SARS-CoV-2 in Urine May Predict a Severe Evolution of COVID-19
Abstract
1. Introduction
2. Material and Methods
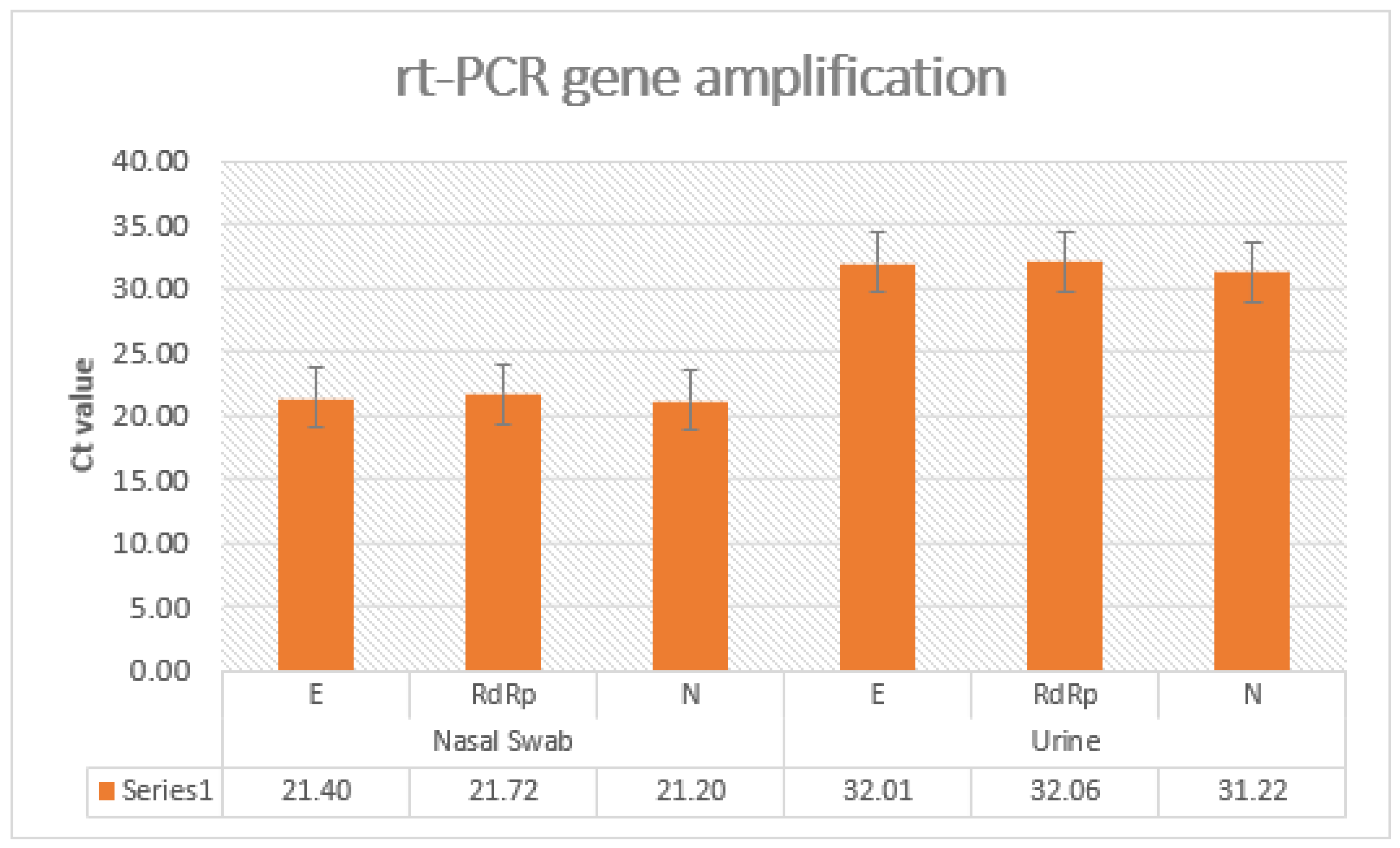
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Perrella, A.; Carannante, N.; Berretta, M.; Rinaldi, M.; Maturo, N.; Rinaldi, L. Novel Coronavirus 2019 (SARS-CoV2): A global emer-gency that needs new approaches? Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 2162–2164. [Google Scholar] [CrossRef] [PubMed]
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): A review. JAMA 2020, 324, 782–793. [Google Scholar] [CrossRef]
- Scully, E.P.; Gupta, A.; Klein, S.L. Sex-biased clinical presentation and outcomes from COVID-19. Clin. Microbiol. Infect. 2021, 27, 1072–1073. [Google Scholar] [CrossRef] [PubMed]
- Yassin, A.; Nawaiseh, M.; Shaban, A.; Alsherbini, K.; El-Salem, K.; Soudah, O.; Abu-Rub, M. Neurological manifestations and com-plications of coronavirus disease 2019 (COVID-19): A systematic review and meta-analysis. BMC Neurol. 2021, 21, 138. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.W.; Wang, S.F. SARS-CoV-2 entry related viral and host genetic variations: Implications on COVID-19 severity, immune escape, and infectivity. Int. J. Mol. Sci. 2021, 22, 3060. [Google Scholar] [CrossRef] [PubMed]
- Shirato, K.; Nao, N.; Katano, H.; Takayama, I.; Saito, S.; Kato, F.; Katoh, H.; Sakata, M.; Nakatsu, Y.; Mori, Y.; et al. Development of henetic diagnostic methods for novel corona virus 2019(nCoV-2019) in Japan. Jpn. J. Infect. Dis. 2020, 73, 304e7. [Google Scholar] [CrossRef]
- Tang, Y.-W.; Schmitz, J.E.; Persing, D.H.; Stratton, C.W. Laboratory diagnosis of COVID-19: Current issues and challenges. J. Clin. Microbiol. 2020, 58, e00512–e00520. [Google Scholar] [CrossRef] [PubMed]
- Quiroga, S.A.; Hernández, C.; Castañeda, S.; Jimenez, P.; Vega, L.; Gomez, M.; Martinez, D.; Ballesteros, N.; Muñoz, M.; Cifuentes, C.; et al. Contrasting SARS-CoV-2 RNA copies and clinical symptoms in a large cohort of Colombian patients during the first wave of the COVID-19 pandemic. Ann. Clin. Microbiol. Antimicrob. 2021, 20, 39. [Google Scholar] [CrossRef]
- Zheng, S.; Fan, J.; Yu, F.; Feng, B.; Lou, B.; Zou, Q.; Xie, G.; Lin, S.; Wang, R.; Yang, X.; et al. Viral load dynamics and disease severity in patients infected with SARS-CoV-2 in Zhejiang province, China, January–March 2020: A retrospective cohort study. BMJ 2020, 369, m1443. [Google Scholar] [CrossRef]
- Rao, S.N.; Manissero, D.; Steele, V.R.; Pareja, J. A narrative systematic review of the clinical utility of cycle threshold values in the context of COVID-19. Infect. Dis. 2020, 9, 573–586. [Google Scholar] [CrossRef]
- Hossain, M.E.; Lister, D.; Bartolo, C.; Kinsella, P.M.; Knox, J.; Aldrich, R.; Cowan, R.; Commons, R.J. Prolonged viral shedding in patients with mild to moderate COVID-19 disease: A regional perspective. Infect. Dis. Res. Treat. 2021, 14, 11786337211010428. [Google Scholar] [CrossRef]
- Váncsa, S.; Dembrovszky, F.; Farkas, N.; Szakó, L.; Teutsch, B.; Bunduc, S.; Nagy, R.; Párniczky, A.; Erőss, B.; Péterfi, Z.; et al. Repeated SARS-CoV-2 positivity: Analysis of 123 cases. Viruses 2021, 13, 512. [Google Scholar] [CrossRef] [PubMed]
- Iba, T.; Connors, J.M.; Levy, J.H. The coagulopathy, endotheliopathy, and vasculitis of COVID-19. Inflamm. Res. 2020, 69, 1181–1189. [Google Scholar] [CrossRef]
- The World Medical Association. Declaration of Helsinki [Web Page]. 2004. Available online: http://www.wma.net/e/policy/b3.htm (accessed on 28 December 2020).
- Lascarrou, J.-B.; Gaultier, A.; Soumagne, T.; Serck, N.; Sauneuf, B.; Piagnerelli, M.; Ly, A.; Lejeune, F.; Lefebvre, L.; Hraiech, S.; et al. Identifying clinical phenotypes in moderate to severe acute respiratory distress syndrome related to COVID-19: The COVADIS study. Front. Med. 2021, 8, 632933. [Google Scholar] [CrossRef]
- Castro, R.A.; Frishman, W.H. Thrombotic complications of COVID-19 infection: A review. Cardiol Rev. 2021, 29, 43–47. [Google Scholar] [CrossRef]
- Stringer, D.; Braude, P.; Myint, P.K.; Evans, L.; Collins, J.T.; Verduri, A.; Quinn, T.J.; Vilches-Moraga, A.; Stechman, M.J.; Pearce, L.; et al. COPE Study Collaborators. The role of C-reactive protein as a prognostic marker in COVID-19. Int. J. Epidemiol. 2021, 50, 420–429. [Google Scholar] [CrossRef]
- Wu, J.; Liu, J.; Li, S.; Peng, Z.; Xiao, Z.; Wang, X.; Yan, R.; Luo, J. Detection and analysis of nucleic acid in various biological samples of COVID-19 patients. Travel Med. Infect. Dis. 2020, 37, 101673. [Google Scholar] [CrossRef]
- Wang, Y.; Dong, C.; Hu, Y.; Li, C.; Ren, Q.; Zhang, X.; Shi, H.; Zhou, M. Temporal changes of CT findings in 90 patients with COVID-19 pneumonia: A longitudinal study. Radiology 2020, 296, E55–E64. [Google Scholar] [CrossRef]
- Ramos-Casals, M.; Brito-Zerón, P.; Mariette, X. Systemic and organ-specific immune-related manifestations of COVID-19. Nat. Rev. Rheumatol. 2021, 17, 315–332. [Google Scholar] [CrossRef] [PubMed]
- Levi, M.; van der Poll, T. Coagulation and sepsis. Thromb. Res. 2017, 149, 38–44. [Google Scholar] [CrossRef]
- Frithiof, R.; Bergqvist, A.; Järhult, J.D.; Lipcsey, M.; Hultström, M. Presence of SARS-CoV-2 in urine is rare and not associated with acute kidney injury in critically ill COVID-19 patients. Crit. Care 2020, 24, 587. [Google Scholar] [CrossRef] [PubMed]
- Phanthanawiboon, S.; Limkittikul, K.; Sakai, Y.; Takakura, N.; Saijo, M.; Kurosu, T. Acute systemic infection with dengue virus leads to vascular leakage and death through tumor necrosis factor-α and Tie2/Angiopoietin signaling in mice lacking type I and II interferon receptors. PLoS ONE 2016, 11, e0148564. [Google Scholar] [CrossRef] [PubMed]
| Scoring System | |||
|---|---|---|---|
| Score 1 | Score 2 | Score 3 | |
| Lymphocytes (>1000 cell/mmc) | 601–1000 | 401–600 | <400 |
| CRP (<5 mg) | >10 x n.v. | >20 x n.v. | <25 x n.v. |
| P/F ratio | ≥250 | 250–150 | <150 |
| CT Scan Score * | <10/25 | 11–18/25 | >18/25 |
| Fever for more than 3 days | <38 °C | 38–39 °C | >39 °C |
| Dyspnea | mild | moderate | severe |
| Variables | Total | Urine Negative (Acute Infection) | Urine Positive (Acute Infection) |
|---|---|---|---|
| (n = 60) | (n = 53) | (n = 7) | |
| Demographic data | |||
| Age (years) Sex | 68 ± 5 | 65 ± 6 | 67 ± 3 |
| Male | 16 (84%) | 9 (75%) | 7 (100%) |
| Female | 3 (16%) | 3 (25%) | 0 (0%) |
| Smoking history | |||
| Yes | 13 (68%) | 7 (58%) | 6 (86%) |
| No | 6 (32%) | 5 (42%) | 1 (14%) |
| Comorbidities | |||
| Hypertension | 12 | 4 | 2 |
| Diabetes | 6 | 3 | 1 |
| Cardiovascular disease | 8 | 4 | 3 |
| Chronic liver disease | 2 | 0 | 0 |
| Chronic lung disease | 4 | 2 | 0 |
| Chronic kidney | 3 | 0 | 0 |
| disease Cancer | 5 | 2 | 1 |
| Immunocompromising conditions | 3 | 1 | 0 |
| Signs and symptoms | |||
| Fever | 12 | 8 | 4 |
| Cough | 9 | 6 | 3 |
| Fatigue | 8 | 3 | 5 |
| Diarrhea | 3 | 2 | 1 |
| Shortness of breath | 13 | 9 | 4 |
| Laboratory findings | 5.753 (±204) | 6.355 (±159) | 4.673 (±109) |
| WBC (cells/mmc) | 1530 (±71) | 996 (±52) | 628 (±64) |
| Lymphocyte (cells/mmc) | 4.0 (5.2) | 4.3 (4.2) | 3.2 (14.2) |
| NLR (Neutropylete/lymphocyte ratio) | 88.1 | 72.1 | 85 |
| CRP (mg/L) | 25 (±15) | 24 (±11) | 35 (±12) |
| AST (U/L < 50) | 267 (±43) | 312 (±15) | 318 (±29) |
| ALT (U/L < 40) | 273 (±159) | 300 (±192) | 234 (±711) |
| LDH (U/L < 250) | 602 (±117) | 425 (±231) | 835 (±194) |
| Fibrinogen (ng/mL) | 2.6 (4.9) | 3.2 (7.6) | 1.8 (3.4) |
| D-dimer (ng/mL) | 257 (12.1) | 28.4 (9.4) | 22.2 (5.4) |
| Creatinine Clearance (MDRD 6 variable) | 62 ± 2 mL/min | 61 ± 2 mL/min | 58 ± 3 mL/min |
| Ct 4 gens mean value of RT-PCR | 20.8 | 18.2 | 21.3 |
| Respiratory Function | 25 (±15) | 24 (±11) | 35 (±12) |
| P/F Ratio | 172 ± 14 | 170 ± 12 | 145 ± 22 |
| NIV (Non-Invasive ventilation) (as %) | 44% | 45% | 43% |
| Venturi Mask (as %) | 32% | 40% | 0% |
| Intubation (as %) | 24% | 15% | 57% |
| CT scan findings | |||
| T Score Wang (x/24) | 19/24 (±1) | 18/254 (±1) | 21/24 (±2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perrella, A.; Brita, M.; Coletta, F.; Cotena, S.; De Marco, G.; Longobardi, A.; Sala, C.; Sannino, D.; Tomasello, A.; Perrella, M.; et al. SARS-CoV-2 in Urine May Predict a Severe Evolution of COVID-19. J. Clin. Med. 2021, 10, 4061. https://doi.org/10.3390/jcm10184061
Perrella A, Brita M, Coletta F, Cotena S, De Marco G, Longobardi A, Sala C, Sannino D, Tomasello A, Perrella M, et al. SARS-CoV-2 in Urine May Predict a Severe Evolution of COVID-19. Journal of Clinical Medicine. 2021; 10(18):4061. https://doi.org/10.3390/jcm10184061
Chicago/Turabian StylePerrella, Alessandro, Mario Brita, Francesco Coletta, Simona Cotena, GiamPaola De Marco, Adele Longobardi, Crescenzo Sala, Dania Sannino, Antonio Tomasello, Marco Perrella, and et al. 2021. "SARS-CoV-2 in Urine May Predict a Severe Evolution of COVID-19" Journal of Clinical Medicine 10, no. 18: 4061. https://doi.org/10.3390/jcm10184061
APA StylePerrella, A., Brita, M., Coletta, F., Cotena, S., De Marco, G., Longobardi, A., Sala, C., Sannino, D., Tomasello, A., Perrella, M., Russo, G., Tarsitano, M., Chetta, M., Della Monica, M., Orlando, V., Coscioni, E., & Villani, R. (2021). SARS-CoV-2 in Urine May Predict a Severe Evolution of COVID-19. Journal of Clinical Medicine, 10(18), 4061. https://doi.org/10.3390/jcm10184061







