surg. 2015, 53, 976–981. [Google Scholar] [CrossRef]
- Baek, K.-W.; Dard, M.; Dmd, H.Z.; Cattin, P.; Juergens, P.; Zeilhofer, H.-F. Comparing the Bone Healing After Cold Ablation Robot-Guided Er:YAG Laser Osteotomy and Piezoelectric Osteotomy—A Pilot Study in a Minipig Mandible. Lasers Surg. Med. 2020. [Google Scholar] [CrossRef]
- Stübinger, S.; Nuss, K.; Landes, C.; Von Rechenberg, B.; Sader, R. Harvesting of intraoral autogenous block grafts from the chin and ramus region: Preliminary results with a variable square pulse Er:YAG laser. Lasers Surg. Med. 2008, 40, 312–318. [Google Scholar] [CrossRef]
- Stübinger, S.; Kober, C.; Zeilhofer, H.-F.; Sader, R.; Stübinger, S. Er:YAG Laser Osteotomy Based on Refined Computer-Assisted Presurgical Planning: First Clinical Experience in Oral Surgery. Photomed. Laser Surg. 2007, 25, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Azarmehr, I.; Stokbro, K.; Bell, R.B.; Thygesen, T. Surgical Navigation: A Systematic Review of Indications, Treatments, and Outcomes in Oral and Maxillofacial Surgery. J. Oral Maxillofac. Surg. 2017, 75, 1987–2005. [Google Scholar] [CrossRef] [PubMed]
- Heufelder, M.; Wilde, F.; Pietzka, S.; Mascha, F.; Winter, K.; Schramm, A.; Rana, M. Clinical accuracy of waferless maxillary positioning using customized surgical guides and patient specific osteosynthesis in bimaxillary orthognathic surgery. J. Cranio-Maxillofac. Surg. 2017, 45, 1578–1585. [Google Scholar] [CrossRef] [PubMed]
- Rodby, K.A.; Turin, S.; Jacobs, R.J.; Cruz, J.F.; Hassid, V.J.; Kolokythas, A.; Antony, A.K. Advances in oncologic head and neck reconstruction: Systematic review and future considerations of virtual surgical planning and computer aided design/computer aided modeling. J. Plast. Reconstr. Aesthetic Surg. 2014, 67, 1171–1185. [Google Scholar] [CrossRef] [PubMed]
- Alasseri, N.; Swennen, G. Minimally invasive orthognathic surgery: A systematic review. Int. J. Oral Maxillofac. Surg. 2018, 47, 1299–1310. [Google Scholar] [CrossRef]
- Swennen, G.R. Surgical Efficiency and Minimizing Patient Morbidity by Using a Novel Surgical Algorithm in Orthognathic Surgery. Atlas Oral Maxillofac. Surg. Clin. 2020, 28, 95–109. [Google Scholar] [CrossRef]
- Hernández-Alfaro, F.; Guijarro-Martínez, R. “Twist Technique” for Pterygomaxillary Dysjunction in Minimally Invasive Le Fort I Osteotomy. J. Oral Maxillofac. Surg. 2013, 71, 389–392. [Google Scholar] [CrossRef]
| Advantages | Limitations |
|---|---|
|
|
|
|
|
|
|
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ureel, M.; Augello, M.; Holzinger, D.; Wilken, T.; Berg, B.-I.; Zeilhofer, H.-F.; Millesi, G.; Juergens, P.; Mueller, A.A. Cold Ablation Robot-Guided Laser Osteotome (CARLO®): From Bench to Bedside. J. Clin. Med. 2021, 10, 450. https://doi.org/10.3390/jcm10030450
Ureel M, Augello M, Holzinger D, Wilken T, Berg B-I, Zeilhofer H-F, Millesi G, Juergens P, Mueller AA. Cold Ablation Robot-Guided Laser Osteotome (CARLO®): From Bench to Bedside. Journal of Clinical Medicine. 2021; 10(3):450. https://doi.org/10.3390/jcm10030450
Chicago/Turabian StyleUreel, Matthias, Marcello Augello, Daniel Holzinger, Tobias Wilken, Britt-Isabelle Berg, Hans-Florian Zeilhofer, Gabriele Millesi, Philipp Juergens, and Andreas A. Mueller. 2021. "Cold Ablation Robot-Guided Laser Osteotome (CARLO®): From Bench to Bedside" Journal of Clinical Medicine 10, no. 3: 450. https://doi.org/10.3390/jcm10030450
APA StyleUreel, M., Augello, M., Holzinger, D., Wilken, T., Berg, B.-I., Zeilhofer, H.-F., Millesi, G., Juergens, P., & Mueller, A. A. (2021). Cold Ablation Robot-Guided Laser Osteotome (CARLO®): From Bench to Bedside. Journal of Clinical Medicine, 10(3), 450. https://doi.org/10.3390/jcm10030450





