A Reperfusion BOLD-MRI Tissue Perfusion Protocol Reliably Differentiate Patients with Peripheral Arterial Occlusive Disease from Healthy Controls
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. BOLD-MRI Examinations
2.3. Image Post Processing
2.4. Statistical Analysis
3. Results
3.1. Study Population
3.2. BOLD-MRI Examination Results
3.3. Correlations between Reperfusion BOLD-MRI Parameters and ABI
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Song, P.; Rudan, D.; Zhu, Y.; Fowkes, F.J.I.; Rahimi, K.; Fowkes, F.G.R.; Rudan, I. Global, regional, and national prevalence and risk factors for peripheral artery disease in 2015: An updated systematic review and analysis. Lancet Glob. Health 2019, 7, e1020–e1030. [Google Scholar] [CrossRef] [Green Version]
- Aboyans, V.; Ricco, J.B.; Bartelink, M.E.L.; Bjorck, M.; Brodmann, M.; Cohnert, T.; Collet, J.P.; Czerny, M.; De Carlo, M.; Debus, S.; et al. Editor’s choice-2017 esc guidelines on the diagnosis and treatment of peripheral arterial diseases, in collaboration with the european society for vascular surgery (esvs). Eur. J. Vasc. Endovasc. Surg. 2018, 55, 305–368. [Google Scholar] [CrossRef] [Green Version]
- Roy, T.L.; Forbes, T.L.; Dueck, A.D.; Wright, G.A. Mri for peripheral artery disease: Introductory physics for vascular physicians. Vasc. Med. 2018, 23, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Grozinger, G.; Pohmann, R.; Schick, F.; Grosse, U.; Syha, R.; Brechtel, K.; Rittig, K.; Martirosian, P. Perfusion measurements of the calf in patients with peripheral arterial occlusive disease before and after percutaneous transluminal angioplasty using mr arterial spin labeling. J. Magn. Reson. Imaging 2014, 40, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, S.; Lee, T.M.; Kay, A.R.; Tank, D.W. Brain magnetic resonance imaging with contrast dependent on blood oxygenation. Proc. Natl. Acad. Sci. USA 1990, 87, 9868–9872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bajwa, A.; Wesolowski, R.; Patel, A.; Saha, P.; Ludwinski, F.; Ikram, M.; Albayati, M.; Smith, A.; Nagel, E.; Modarai, B. Blood oxygenation level-dependent cmr-derived measures in critical limb ischemia and changes with revascularization. J. Am. Coll. Cardiol. 2016, 67, 420–431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Partovi, S.; von Tengg-Kobligk, H.; Bhojwani, N.; Karmonik, C.; Maurer, M.; Robbin, M.R. Advanced noncontrast mr imaging in musculoskeletal radiology. Radiol. Clin. N. Am. 2015, 53, 549–567. [Google Scholar] [CrossRef]
- Glover, G.H. Overview of functional magnetic resonance imaging. Neurosurg. Clin. 2011, 22, 133–139, vii. [Google Scholar] [CrossRef] [Green Version]
- Stacy, M.R.; Qiu, M.; Papademetris, X.; Caracciolo, C.M.; Constable, R.T.; Sinusas, A.J. Application of bold magnetic resonance imaging for evaluating regional volumetric foot tissue oxygenation: A feasibility study in healthy volunteers. Eur. J. Vasc. Endovasc. Surg. 2016, 51, 743–749. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ledermann, H.P.; Schulte, A.C.; Heidecker, H.G.; Aschwanden, M.; Jager, K.A.; Scheffler, K.; Steinbrich, W.; Bilecen, D. Blood oxygenation level-dependent magnetic resonance imaging of the skeletal muscle in patients with peripheral arterial occlusive disease. Circulation 2006, 113, 2929–2935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakermans, A.J.; Wessel, C.H.; Zheng, K.H.; Groot, P.F.C.; Stroes, E.S.G.; Nederveen, A.J. Dynamic magnetic resonance measurements of calf muscle oxygenation and energy metabolism in peripheral artery disease. J. Magn. Reson. Imaging 2020, 51, 98–107. [Google Scholar] [CrossRef]
- Green, S. Haemodynamic limitations and exercise performance in peripheral arterial disease. Clin. Physiol. Funct. Imaging 2002, 22, 81–91. [Google Scholar] [CrossRef]
- Jacobi, B.; Bongartz, G.; Partovi, S.; Schulte, A.C.; Aschwanden, M.; Lumsden, A.B.; Davies, M.G.; Loebe, M.; Noon, G.P.; Karimi, S.; et al. Skeletal muscle bold mri: From underlying physiological concepts to its usefulness in clinical conditions. J. Magn. Reson. Imaging 2012, 35, 1253–1265. [Google Scholar] [CrossRef] [PubMed]
- Behnel, S.B.R.; Citro, C.; Dalcin, L.; Seljebotn, D.S.; Smith, K. Cython: The best of both worlds. Comput. Sci. Eng. 2011, 13, 31–39. [Google Scholar]
- Kluyver, T.; Ragan-Kelley, B.; Perez, F.; Granger, B.E.; Bussonnier, M.; Frederic, J. Notebooks—A publishing format for reproducible computational workflows. In Positioning and Power in Academic Publishing: Players, Agents and Agendas; Loizides, F., Schmidt, B., Eds.; IOS Press: Amsterdam, The Netherlands, 2016; pp. 87–90. [Google Scholar]
- Filip Malmberg, R.N.; Robin, S.; Joel, K. Smartpaint: A tool for interactive segmentation of medical volume images. Comput. Methods Biomech. Biomed. Eng. Imaging Vis. 2017, 5, 36–44. [Google Scholar] [CrossRef]
- Klein, S.; Staring, M.; Murphy, K.; Viergever, M.A.; Pluim, J.P.W. Elastix: A toolbox for intensity based medical image registration. IEEE Trans. Med. Imaging 2010, 29, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Yoo, T.S.; Ackerman, M.J.; Lorensen, W.E.; Schroeder, W.; Chalana, V.; Aylward, S.; Metaxas, D.; Whitaker, R. Engineering and algorithm design for an image processing api: A technical report on itk—The insight toolkit. In Medicine Meets Virtual Reality 02/10; IOS Press: Amsterdam, The Netherlands, 2002; pp. 586–592. [Google Scholar]
- Hinkle, D.E.; Wiersma, W.; Jurs, S.G. Applied Statistics for the Behavioral Sciences, 5th ed.; Houghton Mifflin: Boston, MA, USA, 2003. [Google Scholar]
- Potthast, S.; Schulte, A.; Kos, S.; Aschwanden, M.; Bilecen, D. Blood oxygenation level-dependent mri of the skeletal muscle during ischemia in patients with peripheral arterial occlusive disease. Rofo 2009, 181, 1157–1161. [Google Scholar] [CrossRef]
- Stacy, M.R.; Caracciolo, C.M.; Qiu, M.; Pal, P.; Varga, T.; Constable, R.T.; Sinusas, A.J. Comparison of regional skeletal muscle tissue oxygenation in college athletes and sedentary control subjects using quantitative bold mr imaging. Physiol. Rep. 2016, 4, e12903. [Google Scholar] [CrossRef] [Green Version]
- Clyne, C.A.; Mears, H.; Weller, R.O.; O’Donnell, T.F. Calf muscle adaptation to peripheral vascular disease. Cardiovasc. Res. 1985, 19, 507–512. [Google Scholar] [CrossRef]
- McGuigan, M.R.; Bronks, R.; Newton, R.U.; Sharman, M.J.; Graham, J.C.; Cody, D.V.; Kraemer, W.J. Muscle fiber characteristics in patients with peripheral arterial disease. Med. Sci. Sports Exerc. 2001, 33, 2016–2021. [Google Scholar] [CrossRef]
- Nishii, T.; Kono, A.K.; Nishio, M.; Kyotani, K.; Nishiyama, K.; Sugimura, K. Dynamic blood oxygen level-dependent mr imaging of muscle: Comparison of postocclusive reactive hyperemia in young smokers and nonsmokers. Magn. Reson. Med. Sci. 2015, 14, 275–283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guensch, D.P.; Michel, M.C.; Huettenmoser, S.P.; Jung, B.; Gulac, P.; Segiser, A.; Longnus, S.L.; Fischer, K. The blood oxygen level dependent (bold) effect of in-vitro myoglobin and hemoglobin. Sci. Rep. 2021, 11, 11464. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, Z.; Gao, P.; Jin, L.; Li, L.; Zhao, W.; Zhang, W.; Sun, Y.; Zhao, Y.; Cui, J. Quantitative evaluation of postintervention foot blood supply in patients with peripheral artery disease by computed tomography perfusion. J. Vasc. Surg. 2020, 72, 1035–1042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sah, B.R.; Veit-Haibach, P.; Strobel, K.; Banyai, M.; Huellner, M.W. Ct-perfusion in peripheral arterial disease—Correlation with angiographic and hemodynamic parameters. PLoS ONE 2019, 14, e0223066. [Google Scholar] [CrossRef]
- Veit-Haibach, P.; Huellner, M.W.; Banyai, M.; Mafeld, S.; Heverhagen, J.; Strobel, K.; Sah, B.R. Ct perfusion in peripheral arterial disease-hemodynamic differences before and after revascularisation. Eur. Radiol. 2021, 31, 5507–5513. [Google Scholar] [CrossRef] [PubMed]
- Criqui, M.H.; Matsushita, K.; Aboyans, V.; Hess, C.N.; Hicks, C.W.; Kwan, T.W.; McDermott, M.M.; Misra, S.; Ujueta, F. American Heart Association Council on Epidemiology and Prevention; et al. Lower extremity peripheral artery disease: Contemporary epidemiology, management gaps, and future directions: A scientific statement from the american heart association. Circulation 2021. [Google Scholar] [CrossRef] [PubMed]
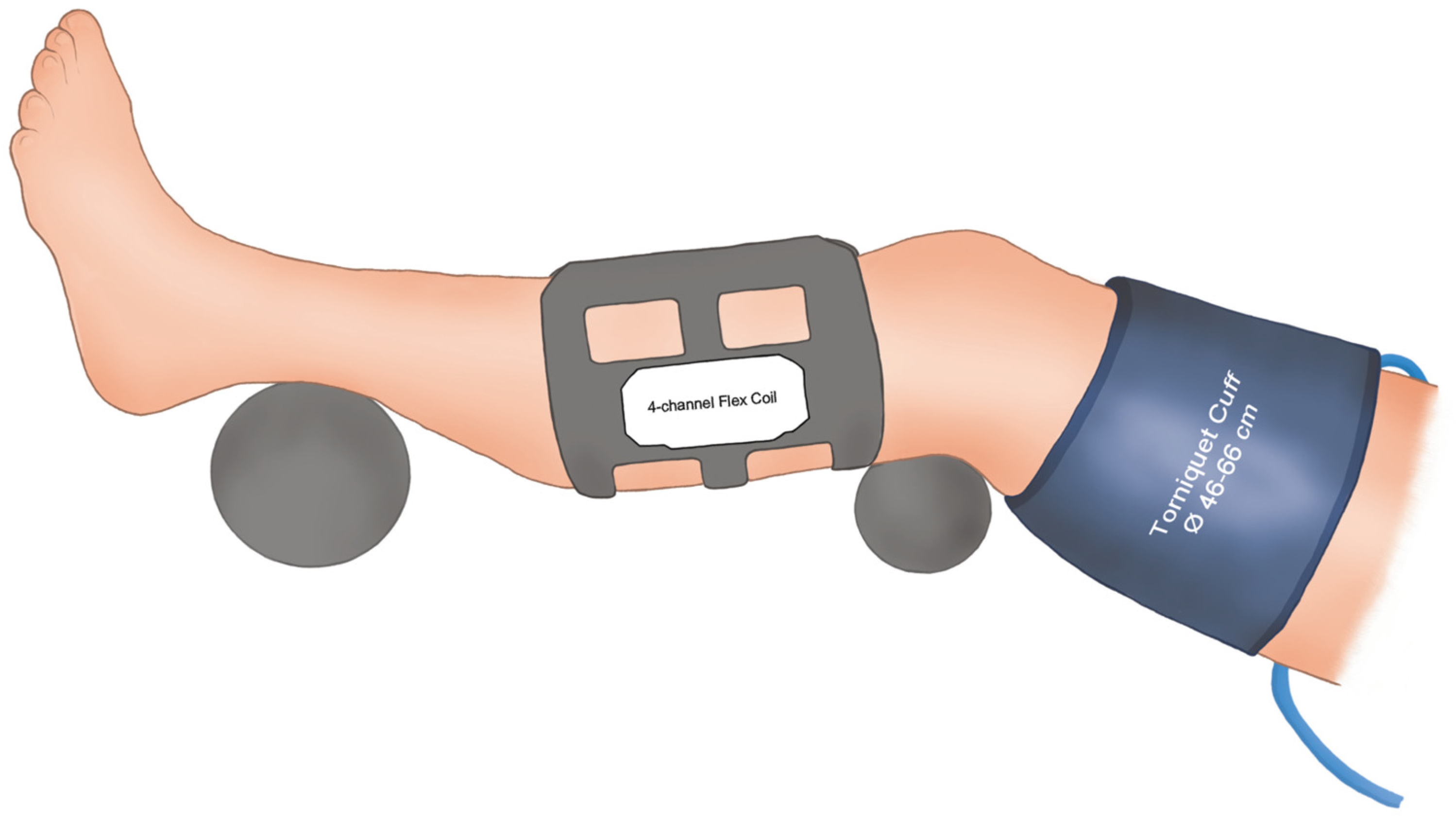

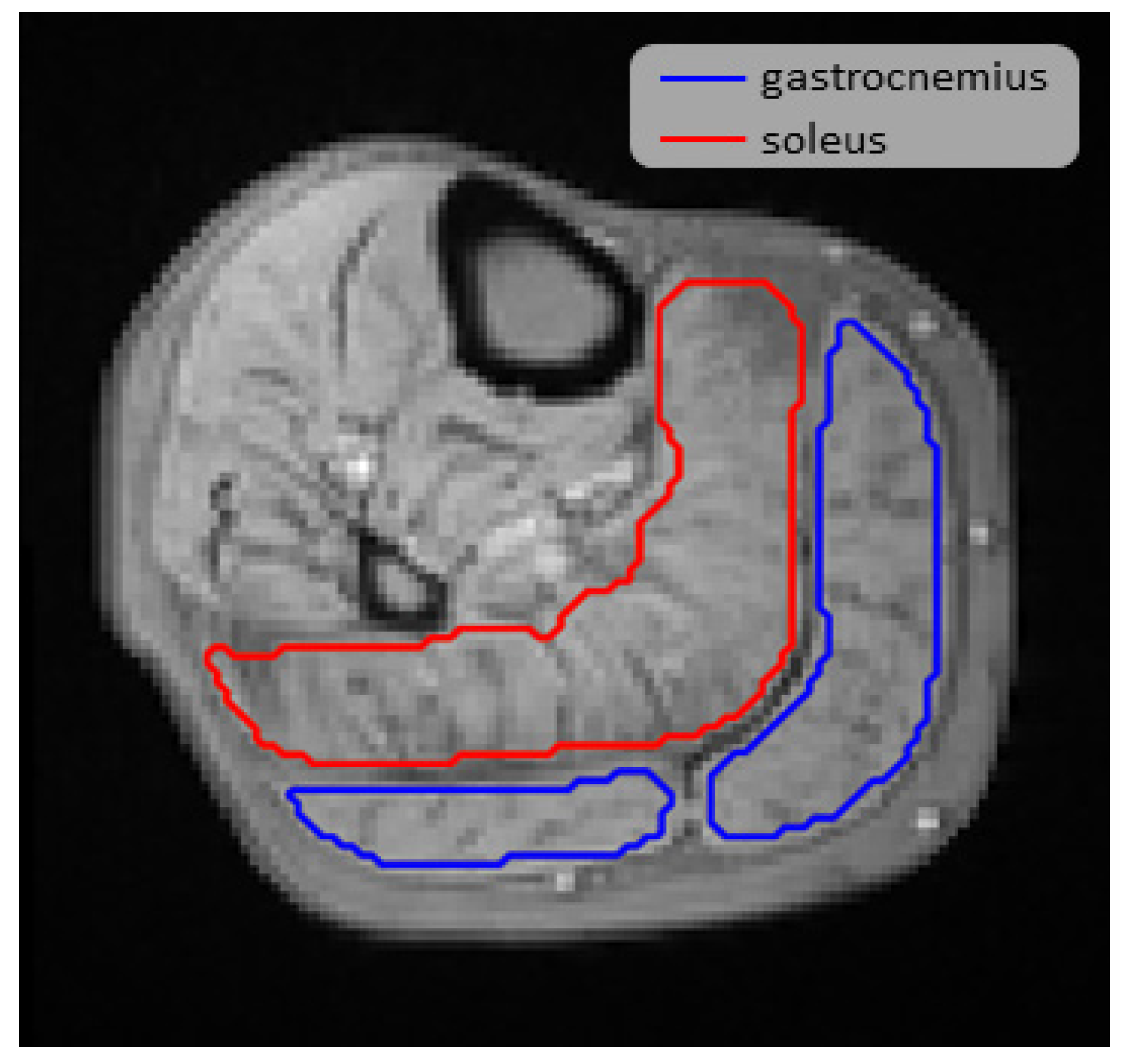

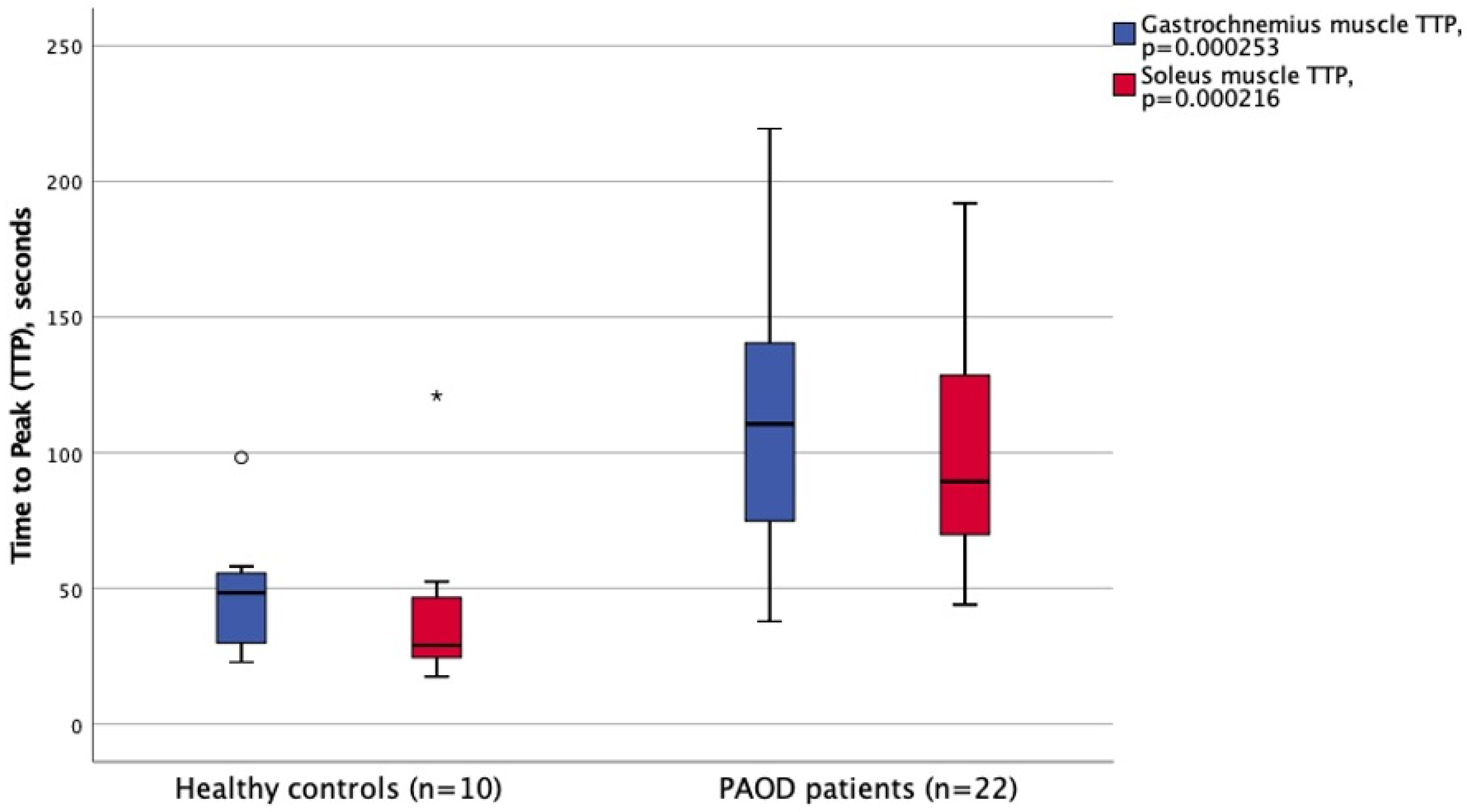
| Parameter | |
|---|---|
| Field-of-view (mm) | 160 × 160 |
| Acquisition matrix | 128 × 119 |
| TR (ms) | 44 |
| TE1–11 (ms) | 2–40 |
| Slice Thickness (mm) | 10 |
| Patients with PAOD (n = 22) | Controls without PAOD (n = 10) | |
|---|---|---|
| Male sex | 12 (55%) | 8 (80%) |
| Smoking: | ||
| active smoker | 2 (9%) | 1 (10%) |
| previous smoker | 17 (77%) | 4 (40%) |
| never smoked | 3 (14%) | 5 (50%) |
| Hypertension | 18 (82%) | 3 (30%) |
| Hyperlipidemia | 11 (50%) | 2 (20%) |
| Diabetes | 9 (41%) | 0 (0%) |
| Coronary artery disease | 11 (50%) | 0 (0%) |
| Chronic obstructive pulmonary disease | 6 (27%) | 0 (0%) |
| Venous insufficiency | 1 (5%) | 3 (30%) |
| Rutherford stage: | ||
| Stage 1 | 5 (23%) | N/A |
| Stage 2 | 14 (64%) | N/A |
| Stage 3 | 3 (14%) | N/A |
| Ankle-brachial index at rest, mean (SD) | 0.65 (0.23) | 1.13 (0.09) |
| BOLD-MRI Perfusion Parameter | Patients with PAOD (n = 22) | Controls without PAOD (n = 10) | p-Value |
|---|---|---|---|
| BL (ms) gastrocnemius | 25.3 ± 2.9 | 24.6 ± 4.1 | 0.59 |
| BL (ms) soleus | 21.2 ± 2.8 | 20.8 ± 3.9 | 0.91 |
| HPV (ms) gastrocnemius | 25.8 ± 2.5 | 26.1 ± 4.3 | 0.31 |
| HPV (ms) soleus | 22.8 ± 2.7 | 22.6 ± 4.3 | 0.96 |
| TTP (s) gastrocnemius | 111 ± 46 | 48 ± 22 | <0.01 |
| TTP (s) soleus | 100 ± 42 | 41 ± 30 | <0.01 |
| Overshoot (ms) gastrocnemius | 0.59 ± 0.93 | 1.49 ± 1.22 | 0.03 |
| Overshoot (ms) soleus | 1.59 ± 0.96 | 1.81 ± 1.86 | 0.46 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Törngren, K.; Eriksson, S.; Arvidsson, J.; Falkenberg, M.; Johnsson, Å.A.; Sjöberg, C.; Lagerstrand, K.; Nordanstig, J. A Reperfusion BOLD-MRI Tissue Perfusion Protocol Reliably Differentiate Patients with Peripheral Arterial Occlusive Disease from Healthy Controls. J. Clin. Med. 2021, 10, 3643. https://doi.org/10.3390/jcm10163643
Törngren K, Eriksson S, Arvidsson J, Falkenberg M, Johnsson ÅA, Sjöberg C, Lagerstrand K, Nordanstig J. A Reperfusion BOLD-MRI Tissue Perfusion Protocol Reliably Differentiate Patients with Peripheral Arterial Occlusive Disease from Healthy Controls. Journal of Clinical Medicine. 2021; 10(16):3643. https://doi.org/10.3390/jcm10163643
Chicago/Turabian StyleTörngren, Kristina, Stefanie Eriksson, Jonathan Arvidsson, Mårten Falkenberg, Åse A. Johnsson, Carl Sjöberg, Kerstin Lagerstrand, and Joakim Nordanstig. 2021. "A Reperfusion BOLD-MRI Tissue Perfusion Protocol Reliably Differentiate Patients with Peripheral Arterial Occlusive Disease from Healthy Controls" Journal of Clinical Medicine 10, no. 16: 3643. https://doi.org/10.3390/jcm10163643
APA StyleTörngren, K., Eriksson, S., Arvidsson, J., Falkenberg, M., Johnsson, Å. A., Sjöberg, C., Lagerstrand, K., & Nordanstig, J. (2021). A Reperfusion BOLD-MRI Tissue Perfusion Protocol Reliably Differentiate Patients with Peripheral Arterial Occlusive Disease from Healthy Controls. Journal of Clinical Medicine, 10(16), 3643. https://doi.org/10.3390/jcm10163643






