Effects of Silica-Particle Coating on a Silica Support for the Fabrication of High-Performance Silicalite-1 Membranes by Gel-Free Steam-Assisted Conversion
Abstract
:1. Introduction
2. Materials and Methods
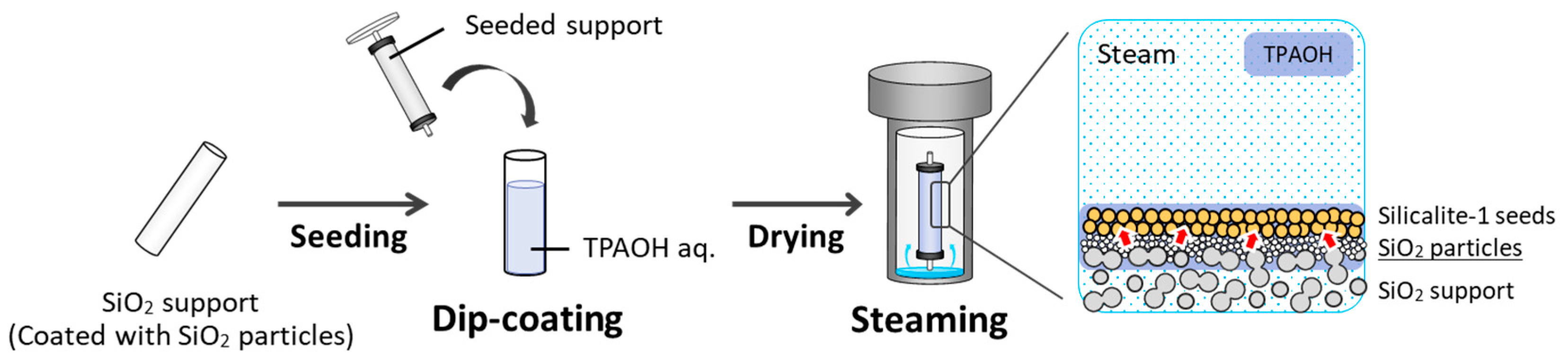
2.1. Formation of Silica-Particle Coating on Silica Support
2.2. Preparation of Seeded Silica-Particle-Coated Support by Electrophoretic Deposition
2.3. Synthesis of Silicalite-1 Membrane by Gel-Free SAC Method
2.4. Membrane Characterization
3. Results and Discussion
3.1. Silica-Particle-Coated Support
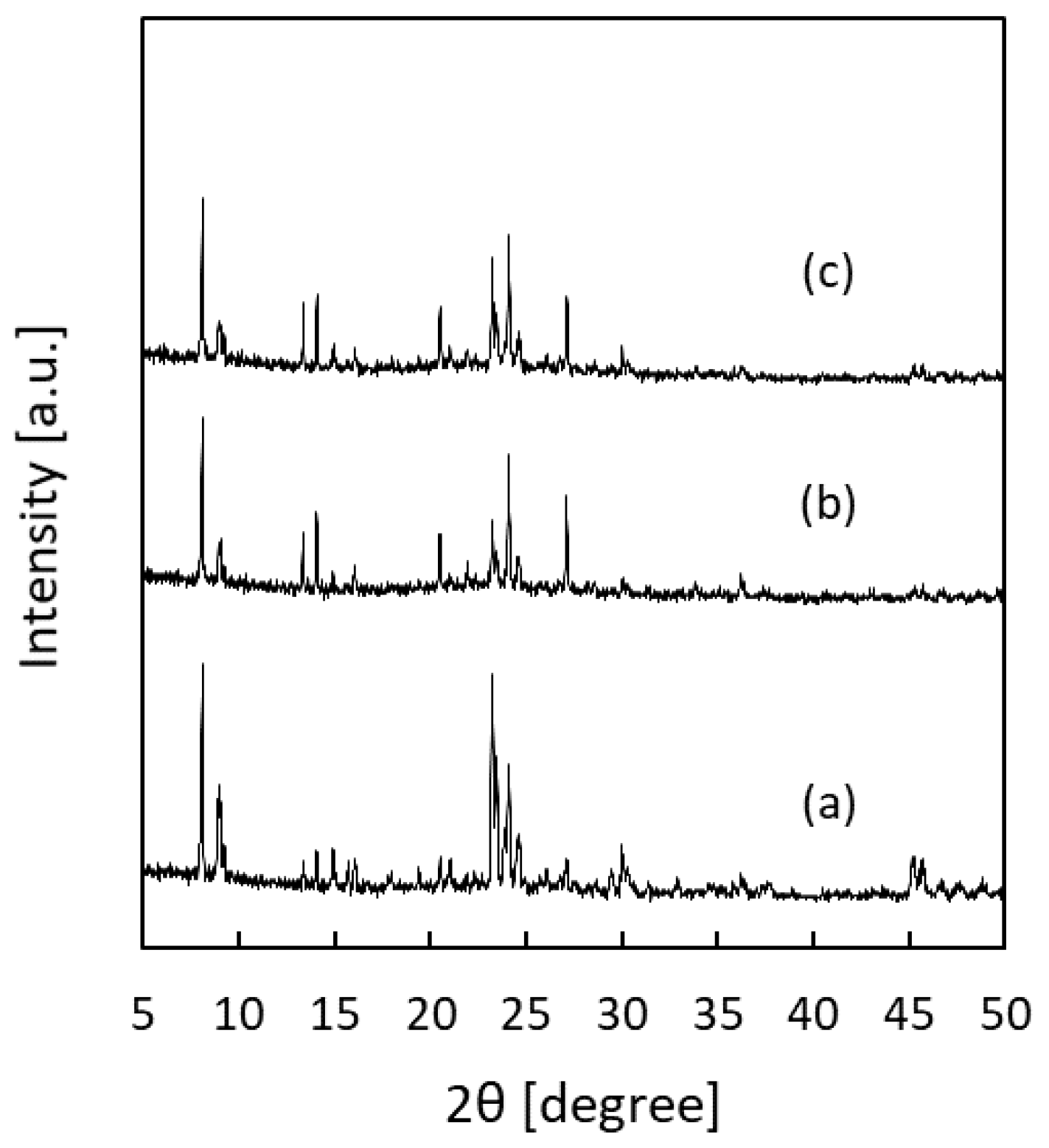
3.2. Silicalite-1 Membrane
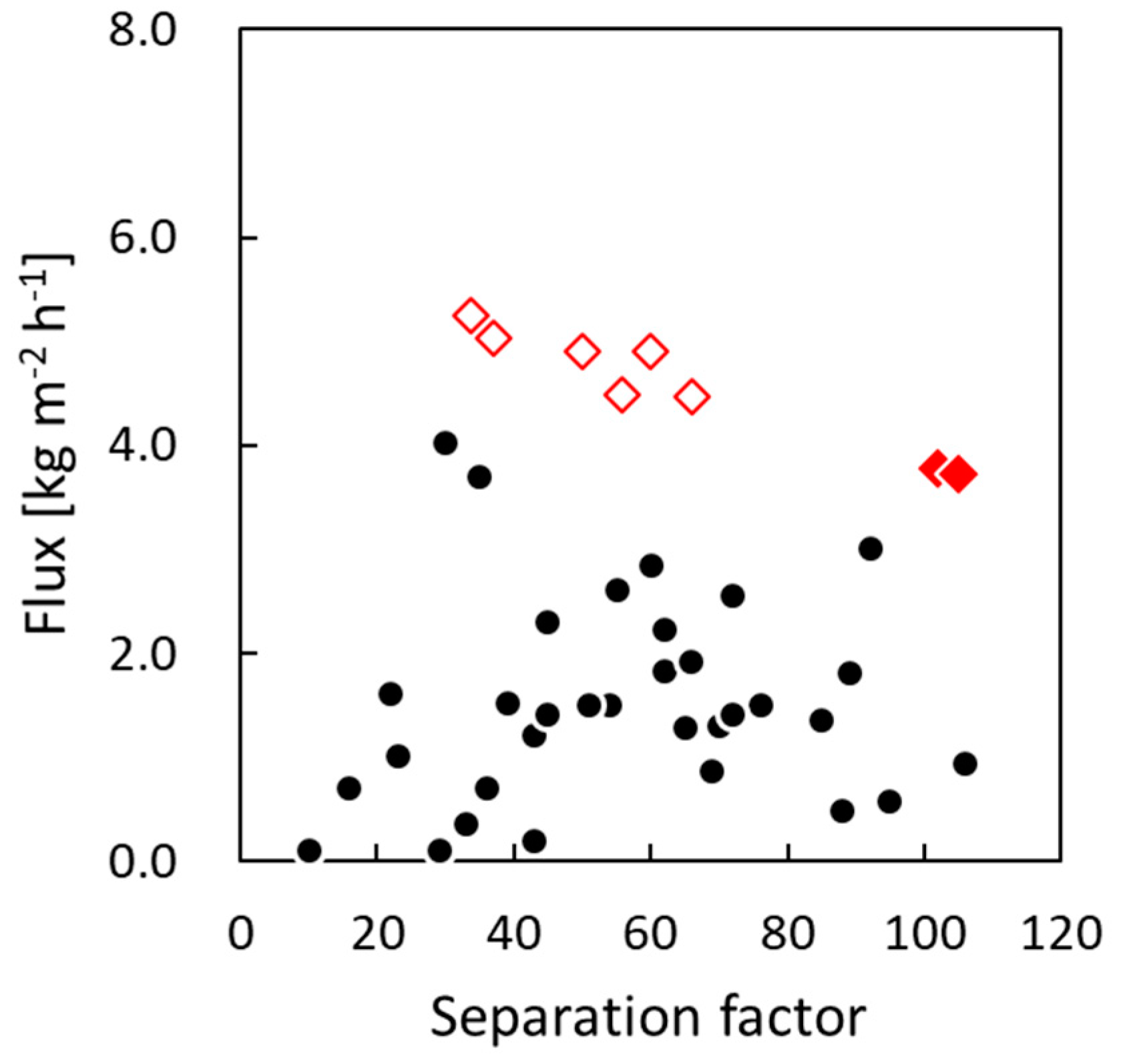
3.3. Pervaporation Performance
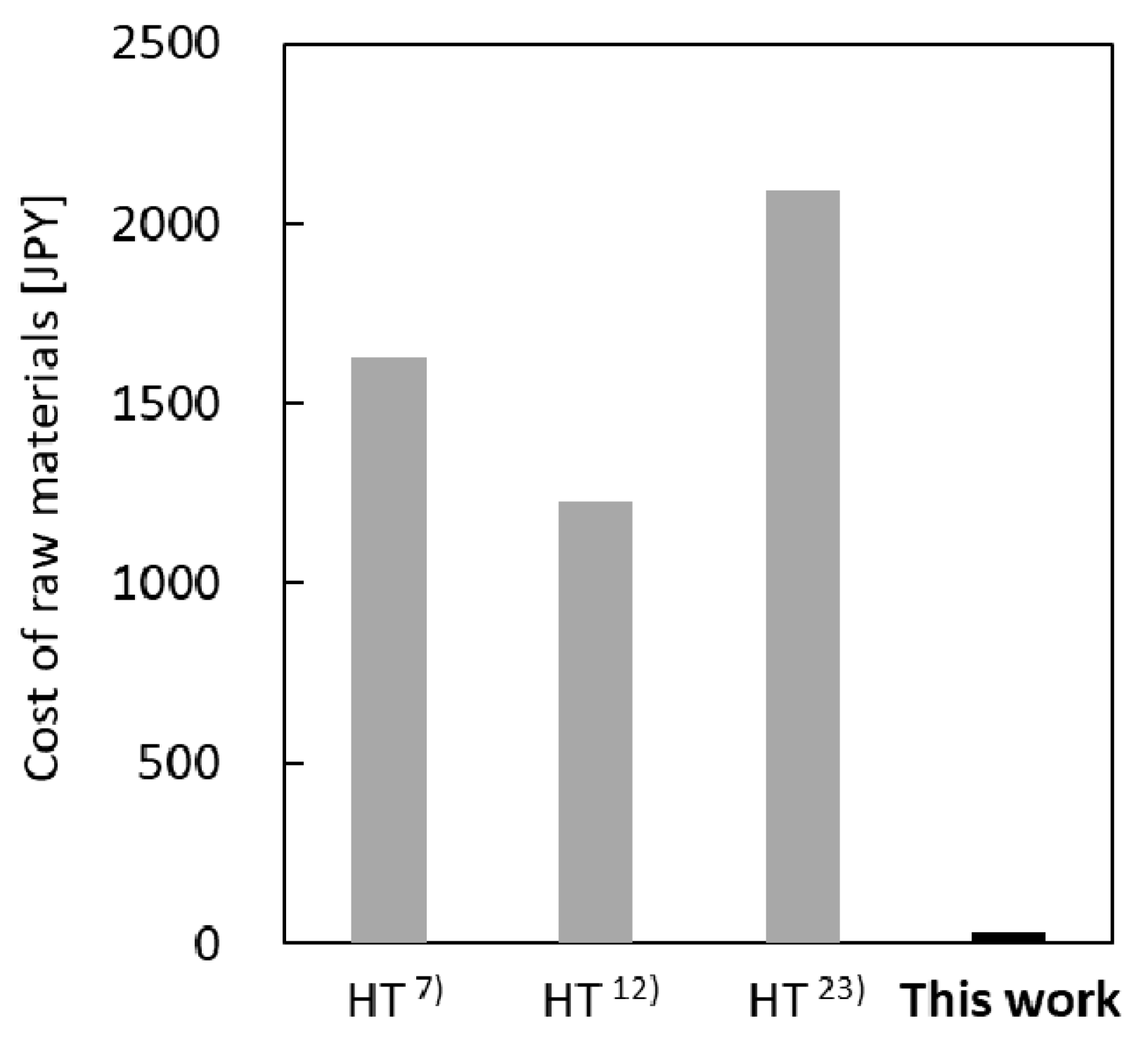
3.4. Cost of Synthesis of Silicalite-1 Membranes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tsapatsis, M. Toward high-throughput zeolite membranes. Science 2011, 334, 767–768. [Google Scholar] [CrossRef]
- Bowen, T.C.; Noble, R.D.; Falconer, J.L. Fundamentals and applications of pervaporation through zeolite membranes. J. Membr. Sci. 2004, 245, 1–33. [Google Scholar] [CrossRef]
- Tuan, V.A.; Li, S.; Falconer, J.L.; Noble, R.D. Separating organics from water by pervaporation with isomorphously-substituted MFI zeolite membranes. J. Membr. Sci. 2002, 196, 111–123. [Google Scholar] [CrossRef]
- Chen, H.; Li, Y.; Yang, W. Preparation of silicalite-1 membrane by solution-filling method and its alcohol extraction properties. J. Membr. Sci. 2007, 296, 122–130. [Google Scholar] [CrossRef]
- Chen, H.; Song, C.; Yang, W. Effects of aging on the synthesis and performance of silicalite membranes on silica tubes without seeding. Microporous Mesoporous Mater. 2007, 102, 249–257. [Google Scholar] [CrossRef]
- Lin, X.; Chen, X.; Kita, H.; Okamoto, K. Synthesis of silicalite tubular membranes by in situ crystallization. AIChE J. 2003, 49, 237–247. [Google Scholar] [CrossRef]
- Lin, X.; Kita, H.; Okamoto, K.I. Silicalite membrane preparation, characterization, and separation performance. Ind. Eng. Chem. Res. 2001, 40, 4069–4078. [Google Scholar] [CrossRef]
- Negishi, H.; Imura, T.; Kitamoto, D.; Ikegami, T.; Yanagishita, H.; Okamoto, M.; Idemoto, Y.; Koura, N.; Sano, T. Preparation of tubular silicalite membranes by hydrothermal synthesis with electrophoretic deposition as a seeding technique. J. Am. Ceram. Soc. 2006, 89, 124–130. [Google Scholar] [CrossRef]
- Stoeger, J.A.; Choi, J.; Tsapatsis, M. Rapid thermal processing and separation performance of columnar MFI membranes on porous stainless steel tubes. Energy Environ. Sci. 2011, 4, 3479–3486. [Google Scholar] [CrossRef]
- Soydas, B.; Dede, O.; Culfaz, A.; Kalipcilar, H. Separation of gas and organic/water mixtures by MFI type zeolite membranes synthesized in a flow system. Microporous Mesoporous Mater. 2010, 127, 96–103. [Google Scholar] [CrossRef]
- Shen, D.; Xiao, W.; Yang, J.; Chu, N.; Lu, J.; Yin, D.; Wang, J. Synthesis of silicalite-1 membrane with two silicon source by secondary growth method and its pervaporation performance. Sep. Purif. Technol. 2011, 76, 308–315. [Google Scholar] [CrossRef]
- Peng, Y.; Zhan, Z.; Shan, L.; Li, X.; Wang, Z.; Yan, Y. Preparation of zeolite MFI membranes on defective macroporous alumina supports by a novel wetting-rubbing seeding method: Role of wetting agent. J. Membr. Sci. 2013, 444, 60–69. [Google Scholar] [CrossRef]
- Chai, L.; Yang, J.; Wang, J.; Lu, J.; Yin, D.; Zhang, Y. Ethanol perm-selective B-ZSM-5 zeolite membranes from dilute solutions. AIChE J. 2016, 62, 2447–2458. [Google Scholar] [CrossRef]
- Peng, Y.; Lu, H.; Wang, Z.; Yan, Y. Microstructural optimization of MFI-type zeolite membranes for ethanol–water separation. J. Mater. Chem. A 2014, 2, 16093–16100. [Google Scholar] [CrossRef]
- Ueno, K.; Negishi, H.; Miyamoto, M.; Uemiya, S.; Oumi, Y. Effect of deposition seed crystal amount on the α-Al2O3 support and separation performance of silicalite-1 membranes for acetic acid/water mixtures. Sep. Purif. Technol. 2017, 174, 57–65. [Google Scholar] [CrossRef]
- Ueno, K.; Negishi, H.; Okuno, T.; Saito, T.; Tawarayama, H.; Ishikawa, S.; Miyamoto, M.; Uemiya, S.; Sawada, Y.; Oumi, Y. High-performance silicalite-1 membranes on porous tubular silica supports for separation of ethanol/water mixtures. Sep. Purif. Technol. 2017, 187, 343–354. [Google Scholar] [CrossRef]
- Lin, X.; Kita, H.; Okamoto, K.I. A novel method for the synthesis of high performance silicalite membranes. Chem. Commun. 2000, 19, 1889–1990. [Google Scholar] [CrossRef]
- Hasegawa, Y.; Kimura, K.; Nemoto, Y.; Nagase, T.; Kiyozumi, Y.; Nishide, T.; Mizukami, F. Real-time monitoring of permeation properties through polycrystalline MFI-type zeolite membranes during pervaporation using mass-spectrometry. Sep. Purif. Technol. 2008, 58, 386–392. [Google Scholar] [CrossRef]
- Zhang, X.; Zhu, M.; Zhou, R.; Chen, X.; Kita, H. Synthesis of silicalite-1 membranes with high ethanol permeation in ultradilute solution containing fluoride. Sep. Purif. Technol. 2011, 81, 480–484. [Google Scholar] [CrossRef]
- Zhang, X.L.; Zhu, M.H.; Zhou, R.F.; Chen, X.S.; Kita, H. Synthesis of a silicalite zeolite membrane in ultradilute solution and its highly selective separation of organic/water mixtures. Ind. Eng. Chem. Res. 2012, 51, 11499–11508. [Google Scholar] [CrossRef]
- Weyd, M.; Richter, H.; Puhlfürß, P.; Voigt, I.; Hamel, C.; Seidel-Morgenstern, A. Transport of binary water-ethanol mixtures through a multilayer hydrophobic zeolite membrane. J. Membr. Sci. 2008, 307, 239–248. [Google Scholar] [CrossRef]
- Ueno, K.; Negishi, H.; Okuno, T.; Saito, T.; Tawarayama, H.; Ishikawa, S.; Miyamoto, M.; Uemiya, S.; Sawada, Y.; Oumi, Y. A simple secondary growth method for the preparation of silicalite-1 membrane on a tubular silica support via gel-free steam-assisted conversion. J. Membr. Sci. 2017, 542, 150–158. [Google Scholar] [CrossRef]
- Shu, X.J.; Wang, X.R.; Kong, Q.Q.; Gu, X.H.; Xu, N.P. High-flux MFI zeolite membrane supported on YSZ hollow fiber for separation of ethanol/water. Ind. Eng. Chem. Res. 2012, 51, 12073–12080. [Google Scholar] [CrossRef]
- Ueno, K.; Horiguchi, Y.; Negishi, H.; Miyamoto, M.; Uemiya, S.; Takeno, A.; Sawada, S.; Oumi, Y. Fabrication of high-performance silicalite-1 membrane by a novel seeding method using zeolite-dispersed polymer film. Microporous Mesoporous Mater. 2018, 261, 58–62. [Google Scholar] [CrossRef]
- Ueno, K.; Negishi, H.; Miyamoto, M.; Uemiya, S.; Oumi, Y. Effect of Si/Al ratio and amount of deposited MFI-type seed crystals on the separation performance of silicalite-1 membranes for ethanol/water mixtures in the presence of succinic acid. Microporous Mesoporous Mater. 2018, 267, 1–8. [Google Scholar] [CrossRef]
- Pham, T.C.T.; Nguyen, T.H.; Yoon, K.B. Gel-free secondary growth of uniformly oriented silica MFI zeolite films and application for xylene separation. Angew. Chem. Int. Ed. 2013, 52, 8693–8698. [Google Scholar] [CrossRef]
- Agrawal, K.V.; Topuz, B.; Pham, T.C.T.; Nguyen, T.H.; Sauer, N.; Rangnekar, N.; Zhang, H.; Narasimharao, K.; Basahel, S.N.; Francis, L.F.; et al. Zeolite membranes: Oriented MFI membranes by gel-less secondary growth of sub-100 nm MFI-nanosheet seed layers. Adv. Mater. 2015, 27, 3243–3249. [Google Scholar] [CrossRef]
- Elyassi, B.; Jeon, M.Y.; Tsapatsis, M.; Narasimharao, K.; Basahel, S.N.; Al Thabaiti, S. Ethanol/water mixture pervaporation performance of b-oriented silicalite-1 membranes made by gel-free secondary growth. AIChE J. 2016, 62, 556–563. [Google Scholar] [CrossRef]
- Xia, S.; Peng, Y.; Wang, Z. Microstructure manipulation of MFI-type zeolite membranes on hollow fibers for ethanol–water separation. J. Membr. Sci. 2016, 498, 324–335. [Google Scholar] [CrossRef]


| Coating Material | Average Primary Particle Diameter [nm] | BET Specific Surface Area [g m−2] |
|---|---|---|
| Spherical silica | 100 | 22 |
| Fumed silica | 7 | 380 |
| Sample Name | Coating Material | Synthesis Time [h] | Separation Factor (α) | Membrane Selectivity (αmem) | Flux [kg m−2 h−1] | Ref. |
|---|---|---|---|---|---|---|
| M1 | Spherical silica | 24 | 105 | 10.6 | 3.72 | This study |
| M2 | Fumed silica | 16 | 102 | 10.3 | 3.76 | This study |
| M3 | – | 24 | 66 | 6.6 | 4.46 | [22] |
| M4 | – | 16 | 56 | 5.6 | 4.49 | [22] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ueno, K.; Negishi, H.; Okuno, T.; Tawarayama, H.; Ishikawa, S.; Miyamoto, M.; Uemiya, S.; Oumi, Y. Effects of Silica-Particle Coating on a Silica Support for the Fabrication of High-Performance Silicalite-1 Membranes by Gel-Free Steam-Assisted Conversion. Membranes 2019, 9, 46. https://doi.org/10.3390/membranes9040046
Ueno K, Negishi H, Okuno T, Tawarayama H, Ishikawa S, Miyamoto M, Uemiya S, Oumi Y. Effects of Silica-Particle Coating on a Silica Support for the Fabrication of High-Performance Silicalite-1 Membranes by Gel-Free Steam-Assisted Conversion. Membranes. 2019; 9(4):46. https://doi.org/10.3390/membranes9040046
Chicago/Turabian StyleUeno, Kyohei, Hideyuki Negishi, Takuya Okuno, Hiromasa Tawarayama, Shinji Ishikawa, Manabu Miyamoto, Shigeyuki Uemiya, and Yasunori Oumi. 2019. "Effects of Silica-Particle Coating on a Silica Support for the Fabrication of High-Performance Silicalite-1 Membranes by Gel-Free Steam-Assisted Conversion" Membranes 9, no. 4: 46. https://doi.org/10.3390/membranes9040046
APA StyleUeno, K., Negishi, H., Okuno, T., Tawarayama, H., Ishikawa, S., Miyamoto, M., Uemiya, S., & Oumi, Y. (2019). Effects of Silica-Particle Coating on a Silica Support for the Fabrication of High-Performance Silicalite-1 Membranes by Gel-Free Steam-Assisted Conversion. Membranes, 9(4), 46. https://doi.org/10.3390/membranes9040046






