Arterial Hypertension and Diabetes Mellitus in COVID-19 Patients: What Is Known by Gender Differences?
Abstract
:1. Introduction
2. Methods
- Mild type (with slight clinical symptoms but no imaging findings of pneumonia);
- Common type (with fever, respiratory tract, and imaging findings of pneumonia);
- Severe type (with any of the following conditions):
- Respiratory distress with respiratory frequency > 30 times/min;
- Finger oxygen saturation at rest 93% AA; or
- Oxygenation index (PaO2/FiO2) < 300 mmHg;
- Critical type (with any of the following conditions):
- c.
- Respiratory failure that requires mechanical ventilation;
- d.
- Shock and other organ failures that require intensive care unit.
3. Statistical Analysis
4. Results
Outcomes
5. Discussion
6. Conclusions and Study Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- OMS. Health Emergency Dashboard. 2021. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019 (accessed on 19 August 2021).
- Fang, L.; Karakiulakis, G.; Roth, M. Are patients with hypertension and diabetes mellitus at increased risk for Covid-19 infection? Lancet Respir. Med. 2020. [Google Scholar] [CrossRef]
- Zhang, J.J.; Dong, X.; Cao, Y.Y.; Yuan, Y.D.; Yang, Y.B.; Yan, Y.Q.; Akdis, C.A.; Gao, Y.D. Clinical characteristics of 140 patients infected with SARS- CoV-2 in Wuhan, China. Allergy 2020, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Yang, J.; Zhao, F.; Zhi, L.; Wang, X.; Liu, L.; Zhao, Y. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin. Res. Cardiol. 2020, 109, 531–538. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.L.; Yang, J.M.; Sun, Y.P.; Su, G.H. Inhibitors of RAS might be a good choice for the therapy of COVID-19 pneumonia. Zhonghua Jie He He Hu Xi Za Zhi 2020, 43, 219–222. [Google Scholar]
- Levy, B.I.; Heusch, G.; Camici, P.G. The many faces of myocardial ischaemia and angina. Cardiovasc 2019, 115, 1460–1470. [Google Scholar] [CrossRef]
- Onder, G.; Rezza, G.; Brusaferro, S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. JAMA 2020. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Wei, C.; Zhang, Y. A meta-analysis of sex differences in severity and mortality among patients with COVID-19. Ann. Med. 2021, in press. [Google Scholar]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D. The Joint European Society of Cardiology/American College of Cardiology/American Heart Association/World Heart Federation Task Force for the Universal Definition of Myocardial Infarction: The Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction. Fourth Universal Definition of Myocardial Infarction. Circulation 2018, 138, e618–e651. [Google Scholar] [CrossRef]
- Travis, W.D.; Costabel, U.; Hansell, D.M.; King, T.E., Jr.; Lynch, D.A.; Nicholson, A.G.; Ryerson, C.J.; Ryu, J.H.; Selman, M.; Wells, A.U.; et al. An official American Thoracic Society/European Respiratory Society statement: Update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. ATS/ERS Committee on Idiopathic Interstitial Pneumonias. Am. J. Respir. Crit. Care Med. 2013, 188, 733–748. [Google Scholar] [CrossRef] [PubMed]
- Webster, A.C.; Nagler, E.V.; Morton, R.L. Philip Masson Chronic Kidney Disease. Lancet 2017, 389, 1238–1252. [Google Scholar] [CrossRef]
- de Leciñana, M.A.; Egido, J.A.; Casado, I.; Ribó, M.; Dávalos, A.; Masjuan, J.; Vivancos, J. Spanish Neurological Society. Guidelines for the treatment of acute ischaemic stroke. Neurologia 2014, 29, 102–122. [Google Scholar] [CrossRef]
- Pasupathy, S.; Air, T.; Dreyer, R.P.; Tavella, R.; Beltrame, J.F. Systematic review of patients presenting with suspected myocardial infarction and nonobstructive coronary arteries. Circulation 2015, 131, 861–870. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swati, S.; Mary, F.; Carol, F.; Allecia, M.W.; Albino, E.; Ilaria, G.; Elisabet, P.; Clare, B.; Fadi, S.; Siraj, M.E.J.; et al. Postmortem Findings Associated with SARS-CoV-2: Systematic Review and Meta-analysis. Am. J. Surg. Pathol. 2021, 45, 587–603. [Google Scholar] [CrossRef]
- Marin, B.G.; Aghagoli, G.; Lavine, K.; Yang, L. Predictors of COVID-19 severity: A literature review. Med. Virol. 2021, 31, 1–10. [Google Scholar] [CrossRef]
- Guzik, T.J.; Mohiddin, S.A.; Dimarco, A.; Patel, V.; Savvatis, K.; Marelli-Berg, F.M.; Madhur, M.S.; Tomaszewski, M.; Maffia, P.; D’Acquisto, F. COVID-19 and the cardiovascular system: Implications for risk assessment, diagnosis, and treatment options. Cardiovasc. Res. 2020, 116, 1666–1668. [Google Scholar] [CrossRef]
- National Health Commission of the People’s Republic of China. Chinese Clinical Guidance for COVID-19 Pneumonia Diagnosis and Treatment (7th Edition). 2020. Available online: http://kjfy.meetingchina.org/msite/news/show/cn/3337.html (accessed on 19 August 2021).
- Lauer, S.A.; Grantz, K.H.; Bi, Q.; Jones, F.K.; Zheng, Q.; Meredith, H.R.; Azman, A.S.; Reich, N.G.; Lessler, J. The incubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: Estimation and application. Ann. Intern Med. 2020. [Google Scholar] [CrossRef] [Green Version]
- Zhou, C.; Gao, C.; Xie, Y.; Xu, M. COVID-19 with spontaneous pneumomediastinum. Lancet Infect. Dis. 2020, 20, 510. [Google Scholar] [CrossRef]
- Sun, R.; Liu, H.; Wang, X. Mediastinal emphysema, giant bulla, and pneumothorax developed during the course of COVID-19 pneumonia. Korean J. Radiol. 2020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grasselli, G.; Zangrillo, A.; Zanella, A.; Antonelli, M.; Cabrini, L.; Castelli, A.; Cereda, D.; Coluccello, A.; Foti, G.; Fumagalli, R.; et al. Baseline Characteristics and Outcomes of 1591 Patients Infected With SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA 2020, 323, 1574–1581. [Google Scholar] [CrossRef] [Green Version]
- Epidemiology Working Group for NCIP Epidemic Response. The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) in China. Chin. J. Epidemiol. 2020, 41, 145–151. [Google Scholar]
- Vaduganathan, M.; Vardeny, O.; Michel, T.; McMurray, J.J.V.; Pfeffer, M.A.; Solomon, S.D. Renin–angiotensin–aldosterone system inhibitors in patients with Covid-19. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Drummond, G.; Vinh, A.; Guzik, T.; Sobey, C.G. Immune mechanisms of hypertension. Nat. Rev. Immunol. 2019, 19, 517–532. [Google Scholar] [CrossRef] [PubMed]
- Loperena, R.; Van Beusecum, J.P.; Itani, H.A.; Engel, N.; Laroumanie, F.; Xiao, L.; Elijovich, F.; Laffer, C.L.; Gnecco, J.S.; Noonan, J.; et al. Hypertension and increased endothelial mechanical stretch promote monocyte differentiation and activation: Roles of STAT3, interleukin 6 and hydrogen peroxide. Cardiovasc. Res. 2018, 114, 1547–1563. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhang, Q.; Wang, P.; Ye, H.; Jing, X.; Zhang, Z.; Zhu, S.; Luo, T.; Zheng, Z. Clinical features of hypertensive patients with COVID-19 compared with a normotensive group: Single-center experience in China. Open Med. 2021, 16, 367–374. [Google Scholar] [CrossRef]
- Siedlinski, M.; Jozefczuk, E.; Xu, X.; Teumer, A.; Evangelou, E.; Schnabel, R.B.; Welsh, P.; Maffia, P.; Erdmann, J.; Tomaszewski, M.; et al. White blood cells and blood pressure: A Mendelian randomization study. Circulation 2020, 141, 1307–1317. [Google Scholar] [CrossRef]
- Steven, S.; Dib, M.; Hausding, M.; Kashani, F.; Oelze, M.; Kroller-Schon, S.; Hanf, A.; Daub, S.; Roohani, S.; Gramlich, Y.; et al. CD40L controls obesity-associated vascular inflammation, oxidative stress, and endothelial dysfunction in high fat diet-treated and db/db mice. Cardiovasc. Res. 2018, 114, 312–332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruan, S. Likelihood of survival of coronavirus disease 2019. Lancet Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Liu, Z.; Bing, X.; Zhi, Z. Novel Coronavirus Pneumonia Emergency Response Epidemiology Team. Epidemiol. Charact. Outbreak 2020, 41, 145–151. [Google Scholar] [CrossRef]
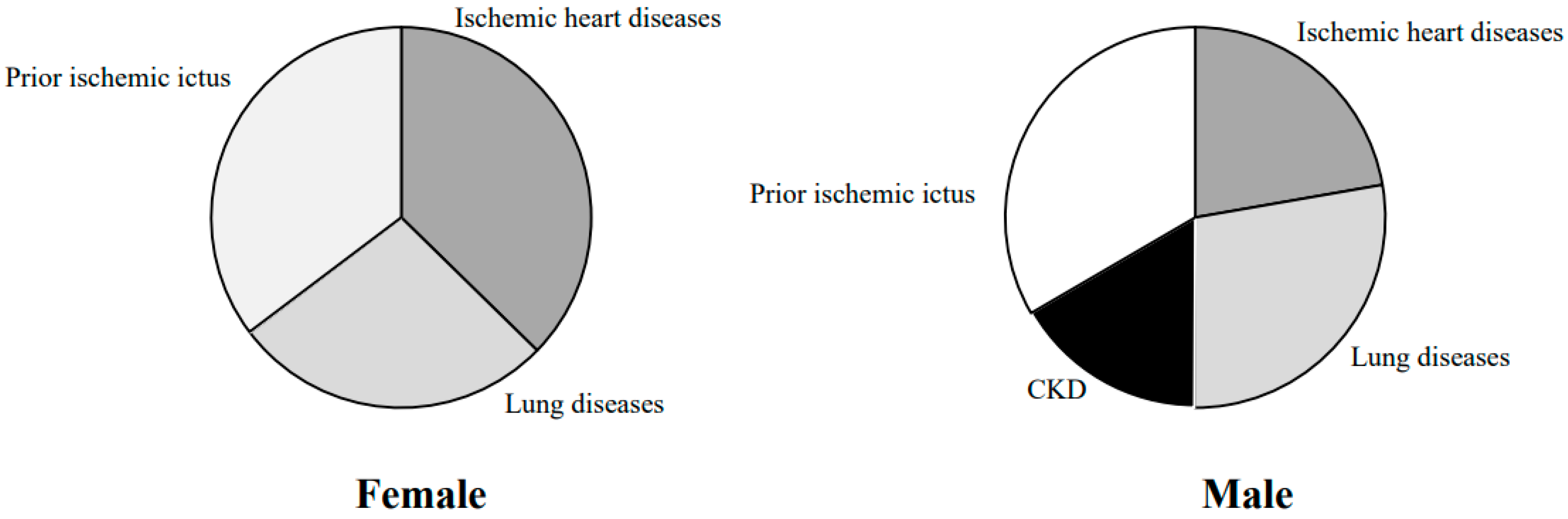
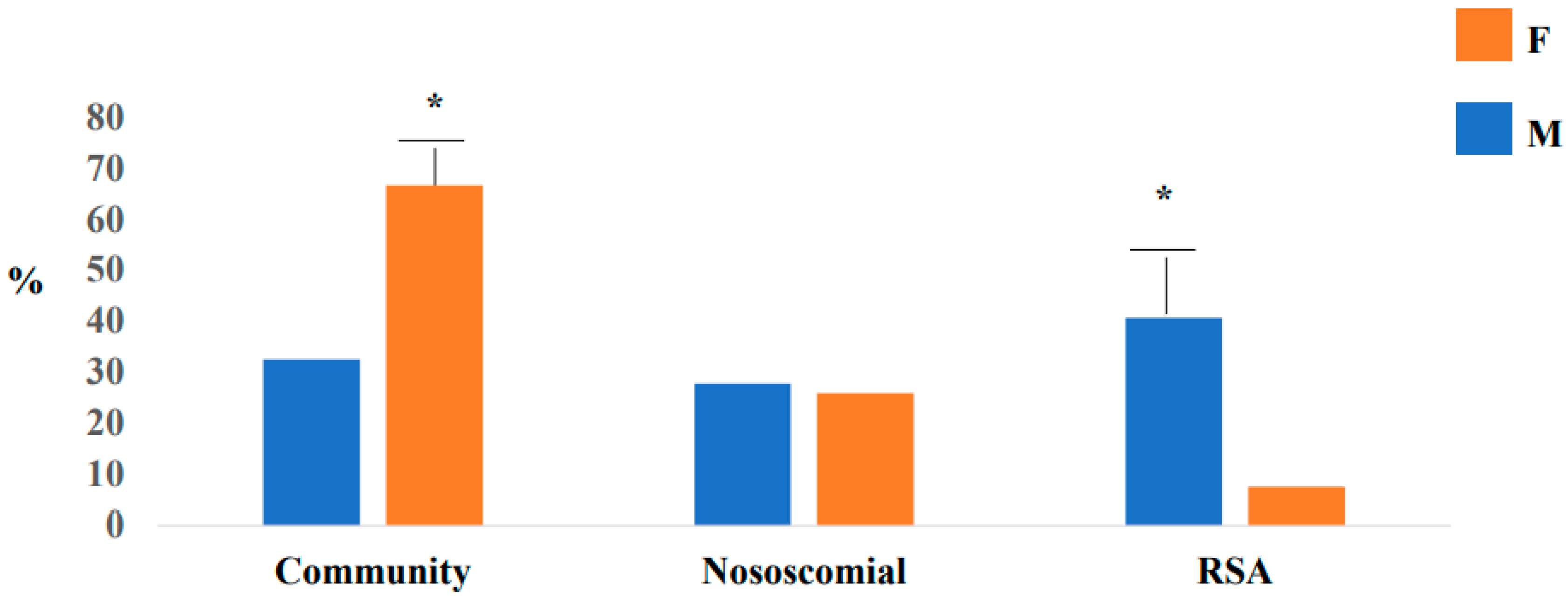
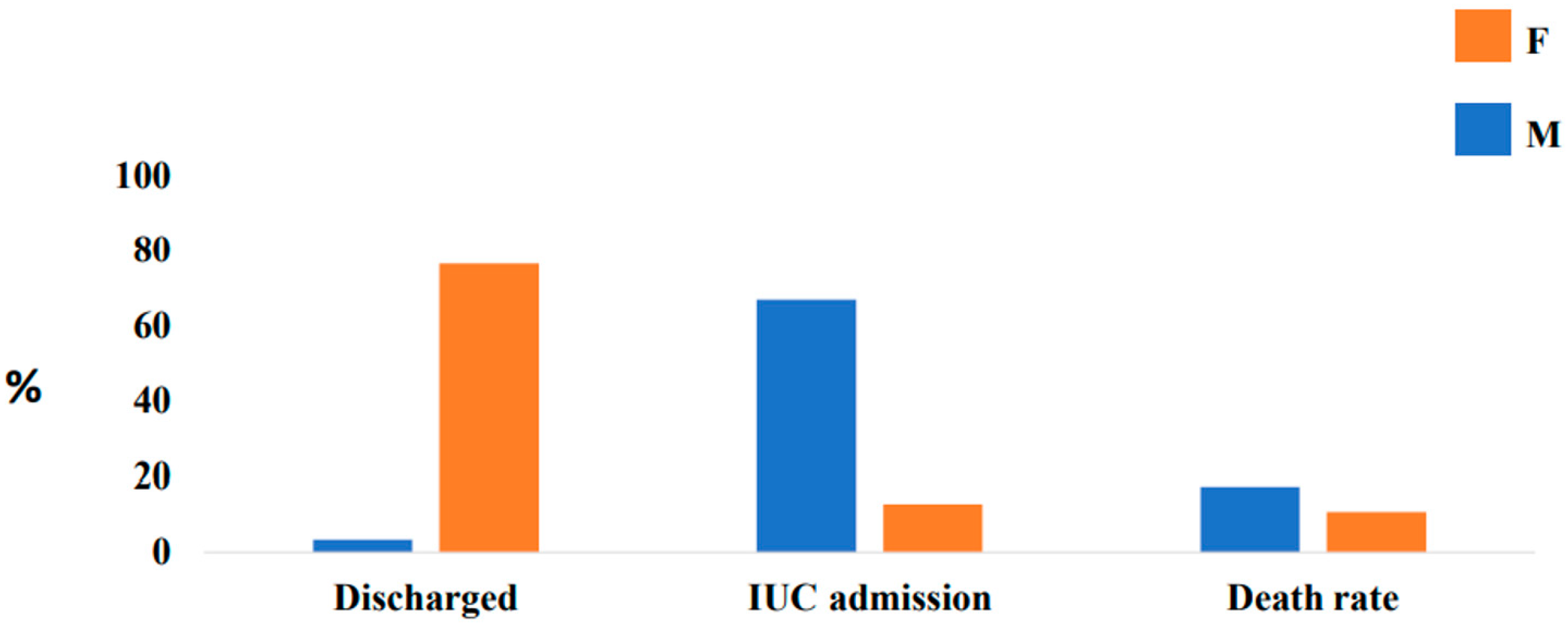
| Female (n. 270) | Male (n. 286) | p Value | |
|---|---|---|---|
| Age (mean) | 76.0 (56.0–86.0) | 63.0 (51.5-70.0) | <0.0001 |
| Ischemic heart disease | 128 (47.5%) | 42 (14.8%) | <0.0001 |
| Lung Disease | 93 (34.8%) | 53 (18.5%) | <0.0001 |
| CKD | 0 (0.01%) | 32 (11.1%) | <0.0001 |
| Prior Ischemic ictus | 121 (44.9%) | 63 (22.2%) | <0.0001 |
| Stay hospital (days) | 17.0 (9.0, 23.0) | 22.0 (16.0, 33.0) | <0.0001 |
| SBP (mmHg) | 135 (130–145) | 130 (125–135) | <0.0001 |
| DBP (mmHg) | 78 (74–88) | 72 (70–84) | <0.0001 |
| Basal glicemia | 135 (140–165) | 130 (135–160) | <0.0001 |
| ICM (kg/m2) | 25 (24.5–25.5) | 26 (25.5–26.5) | 0.024 |
| Smoking status (%) | 40 (15) | 45 (16) | <0.0001 |
| DM-HT Group (n. 556) | Non-DM Non-HT Group (n. 458) | p Value | |
|---|---|---|---|
| Age (mean) | 76.0 (56.0–86.0) | 67 (51.0-83.0) | <0.0001 |
| SBP (mmHg) | 140 (130–145) | 110 (105–115) | <0.0001 |
| DBP (mmHg) | 90 (85–100) | 77 (70–84) | <0.0001 |
| Basal glicemia | 137 (140–165) | 110 (105–115) | <0.0001 |
| ICM (kg/m2) | 27 (26.5–27.5) | 26.0 (25.5–27.5) | 0.024 |
| Smoking status (%) | 100 (18) | 71 (16) | 0.0039 |
| Male (n. 286) | Female (n. 270) | p Value | |
|---|---|---|---|
| Epidemiological Link | |||
| Community | 180 (66.7%) | 87 (32.5%) | <0.0001 |
| Nosocomial | 79 (27.8%) | 70 (25.9%) | 0.0016 |
| RSA | 116 (40.7%) | 20 (7.4%) | <0.0001 |
| Symptoms | |||
| Fever | 120 (42.0%) | 230 (85.2%) | <0.0001 |
| Dyspnea | 148 (51.9%) | 40 (14.8%) | <0.0001 |
| Rx lung | |||
| Atypical | 242 (84.8%) | 69 (25.7%) | <0.0001 |
| Typical | 44 (15.2%) | 200 (74.3%) | <0.0001 |
| CT lung | |||
| Atypical | 286 (100.0%) | 240 (88.9%) | <0.0001 |
| Typical | 0 (0.0%) | 30 (11.1%) | <0.0001 |
| PF ratio < 300 (%) | 58 (20.3%) | 40 (14.8%) | <0.0001 |
| Discharge at home (%) | 9 (3.14%) | 206 (76.5%) | <0.0001 |
| ICU admission (%) | 191 (66.9%) | 34 (12.8%) | <0.0001 |
| Death rate (%) | 49 (17.3%) | 28 (10.7%) | <0.0001 |
| O2 therapy | 140 (51.8%) | 83 (29.1%) | <0.0001 |
| HFNC | 270 (100.0%) | 267 (93.7%) | <0.0001 |
| Hydroxychloroquine | 128 (44.9%) | 260 (96.3%) | <0.0001 |
| Darunavir/ritonavir | 21 (7.4%) | 0 (0.0%) | Ns |
| LMWH therapy | 201 (70.4%) | 82 (30.5%) | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciarambino, T.; Ciaburri, F.; Paoli, V.D.; Caruso, G.; Giordano, M.; D’Avino, M. Arterial Hypertension and Diabetes Mellitus in COVID-19 Patients: What Is Known by Gender Differences? J. Clin. Med. 2021, 10, 3740. https://doi.org/10.3390/jcm10163740
Ciarambino T, Ciaburri F, Paoli VD, Caruso G, Giordano M, D’Avino M. Arterial Hypertension and Diabetes Mellitus in COVID-19 Patients: What Is Known by Gender Differences? Journal of Clinical Medicine. 2021; 10(16):3740. https://doi.org/10.3390/jcm10163740
Chicago/Turabian StyleCiarambino, Tiziana, Filippina Ciaburri, Venere Delli Paoli, Giuseppe Caruso, Mauro Giordano, and Maria D’Avino. 2021. "Arterial Hypertension and Diabetes Mellitus in COVID-19 Patients: What Is Known by Gender Differences?" Journal of Clinical Medicine 10, no. 16: 3740. https://doi.org/10.3390/jcm10163740
APA StyleCiarambino, T., Ciaburri, F., Paoli, V. D., Caruso, G., Giordano, M., & D’Avino, M. (2021). Arterial Hypertension and Diabetes Mellitus in COVID-19 Patients: What Is Known by Gender Differences? Journal of Clinical Medicine, 10(16), 3740. https://doi.org/10.3390/jcm10163740





