Effect of Automated Oxygen Titration during Walking on Dyspnea and Endurance in Chronic Hypoxemic Patients with COPD: A Randomized Crossover Trial
Abstract
1. Introduction
2. Materials and Methods
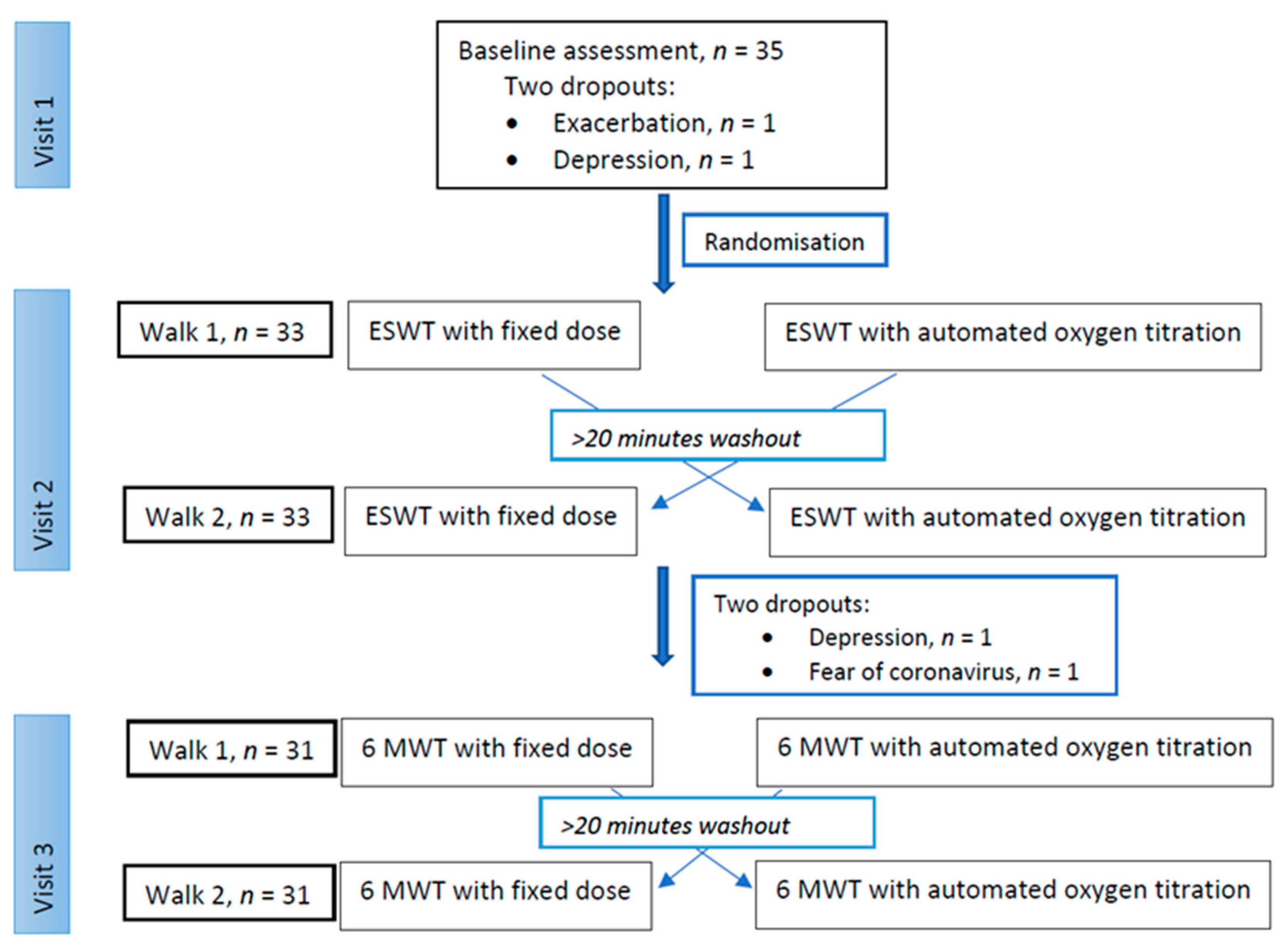
2.1. Patients and Study Design
2.2. Oxygen Equipment and Randomisation
- (a)
- visit 2: ESWT with AOT followed by FD; visit 3: 6MWT with AOT followed by FD, or
- (b)
- visit 2: ESWT with FD followed by AOT; visit 3: 6MWT with AOT followed by FD, or
- (c)
- visit 2: ESWT with AOT followed by FD; visit 3: 6MWT with FD followed by AOT, or
- (d)
- visit 2: ESWT with FD followed by AOT; visit 3: 6MWT with FD followed by AOT.
2.3. Study Visits
2.4. Outcome Measures
- Difference in walking time between arms in ESWT;
- Difference in experienced dyspnea and walking distance in the 6MWT;
- Difference in average oxygen flow (L/min) between arms in both ESWT and 6MWT;
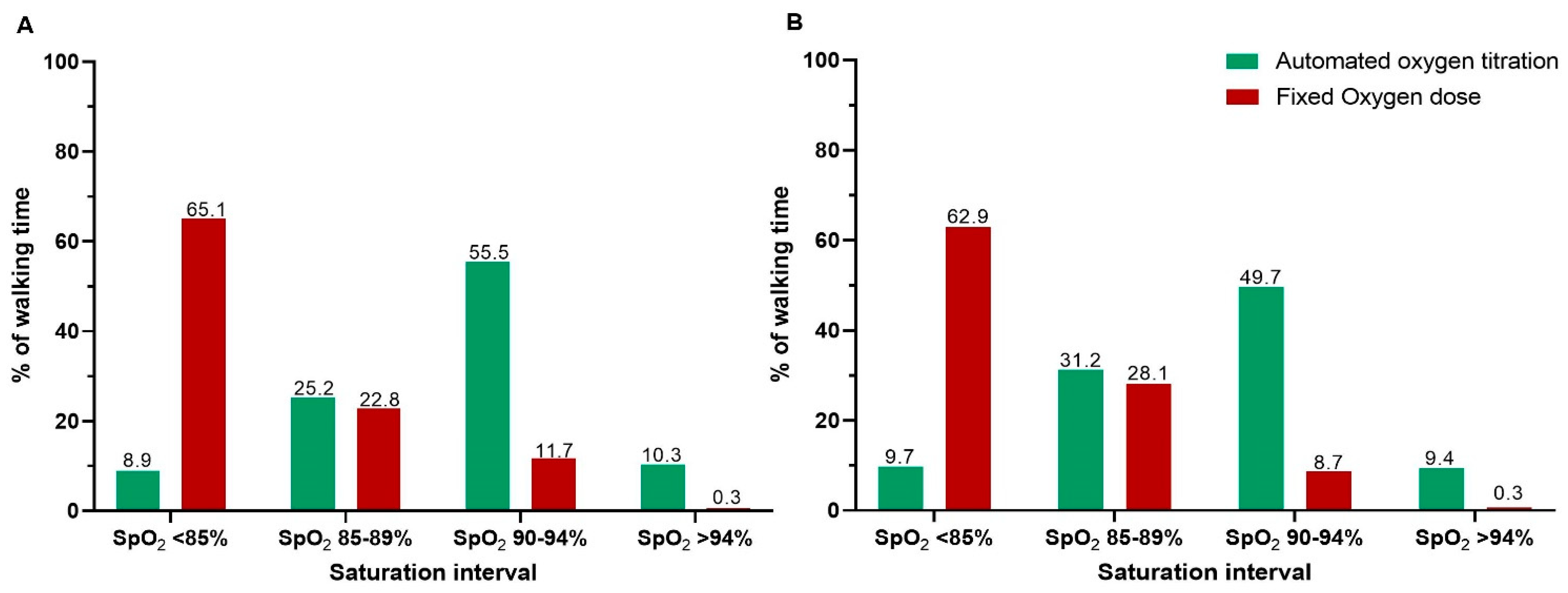
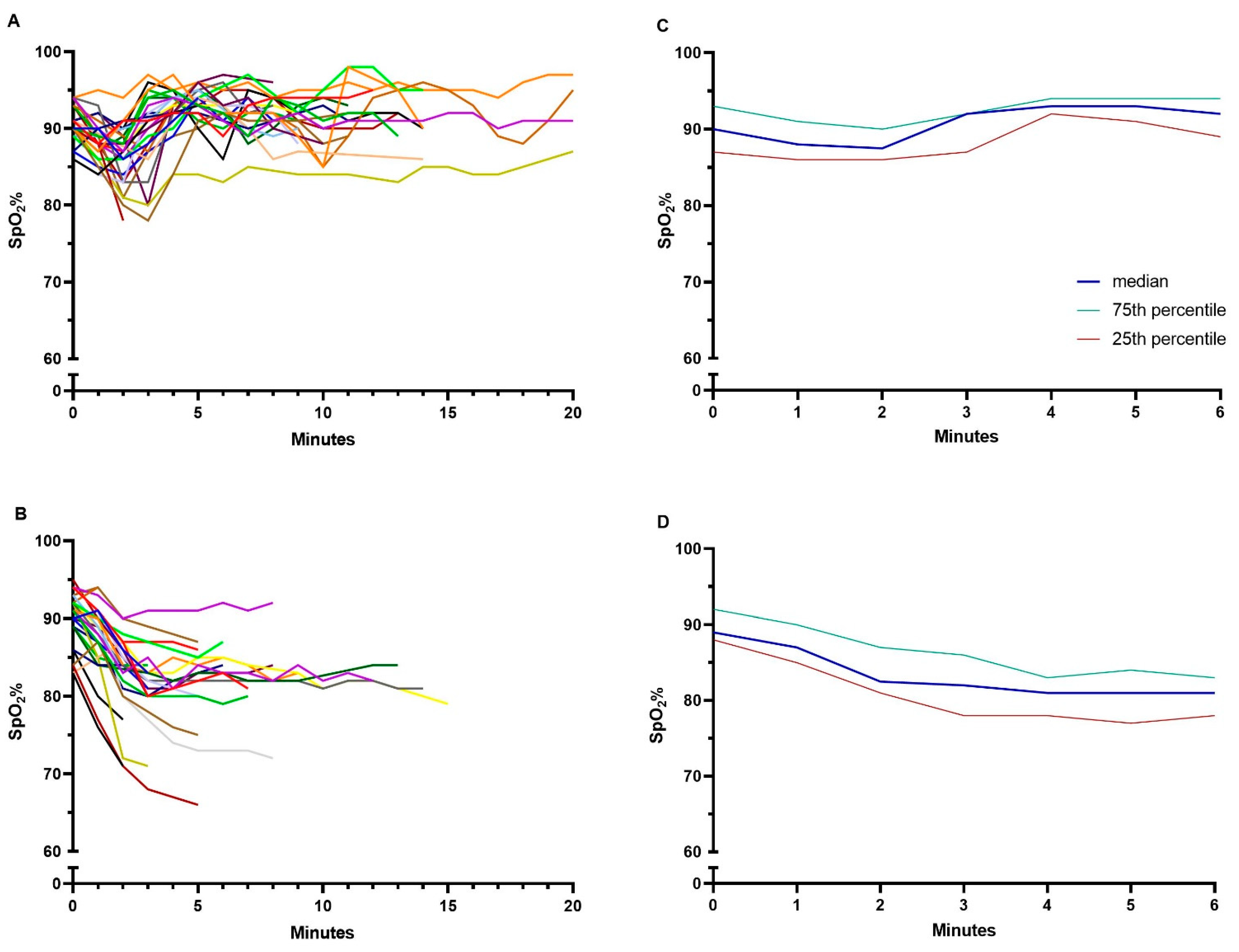
- Time spent within acceptable saturation interval (SpO2 90–94%), and time spent with moderate hypoxemia (SpO2 < 88%) and severe hypoxemia (SpO2 < 85%) in both tests.
2.5. Statistics
3. Results
3.1. Endurance Shuttle Walk Test
3.2. The Six-Minute Walk Test
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GOLD—Guidelines. From the Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD). 2021. Available online: www.goldcopd.org. (accessed on 20 September 2021).
- McCarthy, B.; Casey, D.; Devane, D. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. 2015, 2 (Suppl. S2), CD003793. [Google Scholar] [CrossRef] [PubMed]
- Hardinge, M.; Annandale, J.; Bourne, S.; Cooper, B.; Evans, A.; Freeman, D.; Green, A.; Hippolyte, S.; Knowles, V.; MacNee, W.; et al. British Thoracic Society guidelines for home oxygen use in adults: Accredited by NICE. Thorax 2015, 70, i1–i43. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, S.S.; Krishnan, J.A.; Lederer, D.J.; Ghazipura, M.; Hossain, T.; Tan, A.-Y.M.; Carlin, B.; Drummond, M.B.; Ekström, M.; Garvey, C.; et al. Home Oxygen Therapy for Adults with Chronic Lung Disease. An Official American Thoracic Society Clinical Practice Guideline. Am. J. Respir. Crit. Care Med. 2020, 202, e121–e141. [Google Scholar] [CrossRef] [PubMed]
- Jensen, D.; Schaeffer, M.R.; Guenette, J.A. Pathophysiological mechanisms of exertional breathlessness in chronic obstructive pulmonary disease and interstitial lung disease. Curr. Opin. Support. Palliat. Care 2018, 12, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Wijkstra, P.; Guyatt, G.; Ambrosino, N.; Celli, B.; Guell, R.; Muir, J.; Prefaut, C.; Mendes, E.; Ferreira, I.; Austin, P.; et al. International approaches to the prescription of long-term oxygen therapy. Eur. Respir. J. 2001, 18, 909–913. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Kim, H.I.; Park, J.Y.; Hong, J.Y.; Kim, J.-H.; Min, K.H.; Rhee, C.K.; Park, S.; Lee, C.Y.; Lim, S.Y.; et al. Korean physician prescription patterns for home oxygen therapy in chronic obstructive pulmonary disease patients. Korean J. Intern. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Güell Rous, R. Long-term oxygen therapy: Are we prescribing appropriately? Int. J. Chron. Obstruct. Pulmon. Dis. 2008, 3 (Suppl. S2), 231–237. [Google Scholar] [CrossRef] [PubMed]
- Hansen, E.F.; Hove, J.D.; Bech, C.S.; Jensen, J.S.; Kallemose, T.; Vestbo, J. Automated oxygen control with O2matic((R)) during admission with exacerbation of COPD. Int. J. Chron. Obstruct. Pulmon Dis. 2018, 13, 3997–4003. [Google Scholar] [CrossRef]
- Lellouche, F.; Bouchard, P.-A.; Roberge, M.; Simard, S.; L’Her, E.; Maltais, F.; Lacasse, Y. Automated oxygen titration and weaning with FreeO2 in patients with acute exacerbation of COPD: A pilot randomized trial. Int. J. Chronic Obstr. Pulm. Dis. 2016, 11, 1983–1990. [Google Scholar] [CrossRef]
- Vivodtzev, I.; L’Her, E.; Vottero, G.; Yankoff, C.; Tamisier, R.; Maltais, F.; Lellouche, F.; Pépin, J.-L. Automated O2 titration improves exercise capacity in patients with hypercapnic chronic obstructive pulmonary disease: A randomised controlled cross-over trial. Thorax 2018, 74, 298–301. [Google Scholar] [CrossRef]
- Lellouche, F.; L’Her, E.; Bouchard, P.-A.; Brouillard, C.; Maltais, F. Automatic Oxygen Titration During Walking in Subjects With COPD: A Randomized Crossover Controlled Study. Respir. Care 2016, 61, 1456–1464. [Google Scholar] [CrossRef] [PubMed]
- Spruit, M.A.; Singh, S.J.; Garvey, C.; ZuWallack, R.; Nici, L.; Rochester, C.; Hill, K.; Holland, A.E.; Lareau, S.C.; Man, W.D.-C.; et al. An Official American Thoracic Society/European Respiratory Society Statement: Key Concepts and Advances in Pulmonary Rehabilitation. Am. J. Respir. Crit. Care Med. 2013, 188, e13–e64. [Google Scholar] [CrossRef]
- Holland, A.E.; Spruit, M.A.; Troosters, T.; Puhan, M.A.; Pepin, V.; Saey, D.; McCormack, M.C.; Carlin, B.W.; Sciurba, F.C.; Pitta, F.; et al. An official European Respiratory Society/American Thoracic Society technical standard: Field walking tests in chronic respiratory disease. Eur. Respir. J. 2014, 44, 1428–1446. [Google Scholar] [CrossRef]
- Puhan, M.A.; Chandra, D.; Mosenifar, Z.; Ries, A.; Make, B.; Hansel, N.N.; Wise, R.; Sciurba, F.; for the National Emphysema Treatment Trial (NETT) Research Group. The minimal important difference of exercise tests in severe COPD. Eur. Respir. J. 2010, 37, 784–790. [Google Scholar] [CrossRef]
- Løkke, A.; Titlestad, I.; Marsaa, K. Dansk KOL-Vejledning. 2017, p. 2. Available online: https://lungemedicin.dk/kol-behandling/ (accessed on 20 September 2021).
- Ekström, M. Why treatment efficacy on breathlessness in laboratory but not daily life trials? The importance of standardized exertion. Curr. Opin. Support. Palliat. Care 2019, 13, 179–183. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Minor, B.L.; Elliott, V.; Fernandez, M.; O’Neal, L.; McLeod, L.; Delacqua, G.; Delacqua, F.; Kirby, J.; et al. The REDCap consortium: Building an international community of software platform partners. J. Biomed. Inform. 2019, 95, 103208. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef]
- Ries, A.L. Minimally Clinically Important Difference for the UCSD Shortness of Breath Questionnaire, Borg Scale, and Visual Analog Scale. COPD: J. Chronic Obstr. Pulm. Dis. 2005, 2, 105–110. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, D.E.; Banzett, R.B.; Carrieri-Kohlman, V.; Casaburi, R.; Davenport, P.W.; Gandevia, S.C.; Gelb, A.F.; Mahler, D.A.; Webb, K.A. Pathophysiology of Dyspnea in Chronic Obstructive Pulmonary Disease: A Roundtable. Proc. Am. Thorac. Soc. 2007, 4, 145–168. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, D.E.; D’Arsigny, C.; Webb, K.A. Effects of Hyperoxia on Ventilatory Limitation During Exercise in Advanced Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2001, 163, 892–898. [Google Scholar] [CrossRef]
- Garrod, R.; A Paul, E.; A Wedzicha, J. Supplemental oxygen during pulmonary rehabilitation in patients with COPD with exercise hypoxaemia. Thorax 2000, 55, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Jarosch, I.; Gloeckl, R.; Damm, E.; Schwedhelm, A.L.; Buhrow, D.; Jerrentrup, A.; Spruit, M.A.; Kenn, K. Short-term Effects of Supplemental Oxygen on 6-Min Walk Test Outcomes in Patients With COPD: A Randomized, Placebo-Controlled, Single-blind, Crossover Trial. Chest 2017, 151 (Suppl. S4), 795–803. [Google Scholar] [CrossRef] [PubMed]
- Nasilowski, J.; Przybylowski, T.; Zielinski, J.; Chazan, R. Comparing supplementary oxygen benefits from a portable oxygen concentrator and a liquid oxygen portable device during a walk test in COPD patients on long-term oxygen therapy. Respir. Med. 2008, 102, 1021–1025. [Google Scholar] [CrossRef] [PubMed]
- Soguel Schenkel, N.; Burdet, L.; de Muralt, B.; Fitting, J.W. Oxygen saturation during daily activities in chronic obstructive pulmonary disease. Eur. Respir. J. 1996, 9 (Suppl. S12), 2584–2589. [Google Scholar] [CrossRef] [PubMed]
| Variables | |
|---|---|
| Men | 12 (36%) |
| Women | 21 (64%) |
| Age, years | 72.7 ± 6.5 |
| Body Mass Index, kg/m2 | 25.6 (21.5–29.0) |
| MRC | 4 (3–4) |
| CAT, points 0–40 | 17.3 ± 6.2 |
| LTOT dose, L/min | 1.6 ± 0.9 |
| SpO2 at rest with LTOT, % | 91.7 ± 2.3 |
| Borg CR10 dyspnea at rest | 1 (0–1.5) |
| Hospitalisations last year, no. | 1.0 (0–2.0) |
| Smocking status, no. | |
| • Smoker | 1 (3%) |
| • Ex-smoker | 31 (94%) |
| • Never-smoker | 1 (3%) |
| Pack years | 44.5 (30.7–60.0) |
| Hand grip strength, men, kg | 37.2 ± 9.8 |
| Hand grip strength, women, kg | 21.3 ± 5.7 |
| ISWT, meters | 120 (85–165) |
| Borg CR 10 after ISWT | 7 (5–8) |
| ESWT, walking speed, km/t | 2.44 (2.09–2.72) |
| Usual walking aid, no. | |
| • No walking aid | 12 (36%) |
| • Rollator | 19 (58%) |
| • A walking stick | 2 (6%) |
| FEV1, liter | 0.6 (0.5–0.9) |
| FEV1, % of predicted | 28 (23–41) |
| FEV1/FVC, ratio | 0.43 (0.35–0.48) |
| Comorbidities, no. | |
| • Ischemic heart disease | 4 (12%) |
| • Heart failure | 8 (24%) |
| • Diabetes | 1 (3%) |
| • Osteoporosis | 18 (55%) |
| • Osteoarthritis (hip or knee) | 10 (30%) |
| • No comorbidities | 6 (18%) |
| • ≥2 comorbidities | 13 (39%) |
| Endurance Shuttle Walk Test, n = 33 | |||
| Variables | Fixed-Dose | Automated Dose | p-Value |
| Dyspnea, Borg CR10 score | 8 (5–9) | 4 (3–6) | <0.001 |
| Walking time, minutes | 5.5 (3.3–7.9) | 10.9 (6.5–14.9) | <0.001 |
| Time with target saturation, minutes | 0.7 (0–1.1) | 5.3 (2.6–7.8) | <0.001 |
| Median saturation, % | 82 (82–84) | 91 (90–94) | <0.001 |
| Average flow, L/min | 1.6 ± 0.9 | 7.9 ± 3.1 | <0.001 |
| Six-Minute Walk Test, n = 31 | |||
| Variables | Fixed-Dose | Automated Dose | p-Value |
| Dyspnea, Borg CR10 score | 7 (6–8) | 5 (3–7) | <0.001 |
| Walking distance, meters | 271.3 ± 65.1 | 291.6 ± 67.4 | <0.001 |
| Time with target saturation, minutes | 0.3 (0–1.0) | 3.0 (1.5–3.9) | <0.001 |
| Median saturation, % | 81 (78–85) | 92 (88–93) | <0.001 |
| Average flow, L/min | 1.6 ± 0.9 | 8.1 ± 3.6 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kofod, L.M.; Westerdahl, E.; Kristensen, M.T.; Brocki, B.C.; Ringbæk, T.; Hansen, E.F. Effect of Automated Oxygen Titration during Walking on Dyspnea and Endurance in Chronic Hypoxemic Patients with COPD: A Randomized Crossover Trial. J. Clin. Med. 2021, 10, 4820. https://doi.org/10.3390/jcm10214820
Kofod LM, Westerdahl E, Kristensen MT, Brocki BC, Ringbæk T, Hansen EF. Effect of Automated Oxygen Titration during Walking on Dyspnea and Endurance in Chronic Hypoxemic Patients with COPD: A Randomized Crossover Trial. Journal of Clinical Medicine. 2021; 10(21):4820. https://doi.org/10.3390/jcm10214820
Chicago/Turabian StyleKofod, Linette Marie, Elisabeth Westerdahl, Morten Tange Kristensen, Barbara Cristina Brocki, Thomas Ringbæk, and Ejvind Frausing Hansen. 2021. "Effect of Automated Oxygen Titration during Walking on Dyspnea and Endurance in Chronic Hypoxemic Patients with COPD: A Randomized Crossover Trial" Journal of Clinical Medicine 10, no. 21: 4820. https://doi.org/10.3390/jcm10214820
APA StyleKofod, L. M., Westerdahl, E., Kristensen, M. T., Brocki, B. C., Ringbæk, T., & Hansen, E. F. (2021). Effect of Automated Oxygen Titration during Walking on Dyspnea and Endurance in Chronic Hypoxemic Patients with COPD: A Randomized Crossover Trial. Journal of Clinical Medicine, 10(21), 4820. https://doi.org/10.3390/jcm10214820








