Assessing mPTC Progression during Active Surveillance: Volume or Diameter Increase?
Abstract
:1. Introduction
2. Patients and Methods
2.1. Patients
2.2. Methods
Neck Ultrasound
2.3. Statistical Analysis
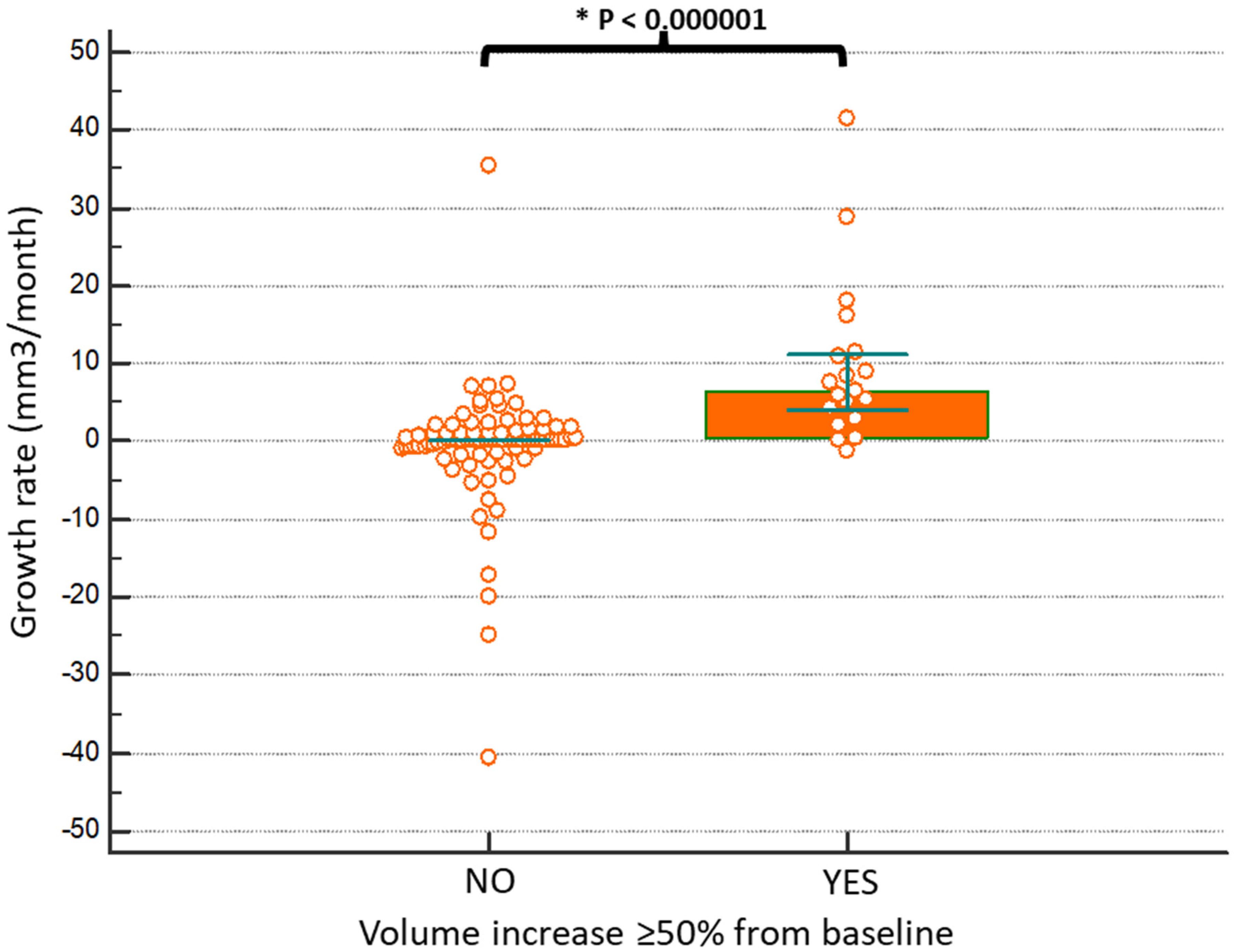
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- National Cancer Institute. Comprehensive Cancer Information. Available online: https://www.cancer.gov/ (accessed on 9 March 2021).
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pacini, F.; Basolo, F.; Bellantone, R.; Boni, G.; Cannizzaro, M.A.; De Palma, M.; Durante, C.; Elisei, R.; Fadda, G.; Frasoldati, A.; et al. Italian consensus on diagnosis and treatment of differentiated thyroid cancer: Joint statements of six Italian societies. J. Endocrinol. Investig. 2018, 41, 849–876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sugitani, I.; Ito, Y.; Takeuchi, D.; Nakayama, H.; Masaki, C.; Shindo, H.; Teshima, M.; Horiguchi, K.; Yoshida, Y.; Kanai, T.; et al. Indications and Strategy for Active Surveillance of Adult Low-Risk Papillary Thyroid Microcarcinoma: Consensus Statements from the Japan Association of Endocrine Surgery Task Force on Management for Papillary Thyroid Microcarcinoma. Thyroid 2020, 31, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Uruno, T.; Nakano, K.; Takamura, Y.; Miya, A.; Kobayashi, K.; Yokozawa, T.; Matsuzuka, F.; Kuma, S.; Kuma, K.; et al. An Observation Trial Without Surgical Treatment in Patients with Papillary Microcarcinoma of the Thyroid. Thyroid 2003, 13, 381–387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ito, Y.; Miyauchi, A.; Kihara, M.; Higashiyama, T.; Kobayashi, K.; Miya, A. Patient Age Is Significantly Related to the Progression of Papillary Microcarcinoma of the Thyroid Under Observation. Thyroid 2014, 24, 27–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sugitani, I.; Toda, K.; Yamada, K.; Yamamoto, N.; Ikenaga, M.; Fujimoto, Y. Three Distinctly Different Kinds of Papillary Thyroid Microcarcinoma should be Recognized: Our Treatment Strategies and Outcomes. World J. Surg. 2010, 32, 1222–1231. [Google Scholar] [CrossRef]
- Fukuoka, O.; Sugitani, I.; Ebina, A.; Toda, K.; Kawabata, K.; Yamada, K. Natural History of Asymptomatic Papillary Thyroid Microcarcinoma: Time-Dependent Changes in Calcification and Vascularity during Active Surveillance. World J. Surg. 2016, 40, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Tuttle, R.M.; Fagin, J.A.; Minkowitz, G.; Wong, R.J.; Roman, B.; Patel, S.; Untch, B.; Ganly, I.; Shaha, A.R.; Shah, J.P.; et al. Natural history and tumor volume kinetics of papillary thyroid cancers during active surveillance. JAMA Otolaryngol. Head Neck Surg. 2017, 143, 1015–1020. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.; Oh, H.-S.; Kim, M.; Park, S.; Jeon, M.J.; Kim, W.G.; Kim, W.B.; Shong, Y.K.; Song, D.E.; Baek, J.H.; et al. Active Surveillance for Patients With Papillary Thyroid Microcarcinoma: A Single Center’s Experience in Korea. J. Clin. Endocrinol. Metab. 2017, 102, 1917–1925. [Google Scholar] [CrossRef]
- Oh, H.S.; Ha, J.; Kim, H.I.; Kim, T.H.; Kim, W.G.; Lim, D.J.; Kim, T.Y.; Kim, S.W.; Kim, W.B.; Shong, Y.K.; et al. Active Surveillance of Low-Risk Papillary Thyroid Microcarcinoma: A Multi-Center Cohort Study in Korea. Thyroid 2018, 28, 1587–1594. [Google Scholar] [CrossRef] [PubMed]
- Sanabria, A. Active Surveillance in Thyroid Microcarcinoma in a Latin-American Cohort. JAMA Otolaryngol. Head Neck Surg. 2018, 144, 947–948. [Google Scholar] [CrossRef] [PubMed]
- Sakai, T.; Sugitani, I.; Ebina, A.; Fukuoka, O.; Toda, K.; Mitani, H.; Yamada, K. Active surveillance for T1bN0M0 papillary thyroid carcinoma. Thyroid 2019, 29, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Smulever, A.; Pitoia, F. Active surveillance in papillary thyroid carcinoma: Not easily accepted but possible in Latin America. Arch. Endocrinol. Metab. 2019, 63, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Molinaro, E.; Campopiano, M.C.; Pieruzzi, L.; Matrone, A.; Agate, L.; Bottici, V.; Viola, D.; Cappagli, V.; Valerio, L.; Giani, C.; et al. Active Surveillance in Papillary Thyroid Microcarcinomas is Feasible and Safe: Experience at a Single Italian Center. J. Clin. Endocrinol. Metab. 2020, 105, 172–180. [Google Scholar] [CrossRef]
- Kim, H.I.; Jang, H.W.; Ahn, H.S.; Ahn, S.; Park, S.Y.; Oh, Y.L.; Hahn, S.Y.; Shin, J.H.; Kim, J.; Kim, J.S.; et al. High serum TSH level is associated with progression of papillary thyroid microcarcinoma during active surveillance. J. Clin. Endocrinol. Metab. 2017, 103, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Miyauchi, A.; Kudo, T.; Higashiyama, T.; Masuoka, H.; Kihara, M.; Miya, A. Kinetic analysis of growth activity in enlarging papillary thyroid microcarcinomas. Thyroid 2019, 29, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Miyauchi, A.; Kudo, T.; Ito, Y.; Oda, H.; Yamamoto, M.; Sasai, H.; Higashiyama, T.; Masuoka, H.; Fukushima, M.; Kihara, M.; et al. Natural history of papillary thyroid microcarcinoma: Kinetic analyses on tumor volume during active surveillance and before presentation. Surgery 2019, 165, 25–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, S.R.; Choi, Y.J.; Lee, S.S.; Kim, S.-O.; Lee, S.-A.; Jeon, M.J.; Kim, W.G.; Lee, J.H.; Baek, J.H. Interobserver Reproducibility in Sonographic Measurement of Diameter and Volume of Papillary Thyroid Microcarcinoma. Thyroid 2021. [Google Scholar] [CrossRef] [PubMed]
- Basolo, F.; Torregrossa, L.; Giannini, R.; Miccoli, M.; Lupi, C.; Sensi, E.; Berti, P.; Elisei, R.; Vitti, P.; Baggiani, A.; et al. Correlation between the BRAF V600E mutation and tumor invasiveness in papillary thyroid carcinomas smaller than 20 millimeters: Analysis of 1060 cases. J. Clin. Endocrinol. Metab. 2010, 95, 4197–4205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, J.; Wu, S.; Xia, X.; Wang, Y.; Fan, Y.; Yang, Z. Cell adhesion-related gene somatic mutations are enriched in aggressive papillary thyroid microcarcinomas. J. Transl. Med. 2018, 16, 269. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Patient A | Patient B | Patient C | Patient D | Patient E | |
|---|---|---|---|---|---|
| Age at diagnosis | 51 years | 36 years | 45 years | 36 years | 38 years |
| Gender | Female | Female | M | Female | Female |
| Kind of progression | LNF MTS | mPTC growth | mPTC growth | LNF MTS | LNF MTS |
| mPTC maximum diameter at baseline | 9 mm | 13 mm | 13 mm | 12 mm | 13 mm |
| mPTC maximum diameter at last evaluation | 9 mm | 18 mm | 18 mm | 11 mm | 13 mm |
| mPTC volume at baseline | 220 mm3 | 1100 mm3 | 800 mm3 | 560 mm3 | 670 mm3 |
| mPTC volume at last evaluation | 260 mm3 | 2100 mm3 | 1600 mm3 | 520 mm3 | 880 mm3 |
| Period of observation | 12 months | 13 months | 23 months | 36 months | 24 months |
| Kind of surgery | TT + LC LNX | TT | TT | TT + LC LNX | TT + LC LNX |
| TMN (8° edition) | T1b(m)N1bMX | T1bNXMX | T1bNXMX | T1b(m)N1bMX | T1b(m)N1bMX |
| Histothype | Tall cell | Classical | Classical | Classical | Classical |
| mPTC maximum diameter at histology | 13 mm | 17 mm | 17 mm | 15 mm | 13 mm |
| Minimal extrathyroidal extension | No | No | No | Yes | No |
| Maximum diameter of LNF MTS | 5 mm | - | - | 5 mm | 3 mm |
| Radioiodine therapy (activity) | Yes (30 mCi) | No | No | Yes (100 mCi) | Yes (130 mCi) |
| Follow-up after surgery | 36 months | 36 months | 18 months | 12 months | 18 months |
| Outcome | Excellent response | Excellent response | Excellent response | Excellent response | Excellent response |
| Period of Observation (Months) | Volume Increase > 50% (n) | Total (n) | % |
|---|---|---|---|
| +6 | 8 | 109 | 7.3 |
| +12 | 6 | 87 | 6.9 |
| +18 | 4 | 66 | 6.1 |
| +24 | 1 | 54 | 1.9 |
| +36 | 2 | 42 | 4.8 |
| +48 | 1 | 30 | 3.3 |
| Clinical, Ultrasound and Cytological Features | Volume Variation < 50% (n 90) Group A | Volume Variation ≥ 50% (n 19) Group B | p |
|---|---|---|---|
| Gender Female n(%) Male n(%) | 66 (73%) 24 (27%) | 13 (68%) 6 (32%) | NS |
| Age (years) Media ±DS | 44 ± 15 | 45 ± 10 | NS |
| Age < 40 years | 33 (37%) | 7 (37%) | NS |
| Hereditary for PTC n(%) | 5 (6%) | 3 (15%) | NS |
| Previous external beam radiation n(%) | 3 (3.3%) | 0 | NS |
| Hypothyroidism treated with levothyroxin n(%) | 11 (12%) | 2 (11%) | NS |
| TSH baseline (mUI/l) mean ± SD | 1.6 ± 0.8 | 1.5 ± 0.8 | NS |
| Tg baseline (mcg/l) mean ± SD ^ | 10.9 ± 10.1 | 10.9 ± 11.4 | NS |
| AbTg baseline (UI/mL) mean ± SD | 2.2 ± 4.7 | 1.6 ± 4.7 | NS |
| Cytological results TIR4 n(%) TIR5 n(%) | 40 (44%) 50 (56%) | 7 (37%) 12 (63%) | NS |
| Hypoechogenicity n(%) | 80 (89%) | 19 (100%) | NS |
| Microcalcification n(%) | 48 (53%) | 10 (53%) | NS |
| Irregular margins n(%) | 58 (64%) | 11 (58%) | NS |
| Thyroid capsular proximity n(%) * | 47 (52%) | 12 (63%) | NS |
| Maximum diameter baseline mm mean ± SD | 9.1 ± 24 | 9 ± 2.5 | NS |
| Nodular volume at baseline mm3 mean ± SD | 305 ± 214 | 262 ± 216 | NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campopiano, M.C.; Matrone, A.; Rago, T.; Scutari, M.; Prete, A.; Agate, L.; Piaggi, P.; Elisei, R.; Molinaro, E. Assessing mPTC Progression during Active Surveillance: Volume or Diameter Increase? J. Clin. Med. 2021, 10, 4068. https://doi.org/10.3390/jcm10184068
Campopiano MC, Matrone A, Rago T, Scutari M, Prete A, Agate L, Piaggi P, Elisei R, Molinaro E. Assessing mPTC Progression during Active Surveillance: Volume or Diameter Increase? Journal of Clinical Medicine. 2021; 10(18):4068. https://doi.org/10.3390/jcm10184068
Chicago/Turabian StyleCampopiano, Maria Cristina, Antonio Matrone, Teresa Rago, Maria Scutari, Alessandro Prete, Laura Agate, Paolo Piaggi, Rossella Elisei, and Eleonora Molinaro. 2021. "Assessing mPTC Progression during Active Surveillance: Volume or Diameter Increase?" Journal of Clinical Medicine 10, no. 18: 4068. https://doi.org/10.3390/jcm10184068
APA StyleCampopiano, M. C., Matrone, A., Rago, T., Scutari, M., Prete, A., Agate, L., Piaggi, P., Elisei, R., & Molinaro, E. (2021). Assessing mPTC Progression during Active Surveillance: Volume or Diameter Increase? Journal of Clinical Medicine, 10(18), 4068. https://doi.org/10.3390/jcm10184068







