Prevention and Management of Complications and Education in Endoscopic Submucosal Dissection
Abstract
1. Introduction
Search Strategy
2. Critical Complications Associated with ESD
2.1. Bleeding after ESD
2.1.1. Drugs
2.1.2. Treatment of the Ulcer Base
2.1.3. Response to Antithrombotic Agents
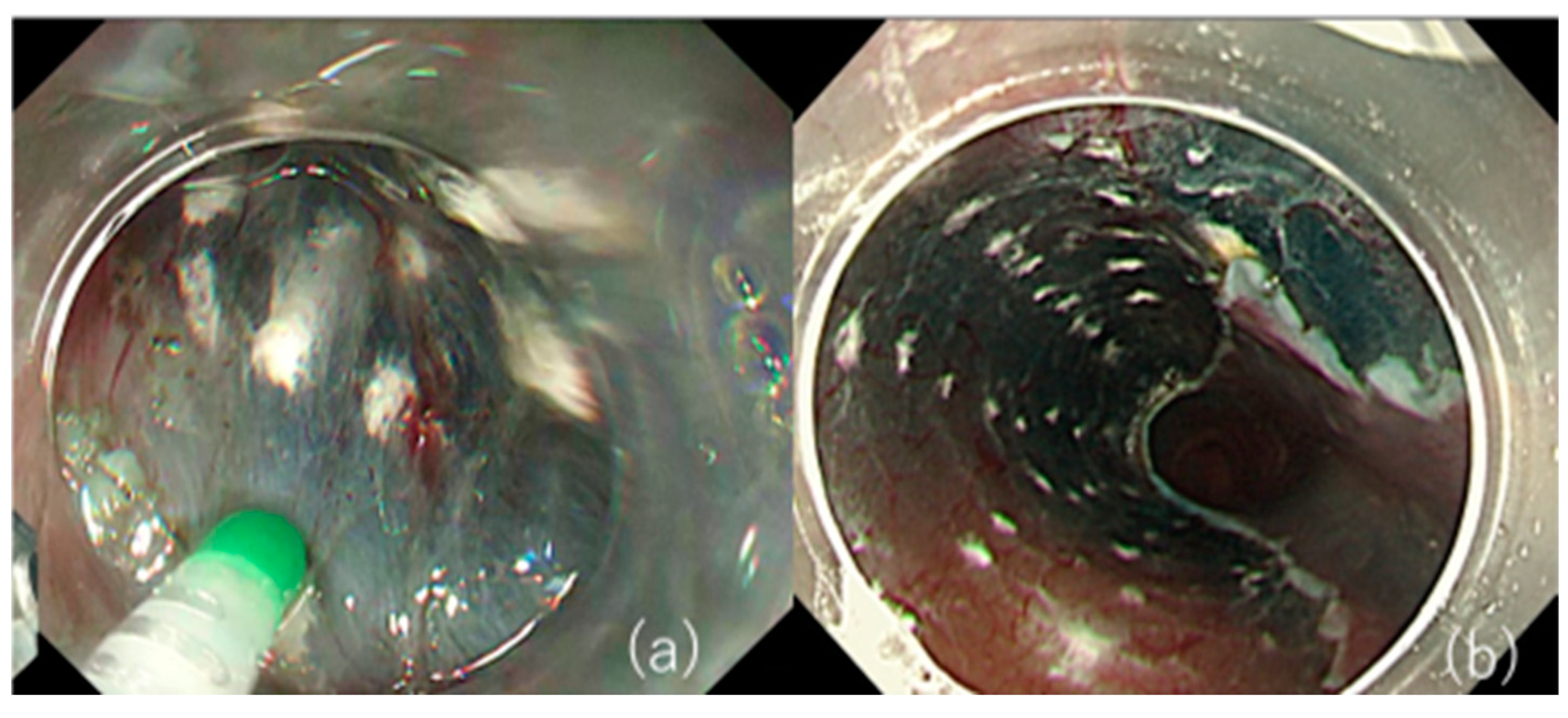
2.2. Perforation
2.3. Stenosis
2.4. Education on ESD Techniques
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gotoda, T.; Yamamoto, H.; Soetikno, R.M. Endoscopic Submucosal Dissection of Early Gastric Cancer. J. Gastroenterol. 2006, 41, 929–942. [Google Scholar] [CrossRef] [PubMed]
- Draganov, P.V.; Wang, A.Y.; Othman, M.O.; Fukami, N. AGA Institute Clinical Practice Update: Endoscopic Submucosal Dissection in the United States. Clin. Gastroenterol. Hepatol. 2019, 17, 16–25.e1. [Google Scholar] [CrossRef]
- Kobayashi, N.; Yoshitake, N.; Hirahara, Y.; Konishi, J.; Saito, Y.; Matsuda, T.; Ishikawa, T.; Sekiguchi, R.; Fujimori, T. Matched Case-Control Study Comparing Endoscopic Submucosal Dissection and Endoscopic Mucosal Resection for Colorectal Tumors. J. Gastroenterol. Hepatol. 2012, 27, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Kim, E.S.; Cho, K.B.; Park, K.S.; Jang, B.K.; Chung, W.J.; Hwang, J.S. Comparison of Clinical Outcomes among Different Endoscopic Resection Methods for Treating Colorectal Neoplasia. Dig. Dis. Sci. 2013, 58, 1727–1736. [Google Scholar] [CrossRef] [PubMed]
- Ono, S.; Fujishiro, M.; Niimi, K.; Goto, O.; Kodashima, S.; Yamamichi, N.; Omata, M. Predictors of Postoperative Stricture after Esophageal Endoscopic Submucosal Dissection for Superficial Squamous Cell Neoplasms. Endoscopy 2009, 41, 661–665. [Google Scholar] [CrossRef]
- Kataoka, Y.; Tsuji, Y.; Sakaguchi, Y.; Minatsuki, C.; Asada-Hirayama, I.; Niimi, K.; Ono, S.; Kodashima, S.; Yamamichi, N.; Fujishiro, M.; et al. Bleeding after endoscopic submucosal dissection: Risk factors and preventive methods. World J. Gastroenterol. 2016, 22, 5927–5935. [Google Scholar] [CrossRef]
- Yang, C.H.; Qiu, Y.; Li, X.; Shi, R.H. Bleeding after endoscopic submucosal dissection of gastric lesions. J. Dig. Dis. 2020, 21, 139–146. [Google Scholar] [CrossRef]
- Shibagaki, K.; Ishimura, N.; Kinoshita, Y. Endoscopic submucosal dissection for duodenal tumors. Ann. Transl. Med. 2017, 5, 188. [Google Scholar] [CrossRef] [PubMed]
- Libânio, D.; Costa, M.N.; Pimentel-Nunes, P.; Dinis-Ribeiro, M. Risk Factors for Bleeding after Gastric Endoscopic Submucosal Dissection: A Systematic Review and Meta-Analysis. Gastrointest. Endosc. 2016, 84, 572–586. [Google Scholar] [CrossRef]
- Green, F.W., Jr.; Kaplan, M.M.; Curtis, L.E.; Levine, P.H. Effect of Acid and Pepsin on Blood Coagulation and Platelet Aggregation. A Possible Contributor Prolonged Gastroduodenal Mucosal Hemorrhage. Gastroenterol. 1978, 74, 38–43. [Google Scholar] [CrossRef]
- Yang, Z.; Wu, Q.; Liu, Z.; Wu, K.; Fan, D. Proton Pump Inhibitors versus histamine-2-Receptor Antagonists for the Management of Iatrogenic Gastric Ulcer after Endoscopic Mucosal Resection or Endoscopic Submucosal Dissection: A Meta-Analysis of Randomized Trials. Digestion 2011, 84, 315–320. [Google Scholar] [CrossRef]
- Uedo, N.; Takeuchi, Y.; Yamada, T.; Ishihara, R.; Ogiyama, H.; Yamamoto, S.; Kato, M.; Tatsumi, K.; Masuda, E.; Tamai, C.; et al. Effect of a Proton Pump Inhibitor or an H2-Receptor Antagonist on Prevention of Bleeding from Ulcer after Endoscopic Submucosal Dissection of Early Gastric Cancer: A Prospective Randomized Controlled Trial. Am. J. Gastroenterol. 2007, 102, 1610–1616. [Google Scholar] [CrossRef]
- Kondo, M.; Kawamoto, M.; Hasuoka, A.; Kajino, M.; Inatomi, N.; Tarui, N. High-throughput screening of potassium-competitive acid blockers. J. Biomol. Screen. 2012, 17, 177–182. [Google Scholar] [CrossRef]
- Gao, H.; Li, L.; Zhang, C.; Tu, J.; Geng, X.; Wang, J.; Zhou, X.; Jing, J.; Pan, W. Comparison of efficacy of pharmacological therapies for gastric endoscopic submucosal dissection-induced ulcers: A systematic review and network meta-analysis. Expert Rev. Gastroenterol. Hepatol. 2020, 14, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Kakushima, N.; Hori, K.; Ono, H.; Horimatsu, T.; Uedo, N.; Ohata, K.; Doyama, H.; Kaneko, K.; Oda, I.; Hikichi, T.; et al. Proton Pump Inhibitor after Endoscopic Resection for Esophageal Squamous Cell Cancer: Multicenter Prospective Randomized Controlled Trial. J. Gastroenterol. 2016, 51, 104–111. [Google Scholar] [CrossRef]
- Choi, K.D.; Jung, H.Y.; Lee, G.H.; Oh, T.H.; Jo, J.Y.; Song, H.J.; Hong, S.S.; Kim, J.H. Application of Metal Hemoclips for Closure of Endoscopic Mucosal Resection-Induced Ulcers of the Stomach to Prevent Delayed Bleeding. Surg. Endosc. 2008, 22, 1882–1886. [Google Scholar] [CrossRef] [PubMed]
- Ogiyama, H.; Tsutsui, S.; Murayama, Y.; Maeda, S.; Satake, S.; Nasu, A.; Umeda, D.; Miura, Y.; Tominaga, K.; Horiki, M.; et al. Prophylactic Clip Closure May Reduce the Risk of Delayed Bleeding after Colorectal Endoscopic Submucosal Dissection. Endosc. Int. Open 2018, 6, E582–E588.e8. [Google Scholar] [CrossRef] [PubMed]
- Hoteya, S.; Kaise, M.; Iizuka, T.; Ogawa, O.; Mitani, T.; Matsui, A.; Kikuchi, D.; Furuhata, T.; Yamashita, S.; Yamada, A.; et al. Delayed Bleeding after Endoscopic Submucosal Dissection for Non-Ampullary Superficial Duodenal Neoplasias Might Be Prevented by Prophylactic Endoscopic Closure: Analysis of Risk Factors. Dig. Endosc. 2015, 27, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, T.; Suzuki, H.; Goto, O.; Ogata, H.; Kanai, T.; Yahagi, N. Effect of prophylactic clipping in colorectal endoscopic resection: A meta-analysis of randomized controlled studies. United Eur. Gastroenterol. J. 2017, 5, 859–867. [Google Scholar] [CrossRef] [PubMed]
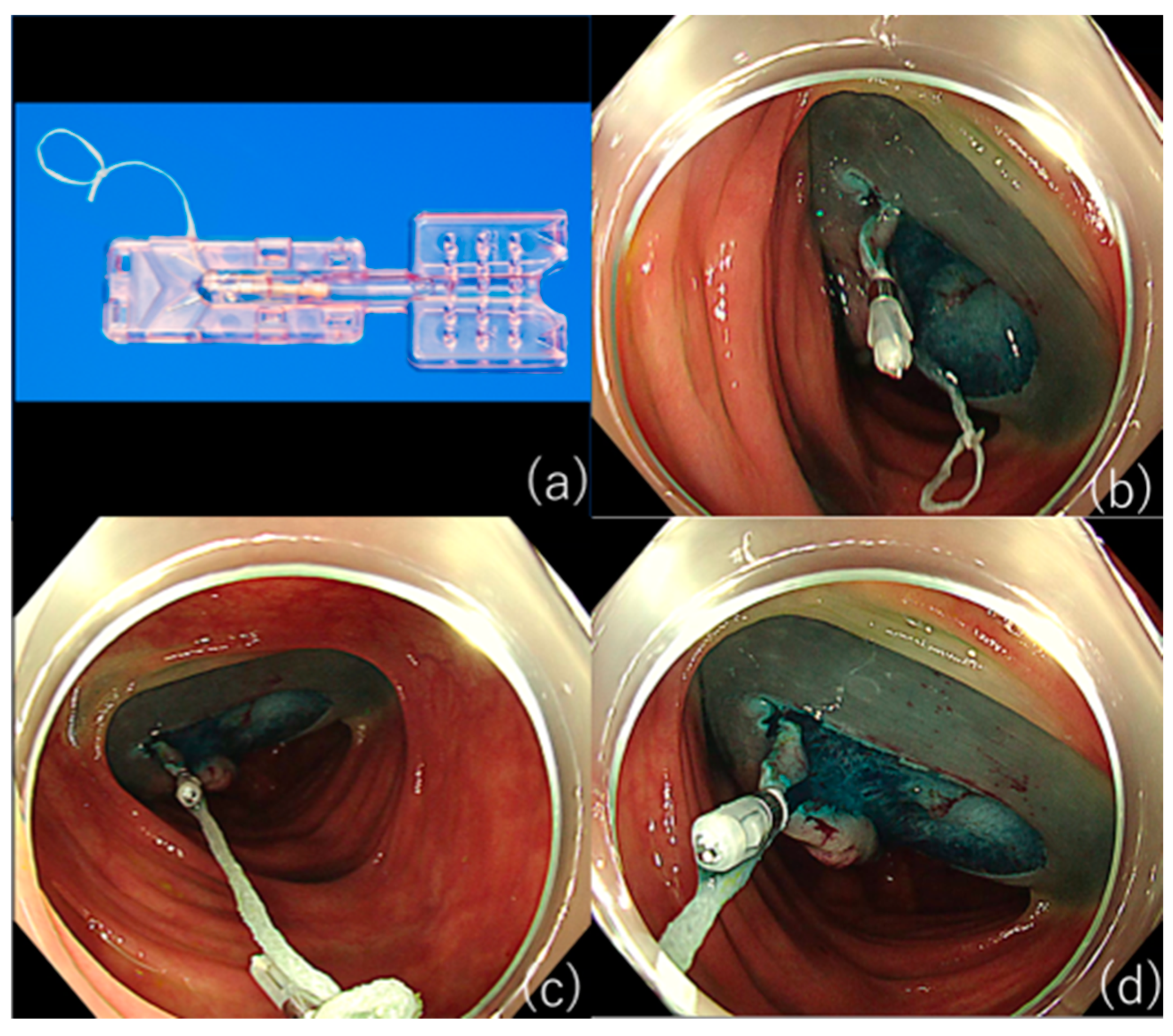
- Otake, Y.; Saito, Y.; Sakamoto, T.; Aoki, T.; Nakajima, T.; Toyoshima, N.; Matsuda, T.; Ono, H. New Closure Technique for Large Mucosal Defects after Endoscopic Submucosal Dissection of Colorectal Tumors (with Video). Gastrointest. Endosc. 2012, 75, 663–667. [Google Scholar] [CrossRef]
- Abe, S.; Oda, I.; Mori, G.; Nonaka, S.; Suzuki, H.; Yoshianaga, S.; Saito, Y. Complete Endoscopic Closure of a Large Gastric Defect with Endoloop and Endoclips after Complex Endoscopic Submucosal Dissection. Endoscopy 2015, 47 (Suppl. 1), E374–E375. [Google Scholar] [CrossRef]
- Kantsevoy, S.V.; Bitner, M.; Mitrakov, A.A.; Thuluvath, P.J. Endoscopic Suturing Closure of Large Mucosal Defects after Endoscopic Submucosal Dissection Is Technically Feasible, Fast, and Eliminates the Need for Hospitalization (with Videos). Gastrointest. Endosc. 2014, 79, 503–507. [Google Scholar] [CrossRef]
- Voermans, R.P.; Le Moine, O.; von Renteln, D.; Ponchon, T.; Giovannini, M.; Bruno, M.; Weusten, B.; Seewald, S.; Costamagna, G.; Deprez, P.; et al. Efficacy of Endoscopic Closure of Acute Perforations of the Gastrointestinal Tract. Clin. Gastroenterol. Hepatol. 2012, 10, 603–608. [Google Scholar] [CrossRef]
- Akimoto, T.; Goto, O.; Sasaki, M.; Mizutani, M.; Tsutsumi, K.; Kiguchi, Y.; Nakayama, A.; Kato, M.; Fujimoto, A.; Ochiai, Y.; et al. Endoscopic Suturing Promotes Healing of Mucosal Defects after Gastric Endoscopic Submucosal Dissection: Endoscopic and Histologic Analyses in Vivo Porcine Models (with Video). Gastrointest. Endosc. 2020, 91, 1172–1182. [Google Scholar] [CrossRef]
- Tsuji, Y.; Fujishiro, M.; Kodashima, S.; Ono, S.; Niimi, K.; Mochizuki, S.; Asada-Hirayama, I.; Matsuda, R.; Minatsuki, C.; Nakayama, C.; et al. Polyglycolic Acid Sheets and Fibrin Glue Decrease the Risk of Bleeding after Endoscopic Submucosal Dissection of Gastric Neoplasms (with Video). Gastrointest. Endosc. 2015, 81, 906–912. [Google Scholar] [CrossRef]
- Kawata, N.; Ono, H.; Takizawa, K.; Kakushima, N.; Tanaka, M.; Igarashi, K.; Yoshida, M.; Kishida, Y.; Iwai, T.; Ito, S.; et al. Efficacy of Polyglycolic Acid Sheets and Fibrin Glue for Prevention of Bleeding after Gastric Endoscopic Submucosal Dissection in Patients under Continued Antithrombotic Agents. Gastric Cancer 2018, 21, 696–702. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, K.; Fujishiro, M.; Kato, M.; Higuchi, K.; Iwakiri, R.; Sakamoto, C.; Uchiyama, S.; Kashiwagi, A.; Ogawa, H.; Murakami, K.; et al. Guidelines for Gastroenterological Endoscopy in Patients Undergoing Antithrombotic Treatment. Dig. Endosc. 2014, 26, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Uedo, N.; Hokimoto, S.; Ieko, M.; Higuchi, K.; Murakami, K.; Fujimoto, K. Guidelines for Gastroenterological Endoscopy in Patients Undergoing Antithrombotic Treatment: 2017 Appendix on Anticoagulants Including Direct Oral Anticoagulants. Dig. Endosc. 2018, 30, 433–440. [Google Scholar] [CrossRef]
- Odagiri, H.; Yasunaga, H. Complications Following Endoscopic Submucosal Dissection for Gastric, Esophageal, and Colorectal Cancer: A Review of Studies Based on Nationwide Large-Scale Databases. Ann. Transl. Med. 2017, 5, 189. [Google Scholar] [CrossRef]
- Hu, J.; Zhao, Y.; Ren, M.; Li, Y.; Lu, X.; Lu, G.; Zhang, D.; Chu, D.; He, S. The Comparison between Endoscopic Submucosal Dissection and Surgery in Gastric Cancer: A Systematic Review and Meta-Analysis. Gastroenterol. Res. Pract. 2018, 2018, 4378945. [Google Scholar] [CrossRef] [PubMed]
- De Ceglie, A.; Hassan, C.; Mangiavillano, B.; Matsuda, T.; Saito, Y.; Ridola, L.; Bhandari, P.; Boeri, F.; Conio, M. Endoscopic Mucosal Resection and Endoscopic Submucosal Dissection for Colorectal Lesions: A Systematic Review. Crit. Rev. Oncol. Hematol. 2016, 104, 138–155. [Google Scholar] [CrossRef]
- Tsuji, K.; Yoshida, N.; Nakanishi, H.; Takemura, K.; Yamada, S.; Doyama, H. Recent Traction Methods for Endoscopic Submucosal Dissection. World J. Gastroenterol. 2016, 22, 5917–5926. [Google Scholar] [CrossRef]
- Oyama, T. Counter Traction Makes Endoscopic Submucosal Dissection Easier. Clin. Endosc. 2012, 45, 375–378. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, Y.; Takeuchi, Y.; Uedo, N.; Kanesaka, T.; Kato, M.; Hamada, K.; Tonai, Y.; Matsuura, N.; Akasaka, T.; Hanaoka, N.; et al. Efficacy of Traction-Assisted Colorectal Endoscopic Submucosal Dissection Using a Clip-and-Thread Technique: A Prospective Randomized Study. Dig. Endosc. 2018, 30, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, N.; Osada, T.; Shibuya, T.; Beppu, K.; Matsumoto, K.; Mori, H.; Kawabe, M.; Nagahara, A.; Otaka, M.; Ogihara, T.; et al. Endoscopic Submucosal Dissection of Large Colorectal Tumors by Using a Novel Spring-Action S-O Clip for Traction (with Video). Gastrointest. Endosc. 2009, 69, 1370–1374. [Google Scholar] [CrossRef] [PubMed]
- Mori, H.; Kobara, H.; Nishiyama, N.; Fujihara, S.; Matsunaga, T.; Masaki, T. Novel Effective and Repeatedly Available Ring-Thread Counter Traction for Safer Colorectal Endoscopic Submucosal Dissection. Surg. Endosc. 2017, 31, 3040–3047. [Google Scholar] [CrossRef] [PubMed]
- Haito-Chavez, Y.; Law, J.K.; Kratt, T.; Arezzo, A.; Verra, M.; Morino, M.; Sharaiha, R.Z.; Poley, J.W.; Kahaleh, M.; Thompson, C.C.; et al. International Multicenter Experience with an Over-the-Scope Clipping Device for Endoscopic Management of GI Defects (with Video). Gastrointest. Endosc. 2014, 80, 610–622. [Google Scholar] [CrossRef] [PubMed]
- Tsunada, S.; Ogata, S.; Mannen, K.; Arima, S.; Sakata, Y.; Shiraishi, R.; Shimoda, R.; Ootani, H.; Yamaguchi, K.; Fujise, T.; et al. Case Series of Endoscopic Balloon Dilation to Treat a Stricture Caused by Circumferential Resection of the Gastric Antrum by Endoscopic Submucosal Dissection. Gastrointest. Endosc. 2008, 67, 979–983. [Google Scholar] [CrossRef] [PubMed]
- Iizuka, H.; Kakizaki, S.; Sohara, N.; Onozato, Y.; Ishihara, H.; Okamura, S.; Itoh, H.; Mori, M. Stricture after Endoscopic Submucosal Dissection for Early Gastric Cancers and Adenomas. Dig. Endosc. 2010, 22, 282–288. [Google Scholar] [CrossRef]
- Hayashi, T.; Kudo, S.E.; Miyachi, H.; Sakurai, T.; Ishigaki, T.; Yagawa, Y.; Toyoshima, N.; Mori, Y.; Misawa, M.; Kudo, T.; et al. Management and Risk Factor of Stenosis after Endoscopic Submucosal Dissection for Colorectal Neoplasms. Gastrointest. Endosc. 2017, 86, 358–369. [Google Scholar] [CrossRef]
- Martínek, J.; Juhas, S.; Dolezel, R.; Walterová, B.; Juhasova, J.; Klima, J.; Rabekova, Z.; Vacková, Z. Prevention of Esophageal Strictures after Circumferential Endoscopic Submucosal Dissection. Minerva Chir. 2018, 73, 394–409. [Google Scholar]
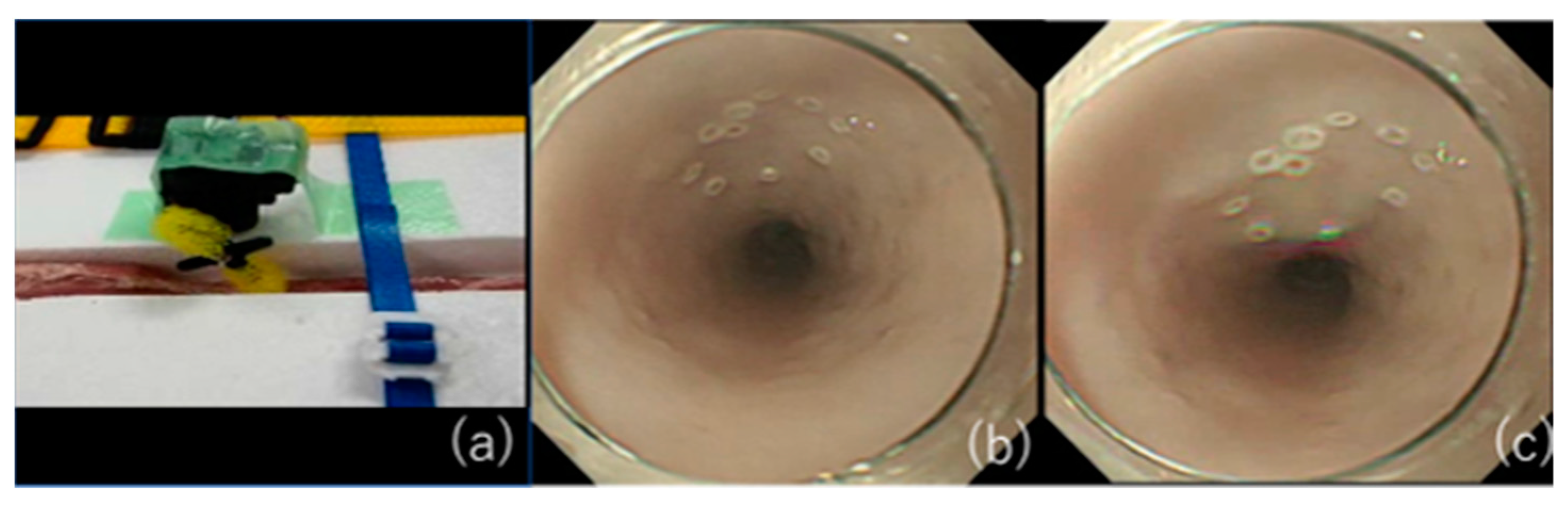
- Nonaka, K.; Miyazawa, M.; Ban, S.; Aikawa, M.; Akimoto, N.; Koyama, I.; Kita, H. Different Healing Process of Esophageal Large Mucosal Defects by Endoscopic Mucosal Dissection between with and without Steroid Injection in an Animal Model. BMC Gastroenterol. 2013, 13, 72. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, K.; Ban, S.; Ryozawa, S. Strictures after Endoscopic Submucosal Dissection of the Esophagus: Are the Histopathological Findings the Same between Human and Porcine Models? Dig. Endosc. 2019, 31, 106. [Google Scholar] [CrossRef]
- Hinz, B.; Phan, S.H.; Thannickal, V.J.; Galli, A.; Bochaton-Piallat, M.L.; Gabbiani, G. The Myofibroblast: One Function, Multiple Origins. Am. J. Pathol. 2007, 170, 1807–1816. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, S.; Kato, M.; Fujimoto, A.; Maehata, T.; Sasaki, M.; Inoshita, N.; Sato, H.; Suzuki, K.; Yahagi, N. Inadequate Steroid Injection after Esophageal ESD Might Cause Mural Necrosis. Endosc. Int. Open 2019, 7, E115–E121.e21. [Google Scholar] [CrossRef]
- Shumeiko, O.; Yoshida, M.; Ono, H. Safe Method of Steroid Injection Following Esophageal Endoscopic Submucosal Dissection for Postoperative Stricture Prevention. Dig. Endosc. 2019, 31, e118–e119. [Google Scholar] [CrossRef] [PubMed]
- Misumi, Y.; Nonaka, K.; Kishino, M. Safer Local Injection of Triamcinolone Acetonide for Preventing Post-Endoscopic Submucosal Dissection Esophageal Stenosis. Dig. Endosc. 2021, 33, e77–e78. [Google Scholar] [CrossRef]
- Yano, T.; Haro, A.; Shikada, Y.; Okamoto, T.; Maruyama, R.; Maehara, Y. A Unique Method for Repairing Intraoperative Pulmonary Air Leakage with Both Polyglycolic Acid Sheets and Fibrin Glue. World J. Surg. 2012, 36, 463–467. [Google Scholar] [CrossRef]
- Maruyama, A.; Tsunoda, A.; Takahashi, M.; Kishimoto, S.; Suzuki, M. Nasopharyngeal Pleomorphic Adenoma Presenting as Otitis Media with Effusion: Case Report and Literature Review. Am. J. Otolaryngol. 2014, 35, 73–76. [Google Scholar] [CrossRef]
- Iizuka, T.; Kikuchi, D.; Yamada, A.; Hoteya, S.; Kajiyama, Y.; Kaise, M. Polyglycolic Acid Sheet Application to Prevent Esophageal Stricture after Endoscopic Submucosal Dissection for Esophageal Squamous Cell Carcinoma. Endoscopy 2015, 47, 341–344. [Google Scholar] [CrossRef]
- Ohki, T.; Yamato, M.; Ota, M.; Takagi, R.; Murakami, D.; Kondo, M.; Sasaki, R.; Namiki, H.; Okano, T.; Yamamoto, M. Prevention of Esophageal Stricture after Endoscopic Submucosal Dissection Using Tissue-Engineered Cell Sheets. Gastroenterology 2012, 143, 582–588.e2. [Google Scholar] [CrossRef] [PubMed]
- Deprez, P.H.; Bergman, J.J.; Meisner, S.; Ponchon, T.; Repici, A.; Dinis-Ribeiro, M.; Haringsma, J. Current Practice with Endoscopic Submucosal Dissection in Europe: Position Statement from a Panel of Experts. Endoscopy 2010, 42, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Casamayor, M.; Morlock, R.; Maeda, H.; Ajani, J. Targeted Literature Review of the Global Burden of Gastric Cancer. Ecancermedicalscience 2018, 12, 883. [Google Scholar] [CrossRef] [PubMed]
- Ohata, K.; Fu, K.; Sakai, E.; Nonaka, K.; Tashima, T.; Minato, Y.; Ohno, A.; Ito, T.; Tsuji, Y.; Chiba, H.; et al. Esophageal Endoscopic Submucosal Dissection Assisted by an Overtube with a Traction Forceps: An Animal Study. Gastroenterol. Res. Pract. 2016, 2016, 3186168. [Google Scholar] [CrossRef]
- Tanimoto, M.A.; Torres-Villalobos, G.; Fujita, R.; Santillan-Doherty, P.; Albores-Saavedra, J.; Chable-Montero, F.; Martin-Del-Campo, L.A.; Vasquez, L.; Bravo-Reyna, C.; Villanueva, O.; et al. Learning Curve in a Western Training Center of the Circumferential en Bloc Esophageal Endoscopic Submucosal Dissection in an In Vivo Animal Model. Diagn. Ther. Endosc. 2011, 2011, 847831. [Google Scholar] [CrossRef] [PubMed]
- Ohata, K.; Nonaka, K.; Misumi, Y.; Tsunashima, H.; Takita, M.; Minato, Y.; Tashima, T.; Sakai, E.; Muramoto, T.; Matsuyama, Y.; et al. Usefulness of Training Using Animal Models for Colorectal Endoscopic Submucosal Dissection: Is Experience Performing Gastric ESD Really Needed? Endosc. Int. Open 2016, 4, E333–E339. [Google Scholar] [CrossRef] [PubMed]
- Misumi, Y.; Nonaka, K.; Kishino, M. Development of an Animal Model That Reproduces Pulsations for Training in Esophageal Endoscopic Submucosal Dissection. Endoscopy 2020. [Google Scholar] [CrossRef]
- Yoshida, N.; Yagi, N.; Inada, Y.; Kugai, M.; Kamada, K.; Katada, K.; Uchiyama, K.; Ishikawa, T.; Takagi, T.; Handa, O.; et al. Possibility of ex vivo animal training model for colorectal endoscopic submucosal dissection. Int. J. Colorectal. Dis. 2013, 28, 49–56. [Google Scholar] [CrossRef]
- Shinozaki, K.; Misumi, Y.; Nonaka, K. Digestive Endoscopic Submucosal Dissection Training Using Konjac. Dig. Endosc. 2021. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, K.; Sato, H.; Hashimoto, S.; Sato, Y.; Terai, S. A Novel Training Model Composed of Nonbiological Materials for Endoscopic Submucosal Dissection. Gastrointest. Endosc. 2016, 84, 373–374. [Google Scholar] [CrossRef] [PubMed]



| Standard Endoscopy | Biopsy | Low Risk of Bleeding | High Risk of Bleeding | |
|---|---|---|---|---|
| Aspirin | ◎ | ○ | ○ | ○or withdraw for 3–5 days |
| Thienopyridine | ◎ | ○ | ○ | ASA/CLZ replacement or withdraw for 5–7 days |
| Antiplatelet agent other than thienopyridine | ◎ | ○ | ○ | withdraw for 1 day |
| Warfarin | ◎ | ○ therapeutic range | ○ therapeutic range | ○therapeutic range or heparin replacement or temporary change in DOAC |
| DOAC | ◎ | ○ | ○ | withdraw on the day of treatment or heparin replacement |
| Aspirin | Thienopyridine | Antiplatelet Agent Other than Thienopyridine | Warfarin | DOAC |
|---|---|---|---|---|
| ○or CLZ replacement | withdraw for 5–7 days | |||
| ○or CLZ replacement | withdraw for 1 day | |||
| ○or CLZ replacement | ○therapeutic range or heparin replacement or temporary change in DOAC | |||
| ○or CLZ replacement | withdraw on the day of treatment | |||
| ASA/CLZ replacement | withdraw for 1 day | |||
| ASA/CLZ replacement | ○therapeutic range or heparin replacement or temporary change in DOAC | |||
| ASA/CLZ replacement | withdraw on the day of treatment | |||
| maintain CLZ or withdraw for 1day | ○therapeutic range or heparin replacement or temporary change in DOAC | |||
| maintain CLZ or withdraw for 1day | withdraw on the day of treatment |
| Aspirin | Thienopyridine | Antiplatelet Agent Other than Thienopyridine | Warfarin | DOAC |
|---|---|---|---|---|
| ○or CLZ replacement | withdraw for 5–7 days | ○therapeutic range or heparin replacement or temporary change in DOAC | ||
| ○or CLZ replacement | withdraw for 5–7 days | withdraw on the day of treatment | ||
| ○or CLZ replacement | Withdraw for 1 day | ○therapeutic range or heparin replacement or temporary change in DOAC | ||
| ○or CLZ replacement | Withdraw for 1 day | withdraw on the day of treatment | ||
| ASA/CLZ replacement | withdraw for 1 day | ○therapeutic range or heparin replacement or temporary change in DOAC | ||
| ASA/CLZ replacement | withdraw for 1 day | withdraw on the day of treatment |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Misumi, Y.; Nonaka, K. Prevention and Management of Complications and Education in Endoscopic Submucosal Dissection. J. Clin. Med. 2021, 10, 2511. https://doi.org/10.3390/jcm10112511
Misumi Y, Nonaka K. Prevention and Management of Complications and Education in Endoscopic Submucosal Dissection. Journal of Clinical Medicine. 2021; 10(11):2511. https://doi.org/10.3390/jcm10112511
Chicago/Turabian StyleMisumi, Yoshitsugu, and Kouichi Nonaka. 2021. "Prevention and Management of Complications and Education in Endoscopic Submucosal Dissection" Journal of Clinical Medicine 10, no. 11: 2511. https://doi.org/10.3390/jcm10112511
APA StyleMisumi, Y., & Nonaka, K. (2021). Prevention and Management of Complications and Education in Endoscopic Submucosal Dissection. Journal of Clinical Medicine, 10(11), 2511. https://doi.org/10.3390/jcm10112511





