Non-Melanoma Skin Cancer Mortality in Spain: A Predictive Model up to 2044
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cameron, M.C.; Lee, E.; Hibler, B.P.; Barker, C.A.; Mori, S.; Cordova, M.; Nehal, K.S.; Rossi, A.M. Basal cell carcinoma: Epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J. Am. Acad. Dermatol. 2019, 80, 303–317. [Google Scholar] [CrossRef] [Green Version]
- Nehal, K.S.; Bichakjian, C.K. Update on Keratinocyte Carcinomas. N. Engl. J. Med. 2018, 379, 363–374. [Google Scholar] [CrossRef]
- Karia, P.S.; Han, J.; Schmults, C.D. Cutaneous squamous cell carcinoma: Estimated incidence of disease, nodal metastasis, and deaths from disease in the United States, 2012. J. Am. Acad. Dermatol. 2013, 68, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Tejera-Vaquerizo, A.; Descalzo-Gallego, M.; Otero-Rivas, M.; Posada-García, C.; Rodríguez-Pazos, L.; Pastushenko, I.; Marcos-Gragera, R.; García-Doval, I. Skin cancer incidence and mortality in Spain: A systematic review and meta-analysis. Actas Dermo-Sifiliogr. 2016, 107, 318–328. [Google Scholar] [CrossRef]
- Kim, D.P.; Kus, K.J.; Ruiz, E. Basal Cell Carcinoma Review. Hematol. Clin. N. Am. 2019, 33, 13–24. [Google Scholar] [CrossRef]
- Rudolph, C.; Schnoor, M.; Eisemann, N.; Katalinic, A. Incidence trends of nonmelanoma skin cancer in Germany from 1998 to 2010. J. Dtsch. Dermatol. Ges. 2015, 13, 788–797. [Google Scholar] [CrossRef] [PubMed]
- Kukko, H.; Böhling, T.; Koljonen, V.; Tukiainen, E.; Haglund, C.; Pokhrel, A.; Sankila, R.; Pukkala, E. Merkel cell carcinoma—A population-based epidemiological study in Finland with a clinical series of 181 cases. Eur. J. Cancer 2012, 48, 737–742. [Google Scholar] [CrossRef]
- Agelli, M.; Clegg, L.X. Epidemiology of primary Merkel cell carcinoma in the United States. J. Am. Acad. Dermatol. 2003, 49, 832–841. [Google Scholar] [CrossRef]
- Martinez, S.R.; Barr, K.L.; Canter, R.J. Rare tumors through the looking glass: An examination of malignant cutaneous adnexal tumors. Arch. Dermatol. 2011, 147, 1058–1062. [Google Scholar] [CrossRef] [Green Version]
- Wu, W.; Weinstock, M.A. Trends of Keratinocyte Carcinoma Mortality Rates in the United States as Reported on Death Certificates, 1999 Through 2010. Dermatol. Surg. 2014, 40, 1395–1401. [Google Scholar] [CrossRef]
- Sendín-Martin, M.; Durán-Romero, A.J.; Martin-Carrasco, P.; Conejo-Mir, J.; Pereyra-Rodriguez, J.-J. Mortality associated with non-melanoma skin cancer in Spain from 1979 to 2018: Trends and age-adjusted rates. Eur. J. Dermatol. 2021, 31, 486–492. [Google Scholar] [CrossRef]
- Møller, B.; Fekjaer, H.; Hakulinen, T.; Sigvaldason, H.; Storm, H.; Talbäck, M.; Haldorsen, T. Prediction of cancer incidence in the Nordic countries: Empirical comparison of different approaches. Stat. Med. 2003, 22, 2751–2766. [Google Scholar] [CrossRef] [PubMed]
- R: The R Project for Statistical Computing n.d. Available online: https://www.r-project.org/ (accessed on 2 September 2021).
- Møller, B.; Fekjaer, H.; Hakulinen, T.; Tryggvadóttir, L.; Storm, H.H.; Talbäck, M.; Haldorsen, T. Prediction of cancer incidence in the Nordic countries up to the year 2020. Eur. J. Cancer Prev. 2002, 11, S1–S96. [Google Scholar]
- Donaldson, M.R.; Coldiron, B.M. No End in Sight: The Skin Cancer Epidemic Continues. Semin. Cutan. Med. Surg. 2011, 30, 3–5. [Google Scholar] [CrossRef] [PubMed]
- Rogers, H.W.; Weinstock, M.A.; Feldman, S.R.; Coldiron, B.M. Incidence Estimate of Nonmelanoma Skin Cancer (Keratinocyte Carcinomas) in the US Population, 2012. JAMA Dermatol. 2015, 151, 1081–1086. [Google Scholar] [CrossRef]
- Páginas—Instituto de Salud Carlos III—ISCIII n.d. Available online: https://www.isciii.es/Paginas/Inicio.aspx (accessed on 3 September 2021).
- Jing, C.; Zhang, H.; Zhang, X.; Yu, S. Dermatofibrosarcoma Protuberans: A Clinicopathologic and Therapeutic Analysis of 254 Cases at a Single Institution. Dermatol. Surg. 2021, 47, e26–e30. [Google Scholar] [CrossRef]
- Maurelli, M.; Tessari, G.; Colato, C.; Schena, D.; Girolomoni, G. Incidence and ten-year follow-up of primary cutaneous lymphomas: A single-centre cohort study. Eur. J. Dermatol. 2017, 28, 44–49. [Google Scholar] [CrossRef]
- Fania, L.; Didona, D.; Morese, R.; Campana, I.; Coco, V.; Di Pietro, F.R.; Ricci, F.; Pallotta, S.; Candi, E.; Abeni, D.; et al. Basal Cell Carcinoma: From Pathophysiology to Novel Therapeutic Approaches. Biomedicines 2020, 8, 449. [Google Scholar] [CrossRef] [PubMed]
- Waldman, A.; Schmults, C. Cutaneous Squamous Cell Carcinoma. Hematol. Clin. N. Am. 2019, 33, 1–12. [Google Scholar] [CrossRef]
- Schmults, C.D.; Karia, P.S.; Carter, J.B.; Han, J.; Qureshi, A.A. Factors Predictive of Recurrence and Death From Cutaneous Squamous Cell Carcinoma. JAMA Dermatol. 2013, 149, 541–547. [Google Scholar] [CrossRef] [Green Version]
- Leiter, U.; Keim, U.; Eigentler, T.; Katalinic, A.; Holleczek, B.; Martus, P.; Garbe, C. Incidence, Mortality, and Trends of Nonmelanoma Skin Cancer in Germany. J. Investig. Dermatol. 2017, 137, 1860–1867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewis, M.A.W.K.G.; Weinstock, M.A. Trends in Nonmelanoma Skin Cancer Mortality Rates in the United States, 1969 through 2000. J. Investig. Dermatol. 2007, 127, 2323–2327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewis, K.G.; Weinstock, M.A. Nonmelanoma Skin Cancer Mortality (1988–2000). Arch. Dermatol. 2004, 140, 837–842. [Google Scholar] [CrossRef] [Green Version]
- Stang, A.; Jöckel, K. Declining mortality rates for nonmelanoma skin cancers in West Germany, 1968–99. Br. J. Dermatol. 2004, 150, 517–522. [Google Scholar] [CrossRef]
- Bennardo, L.; Bennardo, F.; Giudice, A.; Passante, M.; Dastoli, S.; Morrone, P.; Provenzano, E.; Patruno, C.; Nisticò, S. Local Chemotherapy as an Adjuvant Treatment in Unresectable Squamous Cell Carcinoma: What Do We Know So Far? Curr. Oncol. 2021, 28, 2317–2325. [Google Scholar] [CrossRef] [PubMed]
- Soydemir, G.P.; Kandaz, M.; Melikoğlu, M. The results of radiotherapy for squamous cell carcinomas of the skin. Dermatol. Ther. 2019, 32, e13058. [Google Scholar] [CrossRef] [PubMed]
- Migden, M.R.; Rischin, D.; Schmults, C.D.; Guminski, A.; Hauschild, A.; Lewis, K.D.; Chung, C.H.; Hernandez-Aya, L.; Lim, A.M.; Chang, A.L.S.; et al. PD-1 Blockade with Cemiplimab in Advanced Cutaneous Squamous-Cell Carcinoma. N. Engl. J. Med. 2018, 379, 341–351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmed, S.R.; Petersen, E.; Patel, R.; Migden, M.R. Cemiplimab-rwlc as first and only treatment for advanced cutaneous squamous cell carcinoma. Expert Rev. Clin. Pharmacol. 2019, 12, 947–951. [Google Scholar] [CrossRef] [PubMed]
- Alipour, J.; Payandeh, A. Common errors in reporting cause-of-death statement on death certificates: A systematic review and meta-analysis. J. Forensic Leg. Med. 2021, 82, 102220. [Google Scholar] [CrossRef]
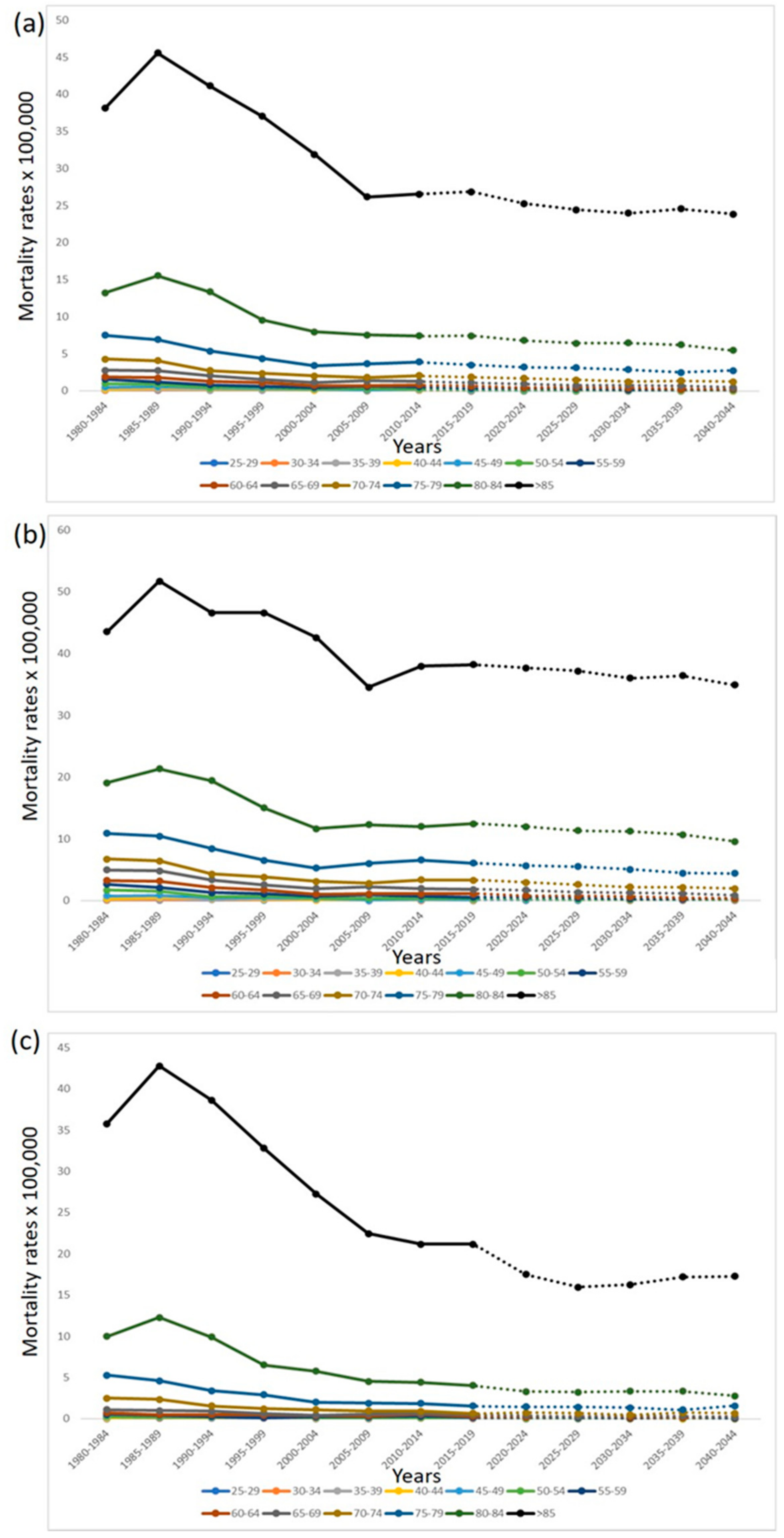
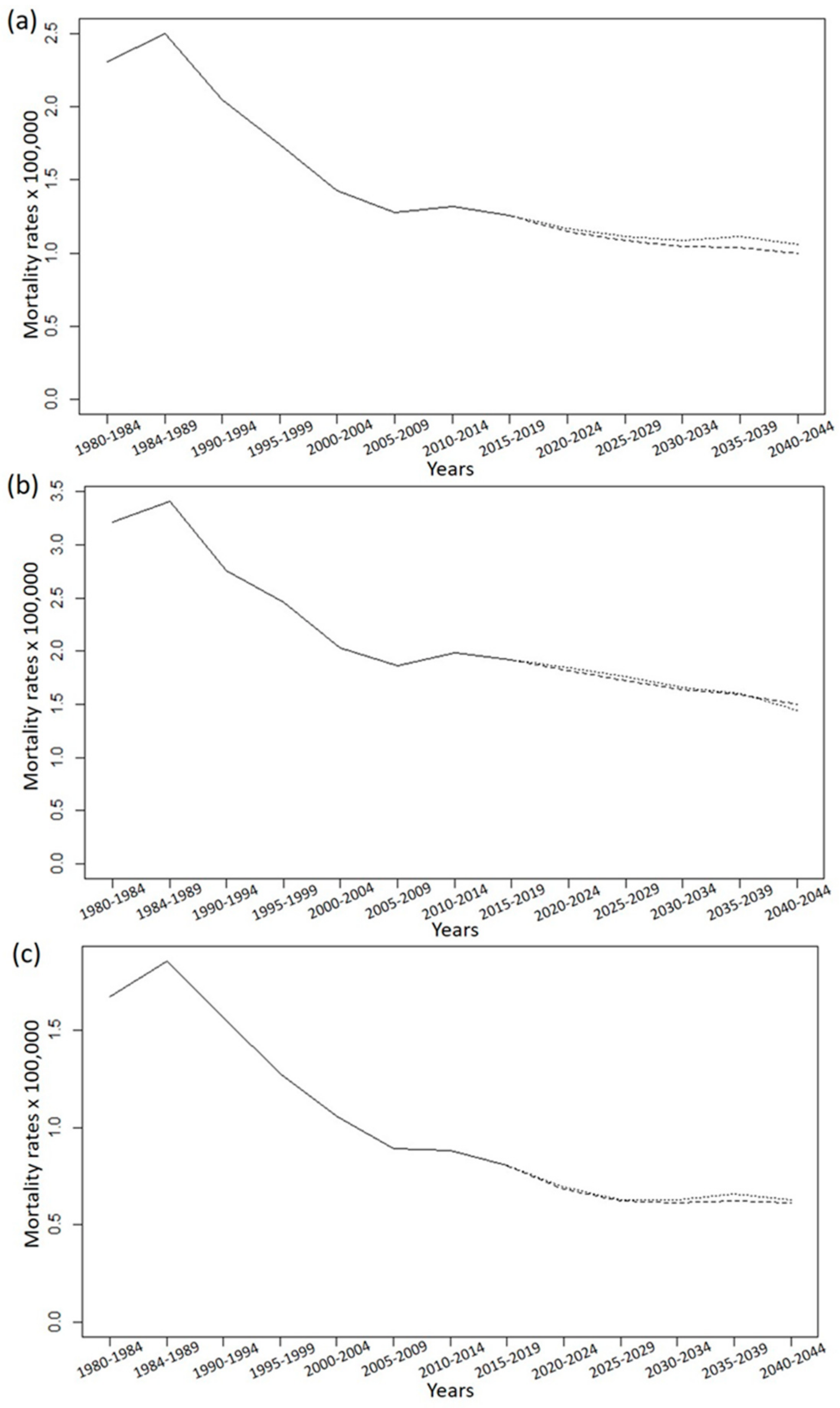
| (A) Both Sexes | |||||||||||||
| Observed NMSC Deaths | Projected NMSC Deaths | ||||||||||||
| Age (Years) | 1980–1984 | 1985–1989 | 1990–1994 | 1995–1999 | 2000–2004 | 2005–2009 | 2010–2014 | 2015–2019 | 2020– 2024 | 2025–2029 | 2030–2034 | 2035–2039 | 2040–2044 |
| 0–4 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5–9 | 1 | 1 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10–14 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 15–19 | 3 | 3 | 3 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 20–24 | 2 | 4 | 1 | 2 | 5 | 1 | 1 | 2 | 2 | 2 | 2 | 2 | 2 |
| 25–29 | 5 | 13 | 13 | 13 | 7 | 3 | 7 | 2 | 4 | 4 | 4 | 5 | 4 |
| 30–34 | 11 | 16 | 21 | 21 | 8 | 6 | 8 | 5 | 3 | 2 | 2 | 2 | 2 |
| 35–39 | 16 | 30 | 18 | 18 | 13 | 12 | 10 | 7 | 5 | 4 | 3 | 3 | 3 |
| 40–44 | 24 | 43 | 32 | 29 | 16 | 20 | 29 | 9 | 10 | 7 | 6 | 5 | 5 |
| 45–49 | 55 | 51 | 38 | 38 | 30 | 17 | 37 | 21 | 12 | 12 | 10 | 8 | 7 |
| 50–54 | 113 | 98 | 41 | 56 | 37 | 35 | 49 | 43 | 34 | 20 | 20 | 18 | 15 |
| 55–59 | 163 | 138 | 86 | 60 | 56 | 76 | 76 | 58 | 64 | 53 | 34 | 37 | 33 |
| 60–64 | 162 | 176 | 135 | 114 | 69 | 82 | 100 | 90 | 74 | 86 | 76 | 53 | 58 |
| 65–69 | 200 | 216 | 193 | 156 | 117 | 129 | 140 | 135 | 127 | 113 | 136 | 127 | 93 |
| 70–74 | 265 | 258 | 196 | 201 | 188 | 172 | 182 | 195 | 195 | 186 | 176 | 218 | 207 |
| 75–79 | 333 | 348 | 283 | 264 | 247 | 303 | 333 | 279 | 302 | 321 | 323 | 324 | 397 |
| 80–84 | 330 | 483 | 484 | 375 | 365 | 433 | 498 | 524 | 446 | 508 | 562 | 590 | 608 |
| >85 | 533 | 834 | 990 | 1128 | 1134 | 1146 | 1540 | 1914 | 2049 | 2057 | 2312 | 2724 | 3006 |
| Total number of deaths | 2217 | 2713 | 2536 | 2475 | 2293 | 2437 | 3010 | 3284 | 3212 | 3373 | 3663 | 4115 | 4440 |
| (B) Men | |||||||||||||
| Observed NMSC Deaths | Projected NMSC Deaths | ||||||||||||
| Age (Years) | 1980–1984 | 1985–1989 | 1990–1994 | 1995–1999 | 2000–2004 | 2005–2009 | 2010–2014 | 2015–2019 | 2020– 2024 | 2025–2029 | 2030–2034 | 2035–2039 | 2040– 2044 |
| 0–4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5–9 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10–14 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 15–19 | 3 | 2 | 2 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 20–24 | 1 | 1 | 0 | 1 | 2 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 |
| 25–29 | 4 | 7 | 11 | 9 | 6 | 1 | 3 | 0 | 1 | 1 | 1 | 2 | 1 |
| 30–34 | 8 | 10 | 19 | 15 | 5 | 5 | 3 | 4 | 3 | 2 | 2 | 2 | 2 |
| 35–39 | 11 | 18 | 14 | 16 | 9 | 8 | 7 | 3 | 4 | 3 | 3 | 3 | 3 |
| 40–44 | 19 | 32 | 25 | 20 | 10 | 13 | 18 | 5 | 4 | 5 | 4 | 4 | 4 |
| 45–49 | 42 | 43 | 28 | 30 | 20 | 11 | 20 | 13 | 8 | 5 | 7 | 6 | 6 |
| 50–54 | 97 | 87 | 32 | 41 | 30 | 26 | 40 | 29 | 25 | 15 | 10 | 14 | 13 |
| 55–59 | 134 | 114 | 70 | 54 | 43 | 60 | 53 | 45 | 46 | 40 | 26 | 19 | 26 |
| 60–64 | 129 | 150 | 107 | 89 | 49 | 61 | 67 | 71 | 58 | 60 | 54 | 39 | 28 |
| 65–69 | 155 | 172 | 146 | 121 | 93 | 101 | 104 | 103 | 101 | 94 | 101 | 96 | 72 |
| 70–74 | 175 | 171 | 132 | 143 | 131 | 122 | 138 | 159 | 152 | 146 | 143 | 158 | 153 |
| 75–79 | 191 | 207 | 174 | 159 | 161 | 213 | 243 | 211 | 232 | 250 | 251 | 259 | 288 |
| 80–84 | 170 | 239 | 254 | 210 | 197 | 272 | 319 | 355 | 321 | 373 | 413 | 432 | 457 |
| >85 | 189 | 294 | 349 | 437 | 456 | 465 | 708 | 910 | 1049 | 1090 | 1247 | 1473 | 1620 |
| Total number of deaths | 1330 | 1548 | 1365 | 1345 | 1213 | 1359 | 1724 | 1908 | 2005 | 2083 | 2262 | 2506 | 2673 |
| (C) Women | |||||||||||||
| Observed NMSC Deaths | Projected NMSC Deaths | ||||||||||||
| Age (Years) | 1980–1984 | 1985–1989 | 1990–1994 | 1995–1999 | 2000–2004 | 2005–2009 | 2010–2014 | 2015–2019 | 2020– 2024 | 2025–2029 | 2030–2034 | 2035–2039 | 2040– 2044 |
| 0–4 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5–9 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10–14 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 15–19 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 20–24 | 1 | 3 | 1 | 1 | 3 | 1 | 0 | 2 | 1 | 1 | 1 | 1 | 1 |
| 25–29 | 1 | 6 | 2 | 4 | 1 | 2 | 4 | 2 | 3 | 3 | 3 | 3 | 3 |
| 30–34 | 3 | 6 | 2 | 6 | 3 | 1 | 5 | 1 | 1 | 0 | 0 | 0 | 0 |
| 35–39 | 5 | 12 | 4 | 2 | 4 | 4 | 3 | 4 | 1 | 1 | 0 | 0 | 0 |
| 40–44 | 5 | 11 | 7 | 9 | 6 | 7 | 11 | 4 | 8 | 2 | 2 | 1 | 1 |
| 45–49 | 13 | 8 | 10 | 8 | 10 | 6 | 17 | 8 | 4 | 8 | 3 | 2 | 2 |
| 50–54 | 16 | 11 | 9 | 15 | 7 | 9 | 9 | 14 | 10 | 5 | 10 | 4 | 3 |
| 55–59 | 29 | 24 | 16 | 6 | 13 | 16 | 23 | 13 | 18 | 11 | 8 | 17 | 7 |
| 60–64 | 33 | 26 | 28 | 25 | 20 | 21 | 33 | 19 | 18 | 28 | 23 | 15 | 30 |
| 65–69 | 45 | 44 | 47 | 35 | 24 | 28 | 36 | 32 | 29 | 22 | 36 | 33 | 23 |
| 70–74 | 90 | 87 | 64 | 58 | 57 | 50 | 44 | 36 | 46 | 45 | 38 | 63 | 59 |
| 75–79 | 142 | 141 | 109 | 105 | 86 | 90 | 90 | 68 | 75 | 81 | 84 | 78 | 123 |
| 80–84 | 160 | 244 | 230 | 165 | 168 | 161 | 179 | 169 | 127 | 148 | 167 | 182 | 175 |
| >85 | 344 | 540 | 641 | 691 | 678 | 681 | 832 | 1004 | 933 | 875 | 1004 | 1211 | 1374 |
| Total number of deaths | 887 | 1165 | 1171 | 1130 | 1080 | 1078 | 1286 | 1376 | 1272 | 1231 | 1378 | 1611 | 1802 |
| (A) Both Sexes | |||||||||||||
| Age (Years) | 1980–1984 | 1985–1989 | 1990–1994 | 1995–1999 | 2000–2004 | 2005–2009 | 2010–2014 | 2015–2019 | 2020– 2024 | 2025–2029 | 2030–2034 | 2035–2039 | 2040–2044 |
| 0–4 | 0 | 0.01 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5–9 | 0.01 | 0.01 | 0.01 | 0 | 0 | 0.01 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10–14 | 0.01 | 0 | 0.01 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 15–19 | 0.02 | 0.02 | 0.02 | 0 | 0.01 | 0.01 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 20–24 | 0.01 | 0.02 | 0.01 | 0.01 | 0.03 | 0.01 | 0.01 | 0.02 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| 25–29 | 0.04 | 0.09 | 0.08 | 0.08 | 0.04 | 0.02 | 0.05 | 0.02 | 0.03 | 0.03 | 0.03 | 0.03 | 0.03 |
| 30–34 | 0.09 | 0.12 | 0.14 | 0.13 | 0.05 | 0.03 | 0.04 | 0.03 | 0.02 | 0.02 | 0.02 | 0.01 | 0.01 |
| 35–39 | 0.14 | 0.24 | 0.14 | 0.12 | 0.08 | 0.06 | 0.05 | 0.04 | 0.03 | 0.03 | 0.02 | 0.02 | 0.02 |
| 40–44 | 0.24 | 0.37 | 0.26 | 0.22 | 0.1 | 0.11 | 0.15 | 0.05 | 0.05 | 0.05 | 0.04 | 0.04 | 0.03 |
| 45–49 | 0.48 | 0.51 | 0.33 | 0.31 | 0.22 | 0.1 | 0.2 | 0.11 | 0.06 | 0.06 | 0.06 | 0.06 | 0.05 |
| 50–54 | 0.99 | 0.88 | 0.42 | 0.49 | 0.3 | 0.25 | 0.3 | 0.24 | 0.18 | 0.1 | 0.11 | 0.11 | 0.11 |
| 55–59 | 1.57 | 1.24 | 0.79 | 0.62 | 0.49 | 0.6 | 0.55 | 0.37 | 0.37 | 0.29 | 0.17 | 0.2 | 0.21 |
| 60–64 | 1.9 | 1.77 | 1.26 | 1.08 | 0.72 | 0.72 | 0.81 | 0.67 | 0.48 | 0.5 | 0.42 | 0.28 | 0.32 |
| 65–69 | 2.78 | 2.72 | 2.06 | 1.54 | 1.15 | 1.37 | 1.26 | 1.13 | 0.99 | 0.76 | 0.82 | 0.73 | 0.5 |
| 70–74 | 4.28 | 4.05 | 2.74 | 2.37 | 2.01 | 1.8 | 2.06 | 1.88 | 1.73 | 1.53 | 1.25 | 1.39 | 1.25 |
| 75–79 | 7.49 | 6.92 | 5.36 | 4.38 | 3.39 | 3.66 | 3.91 | 3.54 | 3.24 | 3.14 | 2.91 | 2.51 | 2.77 |
| 80–84 | 13.24 | 15.54 | 13.35 | 9.55 | 7.94 | 7.52 | 7.42 | 7.44 | 6.83 | 6.47 | 6.49 | 6.25 | 5.51 |
| >85 | 38.18 | 45.57 | 41.12 | 37.05 | 31.87 | 26.16 | 26.55 | 26.87 | 25.27 | 24.46 | 23.98 | 24.57 | 23.87 |
| SMR | 2.31 | 2.5 | 2.05 | 1.74 | 1.43 | 1.28 | 1.32 | 1.25 | 1.15 | 1.09 | 1.04 | 1.03 | 0.99 |
| (B) Men | |||||||||||||
| Age (Years) | 1980–1984 | 1985–1989 | 1990–1994 | 1995–1999 | 2000–2004 | 2005–2009 | 2010–2014 | 2015–2019 | 2020– 2024 | 2025–2029 | 2030–2034 | 2035–2039 | 2040–2044 |
| 0–4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5–9 | 0.01 | 0.01 | 0.02 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10–14 | 0.01 | 0 | 0.01 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 15–19 | 0.04 | 0.02 | 0.02 | 0 | 0.02 | 0.02 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 20–24 | 0.01 | 0.01 | 0 | 0.01 | 0.02 | 0 | 0.02 | 0 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| 25–29 | 0.06 | 0.09 | 0.14 | 0.11 | 0.07 | 0.01 | 0.04 | 0 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 |
| 30–34 | 0.13 | 0.15 | 0.26 | 0.18 | 0.06 | 0.05 | 0.03 | 0.05 | 0.04 | 0.04 | 0.03 | 0.03 | 0.03 |
| 35–39 | 0.19 | 0.29 | 0.21 | 0.21 | 0.11 | 0.08 | 0.07 | 0.03 | 0.06 | 0.05 | 0.04 | 0.04 | 0.04 |
| 40–44 | 0.38 | 0.55 | 0.41 | 0.3 | 0.13 | 0.14 | 0.18 | 0.05 | 0.04 | 0.07 | 0.06 | 0.06 | 0.05 |
| 45–49 | 0.75 | 0.87 | 0.49 | 0.49 | 0.3 | 0.13 | 0.22 | 0.14 | 0.08 | 0.05 | 0.09 | 0.08 | 0.08 |
| 50–54 | 1.73 | 1.59 | 0.66 | 0.73 | 0.49 | 0.38 | 0.49 | 0.33 | 0.26 | 0.15 | 0.1 | 0.18 | 0.18 |
| 55–59 | 2.67 | 2.11 | 1.33 | 1.14 | 0.77 | 0.97 | 0.78 | 0.58 | 0.53 | 0.43 | 0.27 | 0.2 | 0.34 |
| 60–64 | 3.28 | 3.18 | 2.1 | 1.77 | 1.07 | 1.1 | 1.12 | 1.1 | 0.77 | 0.71 | 0.61 | 0.41 | 0.32 |
| 65–69 | 4.96 | 4.83 | 3.39 | 2.57 | 1.96 | 2.28 | 1.97 | 1.82 | 1.65 | 1.32 | 1.27 | 1.13 | 0.8 |
| 70–74 | 6.73 | 6.45 | 4.31 | 3.82 | 3.13 | 2.83 | 3.4 | 3.32 | 2.91 | 2.58 | 2.17 | 2.14 | 1.93 |
| 75–79 | 10.92 | 10.47 | 8.46 | 6.56 | 5.3 | 6.06 | 6.6 | 6.1 | 5.62 | 5.46 | 5.02 | 4.42 | 4.37 |
| 80–84 | 19.08 | 21.34 | 19.46 | 15.04 | 11.66 | 12.32 | 12 | 12.47 | 11.97 | 11.29 | 11.23 | 10.65 | 9.54 |
| >85 | 43.55 | 51.74 | 46.62 | 46.59 | 42.59 | 34.53 | 37.94 | 38.19 | 37.67 | 37.14 | 36 | 36.41 | 34.9 |
| SMR | 3.21 | 3.41 | 2.76 | 2.46 | 2.03 | 1.87 | 1.98 | 1.92 | 1.82 | 1.73 | 1.64 | 1.59 | 1.5 |
| (C) Women | |||||||||||||
| Age (Years) | 1980–1984 | 1985–1989 | 1990–1994 | 1995–1999 | 2000–2004 | 2005–2009 | 2010–2014 | 2015–2019 | 2020– 2024 | 2025–2029 | 2030–2034 | 2035–2039 | 2040–2044 |
| 0–4 | 0 | 0.02 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5–9 | 0 | 0 | 0 | 0 | 0 | 0.02 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10–14 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 15–19 | 0 | 0.01 | 0.01 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 20–24 | 0.01 | 0.04 | 0.01 | 0.01 | 0.04 | 0.01 | 0 | 0.04 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 |
| 25–29 | 0.02 | 0.08 | 0.03 | 0.05 | 0.01 | 0.02 | 0.05 | 0.03 | 0.04 | 0.04 | 0.04 | 0.04 | 0.04 |
| 30–34 | 0.05 | 0.09 | 0.03 | 0.07 | 0.04 | 0.01 | 0.05 | 0.01 | 0.01 | 0 | 0 | 0 | 0 |
| 35–39 | 0.09 | 0.2 | 0.06 | 0.03 | 0.05 | 0.04 | 0.03 | 0.04 | 0.01 | 0.01 | 0.01 | 0 | 0 |
| 40–44 | 0.1 | 0.19 | 0.11 | 0.14 | 0.08 | 0.08 | 0.12 | 0.04 | 0.08 | 0.03 | 0.02 | 0.02 | 0.02 |
| 45–49 | 0.22 | 0.16 | 0.17 | 0.13 | 0.15 | 0.07 | 0.19 | 0.09 | 0.04 | 0.09 | 0.03 | 0.03 | 0.03 |
| 50–54 | 0.27 | 0.19 | 0.18 | 0.26 | 0.11 | 0.13 | 0.11 | 0.16 | 0.1 | 0.05 | 0.11 | 0.04 | 0.04 |
| 55–59 | 0.54 | 0.42 | 0.29 | 0.12 | 0.22 | 0.25 | 0.33 | 0.16 | 0.21 | 0.15 | 0.08 | 0.18 | 0.08 |
| 60–64 | 0.72 | 0.5 | 0.5 | 0.45 | 0.4 | 0.36 | 0.52 | 0.28 | 0.22 | 0.32 | 0.25 | 0.16 | 0.32 |
| 65–69 | 1.11 | 1 | 0.93 | 0.64 | 0.44 | 0.56 | 0.62 | 0.51 | 0.43 | 0.28 | 0.42 | 0.36 | 0.24 |
| 70–74 | 2.51 | 2.34 | 1.57 | 1.23 | 1.1 | 0.96 | 0.92 | 0.65 | 0.77 | 0.69 | 0.5 | 0.77 | 0.67 |
| 75–79 | 5.27 | 4.62 | 3.38 | 2.91 | 2.02 | 1.89 | 1.86 | 1.54 | 1.46 | 1.44 | 1.38 | 1.11 | 1.59 |
| 80–84 | 9.99 | 12.27 | 9.92 | 6.52 | 5.78 | 4.53 | 4.41 | 4.02 | 3.31 | 3.24 | 3.35 | 3.37 | 2.81 |
| >85 | 35.76 | 42.79 | 38.63 | 32.8 | 27.26 | 22.45 | 21.15 | 21.18 | 17.52 | 15.98 | 16.26 | 17.2 | 17.28 |
| SMR | 1.67 | 1.86 | 1.57 | 1.28 | 1.06 | 0.89 | 0.88 | 0.81 | 0.69 | 0.63 | 0.63 | 0.65 | 0.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sendín-Martin, M.; Hernández-Rodríguez, J.C.; Durán-Romero, A.-J.; Ortiz-Álvarez, J.; Conejo-Mir, J.; Pereyra-Rodríguez, J.-J. Non-Melanoma Skin Cancer Mortality in Spain: A Predictive Model up to 2044. J. Clin. Med. 2021, 10, 5750. https://doi.org/10.3390/jcm10245750
Sendín-Martin M, Hernández-Rodríguez JC, Durán-Romero A-J, Ortiz-Álvarez J, Conejo-Mir J, Pereyra-Rodríguez J-J. Non-Melanoma Skin Cancer Mortality in Spain: A Predictive Model up to 2044. Journal of Clinical Medicine. 2021; 10(24):5750. https://doi.org/10.3390/jcm10245750
Chicago/Turabian StyleSendín-Martin, Mercedes, Juan Carlos Hernández-Rodríguez, Antonio-José Durán-Romero, Juan Ortiz-Álvarez, Julian Conejo-Mir, and José-Juan Pereyra-Rodríguez. 2021. "Non-Melanoma Skin Cancer Mortality in Spain: A Predictive Model up to 2044" Journal of Clinical Medicine 10, no. 24: 5750. https://doi.org/10.3390/jcm10245750
APA StyleSendín-Martin, M., Hernández-Rodríguez, J. C., Durán-Romero, A.-J., Ortiz-Álvarez, J., Conejo-Mir, J., & Pereyra-Rodríguez, J.-J. (2021). Non-Melanoma Skin Cancer Mortality in Spain: A Predictive Model up to 2044. Journal of Clinical Medicine, 10(24), 5750. https://doi.org/10.3390/jcm10245750







