A 3-Year Observational Study of Patients with Progressive Systemic Sclerosis Treated with an Intensified B Lymphocyte Depletion Protocol: Clinical and Immunological Response
Abstract
1. Introduction
2. Aim
3. Patients and Methods
3.1. Protocol
Therapeutic Schedules
3.2. Statistical Analysis
3.3. Outcomes
Lung and Skin Fibrosis Assessment
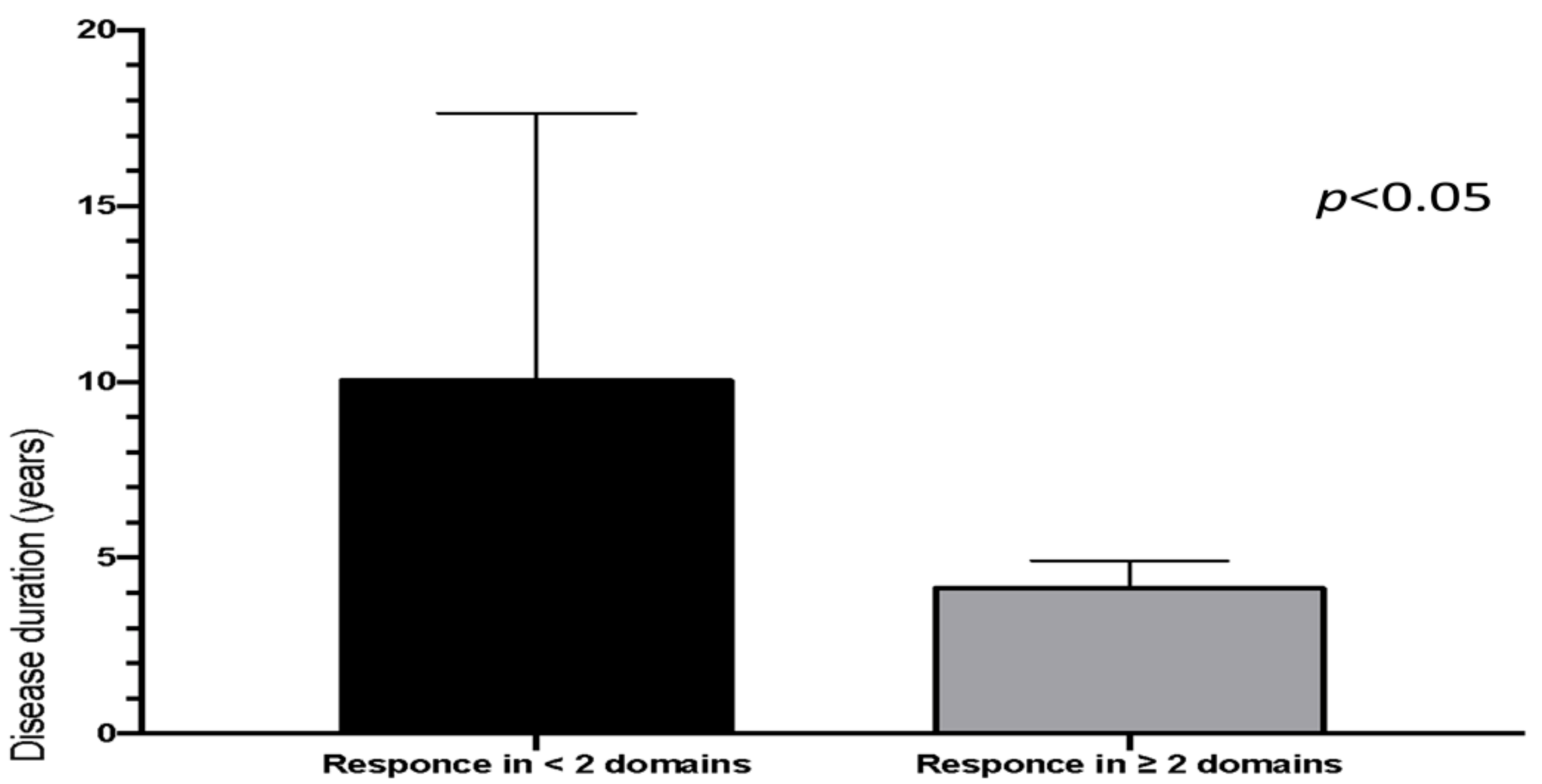
4. Results
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Elhai, M.; Meune, C.; Avouac, J.; Kahan, Y. Allanore Trends in mortality in patients with systemic sclerosis over 40 years: A systematic review and meta-analysis of cohort studies. Rheumatology 2012, 51, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, L.; Chikanza, I.C.; Platsoucas, C.D. Mechanisms of Disease: The role of immune cells in the pathogenesis of systemic sclerosis. Nat. Clin. Pract. Rheumatol. 2006, 2, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Denton, C.P.; Khanna, D. Systemic sclerosis. Lancet 2017, 390, 1685–1699. [Google Scholar] [CrossRef]
- Hao, Y.; Hudson, M.; Baron, M.; Carreira, P.; Stevens, W.; Rabusa, C.; Tatibouet, S.; Carmona, L.; Joven, B.E.; Huq, M.; et al. Early mortality in a multinational systemic sclerosis inception cohort. Arthritis Rheumatol. 2017, 69, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- Tashkin, D.P.; Elashoff, R.; Clements, P.J.; Goldin, J.; Roth, M.D.; Furst, D.E.; Arriola, E.; Silver, R.; Strange, C.; Bolster, M.; et al. Cyclophosphamide versus placebo in scleroderma lung disease. N. Engl. J. Med. 2006, 354, 2655–2666. [Google Scholar] [CrossRef] [PubMed]
- Tashkin, D.P.; Elashoff, R.; Clements, P.J.; Roth, M.D.; Furst, D.E.; Silver, R.M.; Goldin, J.; Arriola, E.; Strange, C.; Bolster, M.B.; et al. Effects of 1-year treatment with cyclophosphamide on outcomes at 2 years in scleroderma lung disease. Am. J. Respir. Crit. Care Med. 2007, 176, 1026–1034. [Google Scholar] [CrossRef] [PubMed]
- Liossis, S.N.; Bounas, A.; Andonopoulos, A.P. Mycophenolate mofetil as first-line treatment improves clinically evident early scleroderma lung disease. Rheumatology 2006, 45, 1005–1008. [Google Scholar] [CrossRef] [PubMed]
- Nihtyanova, S.I.; Brough, G.M.; Black, C.M.; Denton, C.P. Mycophenolate mofetil in diffuse cutaneous systemic sclerosis—A retrospective analysis. Rheumatology 2007, 46, 442–445. [Google Scholar] [CrossRef] [PubMed]
- Smith, V.; Van Praet, J.T.; Vandooren, B.; Van Der Cruyssen, B.; Naeyaert, J.-M.; Decuman, S.; Elewaut, D.; De Keyser, F. Rituximab in diffuse cutaneous systemic sclerosis: An open-label clinical and histopathological study. Ann. Rheum. Dis. 2008, 69, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Smith, V.; Piette, Y.; Van Praet, J.T.; Decuman, S.; Deschepper, E.; Elewaut, D.; De Keyser, F. Two-year results of an open pilot study of a 2-treatment course with rituximab in patients with early systemic sclerosis with diffuse skin involvement. J. Rheumatol. 2012, 40, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Volkmann, E.R.; Varga, J. Emerging targets of disease-modifying therapy for systemic sclerosis. Nat. Rev. Rheumatol. 2019, 15, 208–224. [Google Scholar] [CrossRef] [PubMed]
- Daoussis, D.; Melissaropoulos, K.; Sakellaropoulos, G.; Antonopoulos, I.; Markatseli, T.E.; Simopoulou, T.; Georgiou, P.; Andonopoulos, A.P.; Drosos, A.; Sakkas, L.; et al. A multicenter, open-label, comparative study of B-cell depletion therapy with Rituximab for systemic sclerosis-associated interstitial lung disease. Semin. Arthritis Rheum. 2017, 46, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Steen, V.D.; Conte, C.; Owens, G.R.; Medsger, T.A. Severe restrictive lung disease in systemic sclerosis. Arthritis Rheum. 1994, 37, 1283–1289. [Google Scholar] [CrossRef] [PubMed]
- Keir, G.J.; Maher, T.M.; Ming, D.; Abdullah, R.; De Lauretis, A.; Wickremasinghe, M.; Nicholson, A.G.; Hansell, D.M.; Wells, A.U.; Renzoni, E.A. Rituximab in severe, treatment-refractory interstitial lung disease. Respirology 2014, 19, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Jordan, S.; Distler, J.H.W.; Maurer, B.; Huscher, D.; Van Laar, J.M.; Allanore, Y.; Distler, O. Effects and safety of rituximab in systemic sclerosis: An analysis from the European scleroderma trial and research (EUSTAR) group. Ann. Rheum. Dis. 2014, 74, 1188–1194. [Google Scholar] [CrossRef]
- Roccatello, D. “How I treat” autoimmune diseases: State of the art on themanagement of rare rheumatic diseases and ANCA-associated systemic idiopathicvasculitis. Autoimmun. Rev. 2017, 16, 995–998. [Google Scholar] [CrossRef][Green Version]
- Roccatello, D.; Sciascia, S.; Baldovino, S.; Rossi, D.; Alpa, M.; Naretto, C.; Di Simone, D.; Menegatti, E. Improved (4 plus 2) Rituximab protocol for severe cases of mixed cryoglobulinemia: A 6-year observational study. Am. J. Nephrol. 2016, 43, 251–260. [Google Scholar] [CrossRef]
- Roccatello, D.; Sciascia, S.; Di Simone, D.; Solfietti, L.; Naretto, C.; Fenoglio, R.; Baldovino, S.; Menegatti, E. New insights into immune mechanisms underlying response to Rituximab in patients with membranous nephropathy: A prospective study and a review of the literature. Autoimmun. Rev. 2016, 15, 529–538. [Google Scholar] [CrossRef]
- Roccatello, D.; Sciascia, S.; Baldovino, S.; Rossi, D.; Alpa, M.; Naretto, C.; Di Simone, D.; Simoncini, M.; Menegatti, E. A 4-year observation in lupus nephritis patients treated with an intensified B-lymphocyte depletion without immunosoppressive maintenance treatment—Clinical response compared to literature and immunological re-assessment. Autoimmun. Rev. 2015, 14, 1123–1130. [Google Scholar] [CrossRef]
- Ciurzyński, M.; Bienias, P.; Ciesielska, K.; Chrzanowska, A.; Dudzik-Niewiadomska, I.; Kurnicka, K.; Domienik-Karłowicz, J.; Siwicka, M.; Sobieraj, P.; Kalińska-Bienias, A.; et al. Accuracy of Doppler echocardiography in the hemo-dynamic assessment of pulmonary circulation in patients with systemic sclerosis. Adv. Med. Sci. 2019, 64, 309–314. [Google Scholar] [CrossRef]
- Goldin, J.G.; Lynch, D.A.; Strollo, D.C.; Suh, R.D.; Schraufnagel, D.E.; Clements, P.J.; Elashoff, R.M.; Furst, D.E.; Vasunilashorn, S.; McNitt-Gray, M.F.; et al. High-resolution CT scan findings in patients with symptomatic scleroderma-related interstitial lung disease. Chest 2008, 134, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Mavropoulo, A.; Simopoulou, T.; Varna, A.; Liaskos, C.; Katsiari, C.G.; Bogdanos, D.P.; Sakkas, L.I. Breg cells are numerically decreased and functionally impaired in patients with systemic sclerosis. Arthritis Rheumatol. 2016, 68, 494–504. [Google Scholar] [CrossRef] [PubMed]
- Asano, N.; Fujimoto, M.; Yazawa, N.; Shirasawa, S.; Hasegawa, M.; Okochi, H.; Tamaki, K.; Tedder, T.F.; Sato, S. B lymphocyte signaling established by the CD19/CD22 loop regulates autoimmunity in the tight-skin mouse. Am. J. Pathol. 2004, 165, 641–650. [Google Scholar] [CrossRef][Green Version]
- Sato, S.; Fujimoto, M.; Hasegawa, M.; Takehara, K. Altered blood B lymphocyte homeostasis in systemic sclerosis: Expanded naive B cells and diminished but activated memory B cells. Arthritis Rheum. 2004, 50, 1918–1927. [Google Scholar] [CrossRef]
- Francois, A.; Chatelus, E.; Wachsmann, S.; Sibilia, J.; Bahram, S.; Alsaleh, G.; Gottenberg, J.-E. Blymphocytes and B-cell activating factor promote collagen and profibrotic markers expression by dermal fibroblasts in systemic sclerosis. Arthritis. Res. Ther. 2013, 15, R168. [Google Scholar] [CrossRef]
- Hasegawa, M.; Hamaguchi, Y.; Yanaba, K.; Bouaziz, J.D.; Uchida, J.; Fujimoto, M.; Matsushita, T.; Matsushita, Y.; Horikawa, M.; Komura, K.; et al. B-lymphocyte depletion reduces skin fibrosis and autoimmunity in the tight-skin mouse model for systemic sclerosis. Am. J. Pathol. 2006, 169, 954–966. [Google Scholar] [CrossRef]
- Whitfield, M.L.; Finlay, D.R.; Murray, J.I.; Troyanskaya, O.G.; Chi, J.T.; Pergamenschikov, A.; McCalmont, T.H.; Brown, P.O.; Botstein, D.; Connolly, M.K. Systemic and cell type-specific gene expression patterns in scleroderma skin. Proc. Natl. Acad. Sci. USA 2003, 100, 12319–12324. [Google Scholar] [CrossRef]
- Lafyatis, R.; O’Hara, C.; Feghali-Bostwick, C.A.; Matteson, E. B cell infiltration in systemic sclerosis-associated interstitial lung disease. Arthritis Rheum. 2007, 56, 3167–3168. [Google Scholar] [CrossRef]
- Rossi, D.; Cecchi, I.; Radin, M.; Rubini, E.; Sciascia, S.; Roccatello, D. SAT0477 Intensified B-cell depletion therapy in progressive systemic sclerosis patients: 24 months follow-up. Ann. Rheum. Dis. 2018, 77, 1096. [Google Scholar]
- Hughes, M.; Denton, C.P.; Khanna, D. Rituximab for the treatment of systemic sclerosis-interstitial lung disease. Rheumatology 2020, keaa675. [Google Scholar] [CrossRef]
- Goswami, R.P.; Ray, A.; Chatterjee, M.; Mukherjee, A.; Sircar, G.; Ghosh, P. OUP accepted manuscript. Rheumatology 2020, keaa550. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Chung, M.P.; Gagne, L.; Guo, H.H.; Guenther, Z.; Li, S.; Jacobs, S.; Morisset, J.; Mooney, J.J.; Raj, R.; et al. Rituximab versus mycophenolate in the treatment of recalcitrant connective tissue disease–associated interstitial lung disease. ACR Open Rheumatol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Giuggioli, D.; Lumetti, F.; Colaci, M.; Fallahi, P.; Antonelli, A.; Ferri, C. Rituximab in the treatment of patients with systemic sclerosis. Our experience and review of the literature. Autoimmun. Rev. 2015, 14, 1072–1078. [Google Scholar] [CrossRef] [PubMed]
- Narváez, J.; Pirola, J.P.; LLuch, J.; Juarez, P.; Nolla, J.M.; Valenzuela, A. Effectiveness and safety of rituximab for the treatment of refractory systemic sclerosis associated calcinosis: A case series and systematic review of the literature. Autoimmun. Rev. 2019, 18, 262–269. [Google Scholar] [CrossRef]
- Daoussis, D.; Tsamandas, A.; Antonopoulos, I.; Filippopoulou, A.; Papachristou, D.J.; Papachristou, N.I.; Andonopoulos, A.P.; Liossis, S.N. B cell depletion therapy upregulates Dkk-1 skin expression in patients with systemic sclerosis: Association with enhanced resolution of skin fibrosis. Arthritis Res. Ther. 2016, 18, 118. [Google Scholar] [CrossRef]
- Trang, G.; Steele, R.; Baron, M.; Hudson, M. Corticosteroids and the risk of scleroderma renal crisis: A systematic review. Rheumatol. Int. 2010, 32, 645–653. [Google Scholar] [CrossRef]
- Melissaropoulos, K.; Kraniotis, P.; Bogdanos, D.P.; Dimitroulas, T.; Sakkas, L.; Daoussis, D. Targeting very early systemic sclerosis: A case-based review. Rheumatol. Int. 2019, 39, 1961–1970. [Google Scholar] [CrossRef]
- Alvis, B.D.; Hughes, C.G. Physiology considerations in geriatric patients. Anesthesiol. Clin. 2015, 33, 447–456. [Google Scholar] [CrossRef]
- Kolb, M.; Vašáková, M. The natural history of progressive fibrosing interstitial lung diseases. Respir. Res. 2019, 20, 57. [Google Scholar] [CrossRef]
| n | % | |
|---|---|---|
| Female | 18 | 90 |
| Age (Mean ± SD, Years) | 66.7 ± 11.0 | |
| Less than 5 Years Disease Duration | 9 | 45 |
| Raynaud’s | 20 | 100 |
| ILD | 20 | 100 |
| PAH | 12 | 60 |
| Skin Thickening | 17 | 85 |
| Esophagus Involvement | 9 | 45 |
| Polyarthritis | 5 | 25 |
| ANA | 20 | 100 |
| Anti-Topoisomerase I Antibodies (Scl-70) | 19 | 95% |
| NT-proBNPLevels (Baseline, Mean ± SD, pg/mL) | 385.4 ± 517 | |
| mRSS (Baseline, Mean ± SD) | 14.4 ± 10.5 | |
| FVC (Baseline, Mean ± SD) | 93.6 ± 19.3 | |
| FEV1 (Baseline, Mean ± SD) | 89.5 ± 15.6 | |
| DLCO (B, Mean ± SD) | 58.8 ± 8.6 | |
| Ejection Fraction (Baseline, Mean ± SD) | 62.8 ± 6.4 | |
| Pulmonary Artery Pressure (Baseline, Mean ± SD) | 30.2 ± 10.5 | |
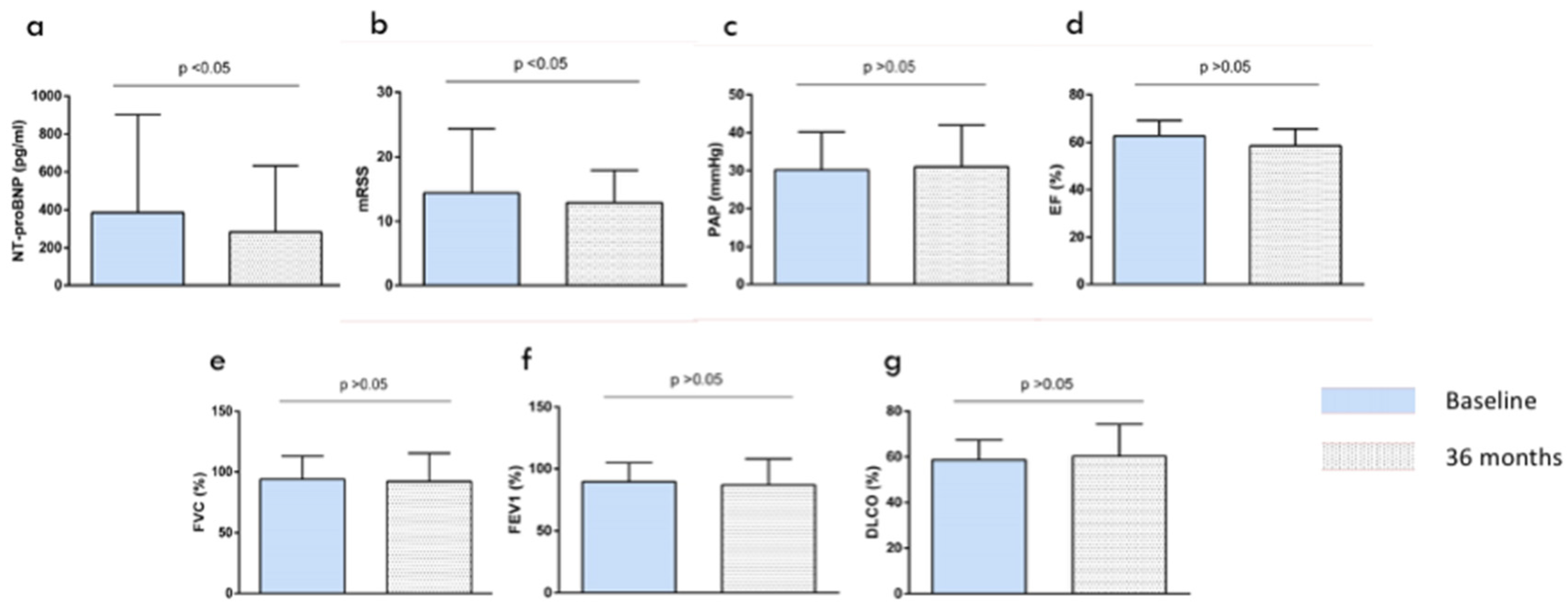
| Baseline | 24 Months | p-Value * | 36 Months | p-Value ** | |
|---|---|---|---|---|---|
| Rodnan Skin Score | 14.4 ± 10.5 | 12.9 ± 10 | p = 0.073 | 11.9 ± 9 | p < 0.05 |
| NT-proBNP (mean ± SD, pg/mL) | 385.4 ± 517 | 283 ± 648 | p < 0.05 | 279 ± 543 | p < 0.05 |
| FVC (mean ± SD) | 93.6 ± 19.3 | 92.2 ± 23.3 | p = 0.77 | 91.2 ± 22.7 | p = 0.63 |
| FEV1 (mean ± SD) | 89.5 ± 15.6 | 87 ± 21.2 | p = 0.59 | 88 ± 23.1 | p = 0.57 |
| DLCO | 58.8 ± 8.6 | 60.3 ± 14 | p = 0.71 | 59.9 ± 13.2 | p = 0.77 |
| Ejection fraction (mean ± SD) | 62.8 ± 6.4 | 58.6 ± 7.1 | p = 0.45 | 59.5 ± 7.9 | p = 0.34 |
| pulmonary artery pressure (mean ± SD) | 30.2 ± 10.5 | 31.1 ± 11.05 | p = 0.51 | 32.2 ± 12.1 | p = 0.49 |
| eGFR (mL/min/1.73 m2) | 71.4 ± 9.7 | 72.3 ± 10.6 | p = 0.75 | 70.2 ± 11.3 | p = 0.71 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossi, D.; Sciascia, S.; Cecchi, I.; Saracco, M.; Montabone, E.; Modena, V.; Pellerito, R.; Carignola, R.; Roccatello, D. A 3-Year Observational Study of Patients with Progressive Systemic Sclerosis Treated with an Intensified B Lymphocyte Depletion Protocol: Clinical and Immunological Response. J. Clin. Med. 2021, 10, 292. https://doi.org/10.3390/jcm10020292
Rossi D, Sciascia S, Cecchi I, Saracco M, Montabone E, Modena V, Pellerito R, Carignola R, Roccatello D. A 3-Year Observational Study of Patients with Progressive Systemic Sclerosis Treated with an Intensified B Lymphocyte Depletion Protocol: Clinical and Immunological Response. Journal of Clinical Medicine. 2021; 10(2):292. https://doi.org/10.3390/jcm10020292
Chicago/Turabian StyleRossi, Daniela, Savino Sciascia, Irene Cecchi, Marta Saracco, Erika Montabone, Vittorio Modena, Raffaele Pellerito, Renato Carignola, and Dario Roccatello. 2021. "A 3-Year Observational Study of Patients with Progressive Systemic Sclerosis Treated with an Intensified B Lymphocyte Depletion Protocol: Clinical and Immunological Response" Journal of Clinical Medicine 10, no. 2: 292. https://doi.org/10.3390/jcm10020292
APA StyleRossi, D., Sciascia, S., Cecchi, I., Saracco, M., Montabone, E., Modena, V., Pellerito, R., Carignola, R., & Roccatello, D. (2021). A 3-Year Observational Study of Patients with Progressive Systemic Sclerosis Treated with an Intensified B Lymphocyte Depletion Protocol: Clinical and Immunological Response. Journal of Clinical Medicine, 10(2), 292. https://doi.org/10.3390/jcm10020292







