Oxidative Stress and Inflammation Biomarker Expression in Obstructive Sleep Apnea Patients
Abstract
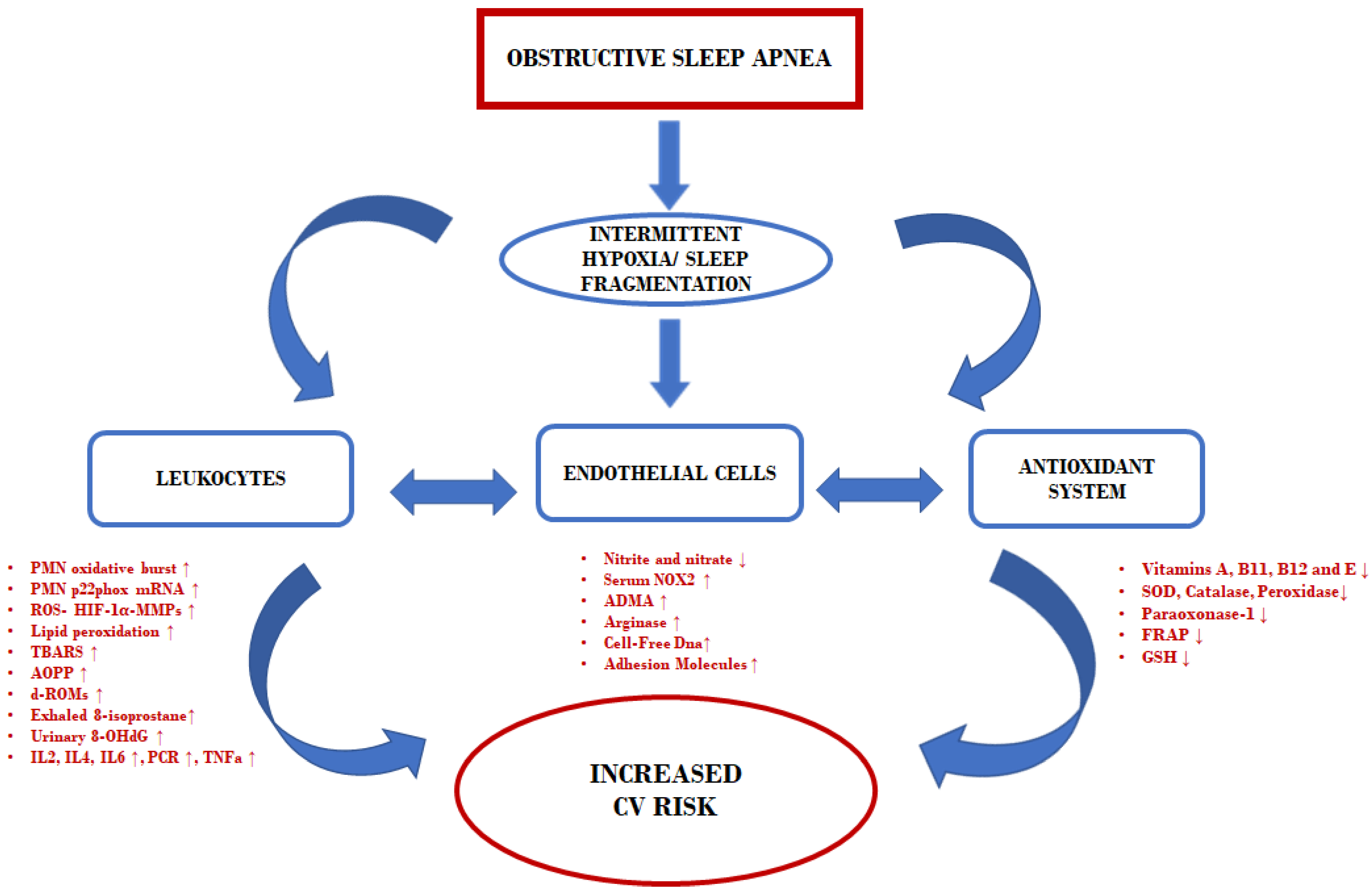
1. Introduction
2. Materials and Methods
Study Protocol
3. Results and Discussion
3.1. Oxidative Stress Markers
3.1.1. Leukocytes’ Oxidative Derived
3.1.2. NADPH Oxidase
3.1.3. Liperoxidation Products
3.1.4. Advanced Oxidation Protein Products (AOPP)
3.1.5. Circulating Free DNA
3.1.6. 8-Hydroxy-2-deoxyguanosine
3.2. Systemic Inflammation Markers and Circulating Cytokines
3.2.1. Tumour Necrosis Alpha Factor (TNF-alpha)
3.2.2. Protein C Reactive
3.2.3. Endothelial Related Markers
3.3. Antioxidant System Impairment
3.4. Physiopathological Features and Inflammatory Profiles
4. Data Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kapur, V.K.; Auckley, D.H.; Chowdhuri, S.; Kuhlmann, D.C.; Mehra, R.; Ramar, K.; Harrod, C.G. Clinical Practice Guideline for Diagnostic Testing for Adult Obstructive Sleep Apnea: An American Academy of Sleep Medicine Clinical Practice Guideline. J. Clin. Sleep Med. 2017, 13, 479–504. [Google Scholar] [CrossRef] [PubMed]
- Paruthi, S.; Rosen, C.L.; Wang, R.; Weng, J.; Marcus, C.L.; Chervin, R.D.; Stanley, J.J.; Katz, E.S.; Amin, R.; Redline, S. End-Tidal Carbon Dioxide Measurement during Pediatric Polysomnography: Signal Quality, Association with Apnea Severity, and Prediction of Neurobehavioral Outcomes. Sleep 2015, 38, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Cuspidi, C.; Tadic, M.; Sala, C.; Gherbesi, E.; Grassi, G.; Mancia, G. Blood Pressure Non-Dipping and Obstructive Sleep Apnea Syndrome: A Meta-Analysis. J. Clin. Med. 2019, 8, 1367. [Google Scholar] [CrossRef] [PubMed]
- Drager, L.F.; McEvoy, R.D.; Barbe, F.; Lorenzi-Filho, G.; Redline, S. INCOSACT Initiative (International Collaboration of Sleep Apnea Cardiovascular Trialists). Sleep Apnea and Cardiovascular Disease: Lessons from Recent Trials and Need for Team Science. Circulation 2017, 136, 1840–1850. [Google Scholar] [CrossRef] [PubMed]
- Iannella, G.; Maniaci, A.; Magliulo, G.; Cocuzza, S.; La Mantia, I.; Cammaroto, G.; Greco, A.; Vicini, C. Current challenges in the diagnosis and treatment of obstructive sleep apnea syndrome in the elderly [published online ahead of print, 2020 Apr 6]. Pol. Arch. Intern. Med. 2020. [Google Scholar] [CrossRef]
- Vicini, C.; De Vito, A.; Iannella, G.; Gobbi, R.; Corso, R.M.; Montevecchi, F.; Polimeni, A.; De Vincentiis, M.; Meccariello, G.; D’Agostino, G.; et al. The aging effect on upper airways collapse of patients with obstructive sleep apnea syndrome. Eur. Arch. Otorhinolaryngol. 2018, 275, 2983–2990. [Google Scholar] [CrossRef]
- Iannella, G.; Vicini, C.; Colizza, A.; Meccariello, G.; Polimeni, A.; Greco, A.; de Vincentiis, M.; de Vito, A.; Cammaroto, G.; Gobbi, R.; et al. Aging effect on sleepiness and apneas severity in patients with obstructive sleep apnea syndrome: A meta-analysis study. Eur. Arch. Otorhinolaryngol. 2019, 276, 3549–3556. [Google Scholar] [CrossRef]
- Baratta, F.; Pastori, D.; Fabiani, M.; Fabiani, V.; Ceci, F.; Lillo, R.; Lolli, V.; Brunori, M.; Pannitteri, G.; Cravotto, E.; et al. Severity of OSAS, CPAP and cardiovascular events: A follow-up study. Eur. J. Clin. Investig. 2018, 48, e12908. [Google Scholar] [CrossRef]
- Bradley, T.D.; Floras, J.S. Obstructive sleep apnoea and its cardiovascular consequences. Lancet 2009, 373, 82–93. [Google Scholar] [CrossRef]
- Vanek, J.; Prasko, J.; Genzor, S.; Ociskova, M.; Kantor, K.; Holubova, M.; Slepecky, M.; Nesnidal, V.; Kolek, A.; Sova, M. Obstructive sleep apnea, depression and cognitive impairment [published online ahead of print, 2020 Mar 23]. Sleep Med. 2020, 72, 50–58. [Google Scholar] [CrossRef]
- Di Luca, M.; Iannella, G.; Montevecchi, F.; Magliulo, G.; De Vito, A.; Cocuzza, S.; Maniaci, A.; Meccariello, G.; Cammaroto, G.; Sgarzani, R.; et al. use of the transoral robotic surgery to treat patients with recurrent lingual tonsillitis. Int. J. Med. Robot. Comput. Assist. Surg. 2020, 16, 2106. [Google Scholar] [CrossRef] [PubMed]
- Iannella, G.; Magliulo, G.; Di Luca, M.; De Vito, A.; Meccariello, G.; Cammaroto, G.; Pelucchi, S.; Bonsembiante, A.; Maniaci, A.; Vicini, C. Lateral pharyngoplasty techniques for obstructive sleep apnea syndrome: A comparative experimental stress test of two different techniques. Eur. Arch. Otorhinolaryngol. 2020, 277, 1793–1800. [Google Scholar] [CrossRef] [PubMed]
- Iannella, G.; Vallicelli, B.; Magliulo, G.; Cammaroto, G.; Meccariello, G.; De Vito, A.; Greco, A.; Pelucchi, S.; Sgarzani, R.; Corso, R.; et al. Long-Term Subjective Outcomes of Barbed Reposition Pharyngoplasty for Obstructive Sleep Apnea Syndrome Treatment. Int. J. Environ. Res. Public Heal. 2020, 17, 1542. [Google Scholar] [CrossRef] [PubMed]
- Vgontzas, A.N.; Bixler, E.O.; Chrousos, G.P. Sleep apnea is a manifestation of the metabolic syndrome. Sleep Med. Rev. 2005, 9, 211–224. [Google Scholar] [CrossRef]
- Alves, E.; Ackel-D’Elia, C.; Luz, G.P.; Cunha, T.C.; Carneiro, G.; Tufik, S.; Bittencourt, L.R.; de Mello, M.T. Does physical exercise reduce excessive daytime sleepiness by improving inflammatory profiles in obstructive sleep apnea patients? Sleep Breath. 2012, 17, 505–510. [Google Scholar] [CrossRef]
- Pace, A.; Iannella, G.; Rossetti, V.; Visconti, I.C.; Gulotta, G.; Cavaliere, C.; De Vito, A.; Maniaci, A.; Cocuzza, S.; Magliulo, G.; et al. Diagnosis of Obstructive Sleep Apnea in Patients with Allergic and Non-Allergic Rhinitis. Medicina 2020, 56, 454. [Google Scholar] [CrossRef]
- Kheirandish-Gozal, L.; Gozal, D. Obstructive Sleep Apnea and Inflammation: Proof of Concept Based on Two Illustrative Cytokines. Int. J. Mol. Sci. 2019, 20, 459. [Google Scholar] [CrossRef]
- Borges, Y.G.; Cipriano, L.; Aires, R.; Zovico, P.; Campos, F.V.; de Araújo, M.; Gouvea, S.A. Oxidative stress and inflammatory profiles in obstructive sleep apnea: Are short-term CPAP or aerobic exercise therapies effective? Sleep Breath. 2019, 24, 541–549. [Google Scholar] [CrossRef]
- Wang, F.; Liu, Y.; Xu, H.; Qian, Y.; Zou, J.; Yi, H.; Guan, J.; Yin, S. Association between Upper-airway Surgery and Ameliorative Risk Markers of Endothelial Function in Obstructive Sleep Apnea. Sci. Rep. 2019, 9, 20157. [Google Scholar] [CrossRef]
- Lino, D.; Freitas, I.A.; Meneses, G.C.; Martins, A.; Daher, E.F.; Rocha, J.; Silva Junior, G.B. Interleukin-6 and adhesion molecules VCAM-1 and ICAM-1 as biomarkers of post-acute myocardial infarction heart failure. Braz. J. Med. Biol. Res. 2019, 52, e8658. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, W.; Wang, T.; Wu, J.; Zhong, X.; Gao, K.; Liu, Y.; He, X.; Zhou, Y.; Wang, H.; et al. Obstructive sleep apnea syndrome promotes the progression of aortic dissection via a ROS- HIF-1α-MMPs associated pathway. Int. J. Biol. Sci. 2019, 15, 2774–2782. [Google Scholar] [CrossRef] [PubMed]
- Düger, M.; Seyhan, E.C.; Günlüoğlu, M.Z.; Bolatkale, M.; Ozgul, M.A.; Turan, D.; Uğur, E.; Ülfer, G. Does ischemia-modified albumin level predict severity of obstructive sleep apnea? Sleep Breath. 2020, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Passali, D.; Corallo, G.; Yaremchuk, S.; Longini, M.; Proietti, F.; Passali, G.C.; Bellussi, L. Oxidative stress in patients with obstructive sleep apnoea syndrome. Acta Otorhinolaryngol. Ital. 2015, 35, 420–425. [Google Scholar]
- Ntalapascha, M.; Makris, D.; Kyparos, A.; Tsilioni, I.; Kostikas, K.; Gourgoulianis, K.; Kouretas, D.; Zakynthinos, E. Oxidative stress in patients with obstructive sleep apnea syndrome. Sleep Breath. 2012, 17, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Lira, A.B.; de Sousa Rodrigues, C.F. Evaluation of oxidative stress markers in obstructive sleep apnea syndrome and additional antioxidant therapy: A review article. Sleep Breath. 2016, 20, 1155–1160. [Google Scholar] [CrossRef]
- Franco, C.M.R.; Lima, A.M.J.; Ataide, L., Jr.; Lins, O.G.; Castro, C.M.M.; Bezerra, A.A.; de Oliveira, M.F.; Oliveira, J.R.M. Obstructive sleep apnea severity correlates with cellular and plasma oxidative stress parameters and affective symptoms. J. Mol. Neurosci. 2012, 47, 300–310. [Google Scholar] [CrossRef]
- Schulz, R.; Mahmoudi, S.; Hattar, K.; Sibelius, U.; Olschewski, H.; Mayer, K.; Seeger, W.; Grimminger, F. Enhanced release of superoxide from polymorphonuclear neutrophils in obstructive sleep apnea. Impact of continuous positive airway pressure therapy. Am. J. Respir. Crit. Care Med. 2000, 162, 566–570. [Google Scholar] [CrossRef]
- Liu, H.G.; Zhou, Y.N.; Liu, K.; Xu, Y.J. Nicotinamide-adenine dinucleotide phosphate oxidase p22phox expression in induced sputum cells for patients with obstructive sleep apnea hypopnea syndrome. Chin. J. Tuberc. Respir. Dis. 2010, 33, 118–122. [Google Scholar]
- Hopps, E.; Canino, B.; Calandrino, V.; Montana, M.; Lo Presti, R.; Caimi, G. Lipid peroxidation and protein oxidation are related to the severity of OSAS. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 3773–3778. [Google Scholar]
- Biller, J.D.; Takahashi, L.S. Oxidative stress and fish immune system: Phagocytosis and leukocyte respiratory burst activity. An. Acad. Bras. Ciências 2018, 90, 3403–3414. [Google Scholar] [CrossRef]
- Alzoghaibi, M.A.; Bahammam, A.S. Lipid peroxides, superoxide dismutase and circulating IL-8 and GCP-2 in patients with severe obstructive sleep apnea: A pilot study. Sleep Breath. 2005, 9, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Ifergane, G.; Ovanyan, A.; Toledano, R.; Goldbart, A.; Abu-Salame, I.; Tal, A.; Stavsky, M.; Novack, V. Obstructive Sleep Apnea in Acute Stroke: A Role for Systemic Inflammation. Stroke 2016, 47, 1207–1212. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Liaw, S.F.; Chiu, C.H.; Chen, W.J.; Lin, M.W.; Chang, F.T. Effects of nasal CPAP on exhaled SIRT1 and tumor necrosis factor-α in patients with obstructive sleep apnea. Respir. Physiol. Neurobiol. 2016, 228, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Volná, J.; Kemlink, D.; Kalousová, M.; Vávrová, J.; Majerová, V.; Mestek, O.; Svarcová, J.; Sonka, K.; Zima, T. Biochemical oxidative stress-related markers in patients with obstructive sleep apnea. Med. Sci. Monit. 2011, 17, CR491–CR497. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.-F.; Chen, Y.-H.; Chen, H.-C.; Huang, W.-C. Interactions among Obstructive Sleep Apnea Syndrome Severity, Sex, and Obesity on Circulatory Inflammatory Biomarkers in Patients with Suspected Obstructive Sleep Apnea Syndrome: A Retrospective, Cross-Sectional Study. Int. J. Environ. Res. Public Heal. 2020, 17, 4701. [Google Scholar] [CrossRef] [PubMed]
- Yokoe, T.; Minoguchi, K.; Matsuo, H.; Oda, N.; Minoguchi, H.; Yoshino, G.; Hirano, T.; Adachi, M. Elevated levels of C-reactive protein and interleukin-6 in patients with obstructive sleep apnea syndrome are decreased by nasal continuous positive airway pressure. Circulation 2003, 107, 1129–1134. [Google Scholar] [CrossRef]
- Mancuso, M.; Bonanni, E.; LoGerfo, A.; Orsucci, D.; Maestri, M.; Chico, L.; DiCoscio, E.; Fabbrini, M.; Siciliano, G.; Murri, L. Oxidative stress biomarkers in patients with untreated obstructive sleep apnea syndrome. Sleep Med. 2012, 13, 632–636. [Google Scholar] [CrossRef]
- Katsoulis, K.; Kontakiotis, T.; Spanogiannis, D.; Vlachogiannis, E.; Kougioulis, M.; Gerou, S.; Daskalopoulou, E. Total antioxidant status in patients with obstructive sleep apnea without comorbidities: The role of the severity of the disease. Sleep Breath. 2011, 15, 861–866. [Google Scholar] [CrossRef]
- Simiakakis, M.; Kapsimalis, F.; Chaligiannis, E.; Loukides, S.; Sitaras, N.; Alchanatis, M. Lack of effect of sleep apnea on oxidative stress in obstructive sleep apnea syndrome (OSAS) patients. PLOS ONE 2012, 7, e39172. [Google Scholar] [CrossRef]
- Sales, L.V.; Bruin, V.M.; D’Almeida, V.; Pompéia, S.; Bueno, O.F.; Tufik, S.; Bittencourt, L. Cognition and biomarkers of oxidative stress in obstructive sleep apnea. Clinics 2013, 68, 449–455. [Google Scholar] [CrossRef]
- Del Ben, M.; Fabiani, M.; Loffredo, L.; Polimeni, L.; Carnevale, R.; Baratta, F.; Brunori, M.; Albanese, F.; Augelletti, T.; Violi, F.; et al. Oxidative stress mediated arterial dysfunction in patients with obstructive sleep apnoea and the effect of continuous positive airway pressure treatment. BMC Pulm. Med. 2012, 12, 36. [Google Scholar] [CrossRef] [PubMed]
- Yağmur, A.R.; Çetin, M.A.; Karakurt, S.E.; Turhan, T.; Dere, H.H. The levels of advanced oxidation protein products in patients with obstructive sleep apnea syndrome. Ir. J. Med. Sci. 2020, 1–7. [Google Scholar] [CrossRef]
- Celec, P.; Hodosy, J.; Behuliak, M.; Pálffy, R.; Gardlik, R.; Halčák, L.; Mucska, I. Oxidative and carbonyl stress in patients with obstructive sleep apnea treated with continuous positive airway pressure. Sleep Breath. 2011, 16, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Ma, G.H.; Chen, L.; Li, M.; Liu, J.L.; Yang, K.; Li, Q.Y.; Li, N.; Wan, H.Y. Quantification of circulating cell-free DNA in the serum of patients with obstructive sleep apnea-hypopnea syndrome. Lung 2010, 188, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Hernandez, R.; Vallejo-Vaz, A.J.; ArmengolÁngeles, S.; Moreno-Luna, R.; Caballero-Eraso, C.; Macher, H.C.; Villar, J.; Merino, A.M.; Castell, J.; Capote, F.; et al. Obstructive Sleep Apnoea Syndrome, Endothelial Function and Markers of Endothelialization. Changes after CPAP. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Karamanlı, H.; Özol, D.; Ugur, K.S.; Yıldırım, Z.; Armutçu, F.; Bozkurt, B.; Yigitoglu, R. Influence of CPAP treatment on airway and systematic inflammation in OSAS patients. Sleep Breath. 2014, 18, 251–256. [Google Scholar] [CrossRef]
- Bauça, J.M.; Yañez, A.; Fueyo, L.; de la Peña, M.; Pierola, J.; Sánchez-de-la-Torre, A.; Mediano, O.; Cabriada-Nuño, V.; Masdeu, M.J.; Duran-Cantolla, J.; et al. Cell Death Biomarkers and Obstructive Sleep Apnea: Implications in the Acute Coronary Syndrome. Sleep 2017, 40. [Google Scholar] [CrossRef]
- Morel, F.; Doussiere, J.; Vignais, P.V. The superoxide-generating oxidase of phagocytic cells. Physiological, molecular and pathological aspects. JBIC J. Biol. Inorg. Chem. 1991, 201, 523–546. [Google Scholar] [CrossRef]
- Zeng, M.Y.; Miralda, I.; Armstrong, C.L.; Uriarte, S.M.; Bagaitkar, J. The roles of NADPH oxidase in modulating neutrophil effector responses. Mol. Oral Microbiol. 2019, 34, 27–38. [Google Scholar] [CrossRef]
- Lemineur, T.; Deby-Dupont, G.; Preiser, J.C. Biomarkers of oxidative stress in critically ill patients: What should be measured, when and how? Curr. Opin. Clin. Nutr. Metab. Care 2006, 9, 704–710. [Google Scholar] [CrossRef]
- Liu, H.G.; Liu, K.; Zhou, Y.N.; Xu, Y.J. Relationship between reduced nicotinamide adenine dinucleotide phosphate oxidase subunit p22phox gene polymorphism and obstructive sleep apnea-hypopnea syndrome in the Chinese Han population. Chin. Med. J. 2009, 122, 1369–1374. [Google Scholar] [PubMed]
- Soccio, M.; Toniato, E.; Evangelista, V.; Carluccio, M.; De Caterina, R. Oxidative stress and cardiovascular risk: The role of vascular NAD(P)H oxidase and its genetic variants. Eur. J. Clin. Investig. 2005, 35, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Piérola, J.; Alemany, A.; Yañez, A.M.; de la Peña, M.; Sánchez-de-la-Torre, M.; Esquinas, C.; Pérez-Gutierrez, C.; Burguera, B.; Barbé, F.; Barceló, A. NADPH oxidase p22phox polymorphisms and oxidative stress in patients with obstructive sleep apnoea. Respir. Med. 2011, 105, 1748–1754. [Google Scholar] [CrossRef] [PubMed]
- Di Castelnuovo, A.; Soccio, M.; Iacoviello, L.; Evangelista, V.; Consoli, A.; Vanuzzo, D.; Diviacco, S.; Carluccio, M.; Rignanese, L.; De Caterina, R. The C242T polymorphism of the p22phox component of NAD(P)H oxidase and vascular risk. Two case-control studies and a meta-analysis. Thromb. Haemost. 2008, 99, 594–601. [Google Scholar] [CrossRef] [PubMed]
- Zhan, G.; Serrano, F.; Fenik, P.; Hsu, R.; Kong, L.; Pratico, D.; Klann, E.; Veasey, S.C. NADPH oxidase mediates hypersomnolence and brain oxidative injury in a murine model of sleep apnea. Am. J. Respir. Crit. Care Med. 2005, 172, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Nair, D.; Dayyat, E.A.; Zhang, S.X.; Wang, Y.; Gozal, D. Intermittent hypoxia-induced cognitive deficits are mediated by NADPH oxidase activity in a murine model of sleep apnea. PLOS ONE 2011, 6, e19847. [Google Scholar] [CrossRef]
- Loffredo, L.; Zicari, A.M.; Occasi, F.; Perri, L.; Carnevale, R.; Angelico, F.; Del Ben, M.; Martino, F.; Nocella, C.; Savastano, V.; et al. Endothelial dysfunction and oxidative stress in children with sleep disordered breathing: Role of NADPH oxidase. Atherosclerosis 2015, 240, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Schulz, R.; Murzabekova, G.; Egemnazarov, B.; Kraut, S.; Eisele, H.J.; Dumitrascu, R.; Heitmann, J.; Seimetz, M.; Witzenrath, M.; Ghofrani, H.A.; et al. Arterial hypertension in a murine model of sleep apnea: Role of NADPH oxidase 2. J. Hypertens. 2014, 32, 300–305. [Google Scholar] [CrossRef]
- Lavie, L.; Vishnevsky, A.; Lavie, P. Evidence for lipid peroxidation in obstructive sleep apnea. Sleep 2004, 27, 123–128. [Google Scholar]
- Alzoghaibi, M.A.; Bahammam, A.S. The effect of one night of continuous positive airway pressure therapy on oxidative stress and antioxidant defense in hypertensive patients with severe obstructive sleep apnea. Sleep Breath. 2011, 16, 499–504. [Google Scholar] [CrossRef]
- Oyama, J.; Yamamoto, H.; Maeda, T.; Ito, A.; Node, K.; Makino, N. Continuous positive airway pressure therapy improves vascular dysfunction and decreases oxidative stress in patients with the metabolic syndrome and obstructive sleep apnea syndrome. Clin. Cardiol. 2012, 35, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Caimi, G.; Montana, M.; Canino, B.; Calandrino, V.; Lo Presti, R.; Hopps, E. Erythrocyte deformability, plasma lipid peroxidation and plasma protein oxidation in a group of OSAS subjects. Clin. Hemorheol. Microcirc. 2016, 64, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Mar, H.L.P.Y.; Hazen, S.L.; Tracy, R.P.; Strohl, K.P.; Auckley, D.; Bena, J.; Wang, L.; Walia, H.K.; Patel, S.R.; Mehra, R. Effect of Continuous Positive Airway Pressure on Cardiovascular Biomarkers: The Sleep Apnea Stress Randomized Controlled Trial. Chest 2016, 150, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Papandreou, C. Levels of TBARS are inversely associated with lowest oxygen saturation in obese patients with OSAS. Sleep Breath. 2013, 17, 1319–1322. [Google Scholar] [CrossRef] [PubMed]
- Stradling, J.; Schwarz, E.I.; Schlatzer, C.; Manuel, A.; Lee, R.; Antoniades, C.; Kohler, M. Biomarkers of oxidative stress following continuous positive airway pressure withdrawal: Data from two randomised trials. Eur. Respir. J. 2015, 46, 1065–1071. [Google Scholar] [CrossRef] [PubMed]
- Fernandez Alvarez, R.; Rubinos Cuadrado, G.; Alonso Arias, R.; Cascon Hernandez, J.A.; Palomo Antequera, B.; Iscar Urrutia, M.; Casan Clara, P. Snoring as a Determinant Factor of Oxidative Stress in the Airway of Patients with Obstructive Sleep Apnea. Lung 2016, 194, 469–473. [Google Scholar] [CrossRef]
- Villa, M.P.; Supino, M.C.; Fedeli, S.; Rabasco, J.; Vitelli, O.; Del Pozzo, M.; Gentile, G.; Lionetto, L.; Barreto, M.; Simmaco, M. Urinary concentration of 8-isoprostane as marker of severity of pediatric OSAS. Sleep Breath. 2014, 18, 723–729. [Google Scholar] [CrossRef]
- Murri, M.; Alcázar-Ramírez, J.D.; Garrido-Sánchez, L.; Linde, F.; Alcaide, J.; Cardona, F.; Tinahones, F.J. Oxidative stress and metabolic changes after continuous positive airway pressure treatment according to previous metabolic disorders in sleep apnea-hypopnea syndrome patients. Transl. Res. 2009, 154, 111–121. [Google Scholar] [CrossRef]
- Witko-Sarsat, V.; Friedlander, M.; Capeillère-Blandin, C.; Nguyen-Khoa, T.; Nguyen, A.T.; Zingraff, J.; Jungers, P.; Descamps-Latscha, B. Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int. 1996, 49, 1304–1313. [Google Scholar] [CrossRef]
- He, Y.; Chen, R.; Wang, J.; Pan, W.; Sun, Y.; Han, F.; Wang, Q.; Liu, C. Neurocognitive impairment is correlated with oxidative stress in patients with moderate-to-severe obstructive sleep apnea hypopnea syndrome. Respir. Med. 2016, 120, 25–30. [Google Scholar] [CrossRef]
- Passali, D.; Corallo, G.; Petti, A.; Longini, M.; Passali, F.M.; Buonocore, G.; Bellussi, L.M. A comparative study on oxidative stress role in nasal breathing impairment and obstructive sleep apnoea syndrome. Studio comparativo sul ruolo dello stress ossidativo nei pazienti con insufficienza respiratoria nasale e sindrome delle apnee ostruttive notturne. Acta Otorhinolaryngol. Ital. 2016, 36, 490–495. [Google Scholar] [PubMed]
- Zhou, L.; Chen, P.; Peng, Y.; Ouyang, R. Role of Oxidative Stress in the Neurocognitive Dysfunction of Obstructive Sleep Apnea Syndrome. Oxidative Med. Cell. Longev. 2016, 2016, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Pialoux, V.; Hanly, P.J.; Foster, G.E.; Brugniaux, J.V.; Beaudin, A.E.; Hartmann, S.E.; Pun, M.; Duggan, C.T.; Poulin, M.J. Effect of 4 days of intermittent hypoxia on oxidative stress in healthy men. FASEB J. 2008, 22, 960–963. [Google Scholar]
- Özben, S.; Huseyinoglu, N.; Hanikoglu, F.; Güvenç, T.S.; Yildirim, B.Z.; Cort, A.; Özdem, S.; Ozben, T.; Yıldırım, B.Z. Advanced oxidation protein products and ischaemia-modified albumin in obstructive sleep apnea. Eur. J. Clin. Investig. 2014, 44, 1045–1052. [Google Scholar] [CrossRef] [PubMed]
- Tóthová, L.; Hodosy, J.; Mucska, I.; Celec, P. Salivary markers of oxidative stress in patients with obstructive sleep apnea treated with continuous positive airway pressure. Sleep Breath. 2014, 18, 563–570. [Google Scholar] [CrossRef]
- Tóthová, Ľ.; Celec, P.; Mucska, I.; Hodosy, J. Short-term effects of continuous positive airway pressure on oxidative stress in severe sleep apnea. Sleep Breath. 2019, 23, 857–863. [Google Scholar] [CrossRef]
- Khan, N.; Lambert-Messerlian, G.; Monteiro, J.F.; Hodosy, J.; Tóthová, Ľ.; Celec, P.; Eklund, E.; Curran, P.; Bourjeily, G. Oxidative and carbonyl stress in pregnant women with obstructive sleep apnea. Sleep Breath. 2017, 22, 233–240. [Google Scholar] [CrossRef]
- Galeazzi, M.; Morozzi, G.; Piccini, M.; Chen, J.; Bellisai, F.; Fineschi, S.; Marcolongo, R. Dosage and characterisation of circulating DNA: Present usage and possible applications in systemic autoimmune disorders. Autoimmun. Rev. 2003, 2, 50–55. [Google Scholar] [CrossRef]
- Bakan, E.; Fidan, V.; Alp, H.H.; Baygutalp, N.K.; Cokluk, E. Effect of Modified Fujita Technique Uvulopalatoplasty on Oxidative DNA Damage Levels in Patients with Obstructive Sleep Apnea Syndrome. J. Craniofacial Surg. 2015, 26, e392–e396. [Google Scholar] [CrossRef]
- Ermakov, A.V.; Konkova, M.S.; Kostyuk, S.V.; Izevskaya, V.L.; Baranova, A.; Veiko, N.N. Oxidised extracellular DNA as a stress signal in human cells. Oxidative Med. Cell. Longev. 2013, 2013, 1–12. [Google Scholar] [CrossRef]
- Lo, Y.M.; Tein, M.S.; Lau, T.K.; Haines, C.J.; Leung, T.N.; Poon, P.M.; Wainscoat, J.S.; Johnson, P.J.; Chang, A.M.; Hjelm, N.M. Quantitative analysis of fetal DNA in maternal plasma and serum: Implications for noninvasive prenatal diagnosis. Am. J. Hum. Genet. 1998, 62, 768–775. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, M.; Nakano, H.; Maekawa, J.; Okamoto, Y.; Ohnishi, Y.; Suzuki, T.; Kimura, H. Oxidative stress in obstructive sleep apnea. Chest 2005, 127, 1674–1679. [Google Scholar] [CrossRef] [PubMed]
- Jurado-Gámez, B.; Fernandez-Marin, M.C.; Gómez-Chaparro, J.L.; Muñoz-Cabrera, L.; Lopez-Barea, J.; Perez-Jimenez, F.; Lopez-Miranda, J. Relationship of oxidative stress and endothelial dysfunction in sleep apnoea. Eur. Respir. J. 2010, 37, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Testelmans, D.; Tamisier, R.; Barone-Rochette, G.; Baguet, J.P.; Roux-Lombard, P.; Pépin, J.L.; Lévy, P. Profile of circulating cytokines: Impact of OSA, obesity and acute cardiovascular events. Cytokine 2013, 62, 210–216. [Google Scholar] [CrossRef]
- Li, K.; Wei, P.; Qin, Y.; Wei, Y. Is C-reactive protein a marker of obstructive sleep apnea? A meta-analysis. Medicine 2017, 96, e6850. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Chen, C.L.; Yu, C.C.; Chen, T.T.; Tseng, S.T.; Ho, C.H. Association of inflammation and oxidative stress with obstructive sleep apnea in ischemic stroke patients. Sleep Med. 2015, 16, 113–118. [Google Scholar] [CrossRef]
- Arnardottir, E.S.; Maislin, G.; Schwab, R.J.; Staley, B.; Benediktsdottir, B.; Olafsson, I.; Juliusson, S.; Romer, M.; Gislason, T.; Pack, A.I. The interaction of obstructive sleep apnea and obesity on the inflammatory markers C-reactive protein and interleukin-6: The Icelandic Sleep Apnea Cohort. Sleep 2012, 35, 921–932. [Google Scholar] [CrossRef]
- Nadeem, R.; Molnar, J.; Madbouly, E.M.; Nida, M.; Aggarwal, S.; Sajid, H.; Naseem, J.; Loomba, R. Serum inflammatory markers in obstructive sleep apnea: A meta-analysis. J. Clin. Sleep Med. 2013, 9, 1003–1012. [Google Scholar] [CrossRef]
- Campos-Rodriguez, F.; Asensio-Cruz, M.I.; Cordero-Guevara, J.; Jurado-Gamez, B.; Carmona-Bernal, C.; Gonzalez-Martinez, M.; Troncoso, M.F.; Sanchez-Lopez, V.; Arellano-Orden, E.; Garcia-Sanchez, M.I.; et al. Effect of continuous positive airway pressure on inflammatory, antioxidant, and depression biomarkers in women with obstructive sleep apnea: A randomised controlled trial. Sleep 2019, 42, 145. [Google Scholar] [CrossRef]
- Brånén, L.; Hovgaard, L.; Nitulescu, M.; Bengtsson, E.; Nilsson, J.; Jovinge, S. Inhibition of tumor necrosis factor-alpha reduces atherosclerosis in apolipoprotein E knockout mice. Arter. Thromb. Vasc. Biol. 2004, 24, 2137–2142. [Google Scholar] [CrossRef]
- Li, Q.; Zheng, X. Tumor necrosis factor alpha is a promising circulating biomarker for the development of obstructive sleep apnea syndrome: A meta-analysis. Oncotarget 2017, 8, 27616–27626. [Google Scholar] [CrossRef] [PubMed]
- Vgontzas, A.N.; Zoumakis, E.; Lin, H.M.; Bixler, E.O.; Trakada, G.; Chrousos, G.P. Marked decrease in sleepiness in patients with sleep apnea by etanercept, a tumor necrosis factor-alpha antagonist. J. Clin. Endocrinol. Metab. 2004, 89, 4409–4413. [Google Scholar] [CrossRef] [PubMed]
- McNicholas, W.T. Obstructive sleep apnea and inflammation. Prog. Cardiovasc. Dis. 2009, 51, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Sahlman, J.; Miettinen, K.; Peuhkurinen, K.; Seppä, J.; Peltonen, M.; Herder, C.; Punnonen, K.; Vanninen, E.; Gylling, H.; Partinen, M.; et al. The activation of the inflammatory cytokines in overweight patients with mild obstructive sleep apnoea. J. Sleep Res. 2009, 19, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.; Lemaire, P.; Lewis, P.; McDonald, F.B.; Lucking, E.; Hogan, S.; Sheehan, D.; Healy, V.; O’Halloran, K.D. Chronic intermittent hypoxia increases rat sternohyoid muscle NADPH oxidase expression with attendant modest oxidative stress. Front. Physiol. 2015, 6, 15. [Google Scholar] [CrossRef]
- In, E.; Özdemir, C.; Kaman, D.; Sökücü, S.N. Heat Shock Proteins, L-Arginine, and Asymmetric Dimethylarginine Levels in Patients With Obstructive Sleep Apnea Syndrome. Arch. Bronconeumol. 2015, 51, 544–550. [Google Scholar] [CrossRef]
- Zinellu, A.; Fois, A.G.; Mangoni, A.A.; Paliogiannis, P.; Sotgiu, E.; Zinellu, E.; Marras, V.; Pirina, P.; Carru, C. Systemic concentrations of asymmetric dimethylarginine (ADMA) in chronic obstructive pulmonary disease (COPD): State of the art. Amino Acids 2018, 50, 1169–1176. [Google Scholar] [CrossRef]
- Eisele, H.J.; Markart, P.; Schulz, R. Obstructive Sleep Apnea, Oxidative Stress, and Cardiovascular Disease: Evidence from Human Studies. Oxidative Med. Cell. Longev. 2015, 2015, 1–9. [Google Scholar] [CrossRef]
- Wysocka, E.; Cofta, S.; Cymerys, M.; Gozdzik, J.; Torlinski, L.; Batura-Gabryel, H. The impact of the sleep apnea syndrome on oxidant-antioxidant balance in the blood of overweight and obese patients. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2008, 59, 761–769. [Google Scholar]
- Singh, T.D.; Patial, K.; Vijayan, V.K.; Ravi, K. Oxidative stress and obstructive sleep apnoea syndrome. Indian J. Chest Dis. Allied. Sci. 2009, 51, 217–224. [Google Scholar]
- Barceló, A.; Barbé, F.; de la Peña, M.; Vila, M.; Pérez, G.; Piérola, J.; Durán, J.; Agustí, A.G. Antioxidant status in patients with sleep apnoea and impact of continuous positive airway pressure treatment. Eur. Respir. J. 2006, 27, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C.L.; Grunstein, R.R. Obstructive sleep apnoea: Time for a radical change? Eur. Respir. J. 2006, 27, 671–673. [Google Scholar] [CrossRef] [PubMed]
- Shahar, E.; Whitney, C.W.; Redline, S.; Lee, E.T.; Newman, A.B.; Nieto, F.J.; O’Connor, G.T.; Boland, L.L.; Schwartz, J.E.; Samet, J.M. Sleep-disordered breathing and cardiovascular disease: Cross-sectional results of the Sleep Heart Health Study. Am. J. Respir. Crit. Care Med. 2001, 163, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.G.; Johnson, D.C. Frequency of sleep apnea in stroke and TIA patients: A meta-analysis. J. Clin. Sleep Med. 2010, 6, 131–137. [Google Scholar] [CrossRef]
- McEvoy, R.D.; Antic, N.A.; Heeley, E.; Luo, Y.; Ou, Q.; Zhang, X.; Mediano, O.; Chen, R.; Drager, L.F.; Liu, Z.; et al. CPAP for prevention of cardiovascular events in obstructive sleep apnea. N. Engl. J. Med. 2016, 375, 919–931. [Google Scholar] [CrossRef]
- Peppard, P.E.; Young, T.; Barnet, J.H.; Palta, M.; Hagen, E.W.; Hla, K.M. Increased prevalence of sleep-disordered breathing in adults. Am. J. Epidemiol. 2013, 177, 1006–1014. [Google Scholar] [CrossRef]
- Narkiewicz, K.; van de Borne, P.J.; Pesek, C.A.; Dyken, M.E.; Montano, N.; Somers, V.K. Selective potentiation of peripheral chemoreflex sensitivity in obstructive sleep apnea. Circulation 1999, 99, 1183–1189. [Google Scholar] [CrossRef]
- Narkiewicz, K.; Somers, V.K. Sympathetic nerve activity in obstructive sleep apnoea. Acta Physiol. Scand. 2003, 177, 385–390. [Google Scholar] [CrossRef]
- Imadojemu, V.A.; Mawji, Z.; Kunselman, A.; Gray, K.S.; Hogeman, C.S.; Leuenberger, U.A. Sympathetic chemoreflex responses in obstructive sleep apnea and effects of continuous positive airway pressure therapy. Chest 2007, 131, 1406–1413. [Google Scholar] [CrossRef]
- Nanduri, J.; Peng, Y.J.; Wang, N.; Khan, S.A.; Semenza, G.L.; Kumar, G.K.; Prabhakar, N.R. Epigenetic regulation of redox state mediates persistent cardiorespiratory abnormalities after long-term intermittent hypoxia. J. Physiol. 2016, 595, 63–77. [Google Scholar] [CrossRef]
- Prabhakar, N.R.; Peng, Y.J.; Nanduri, J. Hypoxia-inducible factors and obstructive sleep apnea. J. Clin. Investig. 2020, 130, 5042. [Google Scholar] [CrossRef] [PubMed]
- Treptow, E.; Pepin, J.L.; Bailly, S.; Levy, P.; Bosc, C.; Destors, M.; Woehrle, H.; Tamisier, R. Reduction in sympathetic tone in patients with obstructive sleep apnoea: Is fixed CPAP more effective than APAP? A randomised, parallel trial protocol. BMJ Open 2019, 9, e024253. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, D.R.; Lasserson, T.J.; Smith, I. Educational, supportive and behavioural interventions to improve usage of continuous positive airway pressure machines in adults with plo. Cochrane Database Syst. Rev. 2014, CD007736. [Google Scholar] [CrossRef]
- Maki-Nunes, C.; Toschi-Dias, E.; Cepeda, F.X.; Rondon, M.U.; Alves, M.J.; Fraga, R.F.; Braga, A.M.F.W.; Aguilar, A.M.; Amaro, A.C.; Drager, L.F.; et al. Diet and exercise improve chemoreflex sensitivity in patients with metabolic syndrome and obstructive sleep apnea. Obesity 2015, 23, 1582–1590. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Cao, Q.; Guo, Z.; Dai, Q. Continuous Positive Airway Pressure in Patients With Obstructive Sleep Apnea and Resistant Hypertension: A Meta-Analysis of Randomized Controlled Trials. J. Clin. Hypertens. 2015, 18, 153–158. [Google Scholar] [CrossRef]
- Hall, A.B.; Ziadi, M.C.; Leech, J.A.; Chen, S.Y.; Burwash, I.G.; Renaud, J.; deKemp, R.A.; Haddad, H.; Mielniczuk, L.M.; Yoshinaga, K.; et al. Effects of short-term continuous positive airway pressure on myocardial sympathetic nerve function and energetics in patients with heart failure and obstructive sleep apnea: A randomised study. Circulation 2014, 130, 892–901. [Google Scholar] [CrossRef]
- Holmqvist, F.; Guan, N.; Zhu, Z.; Kowey, P.R.; Allen, L.A.; Fonarow, G.C.; Hylek, E.M.; Mahaffey, K.W.; Freeman, J.V.; Chang, P.; et al. Impact of obstructive sleep apnea and continuous positive airway pressure therapy on outcomes in patients with atrial fibrillation-Results from the Outcomes Registry for Better Informed Treatment of Atrial Fibrillation (ORBIT-AF). Am. Hear. J. 2015, 169, 647–654.e2. [Google Scholar] [CrossRef]
- Kaneko, Y.; Floras, J.S.; Usui, K.; Plante, J.; Tkacova, R.; Kubo, T.; Ando, S.-I.; Bradley, T.D. Cardiovascular effects of continuous positive airway pressure in patients with heart failure and obstructive sleep apnea. N. Engl. J. Med. 2003, 348, 1233–1241. [Google Scholar] [CrossRef]
- Bradley, T.D.; Logan, A.G.; Kimoff, R.J.; Sériès, F.; Morrison, D.; Ferguson, K.; Belenkie, I.; Pfeifer, M.; Fleetham, J.; Hanly, P.; et al. CANPAP Investigators. Continuous positive airway pressure for central sleep apnea and heart failure. N. Engl. J. Med. 2005, 353, 2025–2033. [Google Scholar] [CrossRef]
- Kaushal, N.; Ramesh, V.; Gozal, D. TNF-α and temporal changes in sleep architecture in mice exposed to sleep fragmentation. PLoS ONE 2012, 7, e45610. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, Q.; Feng, J.; Sun, X. Obstructive sleep apnea and endothelial progenitor cells. Patient Prefer. Adherence 2013, 7, 1077–1090. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Vgontzas, A.N.; Fernandez-Mendoza, J.; Kritikou, I.; Basta, M.; Pejovic, S.; Gaines, J.; Bixler, E.O. Objective, but Not Subjective, Sleepiness is Associated With Inflammation in Sleep Apnea. Sleep 2016, 40, zsw033. [Google Scholar] [CrossRef] [PubMed]
- Kritikou, I.; Basta, M.; Vgontzas, A.N.; Pejovic, S.; Liao, D.; Tsaoussoglou, M.; Bixler, E.O.; Stefanakis, Z.; Chrousos, G.P. Sleep apnoea, sleepiness, inflammation and insulin resistance in middle-aged males and females. Eur. Respir. J. 2013, 43, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, D.J.; Punjabi, N.M.; Mehra, R.; Patel, S.R.; Quan, S.F.; Babineau, D.C.; Tracy, R.P.; Rueschman, M.; Blumenthal, R.S.; Lewis, E.F.; et al. CPAP versus oxygen in obstructive sleep apnea. N. Engl. J. Med. 2014, 370, 2276–2285. [Google Scholar] [CrossRef] [PubMed]
- Stradling, J.R.; Craig, S.E.; Kohler, M.; Nicoll, D.; Ayers, L.; Nunn, A.J.; Bratton, D.J. Markers of inflammation: Data from the MOSAIC randomised trial of CPAP for minimally symptomatic OSA [published correction appears in Thorax. 2015 Apr;70,319]. Thorax 2015, 70, 181–182. [Google Scholar] [CrossRef]
- Martin, K.; Stanchina, M.; Kouttab, N.; Harrington, E.O.; Rounds, S. Circulating endothelial cells and endothelial progenitor cells in obstructive sleep apnea. Lung 2008, 186, 145–150. [Google Scholar] [CrossRef]
- Yun, C.H.; Jung, K.H.; Chu, K.; Kim, S.H.; Ji, K.H.; Park, H.K.; Kim, H.C.; Lee, S.T.; Lee, S.K.; Roh, J.K. Increased Circulating Endothelial Microparticles and Carotid Atherosclerosis in Obstructive Sleep Apnea. J. Clin. Neurol. 2010, 6, 89–98. [Google Scholar] [CrossRef]
- Iannella, G.; Magliulo, G.; Maniaci, A.; Meccariello, G.; Cocuzza, S.; Cammaroto, G.; Gobbi, R.; Sgarzani, R.; Firinu, E.; Corso, R.M.; et al. Olfactory function in patients with obstructive sleep apnea: A meta-analysis study. Eur. Arch. Otorhinolaryngol. 2020, 1–9. [Google Scholar] [CrossRef]
- Chang, C.P.; Chia, R.H.; Wu, T.L.; Tsao, K.C.; Sun, C.F.; Wu, J.T. Elevated cell-free serum DNA detected in patients with myocardial infarction. Clin. Chim. Acta 2003, 327, 95–101. [Google Scholar] [CrossRef]
| OXIDATIVE STRESS MARKERS | Reactive Oxygen Species (Peroxides, Superoxide, Hydroxyl Radical) |
| Nicotinamide adenine dinucleotide phosphate oxidase (NADPH) | |
| Liperoxidation Products | |
| Advanced Oxidation Protein Products (AOPPs) | |
| Circulating free DNA | |
| 8-Hydroxy-2-deoxyguanosine | |
| SYSTEMIC INFLAMMATION MARKERS AND CIRCULATING CYTOKINES | IL2, IL4, IL6 |
| Tumour necrosis alpha factor (TNF-alpha) | |
| Protein C reactive | |
| Endothelial related markers (E-selectin, P-selectin, ICAM-1, VECAM-1) | |
| ANTIOXIDANT SYSTEM | Superoxide dismutase, peroxidase, catalase |
| Glutathione, Ferric reducing/antioxidant power (FRAP) | |
| Vitamin C | |
| Vitamin E Vitamin B11, B12 |
| Leukocytes’ Oxidative Derived | ||
|---|---|---|
| Authors | Study Features | Outcomes Observed |
| R Schulz, S Mahmoudi, K Hattarm et al. (2000) [27] | 18 OSAS patients compared vs. two control groups of 10 healthy volunteers and 10 patients without OSAS | ↑ Superoxide Release markedly increased for each comparison (p < 0.01) |
| Liu HG, Zhou YN, Liu K et al. (2010) [28] | 30 OSAS patients vs. 23 healthy controls | ↑ NADPH oxidase p22phox mRNA in sputum samples was significantly higher in OSAS (p < 0.05). |
| E Hopps, B Canino, V Calandrino et al. (2014) [29] | 48 patients with OSAS, subdivided into two subgroups: Low 21 subjects (AHI < 30) High 27 subjects (AHI > 30) | ↑ TBARS and AHI value (r = 0.88, p < 0.0001) ↑ TBARS and ODI (r = 0.88, p < 0.0001) |
| Lavie L, Vishnevsky A, Lavie P (2004) [30] | 114 patients with OSAS (55 without CVD and 59 with CVD) vs. 30 non-apneic controls. | ↑ TBARS and Peroxides higher in the morning than in controls and positively correlated with RDI (p < 0.01) |
| Alzoghaibi MA, Bahammam SA (2005) [31] | 34 hypertensive patients with severe obstructive sleep apnea syndrome (OSAS). | = SOD concentrations unchanged after CPAP treatment (0.22 ± 0.09 vs. 0.22 ± 0. U/mL). ↓ TBARS levels after CPAP treatment (2.81 ± 0.27 vs. 2.47 ± 0.35 mmol/mL, respectively, p < 0.005). |
| Ntalapascha M, Makris D, Kyparos A et al. (2012) [24] | 18 patients with severe OSAS and 13 controls included in the study. | ↑ GSH/GSSG overnight ratio and GSH significantly different than controls (p = 0.03 and p = 0.048). = Plasma protein carbonyls, erythrocyte catalase activity, 8-isoprostane, SOD, TBARS, and TAC plasma values not different (p > 0.05). |
| Systemic Inflammation Markers and Circulating Cytokines | ||
| Ifergane G, Ovanyan A, Toledano R et al. (2016) [32] | 43 patients with acute stroke and sleep apnea | ↑ correlation between AHI, IL-6 (ρ = 0.37, p = 0.02) and PAI-1 (ρ = 0.31, p = 0.07). |
| Lin CC, Liaw SF, Chiu CH et al. (2016) [33] | 35 patients with moderately severe to severe OSAS vs. 20 healthy controls | ↓ SIRT1 was lower (p < 0.01) ↑ TNF-α was higher (p < 0.01) |
| Volná J, Kemlink D, Kalousová M et al. (2011) [34] | 51 patients suspected for OSAS included | ↑ hsCRP and ODI (R = 0.450, p = 0.001) ↑ hsCRP AHI (R = 0.479, p = 0.001) ↑ hsCRP SpO2 < 90 (R = 0.480, p = 0.001). |
| Wu MF, Chen YH, Chen HC et al. (2020) [35] | 100 patients included in the final analysis (63 Normal to Moderate OSAS while 37Severe OSAS) | ↑IL-6 level for all OSAS severity and sex had an interaction effect on (p = 0.030). ↑ CRP (p = 0.001) and ↑ IL-6 (p = 0.000) levels were higher in the obese group than in the non-obese group independently of OSAS severity and sex. |
| Yokoe T, Minoguchi K, Matsuo H, et al. (2003) [36] | 30 patients with OSAS and 14 obese control subjects. | ↑ Levels of CRP significantly higher in patients with OSAS than in the control group (p < 0.001) ↑ IL-6 significantly higher in patients with OSAS than in the control group (p < 0.05) |
| Antioxidant System Impairment | ||
| Mancuso M, Bonanni E, Lo Gerfo A et al. (2012) [37] | 41 untreated patients with a new diagnosis of OSAS vs. 32 healthy subjects | ↑AOPP higher than in controls (293.4 ± 109.7 mmol/L vs. 203.2 ± 45.2 mmol/L; (p < 0.0005) ↓ FRAP lower (95% CI for the mean 0.518–0.579 mmol/L vs. 0.713–0.875 mmol/L; p < 0.0001). ↓ Total GSH lower (95% CI for the mean 0.389–0.449 nmol/μL vs. 0.574–0.713 nmol/μL; (p < 0.0001). |
| Katsoulis K, Kontakiotis T, Spanogiannis D et al. (2011) [38] | 32 OSAS patients without comorbidities | ↓ TAS significantly decreased compared with the measurement before (1.68 ± 0.11 vs. 1.61 ± 0.10 mmol/l, p < 0.01). |
| Simiakakis M, Kapsimalis F, Chaligiannis E et al. (2012) [39] | 66 total subjects referred (42 patients with OSAS vs. 24 controls) | ↑The levels of d-ROMS were significantly higher (p = 0.005) in the control group ↓ levels of antioxidant capacity in OSAS patients significantly lower (p = 0.004). |
| Sales LV, Bruin VM, D’Almeida V, et al. (2013) [40] | 14 patients with obstructive sleep apnea vs. 13 controls | ↓ vitamin E lower levels of (p < 0.006) ↓ superoxide dismutase (p < 0.001) ↓ vitamin B11 (p < 0.001) ↑ homocysteine higher concentrations (p < 0.02) = Serum concentrations of vitamin C, catalase, glutathione and vitamin B12 unaltered. |
| References | Study Design | Features | Pretreatment Scores | Post Treatment Outcomes |
|---|---|---|---|---|
| Del Ben et al. 2012 [41] | Prospective Study | Patients n = 138 (47diagnosed with primary snoring and 91 OSAS) Serum levels of soluble NOX2-derived peptide urinary 8-iso-PGF2α | Severe OSAS patients: sNOX2-dp (pg/mL) ↑ but NS Serum NOx (uM/mL) ↑ but NS urinary 8-iso-PGF2α ↑ p < 0.001 | Severe OSAS patients after CPAP: Urinary 8-iso-PGF2α (pg/mL) ↓ p = 0.007 sNOX2-dp (pg/mL) ↓ p = 0.003 Serum NOx (uM/mL) ↓ but NS |
| Yagmur et al. 2020 [42] | Prospective Study | Patients n = 165 (125 diagnosed with OSAS, 40 control group) | AOPP: Severe OSAS vs. Mild ↑ p < 0.05 Severe OSAS vs. Control ↑ p < 0.05 | AOPP ↓ p = 0.36 but NS |
| Mancuso et al. 2012 [37] | Prospective Study | Patients n = 73 (41 diagnosed with OSAS, 32 control group) | - AOPP ↑ p <0.0005 - Ferric reducing antioxidant power (FRAP) ↓ p < 0.0001 Total glutathione (GSH) p < 0.0001 | AOPP unchanged NS FRAP levels ↑ p < 0.005 GSH not been re-evaluated |
| Celec et al. 2012 [43] | Prospective Study | Patients n = 89 diagnosed with OSAS TBARS AOPP Carbonyl stress (AGEs) Total antioxidant capacity (TAC) | TBARS AOPP Carbonyl stress (AGEs) Total antioxidant capacity (TAC) | TBARS ↓ p < 0.03 AOPP ↓ but NS Carbonyl stress (AGEs) ↓p < 0.02). Total antioxidant capacity (TAC) NS |
| Ye L et al. 2010 [44] | Prospective Study | Patients n = 179 (127 diagnosed with OSAS, 52 control group) | Serum DNA (ng/mL) ↑ p < 0.01 malonaldehyde (MDA) (nmol/mL) ↑ p < 0.01 IL-6 (pg/mL) ↑ p < 0.01 | Serum DNA (ng/mL) ↓ p < 0.01 malonaldehyde (MDA) (nmol/mL) ↓ p= 0.04 IL-6 (pg/mL) ↓p < 0.01 |
| Muñoz-Hernandez et al. 2015 [45] | Prospective Study | Patients n = 30 diagnosed with OSAS, no control group. | cell free-DNA = 187.93 ± 115.81 ng/mL | cell free-DNA (121.28 ± 78.98 ng/mL) ↓ p < 0.01 |
| Karamanlı et al. 2014 [46] | Prospective Study | Patients n = 35 diagnosed with OSAS and treated with CPAP, no control group. | Nitrotyrosine 17.3 ± 30.7 pg/mL IL-6 1.1 ± 2.3 pg/mL TNF-α 28.9 ± 1.35 pg/mL 8-Isoprostane 5.7 ± 7.9 pg/mL CRP 8.3 ± 8.5 mg/l | Nitrotyrosine 4.6 ± 3.4 ↓ p = 0.037 IL-6 0.3 ± 0.2 ↓ p = 0.000 TNF-α 26.8 ± 1.9 ↓ p = 0.000 8-Isoprostane 3.0 ± 1.6 ↓ p = 0.027 CRP 6.2 ± 4.3 ↓ p = 0.064 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maniaci, A.; Iannella, G.; Cocuzza, S.; Vicini, C.; Magliulo, G.; Ferlito, S.; Cammaroto, G.; Meccariello, G.; De Vito, A.; Nicolai, A.; et al. Oxidative Stress and Inflammation Biomarker Expression in Obstructive Sleep Apnea Patients. J. Clin. Med. 2021, 10, 277. https://doi.org/10.3390/jcm10020277
Maniaci A, Iannella G, Cocuzza S, Vicini C, Magliulo G, Ferlito S, Cammaroto G, Meccariello G, De Vito A, Nicolai A, et al. Oxidative Stress and Inflammation Biomarker Expression in Obstructive Sleep Apnea Patients. Journal of Clinical Medicine. 2021; 10(2):277. https://doi.org/10.3390/jcm10020277
Chicago/Turabian StyleManiaci, Antonino, Giannicola Iannella, Salvatore Cocuzza, Claudio Vicini, Giuseppe Magliulo, Salvatore Ferlito, Giovanni Cammaroto, Giuseppe Meccariello, Andrea De Vito, Alice Nicolai, and et al. 2021. "Oxidative Stress and Inflammation Biomarker Expression in Obstructive Sleep Apnea Patients" Journal of Clinical Medicine 10, no. 2: 277. https://doi.org/10.3390/jcm10020277
APA StyleManiaci, A., Iannella, G., Cocuzza, S., Vicini, C., Magliulo, G., Ferlito, S., Cammaroto, G., Meccariello, G., De Vito, A., Nicolai, A., Pace, A., Artico, M., & Taurone, S. (2021). Oxidative Stress and Inflammation Biomarker Expression in Obstructive Sleep Apnea Patients. Journal of Clinical Medicine, 10(2), 277. https://doi.org/10.3390/jcm10020277











