Incidence of Immune-Mediated Pseudoprogression of Lymphoma Treated with Immune Checkpoint Inhibitors: Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Inclusion and Exclusion Criteria
- Population: studies that included six or more patients with lymphoma treated with ICIs;
- Reference standard: studies that used tumor response criteria based on imaging;
- Study design: clinical trials and observational studies (i.e., retrospective or prospective);
- Outcomes: results were reported in sufficient detail to evaluate the incidence of pseudoprogression among patients treated with ICIs.
- Case reports and series with sample sizes of less than five patients and studies;
- Review articles, editorials, letters, comments, and conference proceedings;
- Studies with topics that deviated from the incidence of pseudoprogression in lymphoma;
- Studies with insufficient data for evaluating the incidence of pseudoprogression.
2.3. Quality Assessment
2.4. Data Extraction
2.5. Statistical Analysis
3. Results
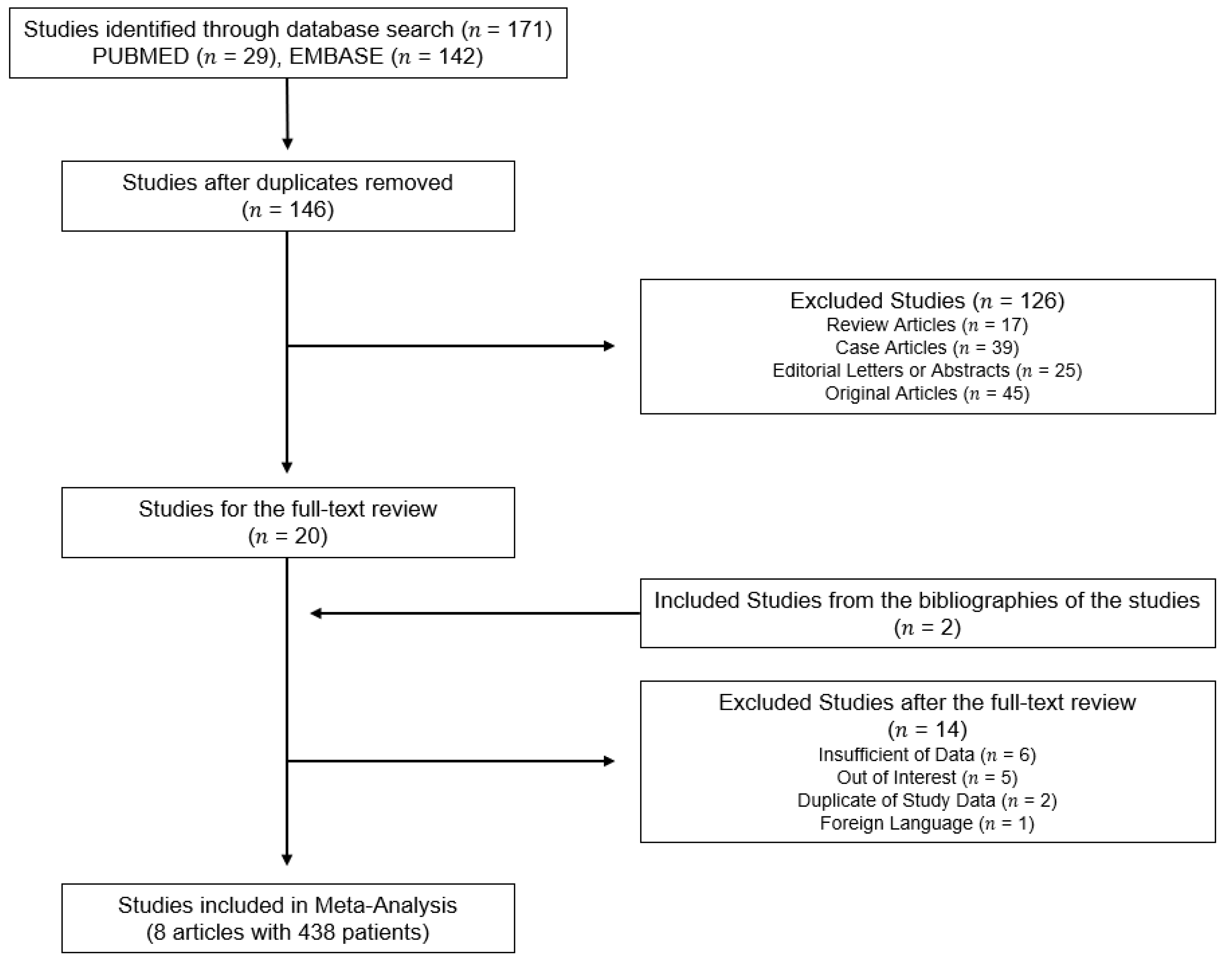
3.1. Literature Search
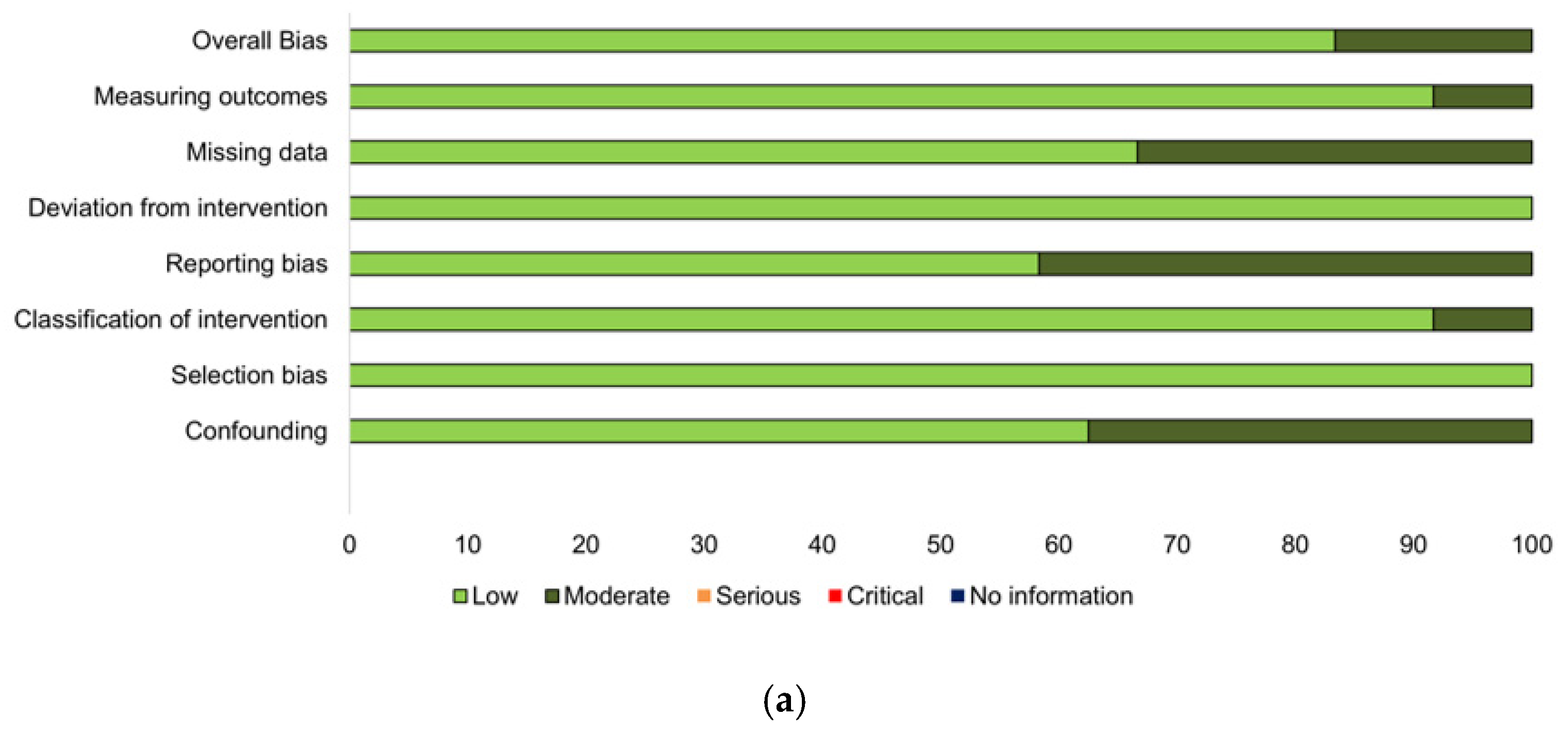
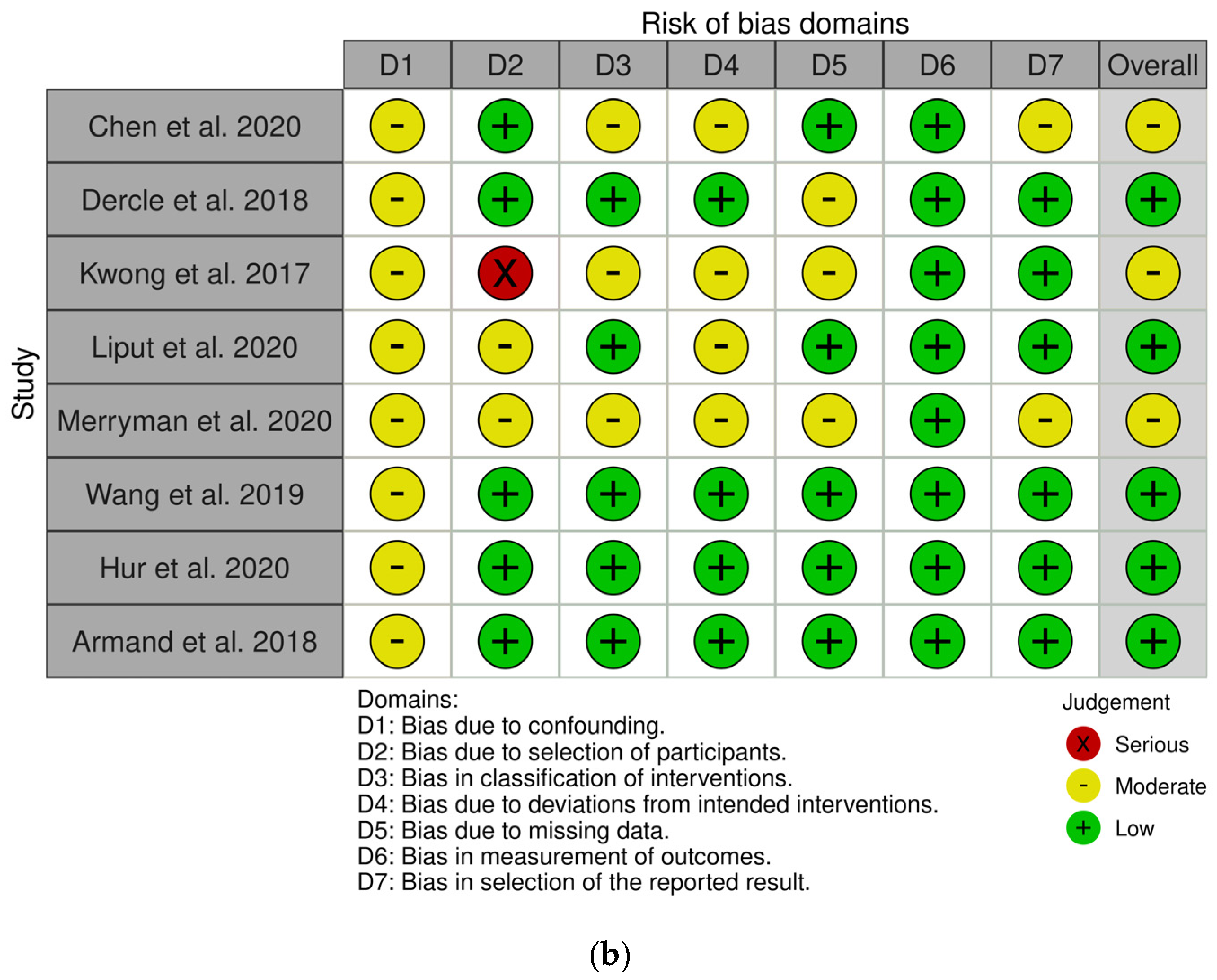
3.2. Study Characteristics and Quality Assessment
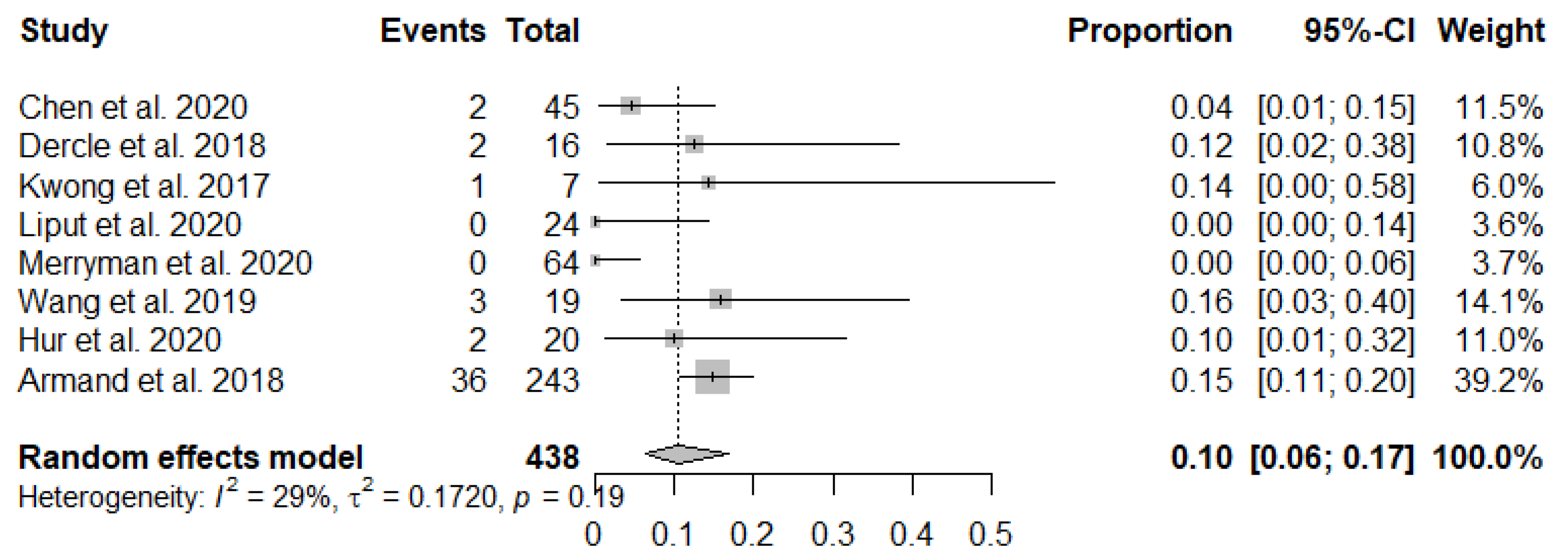
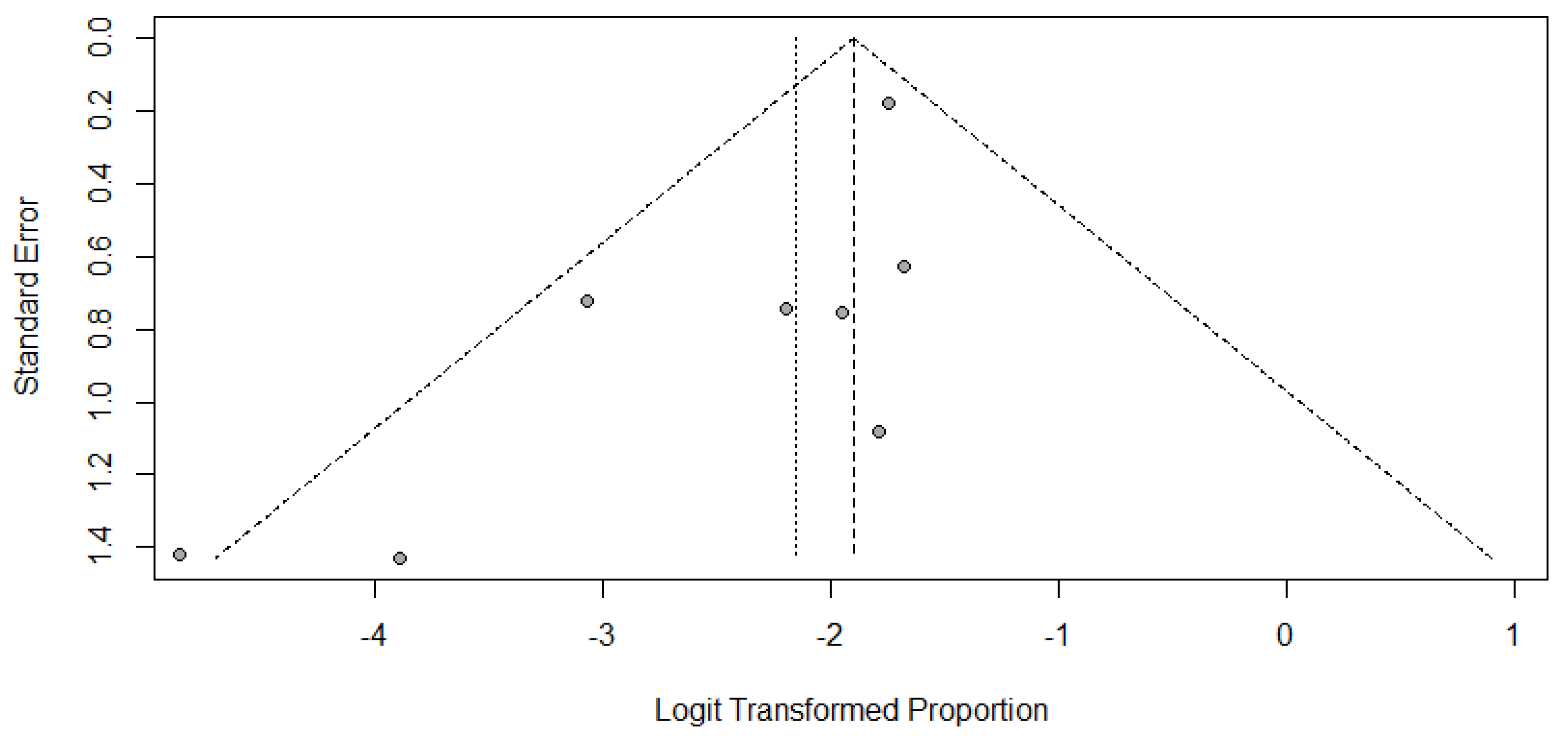
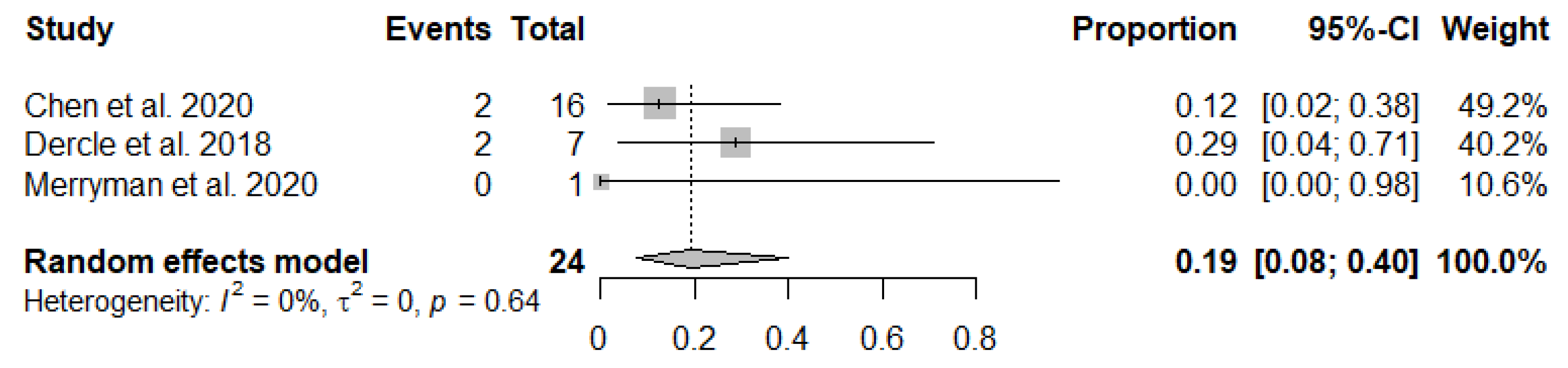
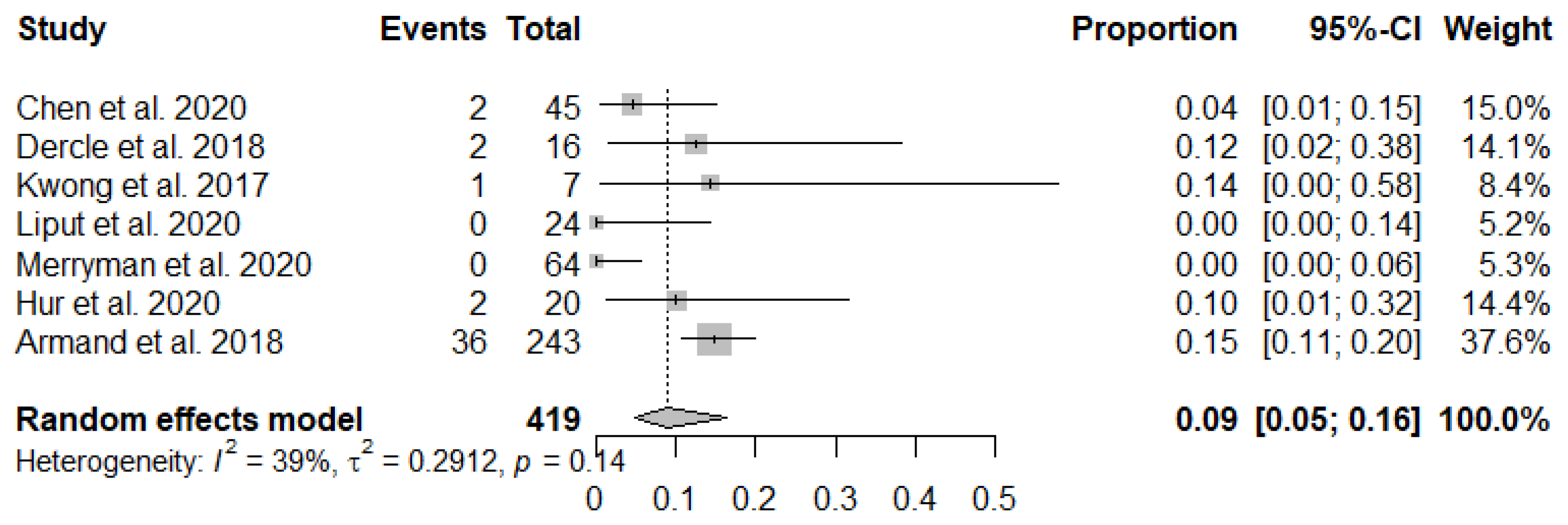
3.3. Meta-Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Galligan, B.M.; Tsao-Wei, D.; Groshen, S.; Kirschabum, M.; O’Donnell, R.; Kaesberg, P.R.; Siddiqui, T.; Popplewell, L.; Sikander, A.; Myo, H.; et al. Efficacy and safety of combined rituximab and ipilimumab to treat patients with relapsed/refractory CD20+ B-cell lymphoma. Blood 2015, 126, 3977. [Google Scholar] [CrossRef]
- Lang, D.; Wahl, G.; Poier, N.; Graf, S.; Kiesl, D.; Lamprecht, B.; Gabriel, M. Impact of PET/CT for Assessing Response to Immunotherapy-A Clinical Perspective. J. Clin. Med. 2020, 9, 3483. [Google Scholar] [CrossRef] [PubMed]
- Lepik, K.V.; Mikhailova, N.B.; Moiseev, I.S.; Kondakova, E.V.; Tsvetkova, L.A.; Zalyalov, Y.R.; Borzenkova, E.S.; Babenko, E.V.; Baykov, V.V.; Markova, I.V.; et al. Nivolumab for the treatment of relapsed and refractory classical Hodgkin lymphoma after ASCT and in ASCT-naive patients. Leuk Lymphoma 2019, 60, 2316–2319. [Google Scholar] [CrossRef] [PubMed]
- Lunning, M.A.; Moskowitz, A.J.; Horwitz, S. Strategies for relapsed peripheral T-cell lymphoma: The tail that wags the curve. J. Clin. Oncol. 2013, 31, 1922–1927. [Google Scholar] [CrossRef][Green Version]
- Seymour, L.; Bogaerts, J.; Perrone, A.; Ford, R.; Schwartz, L.H.; Mandrekar, S.; Lin, N.U.; Litière, S.; Dancey, J.; Chen, A.; et al. iRECIST: Guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol. 2017, 18, e143–e152. [Google Scholar] [CrossRef]
- Park, H.J.; Kim, K.W.; Pyo, J.; Suh, C.H.; Yoon, S.; Hatabu, H.; Nishino, M. Incidence of Pseudoprogression during Immune Checkpoint Inhibitor Therapy for Solid Tumors: A Systematic Review and Meta-Analysis. Radiology 2020, 297, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Cheson, B.D.; Ansell, S.; Schwartz, L.; Gordon, L.I.; Advani, R.; Jacene, H.A.; Hoos, A.; Barrington, S.F.; Armand, P. Refinement of the Lugano Classification lymphoma response criteria in the era of immunomodulatory therapy. Blood 2016, 128, 2489–2496. [Google Scholar] [CrossRef]
- Cheson, B.D.; Fisher, R.I.; Barrington, S.F.; Cavalli, F.; Schwartz, L.H.; Zucca, E.; Lister, T.A. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: The Lugano classification. J. Clin. Oncol. 2014, 32, 3059–3068. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef] [PubMed]
- Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J. Stat. Softw. 2010, 36, 1–48. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Kim, K.W.; Lee, J.; Choi, S.H.; Huh, J.; Park, S.H. Systematic Review and Meta-Analysis of Studies Evaluating Diagnostic Test Accuracy: A Practical Review for Clinical Researchers-Part I. General Guidance and Tips. Korean J. Radiol. 2015, 16, 1175–1187. [Google Scholar] [CrossRef]
- Lee, J.; Kim, K.W.; Choi, S.H.; Huh, J.; Park, S.H. Systematic Review and Meta-Analysis of Studies Evaluating Diagnostic Test Accuracy: A Practical Review for Clinical Researchers-Part II. Statistical Methods of Meta-Analysis. Korean J. Radiol. 2015, 16, 1188–1196. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Mokrane, F.Z.; Schwartz, L.H.; Morschhauser, F.; Stamatoullas, A.; Schiano de Colella, J.M.; Vercellino, L.; Casasnovas, O.; Chauchet, A.; Delmer, A.; et al. Early 18F-FDG PET/CT Response Predicts Survival in Relapsed or Refractory Hodgkin Lymphoma Treated with Nivolumab. J. Nucl. Med.: Off. Publ. Soc. Nucl. Med. 2020, 61, 649–654. [Google Scholar] [CrossRef] [PubMed]
- Dercle, L.; Seban, R.D.; Lazarovici, J.; Schwartz, L.H.; Houot, R.; Ammari, S.; Danu, A.; Edeline, V.; Marabelle, A.; Ribrag, V.; et al. (18)F-FDG PET and CT Scans Detect New Imaging Patterns of Response and Progression in Patients with Hodgkin Lymphoma Treated by Anti-Programmed Death 1 Immune Checkpoint Inhibitor. J. Nucl Med. 2018, 59, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Merryman, R.W.; Carreau, N.A.; Advani, R.H.; Spinner, M.A.; Herrera, A.F.; Chen, R.; Tomassetti, S.; Ramchandren, R.; Hamid, M.; Assouline, S.; et al. Impact of Treatment Beyond Progression with Immune Checkpoint Blockade in Hodgkin Lymphoma. Oncologist 2020, 25, e993–e997. [Google Scholar] [CrossRef]
- Wang, J.; Hu, Y.; Yang, S.; Wei, G.; Zhao, X.; Wu, W.; Zhang, Y.; Zhang, Y.; Chen, D.; Wu, Z.; et al. Role of Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography in Predicting the Adverse Effects of Chimeric Antigen Receptor T Cell Therapy in Patients with Non-Hodgkin Lymphoma. Biol. Blood Marrow Transplant. 2019, 25, 1092–1098. [Google Scholar] [CrossRef]
- Kwong, Y.L.; Chan, T.S.Y.; Tan, D.; Kim, S.J.; Poon, L.M.; Mow, B.; Khong, P.L.; Loong, F.; Au-Yeung, R.; Iqbal, J.; et al. PD1 blockade with pembrolizumab is highly effective in relapsed or refractory NK/T-cell lymphoma failing L-asparaginase. Blood 2017, 129, 2437–2442. [Google Scholar] [CrossRef] [PubMed]
- Liput, J.; Guler, E.; Smith, D.A.; Tirumani, S.H.; Hoimes, C.; Caimi, P.F.; Ramaiya, N.H. Clinical, Imaging Findings, Responses, and Outcomes of Patients With Classical Hodgkin Lymphoma and Non-Hodgkin Lymphoma Undergoing Immune Checkpoint Inhibitor Therapy: A Single-Institution Experience. J. Comput. Assist. Tomogr. 2020, 44, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Armand, P.; Engert, A.; Younes, A.; Fanale, M.; Santoro, A.; Zinzani, P.L.; Ansell, S.M. Nivolumab for Relapsed/Refractory Classic Hodgkin Lymphoma After Failure of Autologous Hematopoietic Cell Transplantation: Extended Follow-Up of the Multicohort Single-Arm Phase II CheckMate 205 Trial. J. Clin. Oncol. 2018, 36. [Google Scholar] [CrossRef] [PubMed]
- Hur, J.Y.; Yoon, S.E.; Kim, S.J.; Kim, W.S. Immune checkpoint inhibitors in patients with pretreated HodgkinEs lymphoma: A Korean single-center, retrospective study. Blood Res. 2020, 55, 85–90. [Google Scholar] [CrossRef] [PubMed]
| Author, Year of Publication | Location | Study Design | Response Criteria | Category of Pseudoprogression | Type of Lymphoma | Agent | No. of Patients | Patients with Pseudoprogression | Patients with IR |
|---|---|---|---|---|---|---|---|---|---|
| Chen et al. 2020 [16] | France | Retrospective | Lugano, LYRIC | Progressive disease followed by transient progression in lesions size and metabolism on LYRIC | Relapsed or Refractory HL | Nivolumab | 45 | 2 | 16 |
| Dercle et al. 2018 [17] | France | Clinical Trial | Lugano, LYRIC | Progressive disease followed by transient progression on LYRIC | Relapsed or Refractory HL | Pembrolizumab or Nivolumab | 16 | 2 | 7 |
| Kwong et al. 2017 [20] | Hong Kong, Singapore, South Korea | Retrospective | 5-point Deauville Score | Progressive Disease followed by CR on 5-point Deauville Score | Relapsed or Refractory NK/T-cell Lymphoma | Pembrolizumab | 7 | 1 | N/A |
| Liput et al. 2020 [21] | USA | Retrospective | iRECIST | N/A | Relapsed or Refractory Classical HL and Non-HL | Pembrolizumab or Nivolumab | 24 | 0 | N/A |
| Merryman et al. 2020 [18] | USA | Retrospective | Lugano, LYRIC | N/A | HL | ICIs (Pembrolizumab, Nivolumab, Ipilimumab, Combination, or other PD-[L]1) | 64 | 0 | 1 |
| Wang et al. 2019 [19] | China | Retrospective | PERCIST, Lugano | Progressive disease followed by PR or CR on PERCIST | Relapsed or Refractory Non-HL | CAR-T cell Therapy | 19 | 3 | N/A |
| Armand et al. 2018 [22] | Europe, North America | Multicenter, Phase II Clinical Trial | IWG Criteria | treated beyond initial progression followed by PR or CR on IWG Criteria | Relapsed or Refractory Classical HL | Nivolumab | 243 | 36 | N/A |
| Hur et al. 2020 [23] | South Korea | Retrospective | The revised response criteria | Progressive disease followed by CR on the revised response criteria | Classical HL | Pembrolizumab or Nivolumab | 20 | 2 | N/A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, A.J.; Kim, K.W.; Cho, Y.C.; Ko, Y.; Sung, Y.S.; Shin, Y.; Lee, J.; Kim, M.-h. Incidence of Immune-Mediated Pseudoprogression of Lymphoma Treated with Immune Checkpoint Inhibitors: Systematic Review and Meta-Analysis. J. Clin. Med. 2021, 10, 2257. https://doi.org/10.3390/jcm10112257
Lee AJ, Kim KW, Cho YC, Ko Y, Sung YS, Shin Y, Lee J, Kim M-h. Incidence of Immune-Mediated Pseudoprogression of Lymphoma Treated with Immune Checkpoint Inhibitors: Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2021; 10(11):2257. https://doi.org/10.3390/jcm10112257
Chicago/Turabian StyleLee, Amy Junghyun, Kyung Won Kim, Young Chul Cho, Yousun Ko, Yu Sub Sung, Youngbin Shin, Jiwoo Lee, and Mi-hyun Kim. 2021. "Incidence of Immune-Mediated Pseudoprogression of Lymphoma Treated with Immune Checkpoint Inhibitors: Systematic Review and Meta-Analysis" Journal of Clinical Medicine 10, no. 11: 2257. https://doi.org/10.3390/jcm10112257
APA StyleLee, A. J., Kim, K. W., Cho, Y. C., Ko, Y., Sung, Y. S., Shin, Y., Lee, J., & Kim, M.-h. (2021). Incidence of Immune-Mediated Pseudoprogression of Lymphoma Treated with Immune Checkpoint Inhibitors: Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 10(11), 2257. https://doi.org/10.3390/jcm10112257






