The Prognostic Role of Neutrophil-to-Lymphocyte Ratio in Patients Hospitalized with Acute Pulmonary Embolism
Abstract
1. Introduction
2. Methods
2.1. Study Design and Population
2.2. Data Collection and Outcomes
2.3. Statistical Analysis
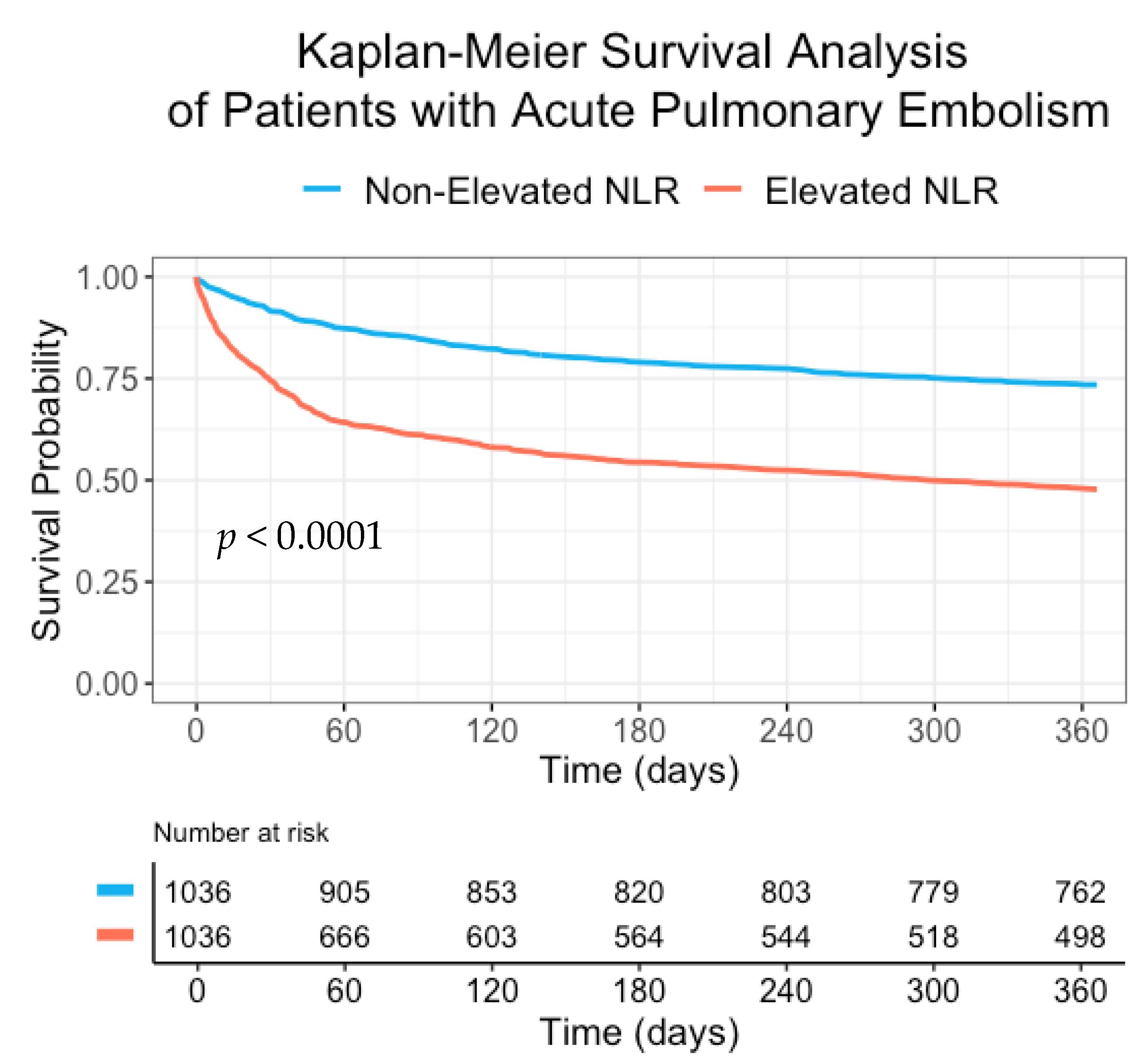
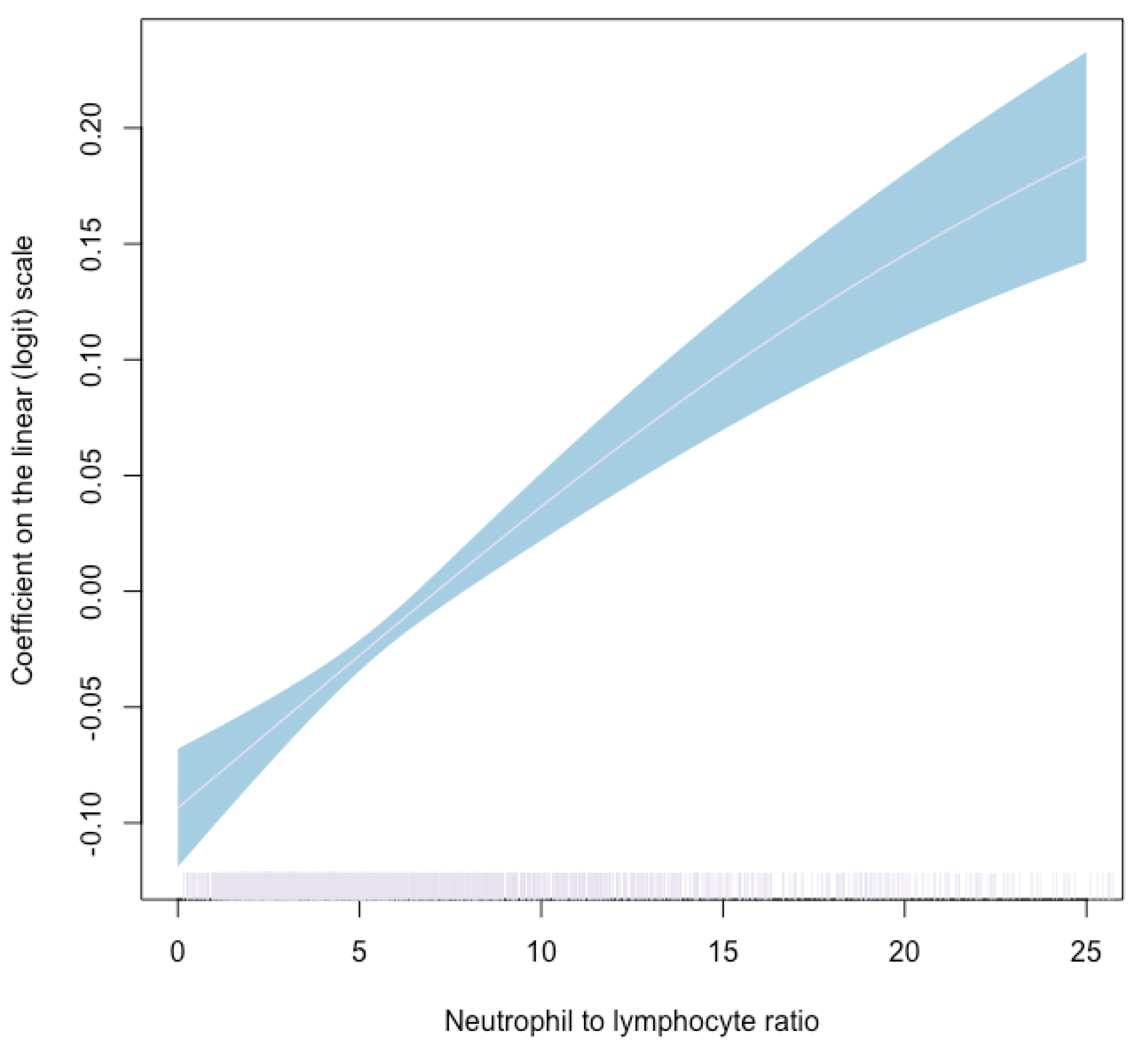
3. Results
4. Discussion
5. Conclusions
6. Study Limitations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wolberg, A.S.; Rosendaal, F.R.; Weitz, J.I.; Jaffer, I.H.; Agnelli, G.; Baglin, T.; Mackman, N. Venous thrombosis. Nat. Rev. Dis. Primers 2015, 1, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Wendelboe, A.M.; Raskob, G.E. Global Burden of Thrombosis: Epidemiologic Aspects. Circ. Res. 2016, 118, 1340–1347. [Google Scholar] [CrossRef]
- Raskob, G.E.; Angchaisuksiri, P.; Blanco, A.N.; Buller, H.; Gallus, A.; Hunt, B.J.; Hylek, E.M.; Kakkar, T.L.; Konstantinides, S.V.; McCumber, M.; et al. Day ISCfWT. Thrombosis: A major contributor to global disease burden. Semin. Thromb. Hemost. 2014, 40, 724–735. [Google Scholar] [CrossRef] [PubMed]
- Carson, J.L.; Kelley, M.A.; Duff, A.; Weg, J.G.; Fulkerson, W.J.; Palevsky, H.I.; Schwartz, J.S.; Thompson, B.T.; Popovich, J., Jr.; Hobbins, T.E. The clinical course of pulmonary embolism. N. Engl. J. Med. 1992, 326, 1240–1245. [Google Scholar] [CrossRef]
- Ng, A.C.C.; Chung, T.; Sze Chiang Yong, A.; Siu Ping Wong, H.; Chow, V.; Celermajer, D.S.; Kritharides, L. Long-term cardiovascular and noncardiovascular mortality of 1023 patients with confirmed acute pulmonary embolism. Circ. Cardiovasc. Qual. Outcomes 2011, 4, 122–128. [Google Scholar] [CrossRef]
- Konstantinides, S.V.; Meyer, G.; Becattini, C.; Bueno, H.; Geersing, G.J.; Harjola, V.P.; Huisman, M.V.; Humbert, M.; Jennings, C.S.; Jimenez, D.; et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). Eur. Heart J. 2020, 41, 543–603. [Google Scholar] [CrossRef]
- Aksu, K.; Donmez, A.; Keser, G. Inflammation-induced thrombosis: Mechanisms, disease associations and management. Curr. Pharm. Des. 2012, 18, 1478–1493. [Google Scholar] [CrossRef]
- Marchena Yglesias, P.J.; Nieto Rodríguez, J.A.; Serrano Martínez, S.; Belinchón Moya, O.; Cortés Carmona, A.; Díaz de Tuesta, A.; Bruscas Alijarde, M.J.; Ruiz Ribó, M.D. Acute-phase reactants and markers of inflammation in venous thromboembolic disease: Correlation with clinical and evolution parameters. Ann. Med. Interna. 2006, 23, 105–110. [Google Scholar] [CrossRef][Green Version]
- Mühl, D.; Füredi, R.; Cristofari, J.; Ghosh, S.; Bogár, L.; Borsiczki, B.; Gasz, B.; Roth, E.; Lantos, J. Evaluation of oxidative stress in the thrombolysis of pulmonary embolism. J. Thromb. Thrombolysis 2006, 22, 221–228. [Google Scholar] [CrossRef]
- Ovechkin, A.V.; Lominadze, D.; Sedoris, K.C.; Robinson, T.W.; Tyagi, S.C.; Roberts, A.M. Lung ischemia-reperfusion injury: Implications of oxidative stress and platelet-arteriolar wall interactions. Arch. Physiol. Biochem. 2007, 113, 1–12. [Google Scholar] [CrossRef]
- von Bruhl, M.L.; Stark, K.; Steinhart, A.; Chandraratne, S.; Konrad, I.; Lorenz, M.; Khandoga, A.; Tirniceriu, A.; Coletti, R.; Kollnberger, M.; et al. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J. Exp. Med. 2012, 209, 819–835. [Google Scholar] [CrossRef]
- Engelmann, B.; Massberg, S. Thrombosis as an intravascular effector of innate immunity. Nat. Rev. Immunol. 2013, 13, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Zahorec, R. Ratio of neutrophil to lymphocyte counts-rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl. Lek. Listy 2001, 102, 5–14. [Google Scholar]
- Minkyo, S.; Graubard, B.I.; Rabkin, C.S.; Engels, E.A. Neutrophil-to-lymphocyte ratio and mortality in the United States general population. Sci. Rep. 2021, 11, 464. [Google Scholar]
- Park, J.-S.; Seo, K.-W.; Choi, B.-J.; Choi, S.-Y.; Yoon, M.-H.; Hwang, G.-S.; Tahk, S.-J.; Shin, J.-H. Importance of prognostic value of neutrophil to lymphocyte ratio in patients with ST-elevation myocardial infarction. Medicine 2018, 97, 48. [Google Scholar] [CrossRef] [PubMed]
- Bhat, T.; Teli, S.; Rijal, J.; Bhat, H.; Raza, M.; Khoueiry, G.; Meghani, M.; Akhtar, M.; Costantino, T. Neutrophil to lymphocyte ratio and cardiovascular diseases: A review. Expert Rev. Cardiovasc. Ther. 2013, 11, 55–59. [Google Scholar] [CrossRef]
- Vazquez-Garza, E.; Jerjes-Sanchez, C.; Navarrete, A.; Joya-Harrison, J.; Rodriguez, D. Venous thromboembolism: Thrombosis, inflammation, and immunothrombosis for clinicians. J. Thromb. Thrombolysis 2017, 44, 377–385. [Google Scholar] [CrossRef]
- Galliazzo, S.; Nigro, O.; Bertù, L.; Guasti, L.; Grandi, A.M.; Ageno, W.; Dentali, F. Prognostic role of neutrophils to lymphocytes ratio in patients with acute pulmonary embolism: A systematic review and meta-analysis of the literature. Intern. Emerg. Med. 2018, 13, 603–608. [Google Scholar] [CrossRef]
- Jia, D.; Liu, F.; Zhang, Q.; Zeng, G.Q.; Li, X.L.; Hou, G. Rapid on-site evaluation of routine biochemical parameters to predict right ventricular dysfunction in and the prognosis of patients with acute pulmonary embolism upon admission to the emergency room. J. Clin. Lab Anal. 2018, 32, e22362. [Google Scholar] [CrossRef]
- Farah, R.; Nseir, W.; Kagansky, D.; Khamisy-Farah, R. The role of neutrophil-lymphocyte ratio, and mean platelet volume in detecting patients with acute venous thromboembolism. J. Clin. Lab Anal. 2020, 34, e23010. [Google Scholar] [CrossRef]
- Kasapoglu, U.S.; Olgun Yildizeli, S.; Arikan, H.; Erer, A.; Kabadayi, F.; Yalcinkaya, E.; Aslan, M.; Cimsit, N.C.; Eryuksel, E.; Karakurt, S. Comparison of neutrophil to lymphocyte ratio with other prognostic markers affecting 30 day mortality in acute pulmonary embolism. Tuberk Toraks. 2019, 67, 179–189. [Google Scholar] [CrossRef]
- Kose, N.; Yildirim, T.; Akin, F.; Yildirim, S.E.; Altun, I. Prognostic role of NLR, PLR, and LMR in patients with pulmonary embolism. Bosn J. Basic Med. Sci. 2020, 20, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Phan, T.; Brailovsky, Y.; Fareed, J.; Hoppensteadt, D.; Iqbal, O.; Darki, A. Neutrophil-to-Lymphocyte and Platelet-to-Lymphocyte Ratios Predict All-Cause Mortality in Acute Pulmonary Embolism. Clin. Appl. Thromb. Hemost. 2020, 26. [Google Scholar] [CrossRef]
- Duman, D.; Sonkaya, E.; Yildirim, E.; Gidik, E.; Tanulku, U.; Salturk, C.; Karakurt, Z. Association of Inflammatory Markers with Mortality in Patients Hospitalized with Non-massive Pulmonary Embolism. Turk. Thorac. J. 2021, 22, 24–30. [Google Scholar] [CrossRef] [PubMed]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gotzsche, P.C.; Vandenbroucke, J.P.; Initiative, S. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. BMJ 2007, 335, 806–808. [Google Scholar] [CrossRef]
- World Health Organization. ICD-10, International Statistical Classification of diseases and Related Health Problems; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Aujesky, D.; Obrosky, D.S.; Stone, R.A.; Auble, T.E.; Perrier, A.; Cornuz, J.; Roy, P.-M.; Fine, M.J. Derivation and validation of a prognostic model for pulmonary embolism. Am. J. Respir. Crit. Care Med. 2005, 172, 1041–1046. [Google Scholar] [CrossRef]
- Hepburn-Brown, M.; Darvall, J.; Hammerschlag, G. Acute pulmonary embolism: A concise review of diagnosis and management. Intern. Med. J. 2019, 49, 15–27. [Google Scholar] [CrossRef] [PubMed]
| Non-Elevated NLR a (≤ 5.12) | Elevated NLR a (>5.12) | p-Value | |
|---|---|---|---|
| n | 1036 | 1036 | |
| Sex [Male, n (%)] | 440 (42.5) | 442 (42.7) | 0.965 |
| Age (years) (median [IQR b]) | 70.59 [56.79, 81.31] | 76.01 [65.12, 84.61] | <0.001 |
| Body mass index (kg/m2) (median [IQR b]) | 26.60 [23.69, 30.87] | 25.70 [22.90, 29.32] | <0.001 |
| Temperature—Celsius (median [IQR b]) | 36.80 [36.60, 37.00] | 36.80 [36.60, 37.20] | <0.001 |
| Pulse rate—beats per minute (median [IQR b]) | 83.00 [74.00, 95.00] | 89.00 [78.00, 102.00] | <0.001 |
| Tachycardia [>100 beats per minute, n (%)] | 172 (16.7) | 283 (27.4) | <0.001 |
| Systolic blood pressure—mmHg (median [IQR b]) | 105.00 [92.00, 118.00] | 97.00 [80.00, 111.00] | <0.001 |
| Low systolic blood pressure [<90 mmHg, n (%)] | 201 (19.4) | 373 (36.1) | <0.001 |
| Oxygen saturation (median [IQR b]) | 96.00 [94.00, 98.00] | 95.00 [92.00, 97.00] | <0.001 |
| Low oxygen saturation [<90%, n (%)] | 96 (9.4) | 170 (16.7) | <0.001 |
| Past PE c or DVT d (%) | 124 (12.0) | 143 (13.8) | 0.238 |
| Hypertension—n (%) | 438 (42.3) | 491 (47.4) | 0.022 |
| Ischemic heart disease—n (%) | 125 (12.1) | 150 (14.5) | 0.120 |
| Diabetes mellitus—n (%) | 205 (19.8) | 186 (18.0) | 0.312 |
| Cerebrovascular accident—n (%) | 86 (8.3) | 78 (7.5) | 0.569 |
| Heart failure—n (%) | 44 (4.2) | 77 (7.4) | 0.003 |
| Malignancy—n (%) | 241 (23.3) | 318 (30.7) | <0.001 |
| Atrial fibrillation—n (%) | 68 (6.6) | 106 (10.2) | 0.003 |
| Chronic obstructive pulmonary disease—n (%) | 63 (6.1) | 127 (12.3) | <0.001 |
| Chronic kidney disease—n (%) | 72 (6.9) | 88 (8.5) | 0.217 |
| Dyslipidemia—n (%) | 279 (26.9) | 297 (28.7) | 0.404 |
| Hemoglobin—g/L (median [IQR b]) | 11.99 [10.50, 13.28] | 11.40 [10.04, 12.96] | <0.001 |
| Anemia e at presentation—n (%) | 603 (59.0) | 688 (67.5) | <0.001 |
| Platelets—109/L (median [IQR b]) | 219.00 [166.00, 284.00] | 214.00 [162.00, 283.00] | 0.767 |
| International normalized ratio—n (median [IQR b]) | 1.07 [0.99, 1.18] | 1.11 [1.02, 1.23] | <0.001 |
| Creatinine—µmol/L (median [IQR b]) | 0.88 [0.72, 1.10] | 0.94 [0.71, 1.29] | 0.001 |
| eGFR f below 60mL/min/1.73m2 at presentation—n (%) | 293 (28.5) | 435 (42.2) | <0.001 |
| Troponin I—µg/L (median [IQR b]) | 0.05 [0.03, 0.20] | 0.05 [0.03, 0.19] | 0.940 |
| D-dimer—nmol/L (median [IQR b]) | 1580.00 [700.00, 2923.00] | 2062.00 [871.00, 4818.50] | <0.001 |
| aOR a for 30-Day Mortality (95% CI b) | p-Value | aOR a for One-Year Mortality (95% CI b) | p-Value | |
|---|---|---|---|---|
| Elevated NLR c (>5.12) | 2.82 (2.14, 3.7) | <0.001 | 2.51 (2.04, 3.08) | <0.001 |
| Sex (males versus females) | 0.95 (0.73, 1.24) | 0.721 | 1.02 (0.83, 1.27) | 0.835 |
| Age | 1.03 (1.02, 1.04) | <0.001 | 1.03 (1.02, 1.04) | <0.001 |
| Atrial fibrillation | 1.08 (0.72, 1.62) | 0.719 | 1.17 (0.82, 1.69) | 0.388 |
| Ischemic heart disease | 1.1 (0.77, 1.56) | 0.614 | 1.13 (0.83, 1.54) | 0.433 |
| Heart failure | 1.13 (0.7, 1.82) | 0.628 | 1.03 (0.67, 1.59) | 0.888 |
| Chronic kidney disease | 1.06 (0.69, 1.62) | 0.796 | 1.06 (0.73, 1.55) | 0.764 |
| Diabetes mellitus | 1.19 (0.87, 1.63) | 0.278 | 1.41 (1.08, 1.84) | 0.011 |
| Cerebrovascular accident | 1.03 (0.67, 1.6) | 0.884 | 0.94 (0.65, 1.35) | 0.735 |
| Hypertension | 1.04 (0.79, 1.37) | 0.804 | 0.94 (0.75, 1.19) | 0.626 |
| Chronic obstructive pulmonary disease | 0.87 (0.59, 1.31) | 0.51 | 1.25 (0.89, 1.76) | 0.19 |
| Malignancy | 1.72 (1.31, 2.25) | <0.001 | 3.4 (2.71, 4.27) | <0.001 |
| Low systolic blood pressure | 2.82 (2.19, 3.64) | <0.001 | 1.79 (1.43, 2.23) | <0.001 |
| Non-Elevated NLR a (≤5.12) | Elevated NLR a (>5.12) | p-Value | |
|---|---|---|---|
| n | 1036 | 1036 | |
| Length of hospital stay (median [IQR b]) | 3.09 [1.67, 7.45] | 5.84 [2.81, 13.99] | <0.001 |
| In-hospital mortality (%) | 35 (3.4) | 149 (14.4) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Efros, O.; Beit Halevi, T.; Meisel, E.; Soffer, S.; Barda, N.; Cohen, O.; Kenet, G.; Lubetsky, A. The Prognostic Role of Neutrophil-to-Lymphocyte Ratio in Patients Hospitalized with Acute Pulmonary Embolism. J. Clin. Med. 2021, 10, 4058. https://doi.org/10.3390/jcm10184058
Efros O, Beit Halevi T, Meisel E, Soffer S, Barda N, Cohen O, Kenet G, Lubetsky A. The Prognostic Role of Neutrophil-to-Lymphocyte Ratio in Patients Hospitalized with Acute Pulmonary Embolism. Journal of Clinical Medicine. 2021; 10(18):4058. https://doi.org/10.3390/jcm10184058
Chicago/Turabian StyleEfros, Orly, Tal Beit Halevi, Eshcar Meisel, Shelly Soffer, Noam Barda, Omri Cohen, Gili Kenet, and Aharon Lubetsky. 2021. "The Prognostic Role of Neutrophil-to-Lymphocyte Ratio in Patients Hospitalized with Acute Pulmonary Embolism" Journal of Clinical Medicine 10, no. 18: 4058. https://doi.org/10.3390/jcm10184058
APA StyleEfros, O., Beit Halevi, T., Meisel, E., Soffer, S., Barda, N., Cohen, O., Kenet, G., & Lubetsky, A. (2021). The Prognostic Role of Neutrophil-to-Lymphocyte Ratio in Patients Hospitalized with Acute Pulmonary Embolism. Journal of Clinical Medicine, 10(18), 4058. https://doi.org/10.3390/jcm10184058







