In the following sections, oxidizing biocides are discussed in detail. Chemical forms and generation, dosing, efficacy, application, advantages and limitations, and removal techniques are described. This provides a complete basis for users of industrial and municipal membrane systems to evaluate options. In some cases, the optimal oxidizing biocide is site specific, depending on factors such as local regulations, capital and operating costs, ease of use/training requirements, space requirements, and permeate quality requirements. It should be emphasized that oxidizing chemicals are not compatible with polyamide membranes, as discussed herein Reaction kinetics of various oxidizers with the polyamide varies considerably such that a few biocides that exhibit slower kinetics, such as chloramine, could, under the appropriate feed water conditions, be used in direct contact with the membrane (but in the case of chloramines, is not recommended). Hence, disinfection with oxidizing biocides is limited to addressing the pretreatment system prior to the RO membranes rather than by direct application to the membrane.
i. Chlorine
As described previously, chlorine is the most commonly used biocide to disinfect a membrane desalination system. It is an oxidizer, and, as such, destroys microbial, cellular membranes and internal content. Specifically, three mechanisms have been proposed for bacterial inactivation via chlorine [
90]. Direct oxidation of the cell wall and the interior components is proposed which leads to leaking of cell components through the oxidized cell wall. Another mechanism is inactivation of key enzymes [
99] that are responsible for enabling bacteria to metabolize glucose. Finally, chlorine disables nucleic acids, making them unable to function which leads to death of the microorganism.
The reaction kinetics and efficacy of chlorine disinfection of specific bacterium is a function of several conditions [
90]. The nature of the type and concentration of the organism(s) present and environmental conditions contribute to efficacy. Environmental conditions impacting efficacy include concentration of chlorine, contact time, solution temperature and pH, and the presence of other, interfering substances that exhibit a chlorine demand (e.g., organics, transitions metals).
Forms of Chlorine
The two commonly used forms of chlorine include sodium hypochlorite (bleach) and gaseous chlorine. Gaseous chlorine is more toxic than hypochlorite and requires careful handling. It is, however, more economical than hypochlorite, special handling not withstanding [
100]. Chlorine gas does not remain in gaseous form in water, which inhibits its ability to penetrate biofilm; it immediately hydrolyzes in water (Equation (1)) to form aqueous hypochlorous acid, HOCl: Liquid sodium hypochlorite also hydrolyzes in water (Equation (2)) to form hypochlorous acid:
The formation of hypochlorous acid using chlorine gas yields hydrochloric acid, while sodium hypochlorite forms caustic, thereby reducing or raising the pH of the water, respectively. These effects may have implications as to the scaling potential of the feed water for calcium carbonate (scales at higher pH) and calcium sulfate (scales at lower pH).
Hypochlorite ion, OCl
−, is formed from the decomposition of hypochlorous acid that is somewhat unstable in solution:
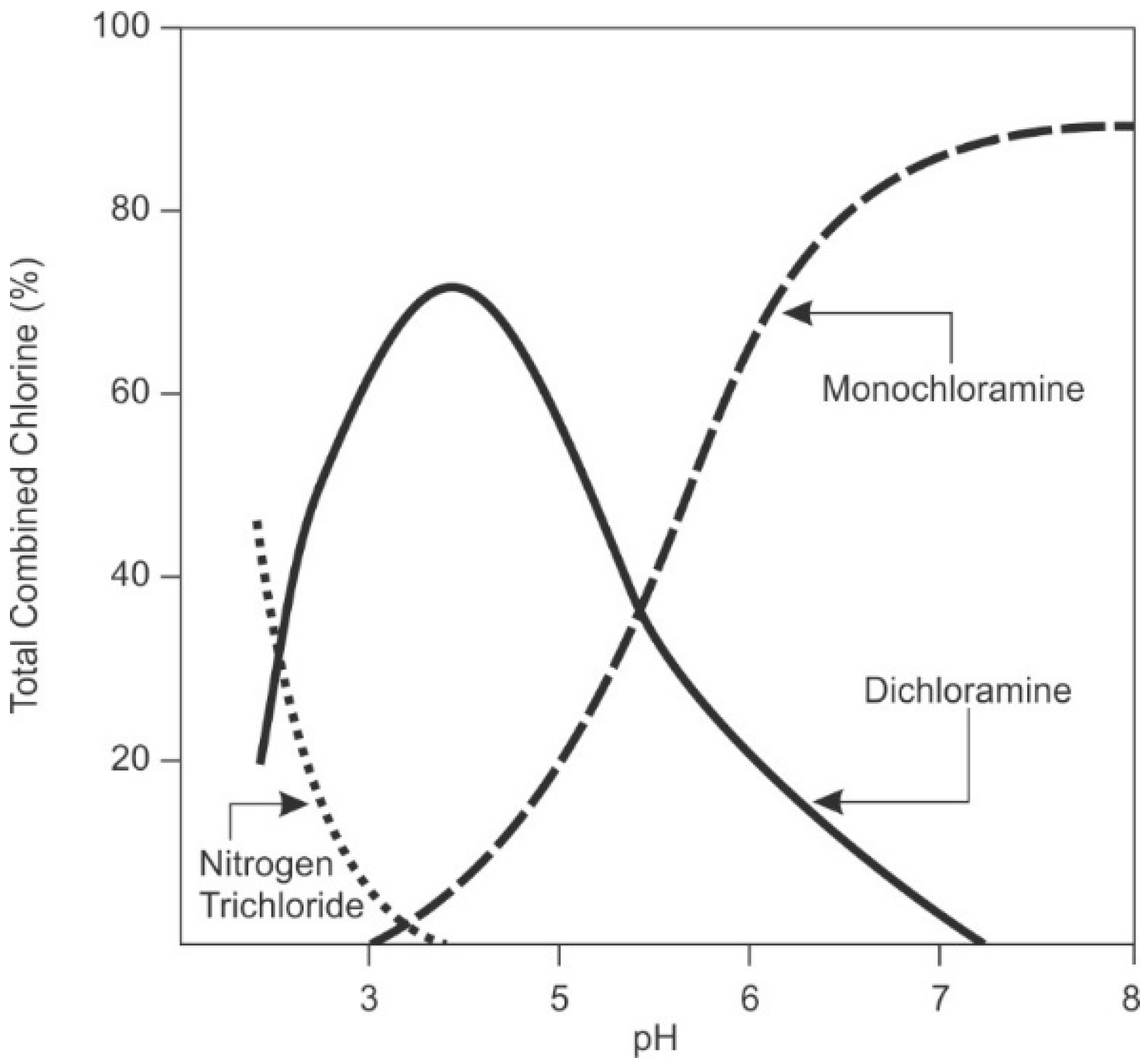
Temperature, salinity, and pH all influence the stability of hypochlorous acid.
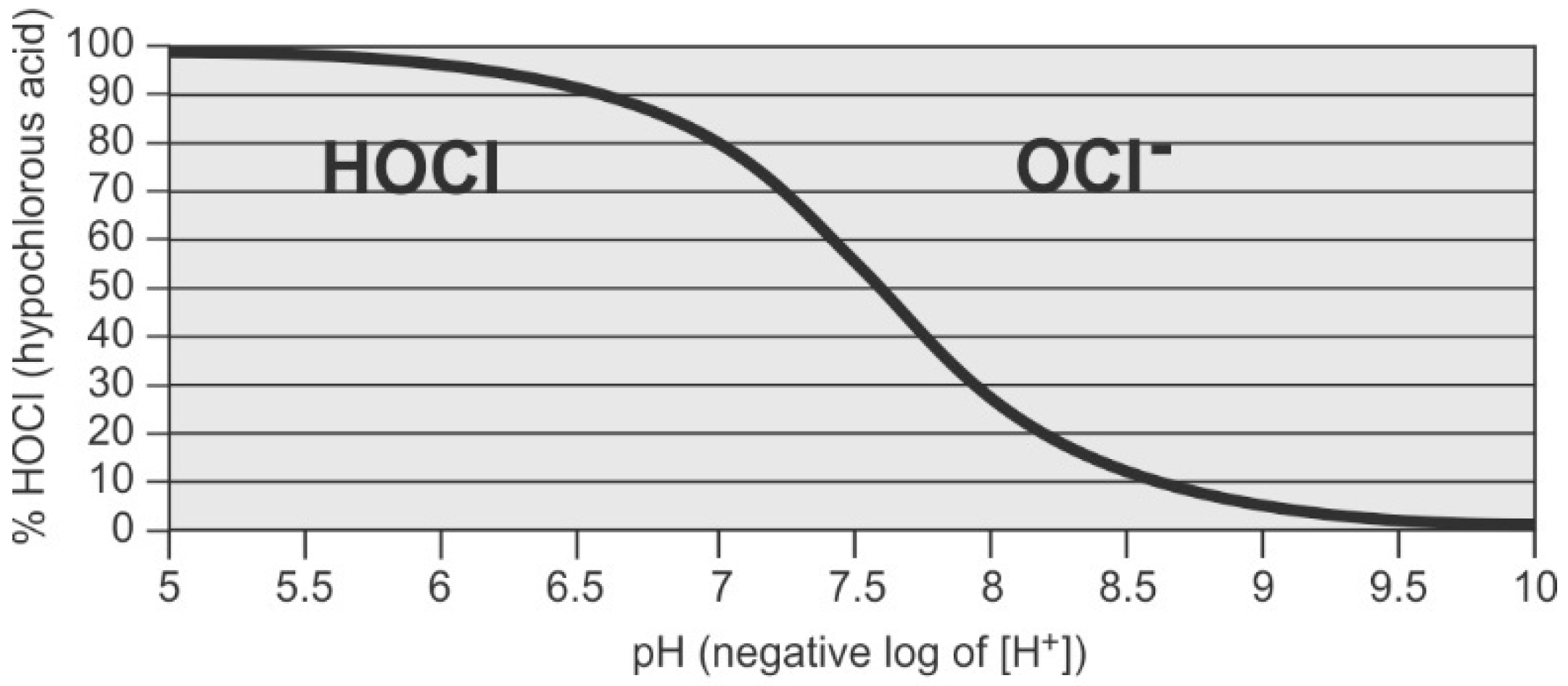
Figure 6 shows the speciation of hypochlorous acid and hypochlorite ion as a function of pH.
Hypochlorite ion has a lower reduction potential than hypochlorous acid (see
Table 3) and is a less powerful disinfectant [
90]. Microorganisms are negatively charged, making entry of the negatively charged hypochlorite ion into the microorganism difficult [
32,
90]. The hydrated ion is much larger in size than the acid and, hence, the resulting kinetics of diffusion into bacterial cells is slower [
90]. Also, disinfection reactions to disrupt the functions of enzymes and nucleic acids are favored at lower pH, where hypochlorous acid, and not the ion, predominates [
101]. Most industrial desalination systems operate between pH 6.5–9, so operators need to be aware of which form of chlorine is present and the relative efficacy for disinfection.
Determination of Chlorine in Water
The most popular methods for determining chlorine residual include amperometric titration and the
N,
N-diethyl-
p-phenylenediamine (DPD) colorimetric tests [
90]. Both are capable to detecting both free (hypochlorite, + hypochlorous acid + chlorine gas + trichloride ion) and total chlorine (free + chloramine (combined chlorine)). Amperometric titration is considered the “gold standard’ to which other methods are compared [
90]. However, it is not well suited for field use, although some portable titrators are available. The DPD method colorimetric testing is typically used in the field for quick determination of free and total chlorine. This technique has a lower detection limit (0.01–4 ppm) than the amperometric titration method (0.1–10 ppm) [
90]. The main advantage of amperometric titration is that it doesn’t suffer the degree of interference with color and particulate matter that hinder the DPD colorimetric test [
90].
Another common method for in-line detection is oxidation-reduction potential (ORP). Specifically, the ORP measures the net potential in an aqueous solution that contains both oxidants, such as chlorine, and reductant, such as bisulfite. The ORP measures the actual activity of the oxidizer or reductant. Free chlorine will register an ORP reading of about 500–700 mV (depending on the pH, chlorine residual and feed water characteristics [
90]). The amperometric and DPD colorimetric methods measure just residuals, which are subject to interferences from other compounds in the solution whereas the ORP measure the actual oxidation potential of the feed water. OPR is commonly used to control dechlorination, and the ORP controller can be tuned to yield a slight residual of sulfite, even under changing chlorine/sulfite activity levels [
90], thereby ensuring the oxidizer has be removed.
Dosing and Efficacy
Critical factors affecting performance of chlorine are exposure time, pH, and method of application. Hypochlorous acid has a higher potential than hypochlorite ion (see
Table 3), and is, therefore, preferred for disinfection. Since pH affects the state of chlorine (hypochlorous acid versus hypochlorite ion,
Figure 6), a pH below 7.5 is desirable for disinfection, as a greater proportion of chlorine is present as hypochlorous acid. Disinfection for the pretreatment system requires an exposure time of 20–30 min with a residual of 0.5–1.0 ppm at the point of dechlorination [
34,
50]. The ASTM method D 1291, “Standard Practice for Determining Chlorine Requirement of Water” is useful to determine the optimal dosage necessary to achieve the required residual.
There are three primary methods of applying chlorine: continuous, shock, and continuous plus shock. Shock treatment can be effective, but continuous feed, which is the most commonly practiced method, has greater efficacy than shock treatment only for disinfection [
34]. For a specific seawater system with a continuous residual of 0.04–0.05 ppm free chlorine in the pretreatment, biofilm was not observed on the RO membranes [
87]. Shock treatment alone is thought to minimize the potential for bacteria to become conditioned to continuous feed. Continuous feed plus shock treatments can help to overcome the conditioning of bacteria to chlorine. This method was shown to be more effective at controlling membrane biofouling than either singular method during warm weather, when bacterial activity is enhanced [
96].
Advantages and Limitations
Chlorine offers several advantages over other oxidizing biocides for disinfection of membrane desalination systems [
102]. Chlorination is a well-established, cost-effective disinfection technique. It allows for flexible dosing control and carries a residual in water. Residuals are important for continued biocontrol downstream of the biocide injection point.
There are some distinct limitations to using this chlorine for disinfection. On a universal level, chlorination forms THMs and HAAs, both of which are carcinogenic species. Hypochlorous acid can also form disinfection by-products (DBPs), such as bromate (in seawater—maximum contaminant level, MCL, of 0.01 ppm) and chlorate (no official MCL, but California has a notification level of 0.8 ppm) [
100,
103].
Cryptosporidium parvum and
Mycobacterium avium, which are pervasive in water system biofilms, are not well controlled using chlorine; endospores and protozoa are also not well controlled with chlorine [
104,
105].
Chlorine also oxidizes organics, such as longer-chained humic acids, cleaving them into shorter-chained molecules. These smaller molecules are transformed into AOC that microbes can more easily digest. This is a particularly serious problem after the point of dechlorination [
50,
87,
89,
106]. Studies have shown increased rates of biofouling following dechlorination [
106,
107,
108]. The absence of disinfectant, the degree of disinfection as opposed to sterilization of the system, and the availability of more AOC post chlorination, lead to greater rates of biofouling post dechlorination.
In seawater systems, chlorination leads to the formation of free bromine (typically present as hypobromite ion and hypobromous acid, both of which are pH dependent similar to hypochlorite ion and hypochlorous acid), due to the high concentration of bromide (about 65 ppm) naturally occurring in seawater [
109]. Research by Shemer and Semiat [
109] suggested that free bromine was more stable and more aggressive to polyamide membranes than free chlorine. As a result, some seawater membrane plants have stopped the use of chlorination all together, with variable results [
110,
111,
112].
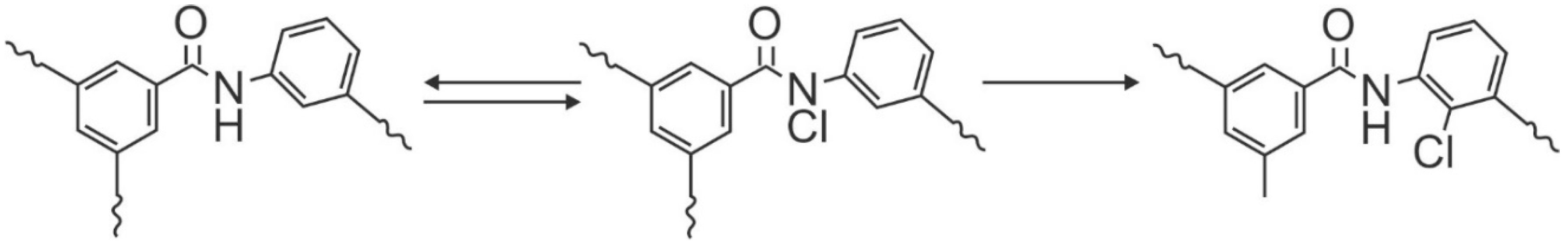
Perhaps the biggest limitation to the use of chlorine is that it oxidizes membranes, destroying the integrity of the polyamide layer. Chlorine substitutes onto the amide functional group followed by destroying the hydrogen-bond linkages in the polymer that ultimately results in ring substitution by the halogen via Orton Rearrangement, as shown in
Figure 7 [
97,
98,
113]. The degraded membrane passes more water and loses its ability to reject solutes; these results are irreversible.
Dechlorination
Due to the damage inflicted on polyamide membranes, free chlorine must be removed prior to the membrane system. In general, ORP is used to determine when free chlorine is no longer present; a reading of less than 200 mV is generally assumed to indicate the absence of free chlorine [
114]. Chlorine analyzers are sometimes used but they are designed to measure the presence of chlorine rather than the absence of it. If an analyzer is used, free chlorine residual should be measured at less than 0.02 ppm to minimize degradation of the membranes [
83]. Dechlorination is relatively straight- forward. Most system rely on sodium metabisulfite to chemically remove free chlorine. Carbon filtration (which relies on an oxidization/reduction reaction to reduce chlorine) is also used frequently. UV radiation is used less commonly for dechlorination.
Sodium Metabisulfite
Dechlorination involving sodium metabisulfite follows a 2-step process, where the sodium metabisulfite first forms sodium bisulfite in water:
Hypochlorous acid is then reduced by sodium bisulfite:
Sodium metabisulfite can be purchased as a dry product and then dissolved in water to form sodium bisulfite. Alternatively, sodium bisulfite, as a liquid, can be used directly for dechlorination. Many systems employ the liquid sodium bisulfite for ease of handling. Non-cobalt catalyzed product is required, as the cobalt in the presence of free chlorine that was not scavenged, will catalyze chlorine destruction of the membrane [
21]. In theory, every ppm of free chlorine requires 1.47 ppm of sodium bisulfite or 3.0 ppm of sodium metabisulfite for reduction [
83]. Since most sodium bisulfite solutions are about 33–37% active, the theoretical dosage would be about 3.5–4.5 ppm per ppm of free chlorine. Often, a safety factor of 1.5 to 2 times theoretical is applied to determine the actual dosage [
83].
ORP is generally used to confirm removal of free chlorine. When using bisulfite, an ORP value of less than 175–200 mV is recommended to ensure protection of the membranes from chlorine attack [
115]. Care should be used to not grossly overfeed bisulfite to the point of a negative ORP, as this produces a reducing environment friendly to proliferation of anaerobic bacteria [
116,
117].
Activated Carbon Filters
Free chlorine can be removed from aqueous solution via an oxidation/reduction process using activated carbon. Chlorine is reduced to chloride while the carbon is oxidized, per the following reactions:
where Carbon-O* is surfaced-oxidized carbon. These reactions are instantaneous, so empty bed contact time (EBCT) can be a low as 3 min for free chlorine removal, however, allowances for up to 5–10 min of EBCT is typical [
83,
118]. Service flow rate for dechlorination prior to a membrane system is 2 gpm/ft
3, with a minimum bed depth of 2.5 ft. General service life for carbon that is used for dechlorination is about 12–18 months.
Carbon filters are known for incubating microbes [
78,
79,
119,
120]. Microbes can slough off with carbon fines or on their own out of the filter and travel downstream to infect the membranes. To eliminate this issue, it is recommended to use sodium bisulfite for dechlorination [
21], unless other circumstances prohibit the use of bisulfite: the feed flow rate is less than about 30 gpm, where control of the bisulfite chemical feed would be difficult; organic (TOC) removal is also required; or there is a process concern with using bisulfite (e.g., in food and beverage applications).
Ultraviolet Radiation (UV)
An alternative method to chemical addition or carbon reduction for dechlorination is UV radiation. High-intensity, broad-spectrum UV (medium-pressure UV), can reduce chlorine gas at peak wavelengths of 180–200 nm, while 292 nm is ideal for hypochlorous acid destruction. A UV dose of 45–90 mJ/cm
2 or about 150% to 300% of that required for general disinfection can dissociate as much as 15 ppm of free chlorine into chloride ions [
121,
122]. (Specific dosage depends on several factors, including organic concentration, total chlorine present, and the ratio of free chlorine to chloramines [
121]). The UV technique offers that advantages of chemical-free dechlorination, and elimination of the problem of sloughing off bacteria that occurs when using a carbon filter.
ii. Combined Chlorine (Chloramine)
Combined chlorine, which includes the oxidizers monochloramine, NH
2Cl; dichloramine, NHCl
2; and nitrogen trichloride (trichloramine), NCl
3, is also used to disinfect membrane-based desalination systems. Monochloramine has a reduction potential similar to hypochlorous acid (as shown in
Table 3) but has lower biocidal power (0.4% of hypochlorous acid) [
123] and slower reaction kinetics (see
Table 4) [
124]. Studies have shown that chloramines require up to 100 times the contact time or 25 times the concentration of free chlorine to inactivate some bacteria, such as
Escherichia coli (
E. coli) [
125,
126,
127]. It can take days or weeks to achieve disinfection [
105]. The slower reaction kinetics of monochloramine leads to lingering residuals in distributions systems (as opposed to chlorine, which dissipates more rapidly). Thus, municipal distribution systems benefit from the lingering monochloramine residual, making it more effective than chlorine in the long term. While water distribution systems benefit from the slower reaction kinetics, membrane pretreatment requires more rapid kinetics. Many municipalities are shifting from using strictly chlorine to chloramine [
128] due to ever stricter regulations on DBPs resulting from chlorination. Operators of membrane systems with chloramine-treated municipal make-up water need to be cognizant of this disinfectant, and its effects on the membrane system.
The mechanism for chloramine attack on microorganisms is not well known but may involve inhibition of proteins or functions such as respiration (a protein-facilitated processes) [
129]. Jancangelo et al. [
129], found that the inactivation process is inconsistent and, therefore, monochloramine should be feed at several discrete intervals to ensure that disinfection occurs.
The comparison of chloramines to free chlorine is not straight forward, as disinfection performance depends on pH and other factors (including temperature and exposure time) [
90]. Monochloramine is more effective at higher pH than the hypochlorite ion, whereas hypochlorous acid is more effective than monochloramine at lower pH. [
101]. In most membrane desalination systems, the feed water pH runs about 6.5–9, so it follows that at the lower end of this range, hypochlorous acid would be more effective, but at the higher end of the range, monochloramine would be more effective.
Chloramine Generation
The combining of free chlorine (as hypochlorous acid) with free ammonia (NH
3(g)) is known as chloramination [
130]:
These reactions are strongly dependent on pH, as shown in
Figure 8, relative concentration of the reactants, and temperature [
131]. The reaction kinetics of formation are minimized at pH 8.4 and 25 °C [
90]. Stoichiometrically, Equation (9) shows that one mole of monochloramine forms in the reaction of one mole of hypochlorous acid with one mole of ammonia. This corresponds to a chlorine, (as ppm Cl
2(g)) to ammonia (as ppm NH
3-N), ratio of about 4.5 to 5.0. Higher ratios will slow the reaction [
90], lead to breakpoint chlorination, and generate dichloramines, which are notorious for taste and odor issues [
90]. Lower ratios increase the potential for nitrification to occur [
90]. Lower pH favors formation of di-and trichloramine [
90], although little trichloramine persists under typical water treatment conditions. Lower temperature will slow the reaction kinetics.
Dichloramine has been shown to be twice as strong of a biocide as monochloramine [
101] but is objectional due to its taste and odor issues [
90]. While being an even stronger oxidizer than dichloramine, little information regarding the biocidal effects of trichloramine are known, as it has highly objectionable taste and odor, and so its formation is avoided in water treatment [
90].
Determination of Chloramine in Water
The most common method of determining chloramine in water is by subtraction of free chlorine (determined by DPD free chlorine method) from total chlorine (determined by the DPD total chlorine method) [
132]. However, many organic chloramines also produce color for the DPD total method [
70], which introduce error into the measurement of chloramine available for disinfection [
132,
133,
134]. Despite this, the DPD subtraction method is the most commonly used field test for chloramine.
A modified indophenol method using MonochlorF (Hach Company, Loveland, CO, USA) reagent does not have interference with organic chloramines or transition metals [
132], and measures only monochloramine, as opposed to all species of chloramine [
135]. Research by Lee et al. [
132], concluded that determination of the actual concentration of monochloramine is a better indicator of the actual disinfection residual than the DPD subtraction method [
132,
136].
Dosing and Efficacy
Ideally, chlorine is added to ammonia to generate chloramine, as this method reduces or eliminates chlorinous taste and odor [
90]. Further, this technique minimizes the potential for damage to the polyamide membrane from free chlorine or the chloramine itself [
137]. For wastewater applications, where ammonia is naturally present, addition of chlorine directly to the wastewater to form chloramines has worked successfully at Water Factory 21 (predecessor to the Ground Water Replenishment System, GWRS, (Orange County, CA, USA)) to minimize biofouling [
138,
139]. This was accomplished
without the need for dechlorination/dechloramination or damage to the polyamide membranes [
96]. The hypothesis for this result is that the high ammonia concentration and organic content of secondary effluent protects the polyamide membrane from Orton Rearrangement and subsequent membrane damage that occurs with chlorination of membrane [
137]. The exact mechanism of interaction between chloramines and polyamide membranes is not yet understood, however.
In industrial and municipal practice, for pretreatment of surface and ground water, chlorine is first added to the water to be treated (known as primary disinfection), and then ammonia is added to form chloramines (secondary disinfection) [
90]. The rationale for adding ammonia to chlorinated water is that initial chlorination will result in more rapid disinfection and the resultant chloramine after ammonia addition will then linger throughout the distribution system [
90]. Good mixing is essential to avoid side reactions with organics that yield organic chloramines, which have no biocidal effect. The full understanding of chloramines to treat water requires the understanding of breakpoint chlorination.
Figure 9 illustrates breakpoint chlorination.
Chloramination occurs in Zone 1 of the figure where chlorine is combined with ammonia to form monochloramine. Note that there may be a lag in the initial curve due to species in the water that put an immediate demand on chlorine, such as iron and manganese. Once the immediate demand has been satisfied, the maximum concentration of monochloramine occurs when the ratio of Cl
2 (gas) to NH
3-N is 5.0. This is the “monochloramine hump.” For systems using monochloramine, the desired operating range is embodied in Zone 1. Addition of chlorine post Zone 1 results in the formation of dichloramine via chlorine reaction with monochloramine and ammonia (Zone 2). A series of dichloramine reactions then follow and continue until “breakpoint” is achieved, where the following equation approximates the situation [
90]:
At breakpoint, the composition of the solution includes primarily dichloramine, with trace amounts of free chlorine, monochloramine, and organic chloramines; total concentrations may be a few tenths of a ppm [
90]. After breakpoint, in Zone 3, the free chlorine residual increases proportionally to the chlorine dose.
Efficacy for control of membrane biofouling with chloramine is questionable [
26]. While some report good biofouling control for wastewater reuse systems [
96], Raffin et al. [
140], found little to no benefit to direct contact of chloramine with polyamide membranes for this application. The theory promoted by Raffin et al. [
140] is that the low rejection of chloramine by the polyamide membrane leads to not enough chloramine residual in the concentrate stream for adequate disinfection [
140]. A literature search found little discussion of efficacy for control of ground or surface water; most research was focused on compatibility with the polyamide membrane. In general, chloramines are not as effective for disinfection compared to hypochlorous acid [
26]. The assumption is, then, that they would be less effective at membrane biofouling control than hypochlorous acid.
Advantages and Limitations
The primary advantage of chloramination is its moderated formation of DBPs relative to chlorination, since they chloramines not as reactive as chlorine is with organics [
136]. Chloramine has proven successful at controlling biofouling of membranes for wastewater reuse applications [
141], where chlorine is added to ammonia naturally present in the feed source. However, other than membrane systems operating on chloraminated, municipal feed water, the use of chloramines has not been widespread to directly treat surface and ground water [
141]. Lozier [
137] studied compatibility of polyamide membranes with chloramine operating on ground water with low concentration of organics, and high-organic loaded surface water. In both cases, the membranes suffered increases in specific flux and loss of salt rejection as compared to control membranes not exposed to chloramine. Lozier [
137] concludes that until further study is undertaken to understand the apparent synergistic effects of the specific water matrix and chloramine on degradation of the polyamide membrane, that chloramines may be best suited to wastewater reuse applications only.
Some report that most polyamide membranes can tolerate longer exposures to
pure chloramine, anywhere from 150,000 to 300,000 ppm-hours [
21,
142,
143]. However, pure chloramine is elusive due to the equilibrium of chloramines with free chlorine and ammonia (Equations (9)–(11)). Research by Lozier [
137], Silva et al. [
144] and Maugin et al. [
145] indicates the that chloramine itself attacks polyamide membranes. Maugin et al. [
145] found that just 20,000 ppm-hours of exposure resulted in a 70% increase in permeability and a linear decrease in salt rejection. The effects of temperature and pH on the equilibrium equations, and any light metals, such as aluminum, iron, and manganese, that may be present can significantly shorten membrane life when exposed to chloramines [
21,
143,
144].
One proposed mechanism for membrane degradation via chloramines is similar to that for chlorine degradation of polyamide membranes [
146]; amidogen radicals (NH
2) are formed (catalyzed by the presence of metals ions) which attack the ring structure and allow for direct chlorination by excess chloramine [
146]. The difference versus free chlorine is that the kinetics of membrane degradation via chloramine are slower and chloramine is less aggressive [
147]. Maugin et al. [
145], on the other hand, found evidence, using Rutherford backscattering spectrometry (RBS) and Fourier transform infrared spectroscopy (FTIR) analyses of virgin, chlorinated, and chloraminated polyamide membranes, that different reaction mechanisms may be involved for degradation via chlorine and chloramine exposure.
The potential for free chlorine to be in equilibrium with monochloramine is great (see Equation (9)). This coupled with the uncertainty regarding polyamide membrane compatibility with chloramine, makes dechlorination a necessity prior to polyamide membranes [
143]. Dechlorination can lead to dechloramination, which can increase the ammonia gas or ammonium ion concentrations, depending on the pH [
21].
Free ammonia (gas) can also be in equilibrium with chloramine (Equation (9)) and will affect the membrane system in negative ways. Nitrification of the system can occur since free ammonia is a nutrient source for nitrifying bacteria [
130,
136]. Gases, including ammonia, are not rejected by an RO membrane. Hence, the concentration of ammonia gas in the feed water will be the same as that in the permeate, which can go on to corrode non-stainless steel metal components and piping downstream. Ammonia gas swells polyamide membranes, causing a
reversible increase in ion passage through the membrane. Swelling occurs at a pH of greater than about 7.5. If the pH is lowered to below 7.5, the ammonia (gas) will be converted primarily to ammonium ion, which does not swell the membrane and is also well rejected by an RO membrane [
83]. Since most membrane desalination systems operate at pH 6.5–9, the presence of free ammonia can be a concern.
Process variables in forming chloramines are difficult to control in actual practice, which can shift Equations (9)–(11). Additionally, if the TOC concentration exceeds about 3 ppm, organic chloramines can be formed in addition to the products shown in Equations (9)–(11). Organic chloramines have little or no disinfecting capabilities. Due to these factors, field testing has been inconclusive as to the efficacy of chloramines in controlling biofouling of polyamide membranes [
87].
Chloramination of seawater systems is not recommended [
96]. The exposure of bromides in seawater to chloramine (ammonia) forms bromamines. Bromamine has several times the oxidation strength of chloramine [
96,
148], and has as much biocidal activity as hypobromous acid [
148]. Severe membrane damage was incurred at West Basin Municipal Water District (Carson, CA, USA), during a pilot study on seawater intake due to the formation of bromamines via chloramination [
100].
Dechloramination
Chloramines are considered a mild oxidizing agent [
143] and, due to the inconclusive data regarding polyamide membrane compatibility, chloraminated water should be removed prior to the membranes There are several methods to remove chloramine, including sodium thiosulfate, ascorbic acid, sodium metabisulfite chemical addition, carbon filtration, and, occasionally, UV radiation., with the later three being the most common methods [
143].
Sodium Metabisulfite
Dechloramination with sodium metabisulfite follows a 2-step process, similar to dechlorination with sodium metabisulfite. The first reaction, Equation (4), shows the reaction of sodium metabisulfite with water to form sodium bisulfite, which then reacts with monochloramine as follows:
The reaction for sodium bisulfite is rapid (albeit slower than the reaction with chlorine), requiring from 1 to 5 min of contact time [
149,
150], with complete mixing essential.
Carbon Filtration
Chloramine is effectively removed from aqueous solution using activated carbon in an oxidation/reduction process. As with dechlorination, the reaction with chloramines is an oxidation reaction process. However, the process for complete chloramine removal, including free chlorine and ammonia, is considerably more complicated. Work by Champlin et al. [
151] and Bauer et al. [
152], demonstrated that removal of chloramine at Cl
2:NH
3-N ratios of less than 7.6 (breakpoint) is governed by the following equation:
This reaction was found to be instantaneous. Note that this process leaves behind free ammonia. As discussed previously, free ammonia, at a pH of greater than about 7.5, will swell the polyamide membranes, resulting in a reversible increase in salt passage. For complete removal of the ammonia, Champlin et al. [
151] and Bauer et al. [
152] determined that additional chlorine must be added to achieve breakpoint. At breakpoint, the reaction is shown in Equation (15) shows complete removal of chloramine (and ammonia) to nitrogen gas [
151]:
Kim and Snoeyink [
153] described an additional reaction, which results in direct removal of chloramine without the intermediate formation of free ammonia:
They determined that after an initial acclimation period about 50% of the monochloramine reacted according to Equation (14) and the remainder reacted according to Equation (16) (Bauer et al. [
152] determined this acclimation period to be about 20 h).
Contact time for removal of chloramine using activated carbon is critical [
153]. Kim and Snoeyink [
153] determined that longer contact time, with flows of less than 1 gpm/ft
3, resulted in the most efficient removal of monochloramine. Potwora [
154] indicates that a flow rate of 0.75 gpm/ft
3 is required for removal using standard activated carbon. Further, Kim and Snoeyink [
153] found that the residual monochloramine concentration was about 0.1 ppm after carbon filtration, and that longer contact times did not change this final, effluent concentration. The age of carbon also impacts contact time; new carbon can require as little ss 5 min of empty bed contact time, while older carbon can require up to 30 min.
Ultraviolet Radiation
As with UV destruction of chlorine, UV can also be used for chloramine destruction. The dose required for chloramine photolysis is about 200% that typically used for disinfection, or about 60 mJ/cm
2 [
122]. Wavelengths of 245–365 can remove up to 5 ppm of chloramine in a single pass [
121]. Wavelengths for optimum photolysis of various forms of chloramine are 245 nm for monochloramine, 297 nm for dichloramine and either 260 nm or 340 nm for trichloramine.
iii. Ozone
Ozone (O
3) or trioxygen is a very strong oxidizing biocide. The use of ozone itself yields minimal DBPs in most applications, except by-products formed by reaction with bromide present in seawater. Seawater by-products include bromate [
155,
156,
157], a carcinogen [
158] that is not well rejected by polyamide membranes [
26], and bromo-organic compounds, such as bromoform [
155].
Ozone was experimentally used to disinfect of water in 1886 [
159], and first employed commercially for this purpose in 1906 operating on the Vesubie River at the Bon-Voyage facility in Nice, France [
160,
161]. Today, thousands of water treatment plants utilize ozone as part of their chemical water treatment [
160], with 72% of ozone users in the United States reporting employing ozone for disinfection [
160].
Ozone is very unstable in water, with the kinetics and reaction products of decomposition depend on the pH and alkalinity of the water, and type and concentration of natural organic matter (NOM) present [
162]. Decomposition of ozone in water forms several free radicals, with hydroxyl radical, OH* being primary [
162]. The free radicals formed are in themselves very strong oxidizers capable of disinfection and react within a few microseconds [
160].
Primary action of ozone with microorganisms follows one of three pathways [
162]: direct reaction with ozone itself, indirect reaction with radicals that form when ozone decomposes in water, or both. For indirect reaction with radicals, ozone in water initially reacts with hydroxide ions to form superoxide anion (O
2−) and hydroperoxyl radical (OH
2*):
In following reactions, ozone and superoxide anion react to form ozonide radical (O
3*), which further reacts to form several radicals, including HO
2*, HO
3*, HO
4*, and the afore mentioned OH* [
160]. Finally, termination reactions occur, during which formation of free radicals is inhibited; these involve the reaction of hydroxyl radical with carbonate or bicarbonate, or the reaction of 2 radicals with each other [
160].
Due to the high oxidation potential of ozone and its radicals, these compounds can oxidize the bacterial cell wall. Once inside the cell, ozone and its radicals oxidize all essential cell components, including enzymes, proteins, deoxyribonucleic acid (DNA), and ribonucleic acid (RNA). The cell lyses when the microbial components and its membrane are damaged in such a manner. Shear forces can then be used to break up the organisms and their biomass [
77]. Ozone is an effective biocide against bacteria, viruses, protozoa (typically resistant to most disinfectants [
155]), and endospores [
163].
Ozone Generation
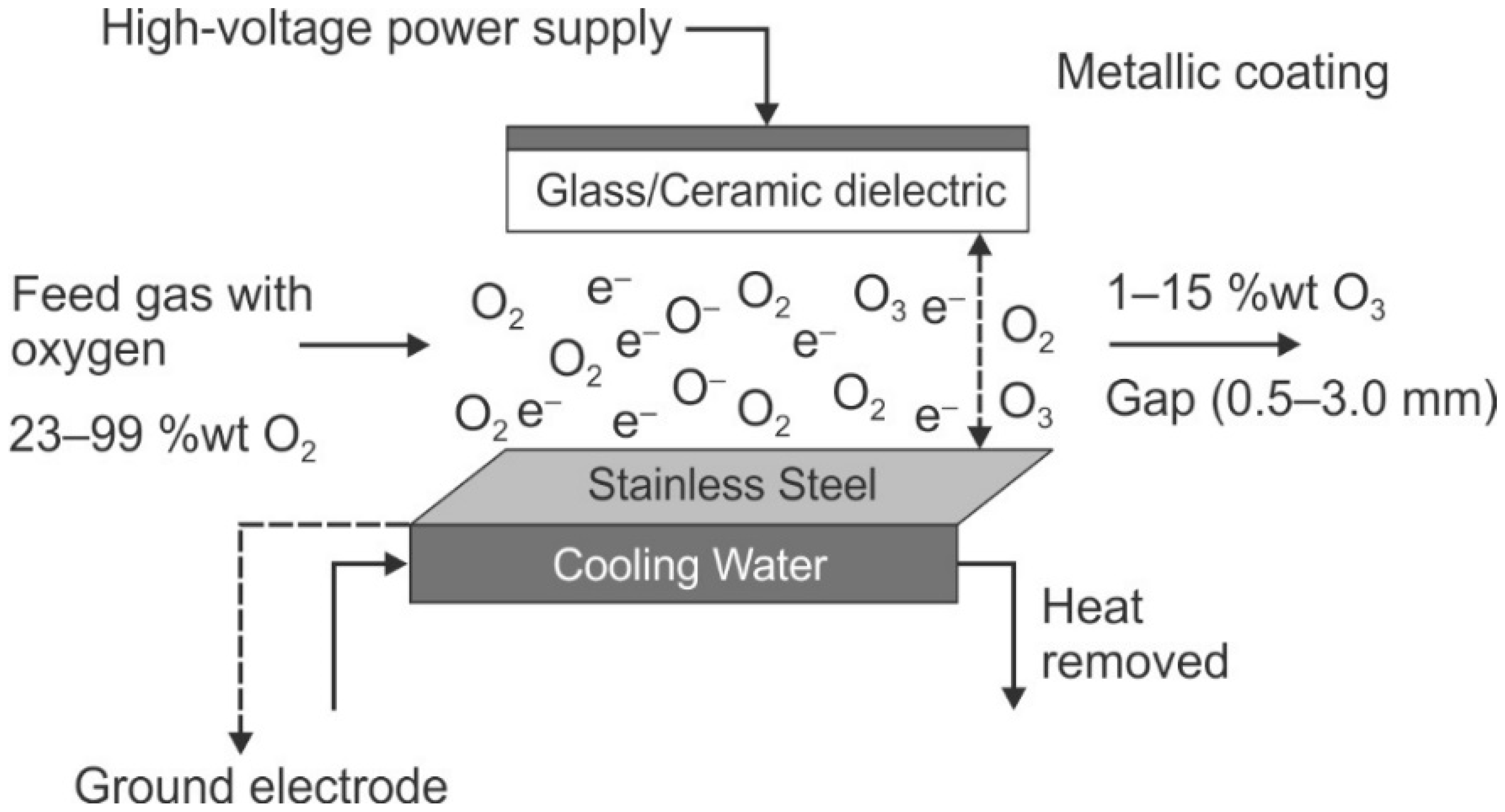
The instability and rapid decomposition of ozone implies that it must be generated on. The most common method of ozone generation is electrical discharge (also known as coronal discharge) [
160,
161,
164]. Electrical generation involves the exposure of air or pure oxygen to a uniform high voltage/high density alternating current, which drives the reactions. A gap between two electrodes, one of which is coated with a dielectric material, allows an electric field to develop, allowing the reactions to proceed (see
Figure 10). Other generation methods include acid electrolysis, UV photochemical reaction, and radiochemically; there are a few other chemical methods that are infrequently used [
160,
161,
164].
The overall chemical reaction for the formation of ozone is shown in Equation (18):
In its simplified form, the initiation reaction (Equation (18)) occurs when free electrons dissociate oxygen molecules into atomic oxygen molecules (O), which then react with additional oxygen molecules in a “three body collision” with any other molecules in the gas, M, to form ozone (Equation (20)) [
165,
166]:
The reactions of ozone with atomic oxygen and free electrons (Equations (21) and (22), respectively) are simultaneously occurring to reform oxygen molecules [
165]:
Further, when air, rather than oxygen, is used as the feed gas, nitrogen species, such as N
+, N
2+, and N, complicate the reactions system [
166]. Nitrogen oxide radicals are formed that consume ozone, as shown in Equation (23) [
166]:
A typical ozone generator yields about 0.5–16 wt% ozone to the carrier gas [
160]. Only 5–10% of the applied energy is used to generate ozone; the remainder dissipates in the form of heat. Decay reactions in Equations (20) and (21) are favored at higher temperatures, so ozone generators include methods to dissipate heat to discourage these ozone decay reactions.
Once generated, there are several transfer methods to available to contact water with ozone. Fine bubble diffusers are most widely used and are the most energy-efficient means of transfer [
153]. Side-stream injectors are commonly used in new installations [
153], which offer transfer efficiencies of greater than 95% that does not decline with time. Smaller systems requiring less than 4 km
3/day of water to be treated typically use packed columns where ozone is bubbled up through a column of water packed with ceramic media. The primary advantage of this technique is that there are no moving parts. U-tube reactors, aspiring turbine mixers, and spray chambers (typically used for oxidation of inorganic compounds rather than disinfection [
160]) are also used.
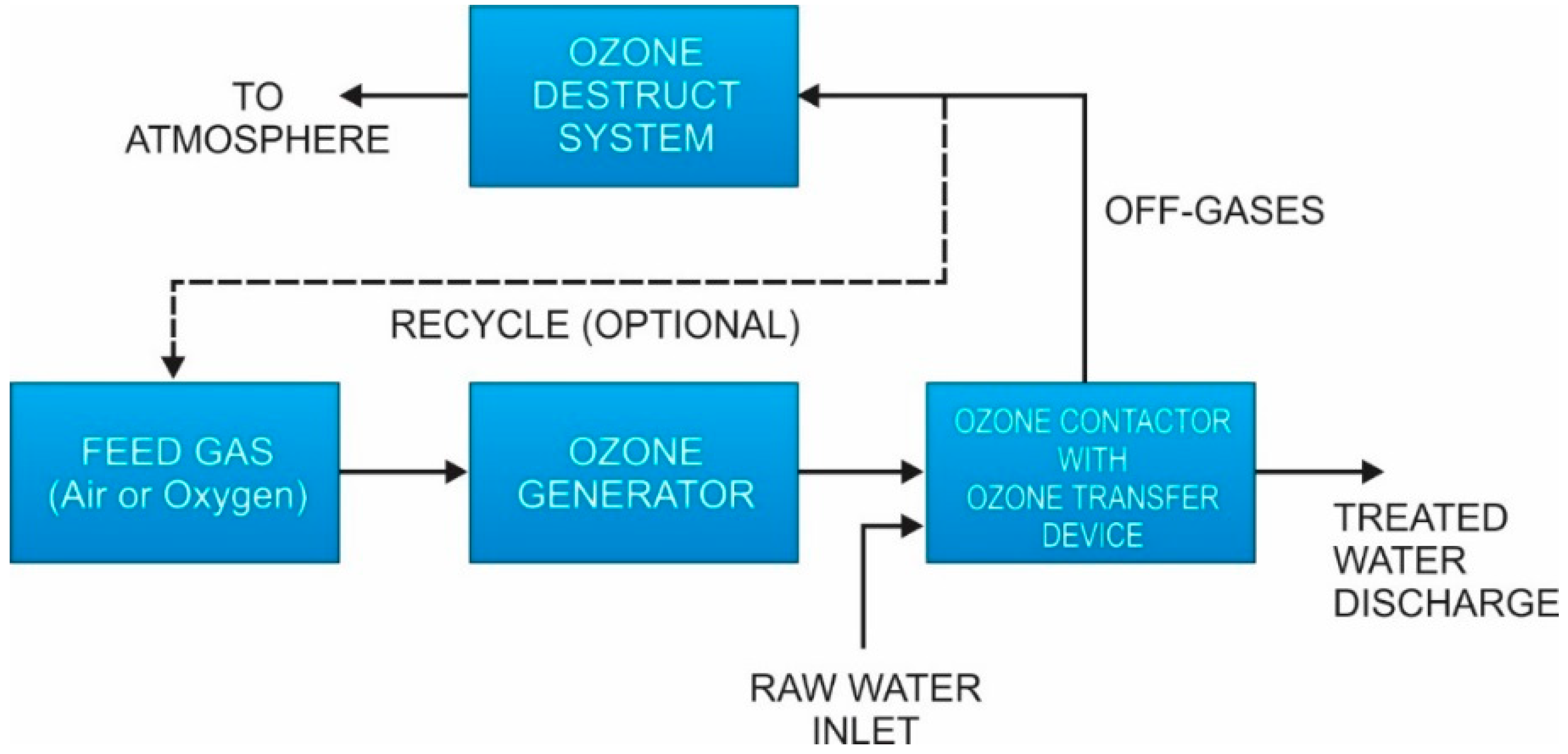
Considerations in selection of transfer technique include the weight percent of ozone in the carrier gas, ozone gas to water ratio, ozone demand, and the nature of the contactor used for to provide residence time for ozone to react with the raw water [
160]. Conventional baffled basins and pipeline contactors are the most common type contactors used for ozone transfer and oxidant contact time [
160]. A generalized ozone process flow diagram is shown in
Figure 11.
Determination of Ozone in Water
Determination of ozone residual in water is problematic [
166,
167,
168]. The volatility in solution and the continuous self-decomposition reactions hamper efforts to determine residual. Further, the reaction of various ozone decomposition products (radicals) and reaction of ozone itself with organic and inorganic contaminants make residual quantification a challenge. The most direct method for measuring ozone residual involves measuring adsorption at 260 nm (adsorption at 258 nm also shows a maximum for ozone, and the values at the two wavelengths are virtually identical [
166]). This method assumes that the water tested has little or no dissolved organic matter (DOM), turbidity, and iron that also adsorb at these wavelengths.
Colorimetric methods, such as DPD and indigo, can be used to field test for ozone residual. The DPD method, however, suffers from interference due to manganese (II) and particulate matter, as described earlier for chlorine testing [
166]. Ozonation forms manganese dioxide colloids that readily oxidize DPD, yielding higher than actual ozone concentration values. And, hypobromous acid formed during ozonation of bromide-containing water also reacts with DPD [
166]. Interferences make the DPD field test for measuring ozone residual unreliable, unless significant pretreatment is employed to eliminate interfering species.
The indigo thiosulfate test is recognized by
Standard Methods [
169] for determination of ozone residual in water. Although manganese (IV) and (VII) can interfere with the indigo method [
167], the interferences are not as troublesome as for the DPD method [
170]. Corrections to the procedures can be made to account for interferences by manganese, chlorine, and bromide [
160].
Iodometric methods were the reference method used for ozone residual detection, but more recent studies have shown that this method measures all oxidative species present, and, therefore, overstates the amount of ozone present [
167,
168,
171]. This test is considered unreliable, and the recommendation is to use this method (and its variants) only for production testing where other species are not usually present [
167].
Amperometric electrodes are used for on-line measurement of ozone residual [
166,
167]. Bare electrodes that were initially used, suffered from interferences due to other oxidants, including bromine and iodine [
167]. The use of gas-permeable membranes, such as Teflon and, more recently, polymeric membranes, have increased selectivity and reduced electrode fouling [
167]. These membrane electrodes exhibit less than 2% interference due to bromine, hypobromous acid, hypochlorous acid, nitrogen trichloride (trichloramine), and chlorine dioxide [
172,
173], and are, therefore, more reliable than other test methods.
Dosing and Efficacy
Dosing and efficacy of ozone as a biocide can be very complex. The keys to successful ozonation are dose, mixing, and contact time [
174]. The design of the transfer mechanism (e.g., fine-bubble diffuser) and the contactor are critical to achieving the proper dosages, mixing, and contact time [
160]. The multifaceted nature of the target water, including the type and quantity of natural organic matter (NOM) present, temperature, alkalinity, and pH all contribute to the complexity [
175]. Suspended solids also impact efficacy, as effectiveness of ozone disinfection directly depends on physical contact of the ozone with the target microbes. Prefiltration is required to remove suspended solids that may obscure microbes from contacting ozone molecules [
176]. Ozone dosage is based the amount of ozone needed to inactivate microorganisms and that required to achieve the required disinfection contact time. Since the exact dosage may be difficult to determine due to variability of the target water source, the general practice is to dose enough ozone to achieve a scarcely measurable residual post disinfection [
160].
The degree of disinfection achieved, known as disinfection credits, is defined as percent log removal, such as 99% removal is equivalent to 2-log removal) [
160]. CT values are used to determine the concentration or contact time required for a specific disinfection credit. CT is calculated as oxidant residual multiplied by contact time, and are reported in mg-min/l. Contact time (in minutes) is defined as the period in which 10% of the feed water through a reactor has passed through, thus ensuring that 90% of the water has been exposed to the disinfectant [
160]. Because ozone decays much more rapidly than chlorine-based oxidants, the CT values for ozone tend to be less precise that those for other disinfectants [
177]. For ozone, the CT is measured at multiple locations within the contactor and the sum of the CT at various locations is used to determine the disinfection credit [
160]. CT values can also be used to compare the efficacy of different types of disinfectants.
Table 5,
Table 6 and
Table 7 show the disinfection credits along with CT values for ozone disinfection of
Giardia cysts, viruses, and
Cryptosporidium, respectively. As shown in the tables, ozone is more effective at higher temperatures. In general, every 10 °C increase in temperature hastens the kinetics of ozone disinfection by a factor of 2–3 [
177].
Table 8 compares the CT values for ozone as compared to chlorine-based disinfectants for 2-log (99%) inactivation of the indicated microorganism at 5 °C. As the table indicates, ozone is a more effective disinfectant for these organisms than chlorine-based compounds commonly used in water treatment.
Advantages and Limitations
There are a few of advantages of ozone over other disinfection methods. Ozone has greater disinfection efficacy than chlorine [
174], and relatively short contact times are required for inactivation of bacteria. Reactions of ozone and its radicals themselves do not persist as hazardous DBP, but do react with other species, primarily bromide and humic compounds, to yield some undesirable compounds.
The primary limitation is the sensitivity of polyamide membranes to oxidation by ozone. While it is well understood that ozone will destroy these membranes upon contact, there is relatively little literature detailing the interaction. Work by Glater et al. [
180] in 1983, is a definitive work on oxidation, including ozonation, of RO membranes, while Maugin [
145] in 2013, describes mechanisms for interaction and performance decay. Maugin [
145] and Glater, et al. [
180] found that ozone is more aggressive to the polyamide membrane than chlorine compounds. Tests with polyamide membranes demonstrated severe damage with exposure to 0.3 ppm ozone within 90 h (27 ppm-hours of exposure) [
180]. Maugin [
145] showed that loss of rejection and increase in permeability of polyamide membranes with ozone at a CT value of 0.015–0.02 millimoles-hour (mM-h) (61.2–81.6 mg-min/L or 1–1.4 ppm-hours) was equivalent to that of a chlorinated (as Cl
2) polyamide membrane at a CT value of 15–20 mM-h (63,000–84,000 mg-min/L or 1040–1400 ppm-hours (the typical value used to estimate membrane life when exposed to chlorine). Hence, the reaction of ozone with the polyamide membrane is 1000 times faster than for chlorine. Ozone destruction of polyamide membranes involves direct attack of the amide bounds in the polymer leading to depolymerization and ultimate destruction/removal of the polyamide layer [
145] (as opposed to the Orton Rearrangement mechanism for chlorine). As a result of ozone destruction of polyamide membranes, ozone removal prior to membrane systems is required [
50,
76].
Ozone generation inherently produces excess ozone (off gas), ozone that is not transferred to solution, and remains in the gases phase. This hazardous, corrosive gas must also be destroyed at the generator before discharge to the environment [
160,
164]. Several options are available for destroying the off-gas (known as ozone destruct systems), but thermal-catalytic destruction with a manganese dioxide catalyst is the most common technique [
160]. Other techniques include thermal destruction without catalytic destruction and catalytic destruction with metal catalysts [
164]. Sometimes the offgas is recycled to the head of the system (as shown in
Figure 11), but the benefits of this can be negligible due to the low concentration of ozone and the cost of the recycle equipment [
160]. Plus, recycling may reduce but not eliminate the need for destruction of the offgas [
160].
As mentioned previously, ozone and its radicals themselves don’t persist as hazardous DBPs, but reactions with other compounds, namely bromide, do yield problematic species. The reaction of ozone with bromide forms hypobromous acid [
155], which then further reacts with NOM [
181] to yield many bromoorganic compounds, including bromoform, bromoactetone, bromopicrin, dibromoacetonitrile, bromoacetic acid, bromoalkanes, and others [
182,
183,
184]. Fortunately, the concentrations of these compounds are generally much lower than current drinking water standards [
155].
One by-product of major concern is bromate, BrO
3−. Bromate is a genotoxic carcinogen that forms via a complicated reaction of ozone and its radicals with bromide [
155]; bromate formation steps include up to six oxidation states of bromine. The World Health Organization (WHO) established a guideline for bromate of 0.025 ppm in drinking water [
185]. The USEPA and European Union have established a maximum contaminant level of 0.010 ppm [
186,
187]. Several studies on bromate formation in ozonation facilities in both Europe and the USA were summarized by Pinkernell and von Guten [
188]. The findings show that most plants had bromate concentrations less than 0.010 ppm, but 6% had greater than this limit.
There are ways to minimize bromate formation, such as adding ammonia, which interferes ozone reactions with hypobromous acid, and pH depression. Both methods yield about a 50% reduction in bromate formation [
188,
189,
190,
191]. Once formed, however, bromate tends to persist, and removal is difficult [
188]. RO has been shown to reject about 96–97% of bromate in ozonated water [
189,
190,
191,
192]. Other removal methods tested are described by von Guten [
155]. These include activated carbon, which reduces bromate to bromide, but other anions and NOM negatively impact the efficacy of the technique. Biological activated carbon and the dosing of iron (II) also reduce bromate, but both techniques require a low-oxygen environment. Finally, UV radiation at 255 nm at a dose 100 times higher than required for disinfection will reduce both hypobromous acid and bromate.
Another concern with the use of ozone is the ozonation of humic compounds (humic and fulvic acids), yielding carboxylic acids and aldehydes [
77]. This oxidation process can result in a slight, net decrease in TOC [
77]. However, a net decrease in the ratio of high-molecular weight organics to low-molecular weight organics typically occurs. Thus, there is a net production of AOC following ozonation that serves to foster microbial growth [
6,
76,
77]. Biofiltration following ozone treatment can be used to reduce AOC and also reduce some of the bromo-organic compounds described earlier [
160]. Chlorine or chloramine would then need to be used downstream of the biofilter for secondary disinfection prior to a membrane system.
Ozone can, under the right conditions, decrease the potential for chloroform formation with secondary chlorination [
77]. However, analyzing and interpreting results from actual systems can be difficult due to the many variables that influence efficacy. These variables include the nature of the humic substances, ozone dosage, pH, chlorine dosage, chlorine pH of application, and bicarbonates that may or may not be present [
77]. Reckhow, et.al. [
193] demonstrated the reduction in formation of chloroform and total organic halides at neutral pH. But this work also shows that the potential for formation of dichloroacetic acid and trichloroacetone may increase [
193]. Despite this, it is believed that ozonation yields fewer undesirable DBPs than chlorination [
77].
Another limitation of ozone is that its high volatility yields little or no residual ozone for preventing regrowth downstream of the contact basin. This is particularly important if the microorganisms are shielded from ozone attack by particulates in the water. This, together with the ability of some microorganisms to regenerate themselves following ozone damage, make secondary disinfection a necessity for drinking water facilities. Secondary disinfection is not practical for pretreatment of membrane systems operating on “fresh” water supplies; however, secondary disinfection (ozone followed by chlorine-based compound(s)) may be encountered in wastewater reuse applications.
Due to the extremely corrosive nature of ozone, materials of construction are limited to 316 L for the ozone generator and wet- and dry-gas piping systems, and type 2 or type 5 Portland cement for the basin structures [
77]. Some plastics have good to excellent compatibility with ozone, including CPVC, HDPE, PVDF, and PTFE, while others, including polyamide and nitrile, have poor resistance to ozone and are “not recommended” for use [
194].
Removal of Ozone
There are a few ways of removing ozone from feed water to membrane systems that can be more reliable than assuming all the oxidant has dissipated. UV radiation and the use of quenching compounds are the most direct and simplistic methods. UV energy at 254 nm converts ozone to water and oxygen [
50]. Quenching compounds include peroxide, calcium thiosulfate, and sodium bisulfite. These stop the ozone and radical reactions [
160]. Two other methods, aeration and carbon filtration, pose hazards. Aeration requires environmental monitoring of the ozone that is stripped to the atmosphere. Additionally, adsorption on carbon media can result in fires and explosions, due to the exponential reaction of ozone with carbon; this hazard is exacerbated when the ozone is generated using pure oxygen [
160,
195].
Hydrogen Peroxide/Peracetic Acid
Hydrogen peroxide (peroxide) and the organic compound peracetic acid (chemical formula CH
3CO
3H, also known as PAA) are both electrophiles/oxidizing compounds that have higher oxidation potentials than free chlorine (see
Table 3). Therefore, they are damaging to polyamide membranes. PAA is an antimicrobial agent that dissociates in water to form peroxide and acetic acid [
196]. Peroxide together with PAA has demonstrated successful mitigation of biofouling on polyamide membranes [
197]. The solution is effective in penetrating stagnant areas within the membrane element [
87]. However, the mixture of peroxide and PAA must be used under very controlled conditions [
92,
198]. Conditions include a solution strength of less than 0.2 wt% peroxide and an application temperature of less than 25 °C. Transition metals and hydrogen sulfide must not be present. And application pH of 3–4 is recommended for optimal disinfection.
Due to the need to very controlled application, the recommendation is to use this solution on a periodic rather than continuous basis, with membrane exposure time limited to about 20 min [
197]. A solution is typically applied in recirculation/soak modes after cleaning. Cleaning should include a high pH solution to remove as much bio-based fouling as possible, followed by a low pH solution to remove any metals that may catalyze the oxidation of the membrane with the peroxide/PAA solution [
197]. While a polyamide membrane is limited to 20 min of exposure to the oxidizing solution [
197], work has shown that a soak of 2 h is required to provide a 90% kill and a 24-h soak is expected to kill 99% of bacteria [
198] with this type of solution. Thus, efficacy of this technique is limited.