A Review on Ionic Liquid Gas Separation Membranes
Abstract
1. Introduction
2. Supported Ionic Liquid Membranes
2.1. Structures and Membrane Preparation Techniques
2.1.1. Precursor Materials and Properties
2.1.2. Membrane Preparation
2.1.3. Long Term Stability
2.2. Pure Gas Transport Properties
- emerging alternatives are for instance alkene/alkane separation;
- the ionic liquids and supports are not changing significantly during the time;
- sometimes there are some trials for some more exotic anions like those based on amino acids or cholinium cations, but imidazolium-based ILs with the Tf2N anion dominate.
2.2.1. Effect Ionic Nature
2.2.2. Effect of Active Groups
2.2.3. Effect of Viscosity
2.2.4. Effect of the Water Vapor Content in the Gas Stream
2.2.5. Effect of Temperature
2.2.6. Effect of Pressure
2.3. Mixed Gas Transport Properties
| Support | Ionic Liquid | PCO2 [Barrer] (a) | α (CO2/N2) [-] | Ref. |
|---|---|---|---|---|
| Nanoporous alumina | [EMIM][Tf2N] | 44 GPU | 4 | [96] |
| [EMIM][TfA] | 43 GPU | 4.5 | ||
| [BMIM][Tf2N] | 44 GPU | 4.4 | ||
| [C3C1MIM][Tf2N] | 30 GPU | 3.0 | ||
| [BMIM][Ac] | 54 GPU | 5.4 | ||
| Porous Polyvinylidene fluoride (PVDF) | [EMIM][Ac] | 878.8 | 33.7 | [91] |
| [BMIM][Ac] | 851.9 | 34.6 | ||
| [Vbtma][Ac] | 1100 | 39 | ||
| Polydimethylsiloxane (PDMS)/ceramic composite | [EMIM][Ac] | 2000 | 152 | [44] |
| Polymer support | [Si-C1-C3-MIM][Tf2N] | 311 | 25 | [31] |
| Polyethersulfone (PES) | [BMIM][Pho] | 2540 | 127 | [92] |
| Porous alumina + phenolic resin | [EMIM][BF4] | 0.182 GPU | 40 | [45] |
| Hydrophilic PES | [Choline][Pro]/PEG200 1:0 | 343 | 35 | [95] |
| [Choline][Pro]/PEG200 1:1 | 810 | 28 | ||
| [Choline][Pro]/PEG200 1:2 | 1138 | 12 | ||
| Hydrophilic Polytetrafluoroethylene (PTFE) | [Choline][Lev] | 18 | 42 | [89] |
| [Choline][Lac] | 7 | 45 | ||
| [Choline][Gly] | 6 | 50 | ||
| [Choline][Mal] | 2 | 40 | ||
| Graphene oxide | [BMIM][BF4] | 68.5 GPU | 382 | [32] |
| MFFK—1 membrane | [BMIM][BF4] | 103 | 10 | [98] |
| [BMIM][doc] | 57 | 5 | ||
| Hydrophilic PES | [DMAPAH][EtOAc] | 3028 | 151 | [99] |
| [DMAPAH][TFA] | 3352 | 168 | ||
| PVDF | [(SiOSi)C1MIM][Tf2N] | 545 | 24 | [93] |
| [(SiOSi)C1MIM][C(CN)3] | 568 | 37 | ||
| Hydrophilic PVDF | [BMIM][DCA] | 4788 | 6 | [97] |
| [BMIM][Ac] | 520 | 23 | ||
| [BMIM][TfO] | 857 | 14 | ||
| [BMIM][Tf2N] | 3883 | 6 |
| Support | Ionic Liquid | Pi [Barrer] (a) | α (CO2/CH4) [-] | Ref. |
|---|---|---|---|---|
| Polypropylene | [EMIM][Tf2N] | 636 | 40.4 | [88] |
| [EMIM][TfO] | 1076 | 32.6 | ||
| [BMIM][Tf2N] | 936 | 74.3 | ||
| [BMIM][TfO] | 754 | 48.1 | ||
| Hydrophilic PTFE | [Ch][Lev] | 18 | 20 | [89] |
| [Ch][Lac] | 7 | 21 | ||
| [Ch][Gly] | 6 | 35 | ||
| [Ch][Mal] | 2 | 25 | ||
| Graphene oxide | [BMIM][BF4] | 68.5 GPU | 234 | [32] |
| PVDF | [BMIM][BF4] | 47 × 10−11 m2 s−1 (b) | 24 | [94] |
| PES | [BMIM][BF4] | 42 | 23 | |
| PVDF | [BMIM][Ac] | 38 | 18 | [94] |
| PES | [BMIM][Ac] | 33 | 15 | |
| PVDF | [OMIM][Ac] | 66 | 21 | [94] |
| PES | [OMIM][Ac] | 60 | 19 | |
| P-84 polyimide | [APTMS][Ac] | 23 GPU | 41 | [90] |
| MFFK—1 membrane | [BMIM][BF4] | 103 | 10 | [98] |
| [BMIM][doc] | 57 | 5 | ||
| hydrophilic PES | [DMAPAH][EtOAc] | 3028 | 72 | [99] |
| [DMAPAH][TFA] | 3352 | 67 | ||
| PVDF | [(SiOSi)C1MIM][Tf2N] | 545 | 10 | [93] |
| [(SiOSi)C1MIM][C(CN)3] | 568 | 11 | ||
| Hydrophilic PVDF | [BMIM][DCA] | 4788 | 2.5 | [97] |
| [BMIM][Ac] | 520 | 5 | ||
| [BMIM][TfO] | 857 | 11 | ||
| [BMIM][Tf2N] | 3883 | 14 | ||
| γ-Alumina | [EMIM][FAP] | 208 GPU | 3 | [100] |
| Mixture, i/j | Support | Ionic Liquid | Pi [Barrer] (a) | α (i/j) [-] | Ref. |
|---|---|---|---|---|---|
| CO2/H2 | Graphene oxide | [BMIM][BF4] | 68.5 GPU | 24 | [32] |
| H2S/CO2 | PVDF | [BMIM][Ac] | 5279 | 11.9 | [87] |
| [BMIM][BF4] | 3708 | 3.5 | |||
| H2S/CH4 | PVDF | [BMIM][Ac] | 5279 | 142 | [87] |
| [BMIM][BF4] | 3708 | 140 |
| Mixture, i/j | Support | Ionic Liquid | Pi [Barrer] (a) | α (i/j) [-] | Ref. |
|---|---|---|---|---|---|
| H2O/Air | Alumina on PVDF | [EMIM][BF4] | 26,000 | 1000 | [103] |
| [EMIM][DCA] | 46,000 | ||||
| [EMIM][ESU] | 46,000 | ||||
| Ethylene/ethane | PVDF | CuCl/[BMIM][Cl] | 3500 | 9 | [104] |
| Ethylene/ethane | PVDF | [EMIM][Me2PO4] +Ag | 100 | 35 | [105] |
| [EMIM][Et2PO4] + Ag | 80 | 40 | |||
| Propylene/propane | Porous alumina | [HMMIM][Tf2N] + Ag carriers | 280 | 9 | [106] |
| [HMIM][Tf2N] + Ag carriers | 450 | 7 |
3. Polymeric Ionic Liquids (PILs)
3.1. Structures of PILs
3.2. Synthesis of PILs
3.2.1. Polymerization of IL Monomers
3.2.2. Post-Functionalization of Existing Polymers
3.3. Pure Gas Transport
3.3.1. Gas Permeation
Influence of backbone
Influence of cation type and post-functionalization
Influence of counter anion
3.3.2. Sorption properties
| PIL | Ct. (a) | An. (b) | Back. (c) | p [bar] | T [°C] | Permeability [Barrer] (d) | Ref. | |||
|---|---|---|---|---|---|---|---|---|---|---|
| CO2 | CH4 | N2 | H2 | |||||||
| p[1-Methyl-3-[2-[(1-oxo-2-propenyl)oxy]ethyl]-imidazolium][Tf2N] | Im | [Tf2N] | Acry | 2 | 20 | 7 | 0.19 | 0.23 | [159] | |
| p[1 -Butyl-3-[2-[(1-oxo-2-propenyl)oxy]ethyl]-imidazolium][Tf2N] | Im | [Tf2N] | Acry | 2 | 20 | 22 | 0.97 | 0.71 | [159] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-methyl-imidazolium][Tf2N] | Im | [Tf2N] | Styr | 2 | 20 | 9.2 | 0.24 | 0.29 | [159] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-butyl-imidazolium][Tf2N] | Im | [Tf2N] | Styr | 2 | 20 | 20 | 0.91 | 0.67 | [159] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-hexyl-imidazolium][Tf2N] | Im | [Tf2N] | Styr | 2 | 20 | 32 | 2.3 | 1.4 | [159] | |
| p[C6 bis imidazolium][Tf2N] | Im | [Tf2N] | PIm | 2 | 20 | 4.4 | 0.16 | 0.2 | [229] | |
| p[PEG short bis imidazolium][Tf2N] | Im | [Tf2N] | PIm | 2 | 20 | 4 | 0.12 | 0.14 | [229] | |
| p[PEG long bis imidazolium][Tf2N] | Im | [Tf2N] | PIm | 2 | 20 | 3.8 | 0.13 | 0.16 | [229] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-[2-methoxyethyl]-imidazolium][Tf2N] | Im | [Tf2N] | Styr | 2 | 22 | 16 | 0.48 | 0.39 | [236] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-[2-(2-methoxy-ethoxy)-ethyl]imidazolium][Tf2N] | Im | [Tf2N] | Styr | 2 | 22 | 22 | 0.74 | 0.5 | [236] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-[4-cyano-butyl]-imidazolium][Tf2N] | Im | [Tf2N] | Styr | 2 | 22 | 4.1 | 0.11 | 0.11 | [236] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-[6-cyanohexyl]-imidazolium][Tf2N] | Im | [Tf2N] | Styr | 2 | 22 | 8.2 | 0.28 | 0.21 | [236] | |
| p[poly(imidazolium)s (imidazolium ionenes)with decyl ][Br] | Im | [Br] | PIm | 4 | RT | 0.13 | 0.84 | [228] | ||
| p[poly(imidazolium)s (imidazolium ionenes)with decyl ][Tf2N] | Im | [Tf2N] | PIm | 2 | RT | 5.3 | 0.26 | 0.22 | 5.3 | [228] |
| p[veIm][DCA] | Im | [DCA] | V | 10 | 35 | 0.09 | [222] | |||
| p[vbIm][DCA] | Im | [DCA] | V | 1 | 35 | 4.24 | 0.16 | [222] | ||
| p[vhIm][DCA] | Im | [DCA] | V | 1 | 35 | 33.5 | 1.6 | [222] | ||
| p[vmIm][Tf2N] | Im | [Tf2N] | V | 2 | RT | 4.8 | 0.1 | 0.2 | [223] | |
| p[vhIm][Tf2N] | Im | [Tf2N] | V | 2 | RT | 69 | 7.0 | 4.1 | [223] | |
| p[1-Vinyl-3-[2-(2-methoxy-ethoxy)-ethyl]imidazolium][Tf2N] | Im | [Tf2N] | V | 2 | RT | 14 | 0.4 | 0.4 | [223] | |
| p[1-Vinyl-3-[2-[2-(2-methoxy-ethoxy)-ethoxy]-ethyl]]- imidazolium][Tf2N] | Im | [Tf2N] | V | 2 | RT | 26 | 1.0 | 0.8 | [223] | |
| p[1-Vinyl-3-(3,3,4,4,5,5,6,6,6-Nonafluorohexyl)-imidazolium][Tf2N] | Im | [Tf2N] | V | 2 | RT | 69 | 4.9 | 6.3 | [223] | |
| p[1-Vinyl-3-(1,1,3,3,3-pentamethyl-disiloxanylmethyl)-imidazolium][Tf2N] | Im | [Tf2N] | V | 2 | RT | 130 | 14.9 | 9.3 | [223] | |
| p[1-[p-(2-(3-methylimidazolium)ethoxy)]phenyl]-2-[p-(trimethylsilyl)phenyl]acetylen][Br] | Im | [Br] | Ac | 1 | 25 | 11 | 0.25 | [226] | ||
| p[1-[p-(2-(3-methylimidazolium)ethoxy)]phenyl]-2-[p-(trimethylsilyl)phenyl]acetylen][Tf2N] | Im | [Tf2N] | Ac | 1 | 25 | 53 | 1.7 | [226] | ||
| p[1-[p-(3-methylimidazolium)ethoxy)phenyl]-2-phenylacetylene][Tf2N] | Im | [Tf2N] | Ac | 1 | 25 | 8.6 | 0.25 | [226] | ||
| p[1-[p-(2-(3-methylimidazolium)ethoxy)]phenyl]-2-[p-(trimethylsilyl)phenyl]acetylen][TfAc] | Im | [TfAc] | Ac | 1 | 25 | 23 | 0.67 | [226] | ||
| p[1-[p-(3-methylimidazolium)ethoxy)phenyl]-2-phenylacetylene][TfAc] | Im | [TfAc] | Ac | 1 | 25 | 1.5 | 0.04 | [226] | ||
| p[diallyldimethylammonium][Tf2N] | Pyrr | [Tf2N] | Pyrr | 1 | 21 | 5.09 | 0.18 | 0.23 | [235] | |
| [TMPBI-BuI][BF4] | BIm | [BF4] | PBI | 19 | 35 | 4.78 | 0.30 | 0.61 | [231] | |
| [TMPBI-BuI][Tf2N] | BIm | [Tf2N] | PBI | 19 | 35 | 17.9 | 1.24 | 2.27 | [231] | |
| pMEBIm[Tf2N] | Im | [Tf2N] | Acry | 1 | 25 | 22.8 | 0.94 | 0.8 | [225] | |
| pMEBIm[Tf2N] | Im | [Tf2N] | Acry | 1 | 35 | 33.9 | 1.99 | 1.41 | [225] | |
| pMEBIm[Tf2N] | Im | [Tf2N] | Acry | 1 | 45 | 46.5 | 3.14 | 2.26 | [225] | |
| pMMA-b-MUBIm[Tf2N] | Im | [Tf2N] | Acry | 1 | 25 | 25.8 | 1.73 | 0.98 | [225] | |
| pMMA-b-MUBIm[Tf2N] | Im | [Tf2N] | Acry | 1 | 35 | 33.7 | 2.73 | 1.64 | [225] | |
| pMMA-b-MUBIm[Tf2N] | Im | [Tf2N] | Acry | 1 | 45 | 47.5 | 4.58 | 2.67 | [225] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-isopropyl-imidazolium][Tf2N] | Im | [Tf2N] | Styr | 3 | 20 | 10.4 | 0.35 | 0.33 | [224] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-sec-butyl-imidazolium][Tf2N] | Im | [Tf2N] | Styr | 3 | 20 | 13.6 | 0.55 | 0.39 | [224] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-methylcyclopropyl-imidazolium][Tf2N] | Im | [Tf2N] | Styr | 3 | 20 | 7.94 | 0.32 | 0.24 | [224] | |
| p[1-[(4-Ethenylphenyl)methyl]-3-cyclopentyl-imidazolium][Tf2N] | Im | [Tf2N] | Styr | 3 | 20 | 6.65 | 0.25 | 0.19 | [224] | |
| p[DPyDBzPBI-BuI][BF4] | BIm | [BF4] | PBI | 20 | 35 | 30 | 0.92 | 1.32 | 29.8 | [232] |
| p[DAnDBzPBI-BuI][BF4] | BIm | [BF4] | PBI | 20 | 35 | 25.1 | 0.75 | 0.92 | 23.3 | [232] |
| p[DPyDBzPBI-BuI][Br] | BIm | [Br] | PBI | 20 | 35 | 23.9 | 0.82 | 1.11 | 24.8 | [232] |
| p[DAnDBzPBI-BuI][Br] | BIm | [Br] | PBI | 20 | 35 | 15 | 0.49 | 0.62 | 15.3 | [232] |
| p[DPyDBzPBI-BuI][Tf2N] | BIm | [Tf2N] | PBI | 20 | 35 | 36.2 | 1.25 | 1.81 | 36.5 | [232] |
| p[DAnDBzPBI-BuI][Tf2N] | BIm | [Tf2N] | PBI | 20 | 35 | 30.9 | 1.04 | 1.43 | 28.7 | [232] |
| p[6FDA-diethyl-Benzimidazolium][Tf2N] | BIm | [Tf2N] | PBI | 1 | 20 | 28.9 | 7.25 | 7.23 | [233] | |
| p[0.5 cross-linked polyether-based 1,2,3-triazolium][I] | Tr | [I] | PEt | RT | 113.2 | 6 | [197] | |||
| p[1 cross-linked polyether-based 1,2,3-triazolium][I] | Tr | [I] | PEt | RT | 86 | 2.1 | [197] | |||
| p[2 cross-linked polyether-based 1,2,3-triazolium][I] | Tr | [I] | PEt | RT | 96.5 | 2.7 | [197] | |||
| p[1 cross-linked polyether-based 1,2,3-triazolium][Tf2N] | Tr | [Tf2N] | PEt | RT | 59.2 | 1.2 | [197] | |||
| p[0.2-PEG-Im-PI][Br] | Im | [Br] | PI | 2 | 30 | 464 | 17.4 | 24.1 | [227] | |
| p[0.5-PEG-Im-PI][Br] | Im | [Br] | PI | 2 | 30 | 177 | 4.72 | 5.87 | [227] | |
| p[0.8-PEG-Im-PI][Br] | Im | [Br] | PI | 2 | 30 | 72 | 1.71 | 1.94 | [227] | |
| p[0.5-Butyl-Im-PI][Br] | Im | [Br] | PI | 2 | 30 | 219 | 7.84 | 11 | [227] | |
| p[1,2,3-triazolium-based linear ionic polyurethane (Isophorone diisocyanate)][Tf2N] | Tr | [Tf2N] | PU | 2 | 20 | 2.31 | [234] | |||
| p[1,2,3-triazolium-based linear ionic polyurethane (toluene diisocyanate)][Tf2N] | Tr | [Tf2N] | PU | 2 | 20 | 2.25 | [234] | |||
| p[voim]/PVA IPN 50%[PF6] | Im | [PF6] | V | 1.2 | 20 | 18.43 | 0.26 | [220] | ||
| p[voim]/PVA IPN 50%[PF6] | Im | [PF6] | V | 1.2 | 30 | 29.47 | 0.46 | [220] | ||
| p[voim]/PVA IPN 50%[PF6] | Im | [PF6] | V | 1.2 | 40 | 38.86 | 0.74 | [220] | ||
| p[voim]/PVA IPN 50%[PF6] | Im | [PF6] | V | 1.2 | 50 | 41.72 | 0.83 | [220] | ||
| p[voim]/PVA IPN 50%[PF6] | Im | [PF6] | V | 1.2 | 60 | 42.72 | 0.89 | [220] | ||
| p[voim]/PVA IPN 50%[PF6] | Im | [PF6] | V | 1.2 | 70 | 43.88 | 0.92 | [220] | ||
| p[Polyimide-imidazolium][Tf2N] | Im | [Tf2N]] | PI | 3 | RT | 0.87 | 0.028 | 0.027 | 1.62 | [230] |
| p[Aromatic polyether copolymer pyridinium ][BF4] | Pyr | [BF4] | Ar-PEt | 30 | 7.2 | 0.099 | <0.1 | 13.5 | [237] | |
| p[Aromatic polyether copolymer pyridinium ][MeSO4] | Pyr | [MeSO4] | Ar-PEt | 30 | 0.9 | 0.05 | <0.1 | 4 | [237] | |
| p[Aromatic polyether copolymer pyridinium ][Tf2N] | Pyr | [Tf2N] | Ar-PEt | 30 | 13.7 | 0.3 | <0.1 | 13.8 | [237] | |
3.4. Mixed Gas Transport
4. Polymer/IL Blends; Ion Gels
4.1. Introduction
4.2. Conventional Glassy and Crystalline Polymers
4.2.1. Glassy High-Performance Polymers
4.2.2. PIM/IL Blends
4.2.3. Semi-Crystalline Fluoropolymers
4.2.4. Thermoplastic Elastomers
4.2.5. Di- and Tri-Block Copolymers
4.3. Chemically Cross-Linked Gels
4.3.1. Acrylic Networks
4.3.2. Epoxy-Based Networks
4.3.3. Cross-Linked Polyether Networks
4.4. Facilitated Transport Polymer/IL Gels
4.4.1. Fixed-Site Carriers
4.4.2. Mobile Carriers
| Polymer | Ionic Liquid | Gases (a) | Ref. |
|---|---|---|---|
| Linear polymers/IL blends | |||
| Various polymers (hydrophilic) | |||
| Gelatin | [EMIM][DCA], [BMIM][DCA], [BMPyr][DCA] | H2, N2, O2, CO2, CH4 | [258] |
| Cellulose Triacetate | [EMIM][BF4], [EMIM][DCA] | N2, CH4, CO2 | [259] |
| Polyvinylalcohol, PVA | [EMIM][DCA] | H2, CO2 | [51] |
| Glassy high-performance polymers | |||
| Polyethersulfone | [EMIM][Tf2N] | CH4, CO2 | [260] |
| Matrimid®5218 polyimide | [EMIM][Tf2N] | CH4, CO2 | [261] |
| 6FDA-TeMPD polyimide | [BMIM][Tf2N] | H2, N2, CH4, CO, CO2 | [262] |
| Polybenzimidazole (PBI) | [BMIM][Tf2N] | H2, N2, CH4, CO, CO2 (Elevated T) | [263] |
| Poly(pyromellitimide-co-4,4′-oxydianiline) (PMDA-ODA PI) | [BMIM][Tf2N] | H2, N2, CH4, CO, CO2 (Elevated T) | [263] |
| Sulfonated polyimide SPI-[Tf2N] | [BMIM][Tf2N] | N2, CO2 | [264] |
| Sulfonated polyimide SPI-[PF6] | [BMIM][PF6] | N2, CO2 | [264] |
| High free volume polymers | |||
| PIM-1 | [C2MIM][Tf2N], [C4MIM][Tf2N], [C6MIM][Tf2N] | O2, N2, CH4, CO2 | [267] |
| PIM-1/PEG functionalized anthracene maleimide comonomer | [C6MIM][Tf2N] | O2, N2, CH4, CO2 | [267] |
| PIM-1 | [BMIM]2[Co(NCS)4] (on PAN) | O2, N2 | [268] |
| Tetrazole-modified PIM-1 | [Methylammonium], [Diisopropylamonium], [N,N-diisopropylethylamonium] | O2, N2, CO2 | [123] |
| Semi-crystalline fluoropolymers | |||
| PVDF | [EMIM][B(CN)4] | H2, N2, CO2 CO2/H2, CO2/N2 | [269] |
| [BMIM][PF6], [BMIM][Tf2N], [BMIM][B(CN)4] | CO2 (modelling) | [270] | |
| Poly(VDF-HFP) | [EMIM][BF4] | CO2/N2 | [255] |
| [EMIM][TFSI] | He, H2, O2, N2, CH4, CO2 | [254] | |
| [EMIM][TFSI] | He, H2, O2, N2, CH4, CO2Butane, pentane, hexane, cyclohexane, heptane, toluene, MeOH, EtOH, PrOH, BuOH, H2O | [256] | |
| [EMIM][Tf2N], [HMIM][Tf2N] | CH4, CO2 | [271] | |
| [EMIM][TFSI]/[HdMIM][TFSI] | He, H2, O2, N2, CH4, CO2 R-(+)-limonene/S-(+)-carvone/EtOH(/H2O) | [272] | |
| [EMIM][Tf2N] | EtOH | [273] | |
| [EMIM][Tf2N] | Hexane, isooctane | [274] | |
| Thermoplastic elastomers | |||
| Pebax® 2533 | [BMIM][CF3SO3] | He, H2, O2, N2, CH4, CO2 | [257] |
| [TETA][Tfa] | CO2/N2 | [275] | |
| [EMIM][EtSO4] | H2S, CH4, CO2, Air | [276] | |
| Pebax® 1657 | [BMIM][CF3SO3] | He, H2, O2, N2, CH4, CO2 | [257] |
| [EMIM][BF4] | H2, N2, CH4, CO2 | [277] | |
| [EMIM][BF4] (on PVDF/PTMSP HF) | N2, CH4, CO2 CO2/N2, CO2/CH4 | [278] | |
| [BMIM][BF4] | N2, CH4, CO2 | [281] | |
| [BMIM][BF4] | He, H2, O2, N2, CH4, CO2, CO2/N2, CO2/CH4 | [280] | |
| [EMIM][Gly], [BMIM][Gly], [HMIM][Gly], [BMIM][Ac], [BMIM][BF4] | N2, CH4, CO2 CO2/N2, CO2/CH4 | [302] | |
| [BMIM][Tf2N] | H2, N2, CH4, CO2 | [279] | |
| [EMIM][SCN], [EMIM][BF4], [EMIM][OTf], [EMIM][Tf2N] | CH2CF2 (R32), CF3CHF2 (R125) CH2CF2/CF3CHF2 (R410A) | [282] | |
| Pebax® 1074 | [BMIM][PF6] | CH4, CO2 | [303] |
| PolyActive™ (4 grades) 1500PEOT77PBT23, 4000PEOT77PBT23, 1000PEOT55PBT45, 4000PEOT50PBT50 | [BMIM][Tf2N] | He, H2, O2, N2, CH4, CO2CO2/N2, CO2/CH4 | [284] |
| Di- and tri-block copolymers | |||
| Poly(styrene-block-ethylene oxide-block-styrene) (SEOS) | [HMIM][Tf2N], [HMIM][BF4], [HMIM][PF6] (on porous PA) | CH4, CO2 | [285] |
| [EMIM][TCM], [BMIM][TCM], [HMIM][TCM] (on porous PA) | N2, CH4, CO2 | [286] | |
| Poly(styrene-b-ethylene oxide-b-styrene) (SOS) | [EMIM][TFSA] (on PVDF) | N2, CH4, CO2 | [287] |
| Poly(styrene-b-Methyl methacrylate-b-styrene) (SMS) | [EMIM][TFSA] (on PVDF porous support) | N2, CH4, CO2 CO2/N2, CO2/CH4 | [287] |
| Polystyrene-b-poly(ethylene oxide) diblock copolymer/polystyrene-b-poly(ethylene oxide)-b-polystyrene triblock copolymer (SO/SOS) | [EMIM][TFSI] | N2, CH4, CO2 | [288] |
| Poly[tert-butylstyrene-b-(ethylene-alt-propylene)-b-(styrene-co-styrenesulfonate)-b-(ethylene-alt-propylene)-b-tert-butylstyrene] pentablock copolymer (Nexar) | [BMIM][BF4] | N2, CO2 (H2O) CO2/N2 (H2O) | [289] |
| Low molecular weight gelators | |||
| Aspartame-based, low molecular-weight organic gelator | [EMIM][Tf2N], [HMIM][Tf2N] | N2, CO2 | [251] |
| 12-hydroxystearic acid | [EMIM][Tf2N]/AgTf2N | C3H6/C3H8 | [301] |
| Network polymer/IL blends | |||
| Poly(dimethylacrylamide) (PDMAAm), poly(vinylpyrrolidone) (PVP), poly(2-hydroxyethylmethacrylate) (PHEMA), poly(methylacrylate) (PMA), poly(ethylacrylate) (PEA), +ethylene glycol dimethacrylate crosslinker | [P4444][Gly], [P4444][Ser], [P4444][Lys], [P4444][Pro] | N2, CO2 CO2/N2 | [290] |
| (1) Poly(2-acrylamido-2-methyl-1-propanesulfonic acid) (PAMPS) (2) Polyvinylpyrrolidone (PVP) or polydimethylacrylamide (PDMAAm) + N,N’-methylenebisacrylamide (MBAA) crosslinker | [P4444][Pro], [P2225][Pro] | CO2/N2 | [291] |
| (1) Poly(2-acrylamido-2-methylpropanesulfonic acid) (PAMPS) (2) Poly(dimethylacrylamide) (PDMAAm) | [P4444][Pro] | CO2/N2 (H2O) | [292] |
| (1) Poly(methacryloylamino propyl trimethylammonium chloride) (PMAPTAC) + N,N′-methylenebis(acrylamide) (MBAA) crosslinker (2) Poly(dimethylacrylamide) (PDMAAm) | [P222(1O1)][Inda] | CO2/N2 | [293] |
| (1) 1,3-bis(2-oxiranyl-ethyl)imidazolium [Tf2N] (2) Tris(aminoethylamine) (TAEA) | [EMIM][Tf2N] | N2, CH4, CO2 | [145] |
| [EMIM][Tf2N], [EMIM][DCA] | CO2/N2 (+H2O) | [188] | |
| [EMIM][Tf2N] | H2, He, N2, O2, CH4, and CO2 CO2/CH4 (+H2O) | [189] | |
| (1) Polydimethylacrylamide (PDMAAm) (2) N,N′-methylenebis(acrylamide) (MBAA) crosslinker (with silica nanoparticles, poly(TEOS)) | [EMIM][Tf2N], [BMIM][Tf2N], [HMIM][Tf2N], [EMIM][B(CN)4], [BMIM][BF4], [P2225][Tf2N], [N2225][Tf2N] | CO2/N2 | [295] |
| [BMIM][Tf2N] | CO2/N2 (+H2O) | [294] | |
| Elvaloy4170, Reactive Ethylene Terpolymer | [EMIM][Tf2N] | [296] | |
| (1) Ethoxylated (20) trimethylolpropane triacrylate(2) 3,6-dioxa-1,8-octanethiol | [EMIM][Tf2N], [HMIM][Tf2N], [iBMIM][Tf2N], [HEMIM][Tf2N], [C3CNMIM][Tf2N], [C5CNMIM][Tf2N], [TMSMIM][Tf2N], [BzMIM][Tf2N], [BzBIM][Tf2N], [EGMIM][Tf2N], [EG2MIM][Tf2N], [EG3MIM][Tf2N] | N2, CO2 | [297] |
| (1) Poly(ethylene glycol) diacrylate (2) Poly(mercaptopropyl) methyl siloxane | [EMIM][AC], [EMIM][DCA], [EMIM][BF4], [EMIM][Tf2N], [BMPyrr][Tf2N], [EMPy][Tf2N], [P1444][Tf2N], [N4111][Tf2N] | N2, CO2 | [299] |
| (1) Poly(ethylene glycol) diacrylate, Bisphenol A ethoxylate diacrylate and Trimethylolpropane ethoxylate triacrylate (2) Di(trimethylolpropane) tetraacrylate and dipentaerythritol penta-/hexa-acrylate | [EMIM][Tf2N] | N2, CO2 | [298] |
| Facilitated transport membranes | |||
| PIM-1 | [BMIM]2[Co(NCS)4] | O2, N2 | [268] |
| Poly([C4vim][Tf2N]) | [C4MIM][Cl], [C4MIM][CuCl2] | CO2, H2, N2, CO N2/CO | [300] |
| Poly[pyridinium-1,4-diyliminocarbonyl-1,4-phenylene-methylene Tf2N−] | [EMIM][Tf2N]/AgTf2N | C3H6/C3H8 | [301] |
| Poly[pyridinium-1,4-diyliminocarbonyl-1,4-phenylene-methylene PF6−] | [EMIM][BF4]/AgBF4 | C3H6/C3H8 | [301] |
| 12-hydroxystearic acid | [EMIM][Tf2N]/AgTf2N | C3H6/C3H8 | [301] |
5. PIL/IL Blends
6. Miscellaneous Uses of Ionic Liquids and Poly(ionic Liquids) Related to Gas Separation
6.1. Mixed Matrix Membranes Incorporating ILs
6.2. Porous Ionic Liquid Materials
6.3. Gas Detection
6.4. CO2 Absorption and Capturing
- (i)
- with ammonium and imidazolium cations and [Tf2N], [BF4], [PF6], [DCA], and acetate anions,
- (ii)
- rigid polystyrene backbone,
- (iii)
- high surface area and porosity, together with high separation efficiency [243].
7. Conclusions
Further Outlook
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Symbols and Abbreviations
| Symbols | |
| D | Diffusion coefficient (m2 s−1) |
| pKa | Acid dissociation constant (-) |
| P | Permeability coefficient (Barrer) |
| S | Solubility coefficient (cm3STP cm−3 bar−1) |
| Vw | Van der Waals volume (cm3 mol−1) |
| α | Permselectivity (-) |
| Miscellaneous Abbreviations | |
| AFM | Atomic force microscopy |
| AIBN | 2,2-azobisisobutyronitrile |
| ATRP | Atom transfer radical polymerization |
| BET | Brunauer-Emmett-Teller |
| C-G | Chain growth polymerization |
| CHI | Chitosan |
| COSMO | Conductor-like screening model |
| DMPA | 2,2-dimethoxy-2-phenylacetophenone |
| EDX | Energy dispersive X-ray analysis |
| FRP | Free radical polymerization |
| GPU | Gas permeation unit or Gas permeance unit |
| HA | Hyaluronic acid or Sodium hyaluronate |
| IL | Ionic liquid |
| ILM | Ionic liquid membranes |
| LMOG | Low molecular-weight organic gelators |
| MBAA | N,N′-methylenebis(acrylamide) |
| MOF | Metalorganic framework |
| MPa | Mega pascal |
| PIL | Polymerized ionic liquid or Poly(ionic liquid) |
| PIM | Polymer of intrinsic microporosity |
| RAFT | Reversible addition-fragmentation chain transfer |
| RH | Relative humidity |
| ROMP | Ring-opening metathesis polymerization |
| RTIL | Room-temperature ionic liquids |
| SCFD | Supercritical fluid deposition |
| SD | Solution-diffusion |
| SEM | Scanning electron microscopy |
| S-G | Step growth polymerization |
| SILM | Supported ionic liquid membrane |
| TGA | Thermogravimetric analysis |
| UV | Ultra-violet radiation |
| VOC | Volatile organic compound |
| XDC | p-Xylylene dichloride |
| XPS | X-ray photoelectron spectroscopy |
| Ionic Liquids | |
| [(SiOSi)C1MIM][C(CN)3] | 1-Methyl-3-pentamethyldisiloxymethylimidazolium Tricyanomethanide |
| [(SiOSi)C1MIM][Tf2N] | 1-Methyl-3-pentamethyldisiloxymethylimidazolium Bis(trifluoromethylsulfonyl)imide |
| [APTMS][Ac] | (3-Aminopropyl)-trimethoxysilane Acetate |
| [BMIM][Ac] | 1-Butyl-3-methylimidazolium Acetate |
| [BMIM][BF4] | 1-Butyl-3-methylimidazolium Tetrafluoroborate |
| [BMIM][DCA] | 1-butyl-3-methylimidazolium Dicyanamide |
| [BMIM][Doc] | 1-Butyl-3-methylimidazolium bis(2-ethylhexyl)sulfosuccinate |
| [BMIM][Pho] | 1-Butyl-3-methylimidazolium Phenolate |
| [BMIM][Tf2N] | 1-butyl-3-methylimidazolium Bis(trifluoromethylsulfonyl)imide |
| [BMIM][TfO] | 1-butyl-3-methylimidazolium Trifluoromethane sulfonate |
| [C1C3MIM][Tf2N] | 1-methyl-3-propylmethylimidazolium Bis(trifluoromethylsulfonyl)imide |
| [C3C1MIM][Tf2N] | 1-propyl-3-methylimidazolium Bis(trifluoromethylsulfonyl)imide |
| [Choline][Gly] | Cholinium Glycolate |
| [Choline][Lac] | Cholinium L(+) lactate |
| [Choline][Lev] | Cholinium Levulinate |
| [Choline][Mal] | Cholinium Malonate |
| [Choline][Pro] | Cholinium Proline |
| [DMAPAH][EtOAc] | 3-(Dimethylamino)-1-propylammonium Ethoxyacetate |
| [DMAPAH][TfA] | 3-Dimethylamino-1-propylammonium Trifluoroacetate |
| [EMIM][Ac] | 1-Ethyl-3-methylimidazolium Acetate |
| [EMIM][C(CN)3] | 1-Ethyl-3-methylimidazolium Tricyanomethanide |
| [EMIM][ESU] | 1-Ethyl-3-methylimidazolium Ethyl sulfate |
| [EMIM][Et2PO4] | 1-Ethyl-3-methylimidazolium Diethylphosphate |
| [EMIM][FAP] | 1-Ethyl-3-methylimidazolium Trifluoro phosphate |
| [EMIM][FAP] | 1-Ethyl-3-methylimidazolium Trifluoro phosphate |
| [EMIM][Me2PO4] | 1-Ethyl-3-methylimidazolium Trimethylphosphate |
| [EMIM][Tf2N] | 1-Ethyl-3-methylimidazolium Bis(trifluoromethylsulfonyl)imide |
| [EMIM][TfA] | 1-Ethyl-3-methylimidazolium Trifluoroacetate |
| [HMIM][Tf2N] | 1-Hexyl-3-methylimidazolium Bis(trifluoromethylsulfonyl)imide |
| [HmMIM][Tf2N] | 1-Hexyl-2-methyl-3-methylimidazolium Bis(trifluoromethylsulfonyl)imide |
| [OMIM][Ac] | 1-Octyl-3-methylimidazolium Acetate |
| [P222(101)][Inda] | Triethyl(2-methoxymethyl) phosphonium Indazolide |
| [P2225][Pro] | Triethyl(pentyl)phosphonium Prolinate |
| [P4444][Pro] | Tetrabutylphosphonium Prolinate |
| [Si-C1-C3-MIM][Tf2N] | 1-Methyl-3-(2-methyl-3-(trimethylsilyl)propyl)imidazolium Bis(trifluoromethylsulfonyl)imide |
| [VBTMA[Ac] | Vinylbenzyl trimethylammonium Acetate |
| [VOIM][PF6] | 1-vinyl-3-octylimidazolium Hexafluorophosphate |
| Cations | |
| P | Phosphonium |
| R1R2Mor | Morpholinium cation with two alkyl substituents |
| R1R2Pip | Piperidinium cation with two alkyl substituents |
| R1R2Pyr | Pyridinium cation with two alkyl substituents |
| R1R2Pyrr | Pyrolidinium cation with two alkyl substituents |
| R1R2R3Im | Imidazolium cation with three alkyl substituents |
| R1R2R3R4A | Ammonium cation with four alkyl substituents |
| R1R2R3R4Thi | Thiazolium cation with four alkyl substituents |
| R1R2R3S | Sulphonium cation with three alkyl substituents |
| R1R2R3Si | Silyl cation with three alkyl substituents |
| Anions | |
| [BF4] | Tetrafluoroborate |
| [C(CN)3] | Tricyanomethanide |
| [DCA] | Dicyanamide |
| [Doc] | bis(2-ethylhexyl)sulfosuccinate |
| [ESU] | Ethyl sulfate |
| [Et2PO4] | Diethylphosphate |
| [EtOAc] | Ethoxyacetate |
| [FAP] | Trifluoro phosphate |
| [Gly] | Glycolate |
| [Inda] | Indazolide |
| [Lac] | L(+) lactate |
| [Lev] | Levulinate |
| [Mal] | Malonate |
| [Me2PO4] | Trimethylphosphate |
| [PF6] | Hexafluorophosphate |
| [Pho] | Phenolate |
| [Pro] | Proline |
| [Pro] | Prolinate |
| [Tf2N] | bis(trifluoromethylsulfonyl)imide |
| [TfA] | Trifluoroacetate |
| [TfO] | Trifluoromethane sulfonate |
| Ac | Acetate |
| B(CN)4 | Tetracyanoborate |
| BF4 | Tetrafluoroborate |
| C(CN)3 | Tricyanomethanide |
| DCA or N(CH)2 | Dicyanamide |
| PF6 | Hexafluorophosphate |
| RPO4 | Alkyl phosphate |
| RSO4 | Alkyl sulfate |
| Tf2N | Bis(trifluoromethylsulfonyl)imide |
| TfO | Trifluoromethylsulfonate |
| TFSI (=Tf2N) | Bis(trifluoromethylsulfonyl)imide |
| Polymers and PILs | |
| 6FDA-TeMPD | (Hexafluoroisopropylidene)dephthalic anhydride and 2,3,5,6-tetramethyl- 1,4-phenylene diamine-based polyimide |
| p(VDF-HFP) | Poly(vinylidene fluoride-co-hexafluoropropylene) fluoroelastomer |
| p[MABI][BF4] | Poly(1-[2-(methacryloyloxy)ethyl]-3-butyl-imidazolium) tetrafluoroborate |
| p[MATMA][BF4] | poly[2-(methacryloyloxy)ethyl]trimethylammonium tetrafluoroborate |
| p[VBBI][BF4] | Poly(1-(p-vinylbenzyl)-3-butyl-imidazolium) tetrafluoroborate |
| p[VBIM][DCA] | 1-vinyl-3-butylimidazolium dicyanamide |
| p[VBTMA][BF4] | poly[1-(para-vinylbenzyl)-triethylammonium tetrafluoroborate |
| p[VBTMA][PF6] | poly[1-(para-vinylbenzyl)-triethylammonium hexafluorophosphate |
| p[VBTMA][Tf2N] | poly[1-(para-vinylbenzyl)-triethylammonium bis(trifluoromethylsulfonyl)imide |
| p[VEIM][DCA] | polymerized 1-vinyl-3-ethylimidazolium dicyanamide |
| p[VHIM][DCA] | 1-vinyl-3-heptylimidazolium dicyanamide |
| P4VP | Poly(4-vinylpyridine) |
| PAA | Poly acrylic acid or Sodium polyacrylate |
| PAH | Poly(allylamine) hydrochloride |
| PAMPS | Poly(2-acrylamido-2-methyl-1-propanesulfonic acid) |
| PAN | Polyacrylonitrile |
| PBI | Polybenzimidazole |
| PC | Polycarbonate |
| PDADMAC | Poly(diallyldimethylammonium) chloride |
| PDMAAm | Polydimethylacrylamide |
| PDMAEMA | Poly(N,N-dimethyl aminoethylmethacrylate) |
| PDMS | Polydimethylsiloxane |
| PEG | Polyethylene glycol |
| PEI | Poly(ethyleneimide) |
| PES | Polyethersulfone |
| PI | Polyimide |
| PMAPTAC | Poly(methacryloylamino propyl trimethylammonium chloride) |
| PMDA-ODA PI | Poly(pyromellitimide-co-4,4′-oxydianiline) polyimide |
| PMMA | Polymethylmethacrylate |
| Poly(SEOS) | Poly(styrene-block-ethylene oxide-block-styrene) |
| PSf | Polysulfone |
| PSS | Polystyrene sulphonate or Sodium polystyrene sulphonate |
| PTFE | Polytetrafluoroethylene |
| PTMEG | Polytetramethylene Ether Glycol |
| PTMSP | Poly(1-trimethylsilyl-1-propyne) |
| PU | Polyurethane |
| PVAc | Poly(vinyl) acetate |
| PVBC | Polyvinyl benzyl chloride |
| PVBTMAC | Poly(4-vinylbenzyltrimethylammonium) chloride |
| PVDF | Polyvinylidene fluoride |
| PVP | Polyvinylpyrrolidone |
References
- Wilkes, J.S.; Leviski, J.A.; Landers, J.S.; Hussey, C.L.; Vaughn, R.L.; Floreani, D.J.; Stech, D.J. A new class of room temperature molten salts for battery applications. In Proceedings of the AFSC/NAVHAT Science and Engineering Symposium, Colorado Springs, CO, USA, 27–29 October 1981; pp. 1–33. [Google Scholar]
- Wilkes, J.S. Molten Salts and Ionic Liquids—Are They not the Same Thing? ECS Trans. 2007, 3, 3–7. [Google Scholar] [CrossRef]
- Torimoto, T.; Tsuda, T.; Okazaki, K.; Kuwabata, S. New Frontiers in Materials Science Opened by Ionic Liquids. Adv. Mater. 2010, 22, 1196–1221. [Google Scholar] [CrossRef]
- Smiglak, M.; Pringle, J.M.; Lu, X.; Han, L.; Zhang, S.; Gao, H.; MacFarlane, D.R.; Rogers, R.D. Ionic liquids for energy, materials, and medicine. Chem. Commun. 2014, 50, 9228–9250. [Google Scholar] [CrossRef] [PubMed]
- Welton, T. Ionic liquids: A brief history. Biophys. Rev. 2018, 10, 691–706. [Google Scholar] [CrossRef] [PubMed]
- Zeng, S.; Cao, Y.; Li, P.; Liu, X.; Zhang, X. Ionic liquid–based green processes for ammonia separation and recovery. Curr. Opin. Green Sustain. Chem. 2020, 25, 100354. [Google Scholar] [CrossRef]
- Haider, J.; Saeed, S.; Qyyum, M.A.; Kazmi, B.; Ahmad, R.; Muhammad, A.; Lee, M. Simultaneous capture of acid gases from natural gas adopting ionic liquids: Challenges, recent developments, and prospects. Renew. Sustain. Energy Rev. 2020, 123, 109771. [Google Scholar] [CrossRef]
- Iulianelli, A.; Drioli, E. Membrane engineering: Latest advancements in gas separation and pre-treatment processes, petrochemical industry and refinery, and future perspectives in emerging applications. Fuel Process. Technol. 2020, 206, 106464. [Google Scholar] [CrossRef]
- Shamair, Z.; Habib, N.; Gilani, M.A.; Khan, A.L. Theoretical and experimental investigation of CO2 separation from CH4 and N2 through supported ionic liquid membranes. Appl. Energy 2020, 268, 115016. [Google Scholar] [CrossRef]
- Bakonyi, P.; Peter, J.; Koter, S.; Mateos, R.; Kumar, G.; Koók, L.; Rózsenberszki, T.; Pientka, Z.; Kujawski, W.; Kim, S.-H.; et al. Possibilities for the biologically-assisted utilization of CO2-rich gaseous waste streams generated during membrane technological separation of biohydrogen. J. CO2 Util. 2020, 36, 231–243. [Google Scholar] [CrossRef]
- Ayati, A.; Ranjbari, S.; Tanhaei, B.; Sillanpää, M. Ionic liquid-modified composites for the adsorptive removal of emerging water contaminants: A review. J. Mol. Liq. 2019, 275, 71–83. [Google Scholar] [CrossRef]
- Krull, F.F.; Fritzmann, C.; Melin, T. Liquid membranes for gas/vapor separations. J. Membr. Sci. 2008, 325, 509–519. [Google Scholar] [CrossRef]
- Kemperman, A.J.B.; Bargeman, D.; van den Boomgaard, T.; Strathmann, H. Stability of supported liquid membranes: State of the art. Sep. Sci. Technol. 1996, 31, 2733–2762. [Google Scholar] [CrossRef]
- Kemperman, A.J.B.; Rolevink, H.H.M.; Bargeman, D.; van den Boomgaard, T.; Strathmann, H. Stabilization of supported liquid membranes by interfacial polymerization top layers. J. Membr. Sci. 1998, 138, 43–55. [Google Scholar] [CrossRef]
- Teramoto, M.; Sakaida, Y.; Fu, S.S.; Ohnishi, N.; Matsuyama, H.; Maki, T.; Fukui, T.; Arai, K. An attempt for the stabilization of supported liquid membrane. Sep. Purif. Technol. 2000, 21, 137–144. [Google Scholar] [CrossRef]
- Karkhanechi, H.; Salmani, S.; Asghari, M. A Review on Gas Separation Applications of Supported Ionic Liquid Membranes. ChemBioEng Rev. 2015, 2, 290–302. [Google Scholar] [CrossRef]
- Meng, Y.; Chen, Y.; Deng, Y.; Zhang, S.; Wang, G. Perspectives on Ionic Liquids and Ionic Liquid Membranes for Natural Gas Sweetening. Prog. Chem. 2015, 27, 1324–1332. [Google Scholar]
- Dai, Z.; Noble, R.D.; Gin, D.L.; Zhang, X.; Deng, L. Combination of ionic liquids with membrane technology: A new approach for CO2 separation. J. Membr. Sci. 2016, 497, 1–20. [Google Scholar] [CrossRef]
- Tomé, L.C.; Marrucho, I.M. Ionic liquid-based materials: A platform to design engineered CO2 separation membranes. Chem. Soc. Rev. 2016, 45, 2785–2824. [Google Scholar] [CrossRef]
- Wang, J.; Luo, J.; Feng, S.; Li, H.; Wan, Y.; Zhang, X. Recent development of ionic liquid membranes. Green Energy Environ. 2016. [Google Scholar] [CrossRef]
- Gao, H.; Bai, L.; Han, J.; Yang, B.; Zhang, S.; Zhang, X. Functionalized ionic liquid membranes for CO2 separation. Chem. Commun. 2018, 54, 12671–12685. [Google Scholar] [CrossRef]
- Luis, P. Chapter 4—Gas permeation and supported liquid membranes. In Fundamental Modeling of Membrane Systems; Elsevier: Amsterdam, The Netherlands, 2018; pp. 103–151. ISBN 978-0-12-813483-2. [Google Scholar]
- Xiong, S.; Yin, D.; Javaid, M.U.; Li, L.; Pan, C.; Tang, J.; Yu, G. Ionic Liquids-Based Membranes for Carbon Dioxide Separation. Isr. J. Chem. 2019, 59, 824–831. [Google Scholar] [CrossRef]
- Zia-ul-Mustafa, M.; Mannan, H.A.; Mukhtar, H.; Nasir, R.; Mohshim, D.F.; Nordin, N. Application of Ionic Liquids in Gas Separation Membranes. In Industrial Applications of Green Solvents; Inamuddin, M.I.A., Asiri, A.M., Eds.; Materials Research Forum: Millersville, PA, USA, 2019; Volume 50, pp. 320–344. ISBN 978-1-64490-023-9. [Google Scholar]
- Zante, G.; Boltoeva, M.; Masmoudi, A.; Barillon, R.; Trébouet, D. Selective separation of cobalt and nickel using a stable supported ionic liquid membrane. Sep. Purif. Technol. 2020, 252, 117477. [Google Scholar] [CrossRef]
- Albo, J.; Yoshioka, T.; Tsuru, T. Porous Al2O3/TiO2 tubes in combination with 1-ethyl-3-methylimidazolium acetate ionic liquid for CO2/N2 separation. Sep. Purif. Technol. 2014, 122, 440–448. [Google Scholar] [CrossRef]
- Tzialla, O.; Labropoulos, A.; Panou, A.; Sanopoulou, M.; Kouvelos, E.; Athanasekou, C.; Beltsios, K.; Likodimos, V.; Falaras, P.; Romanos, G. Phase behavior and permeability of Alkyl-Methyl-Imidazolium Tricyanomethanide ionic liquids supported in nanoporous membranes. Sep. Purif. Technol. 2014, 135, 22–34. [Google Scholar] [CrossRef]
- Cheng, L.H.; Rahaman, M.S.A.; Yao, R.; Zhang, L.; Xu, X.H.; Chen, H.L.; Lai, J.Y.; Tung, K.L. Study on microporous supported ionic liquid membranes for carbon dioxide capture. Int. J. Greenh. Gas Control 2014, 21, 82–90. [Google Scholar] [CrossRef]
- Zhao, W.; He, G.; Nie, F.; Zhang, L.; Feng, H.; Liu, H. Membrane liquid loss mechanism of supported ionic liquid membrane for gas separation. J. Membr. Sci. 2012, 411–412, 73–80. [Google Scholar] [CrossRef]
- Luis, P.; Neves, L.A.; Afonso, C.A.M.; Coelhoso, I.M.; Crespo, J.G.; Garea, A.; Irabien, A. Facilitated transport of CO2 and SO2 through Supported Ionic Liquid Membranes (SILMs). Desalination 2009, 245, 485–493. [Google Scholar] [CrossRef]
- Gouveia, A.S.L.; Soares, B.; Simões, S.; Antonov, D.Y.; Lozinskaya, E.I.; Saramago, B.; Shaplov, A.S.; Marrucho, I.M. Ionic Liquid with Silyl Substituted Cation: Thermophysical and CO2/N2 Permeation Properties. Isr. J. Chem. 2019, 59, 852–865. [Google Scholar] [CrossRef]
- Ying, W.; Cai, J.; Zhou, K.; Chen, D.; Ying, Y.; Guo, Y.; Kong, X.; Xu, Z.; Peng, X. Ionic Liquid Selectively Facilitates CO2 Transport through Graphene Oxide Membrane. ACS Nano 2018, 12, 5385–5393. [Google Scholar] [CrossRef]
- Sánchez Fuentes, C.E.; Guzmán-Lucero, D.; Torres-Rodriguez, M.; Likhanova, N.V.; Bolaños, J.N.; Olivares-Xometl, O.; Lijanova, I.V. CO2/N2 separation using alumina supported membranes based on new functionalized ionic liquids. Sep. Purif. Technol. 2017, 182, 59–68. [Google Scholar] [CrossRef]
- Karousos, D.S.; Labropoulos, A.I.; Tzialla, O.; Papadokostaki, K.; Gjoka, M.; Stefanopoulos, K.L.; Beltsios, K.G.; Iliev, B.; Schubert, T.J.S.; Romanos, G.E. Effect of a cyclic heating process on the CO2/N2 separation performance and structure of a ceramic nanoporous membrane supporting the ionic liquid 1-methyl-3-octylimidazolium tricyanomethanide. Sep. Purif. Technol. 2018, 200, 11–22. [Google Scholar] [CrossRef]
- Chen, D.; Ying, W.; Guo, Y.; Ying, Y.; Peng, X. Enhanced Gas Separation through Nanoconfined Ionic Liquid in Laminated MoS2 Membrane. ACS Appl. Mater. Interfaces 2017, 9, 44251–44257. [Google Scholar] [CrossRef] [PubMed]
- Fortunato, R.; Branco, L.C.; Afonso, C.A.M.; Benavente, J.; Crespo, J.G. Electrical impedance spectroscopy characterisation of supported ionic liquid membranes. J. Membr. Sci. 2006, 270, 42–49. [Google Scholar] [CrossRef]
- Cserjési, P.; Nemestóthy, N.; Bélafi-Bakó, K. Gas separation properties of supported liquid membranes prepared with unconventional ionic liquids. J. Membr. Sci. 2010, 349, 6–11. [Google Scholar] [CrossRef]
- Gomez-Coma, L.; Garea, A.; Rouch, J.C.; Savart, T.; Lahitte, J.F.; Remigy, J.C.; Irabien, A. Membrane modules for CO2 capture based on PVDF hollow fibers with ionic liquids immobilized. J. Membr. Sci. 2016, 498, 218–226. [Google Scholar] [CrossRef]
- Abdelrahim, M.Y.; Martins, C.F.; Neves, L.A.; Capasso, C.; Supuran, C.T.; Coelhoso, I.M.; Crespo, J.G.; Barboiu, M. Supported ionic liquid membranes immobilized with carbonic anhydrases for CO2 transport at high temperatures. J. Membr. Sci. 2017, 528, 225–230. [Google Scholar] [CrossRef]
- Klingberg, P.; Wilkner, K.; Schlüter, M.; Grünauer, J.; Shishatskiy, S. Separation of Carbon Dioxide from Real Power Plant Flue Gases by Gas Permeation Using a Supported Ionic Liquid Membrane: An Investigation of Membrane Stability. Membranes 2019, 9, 35. [Google Scholar] [CrossRef]
- Akhmetshina, A.I.; Gumerova, O.R.; Atlaskin, A.A.; Petukhov, A.N.; Sazanova, T.S.; Yanbikov, N.R.; Nyuchev, A.V.; Razov, E.N.; Vorotyntsev, I.V. Permeability and selectivity of acid gases in supported conventional and novel imidazolium-based ionic liquid membranes. Sep. Purif. Technol. 2017, 176, 92–106. [Google Scholar] [CrossRef]
- Dahi, A.; Fatyeyeva, K.; Langevin, D.; Chappey, C.; Poncin-Epaillard, F.; Marais, S. Effect of cold plasma surface treatment on the properties of supported ionic liquid membranes. Sep. Purif. Technol. 2017, 187, 127–136. [Google Scholar] [CrossRef]
- Zeh, M.; Wickramanayake, S.; Hopkinson, D. Failure Mechanisms of Hollow Fiber Supported Ionic Liquid Membranes. Membranes 2016, 6, 21. [Google Scholar] [CrossRef]
- Ziobrowski, Z.; Rotkegel, A. Enhanced CO2/N2 separation by supported ionic liquid membranes (SILMs) based on PDMS and 1-ethyl-3-methylimidazolium acetate. Chem. Eng. Commun. 2021, 208, 137–147. [Google Scholar] [CrossRef]
- Tan, M.; Lu, J.; Zhang, Y.; Jiang, H. Ionic Liquid Confined in Mesoporous Polymer Membrane with Improved Stability for CO2/N2 Separation. Nanomaterials 2017, 7, 299. [Google Scholar] [CrossRef] [PubMed]
- Chai, S.H.; Fulvio, P.F.; Hillesheim, P.C.; Qiao, Z.A.; Mahurin, S.M.; Dai, S. “Brick-and-mortar” synthesis of free-standing mesoporous carbon nanocomposite membranes as supports of room temperature ionic liquids for CO2-N2 separation. J. Membr. Sci. 2014, 468, 73–80. [Google Scholar] [CrossRef]
- Ola, P.D.; Matsumoto, M. Supported Ionic Liquid Membranes for Metal Separation. In Progress and Developments in Ionic Liquids; Handy, S., Ed.; InTech: London, UK, 2017; pp. 539–555. ISBN 978-953-51-4108-2. [Google Scholar]
- Rogalsky, S.; Bardeau, J.-F.; Makhno, S.; Babkina, N.; Tarasyuk, O.; Cherniavska, T.; Orlovska, I.; Kozyrovska, N.; Brovko, O. New proton conducting membrane based on bacterial cellulose/polyaniline nanocomposite film impregnated with guanidinium-based ionic liquid. Polymer 2018, 142, 183–195. [Google Scholar] [CrossRef]
- Zhu, L.; Guo, L.; Zhang, Z.; Chen, J.; Zhang, S. The preparation of supported ionic liquids (SILs) and their application in rare metals separation. Sci. China Chem. 2012, 55, 1479–1487. [Google Scholar] [CrossRef]
- Wang, H.; Zhu, J.; Tan, L.; Zhou, M.; Zhang, S. Encapsulated ionic liquids for CO2 capture. Mater. Chem. Phys. 2020, 251, 122982. [Google Scholar] [CrossRef]
- Klepić, M.; Setničková, K.; Lanč, M.; Žák, M.; Izák, P.; Dendisová, M.; Fuoco, A.; Jansen, J.C.; Friess, K. Permeation and sorption properties of CO2-selective blend membranes based on polyvinyl alcohol (PVA) and 1-ethyl-3-methylimidazolium dicyanamide ([EMIM][DCA]) ionic liquid for effective CO2/H2 separation. J. Membr. Sci. 2020, 597, 117623. [Google Scholar] [CrossRef]
- Hernández-Fernández, F.J.; de los Ríos, A.P.; Tomás-Alonso, F.; Palacios, J.M.; Víllora, G. Preparation of supported ionic liquid membranes: Influence of the ionic liquid immobilization method on their operational stability. J. Membr. Sci. 2009, 341, 172–177. [Google Scholar] [CrossRef]
- Liu, Y.-F.; Xu, Q.-Q.; Wang, Y.-Q.; Zhu, H.-Y.; Yin, J.-Z. Preparation of Supported Ionic Liquid Membranes Using a Supercritical Fluid Deposition Method and Study of the Capillary Phase Transition of Ionic Liquids in Supercritical CO2. Ind. Eng. Chem. Res. 2019, 58, 19189–19196. [Google Scholar] [CrossRef]
- Khakpay, A.; Scovazzo, P.; Nouranian, S. Homogeneous and biphasic cellulose acetate/room temperature ionic liquid membranes for gas separations: Solvent and phase-inversion casting vs. supported ionic liquid membranes. J. Membr. Sci. 2019, 589, 117228. [Google Scholar] [CrossRef]
- Fortunato, R.; Afonso, C.A.M.; Benavente, J.; Rodriguez-Castellón, E.; Crespo, J.G. Stability of supported ionic liquid membranes as studied by X-ray photoelectron spectroscopy. J. Membr. Sci. 2005, 256, 216–223. [Google Scholar] [CrossRef]
- Cserjési, P.; Nemestóthy, N.; Vass, A.; Csanádi, Z.; Bélafi-Bakó, K. Study on gas separation by supported liquid membranes applying novel ionic liquids. Desalination 2009, 245, 743–747. [Google Scholar] [CrossRef]
- Neves, L.A.; Crespo, J.G.; Coelhoso, I.M. Gas permeation studies in supported ionic liquid membranes. J. Membr. Sci. 2010, 357, 160–170. [Google Scholar] [CrossRef]
- He, W.; Zhang, F.; Wang, Z.; Sun, W.; Zhou, Z.; Ren, Z. Facilitated Separation of \ce{CO2} by Liquid Membranes and Composite Membranes with Task-Specific Ionic Liquids. Ind. Eng. Chem. Res. 2016, 55, 12616–12631. [Google Scholar] [CrossRef]
- Selvam, T.; Machoke, A.; Schwieger, W. Supported ionic liquids on non-porous and porous inorganic materials—A topical review. Appl. Catal. A Gen. 2012, 445–446, 92–101. [Google Scholar] [CrossRef]
- Rahmani, F.; Nouranian, S.; Chiew, Y.C. 3D Graphene as an Unconventional Support Material for Ionic Liquid Membranes: Computational Insights into Gas Separations. Ind. Eng. Chem. Res. 2020, 59, 2203–2210. [Google Scholar] [CrossRef]
- Camper, D.; Bara, J.; Koval, C.; Noble, R. Bulk-fluid solubility and membrane feasibility of Rmim-based room-temperature ionic liquids. Ind. Eng. Chem. Res. 2006, 45, 6279–6283. [Google Scholar] [CrossRef]
- Scovazzo, P. Determination of the upper limits, benchmarks, and critical properties for gas separations using stabilized room temperature ionic liquid membranes (SILMs) for the purpose of guiding future research. J. Membr. Sci. 2009, 343, 199–211. [Google Scholar] [CrossRef]
- Morgan, D.; Ferguson, L.; Scovazzo, P. Diffusivities of Gases in Room-Temperature Ionic Liquids: Data and Correlations Obtained Using a Lag-Time Technique. Ind. Eng. Chem. Res. 2005, 44, 4815–4823. [Google Scholar] [CrossRef]
- Condemarin, R.; Scovazzo, P. Gas permeabilities, solubilities, diffusivities, and diffusivity correlations for ammonium-based room temperature ionic liquids with comparison to imidazolium and phosphonium RTIL data. Chem. Eng. J. 2009, 147, 51–57. [Google Scholar] [CrossRef]
- Kilaru, P.K.; Condemarin, R.A.; Scovazzo, P. Correlations of Low-Pressure Carbon Dioxide and Hydrocarbon Solubilities in Imidazolium-, Phosphonium-, and Ammonium-Based Room-Temperature Ionic Liquids. Part 1. Using Surface Tension. Ind. Eng. Chem. Res. 2008, 47, 900–909. [Google Scholar] [CrossRef]
- Camper, D.; Scovazzo, P.; Koval, C.; Noble, R. Gas solubilities in room-temperature ionic liquids. Ind. Eng. Chem. Res. 2004, 43, 3049–3054. [Google Scholar] [CrossRef]
- Scovazzo, P.; Camper, D.; Kieft, J.; Poshusta, J.; Koval, C.; Noble, R. Regular Solution Theory and CO2 Gas Solubility in Room-Temperature Ionic Liquids. Ind. Eng. Chem. Res. 2004, 43, 6855–6860. [Google Scholar] [CrossRef]
- Camper, D.; Becker, C.; Koval, C.; Noble, R. Low Pressure Hydrocarbon Solubility in Room Temperature Ionic Liquids Containing Imidazolium Rings Interpreted Using Regular Solution Theory. Ind. Eng. Chem. Res. 2005, 44, 1928–1933. [Google Scholar] [CrossRef]
- Finotello, A.; Bara, J.E.; Camper, D.; Noble, R.D.; Che, V. Room-Temperature Ionic Liquids: Temperature Dependence of Gas Solubility Selectivity. Ind. Eng. Chem. Res. 2008, 47, 3453–3459. [Google Scholar] [CrossRef]
- Moganty, S.S.; Baltus, R.E. Regular Solution Theory for Low Pressure Carbon Dioxide Solubility in Room Temperature Ionic Liquids: Ionic Liquid Solubility Parameter from Activation Energy of Viscosity. Ind. Eng. Chem. Res. 2010, 49, 5846–5853. [Google Scholar] [CrossRef]
- Shannon, M.S.; Tedstone, J.M.; Danielsen, S.P.O.; Hindman, M.S.; Irvin, A.C.; Bara, J.E. Free Volume as the Basis of Gas Solubility and Selectivity in Imidazolium-Based Ionic Liquids. Ind. Eng. Chem. Res. 2012, 51, 5565–5576. [Google Scholar] [CrossRef]
- Jeffrey Horne, W.; Shannon, M.S.; Bara, J.E. Correlating fractional free volume to CO2 selectivity in [Rmim][Tf2N] ionic liquids. J. Chem. Thermodyn. 2014, 77, 190–196. [Google Scholar] [CrossRef]
- Gonfa, G.; Bustam, M.A.; Sharif, A.M.; Mohamad, N.; Ullah, S. Tuning ionic liquids for natural gas dehydration using COSMO-RS methodology. J. Nat. Gas Sci. Eng. 2015, 27, 1141–1148. [Google Scholar] [CrossRef]
- Lee, B.-S.; Lin, S.-T. Screening of ionic liquids for CO2 capture using the COSMO-SAC model. Chem. Eng. Sci. 2015, 121, 157–168. [Google Scholar] [CrossRef]
- Mohammadi, M.; Asadollahzadeh, M.; Shirazian, S. Molecular-level understanding of supported ionic liquid membranes for gas separation. J. Mol. Liq. 2018, 262, 230–236. [Google Scholar] [CrossRef]
- Ramli, N.A.; Hashim, N.A.; Aroua, M.K. Prediction of CO2/O2 absorption selectivity using supported ionic liquid membranes (SILMs) for gas–liquid membrane contactor. Chem. Eng. Commun. 2018, 205, 295–310. [Google Scholar] [CrossRef]
- Sullivan-González, F.; Scovazzo, P.; Amos, R.; Bae, S.-K. Hydrogen-bond acceptance’s role in designing room temperature ionic liquid (RTIL) membranes for gas dehumidification, part I: Impacts on permeance and selectivity. J. Membr. Sci. 2017, 533, 190–200. [Google Scholar] [CrossRef]
- Carvalho, P.J.; Coutinho, J.A.P. The polarity effect upon the methane solubility in ionic liquids: A contribution for the design of ionic liquids for enhanced CO2/CH4 and H2S/CH4 selectivities. Energy Environ. Sci. 2011, 4, 4614–4619. [Google Scholar] [CrossRef]
- Khakpay, A.; Scovazzo, P. Reverse-selective behavior of room temperature ionic liquid based membranes for natural gas processing. J. Membr. Sci. 2018, 545, 204–212. [Google Scholar] [CrossRef]
- Grünauer, J.; Shishatskiy, S.; Abetz, C.; Abetz, V.; Filiz, V. Ionic liquids supported by isoporous membranes for CO2/N2 gas separation applications. J. Membr. Sci. 2015, 494, 224–233. [Google Scholar] [CrossRef]
- Akhmetshina, A.; Davletbaeva, I.; Grebenschikova, E.; Sazanova, T.; Petukhov, A.; Atlaskin, A.; Razov, E.; Zaripov, I.; Martins, C.; Neves, L.; et al. The Effect of Microporous Polymeric Support Modification on Surface and Gas Transport Properties of Supported Ionic Liquid Membranes. Membranes 2015, 6, 4. [Google Scholar] [CrossRef]
- Gouveia, A.S.L.; Tomé, L.C.; Marrucho, I.M. Towards the potential of cyano and amino acid-based ionic liquid mixtures for facilitated CO2 transport membranes. J. Membr. Sci. 2016, 510, 174–181. [Google Scholar] [CrossRef]
- Jie, X.; Chau, J.; Obuskovic, G.; Sirkar, K.K. Microporous Ceramic Tubule Based and Dendrimer-Facilitated Immobilized Ionic Liquid Membrane for CO2 Separation. Ind. Eng. Chem. Res. 2015, 54, 10401–10418. [Google Scholar] [CrossRef]
- Zarca, G.; Ortiz, I.; Urtiaga, A. Behaviour of 1-hexyl-3-methylimidazolium chloride-supported ionic liquid membranes in the permeation of CO2, H2, CO and N2 single and mixed gases. Desalin. Water Treat. 2015, 56, 3640–3646. [Google Scholar] [CrossRef]
- Kárászová, M.; Sedláková, Z.; Friess, K.; Izák, P. Effective permeability of binary mixture of carbon dioxide and methane and pre-dried raw biogas in supported ionic liquid membranes. Sep. Purif. Technol. 2015, 153, 14–18. [Google Scholar] [CrossRef]
- Grünauer, J.; Filiz, V.; Shishatskiy, S.; Abetz, C.; Abetz, V. Scalable application of thin film coating techniques for supported liquid membranes for gas separation made from ionic liquids. J. Membr. Sci. 2016, 518, 178–191. [Google Scholar] [CrossRef]
- Zhang, X.; Tu, Z.; Li, H.; Huang, K.; Hu, X.; Wu, Y.; MacFarlane, D.R. Selective separation of H2S and CO2 from CH4 by supported ionic liquid membranes. J. Membr. Sci. 2017, 543, 282–287. [Google Scholar] [CrossRef]
- Cichowska-Kopczyńska, I.; Joskowska, M.; Dębski, B.; Łuczak, J.; Aranowski, R. Influence of Ionic Liquid Structure on Supported Ionic Liquid Membranes Effectiveness in Carbon Dioxide/Methane Separation. J. Chem. 2013, 2013, 980689. [Google Scholar] [CrossRef]
- Tomé, L.C.; Patinha, D.J.S.; Ferreira, R.; Garcia, H.; Silva Pereira, C.; Freire, C.S.R.; Rebelo, L.P.N.; Marrucho, I.M. Cholinium-based Supported Ionic Liquid Membranes: A Sustainable Route for Carbon Dioxide Separation. ChemSusChem 2014, 7, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, A.; Muhammad, N.; Gilani, M.A.; Ayub, K.; Vankelecom, I.F.J.; Khan, A.L. Supported protic ionic liquid membrane based on 3-(trimethoxysilyl)propan-1-aminium acetate for the highly selective separation of CO2. J. Membr. Sci. 2017, 543, 301–309. [Google Scholar] [CrossRef]
- Santos, E.; Albo, J.; Irabien, A. Acetate based Supported Ionic Liquid Membranes (SILMs) for CO2 separation: Influence of the temperature. J. Membr. Sci. 2014, 452, 277–283. [Google Scholar] [CrossRef]
- Zhang, X.; Xiong, W.; Tu, Z.; Peng, L.; Wu, Y.; Hu, X. Supported Ionic Liquid Membranes with Dual-Site Interaction Mechanism for Efficient Separation of CO2. ACS Sustain. Chem. Eng. 2019, 7, 10792–10799. [Google Scholar] [CrossRef]
- Tomé, L.C.; Gouveia, A.S.L.; Ab Ranii, M.A.; Lickiss, P.D.; Welton, T.; Marrucho, I.M. Study on Gas Permeation and CO2 Separation through Ionic Liquid-Based Membranes with Siloxane-Functionalized Cations. Ind. Eng. Chem. Res. 2017, 56, 2229–2239. [Google Scholar] [CrossRef]
- Bui, T.T.L. Synthesis, Characterization, and Impregnation of Some Ionic Liquids on Polymer Membrane for Separation of Carbon Dioxide from Its Mixture with Methane. Chem. Biochem. Eng. Q. 2018, 32, 41–53. [Google Scholar] [CrossRef]
- Fan, T.; Xie, W.; Ji, X.; Liu, C.; Feng, X.; Lu, X. CO2/N2 separation using supported ionic liquid membranes with green and cost-effective [Choline][Pro]/PEG200 mixtures. Chin. J. Chem. Eng. 2016, 24, 1513–1521. [Google Scholar] [CrossRef]
- Close, J.J.; Farmer, K.; Moganty, S.S.; Baltus, R.E. CO2/N2 separations using nanoporous alumina-supported ionic liquid membranes: Effect of the support on separation performance. J. Membr. Sci. 2012, 390–391, 201–210. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, C.; Li, L.; Qin, W.; Xu, A. CO2 separation by supported ionic liquid membranes and prediction of separation performance. Int. J. Greenh. Gas Control 2016, 53, 79–84. [Google Scholar] [CrossRef]
- Akhmetshina, A.I.; Yanbikov, N.R.; Petukhov, A.N.; Vorotyntsev, I.V. Effect of temperature on gas transport properties of supported ionic liquid membranes. Pet. Chem. 2017, 57, 770–778. [Google Scholar] [CrossRef]
- Zhang, X.-M.; Tu, Z.-H.; Li, H.; Li, L.; Wu, Y.-T.; Hu, X.-B. Supported protic-ionic-liquid membranes with facilitated transport mechanism for the selective separation of CO2. J. Membr. Sci. 2017, 527, 60–67. [Google Scholar] [CrossRef]
- Althuluth, M.; Overbeek, J.P.; van Wees, H.J.; Zubeir, L.F.; Haije, W.G.; Berrouk, A.; Peters, C.J.; Kroon, M.C. Natural gas purification using supported ionic liquid membrane. J. Membr. Sci. 2015, 484, 80–86. [Google Scholar] [CrossRef]
- Scovazzo, P.; Havard, D.; McShea, M.; Mixon, S.; Morgan, D. Long-term, continuous mixed-gas dry fed CO2/CH4 and CO2/N2 separation performance and selectivities for room temperature ionic liquid membranes. J. Membr. Sci. 2009, 327, 41–48. [Google Scholar] [CrossRef]
- Hojniak, S.D.; Silverwood, I.P.; Khan, A.L.; Vankelecom, I.F.J.; Dehaen, W.; Kazarian, S.G.; Binnemans, K. Highly Selective Separation of Carbon Dioxide from Nitrogen and Methane by Nitrile/Glycol-Difunctionalized Ionic Liquids in Supported Ionic Liquid Membranes (SILMs). J. Phys. Chem. B 2014, 118, 7440–7449. [Google Scholar] [CrossRef]
- Kudasheva, A.; Kamiya, T.; Hirota, Y.; Ito, A. Dehumidification of air using liquid membranes with ionic liquids. J. Membr. Sci. 2016, 499, 379–385. [Google Scholar] [CrossRef]
- Sun, Y.; Bi, H.; Dou, H.; Yang, H.; Huang, Z.; Wang, B.; Deng, R.; Zhang, L. A Novel Copper(I)-Based Supported Ionic Liquid Membrane with High Permeability for Ethylene/Ethane Separation. Ind. Eng. Chem. Res. 2017, 56, 741–749. [Google Scholar] [CrossRef]
- Dou, H.; Jiang, B.; Xu, M.; Zhou, J.; Sun, Y.; Zhang, L. Supported ionic liquid membranes with high carrier efficiency via strong hydrogen-bond basicity for the sustainable and effective olefin/paraffin separation. Chem. Eng. Sci. 2019, 193, 27–37. [Google Scholar] [CrossRef]
- Sanchez, C.M.; Song, T.; Brennecke, J.F.; Freeman, B.D. Hydrogen Stable Supported Ionic Liquid Membranes with Silver Carriers: Propylene and Propane Permeability and Solubility. Ind. Eng. Chem. Res. 2020, 59, 5362–5370. [Google Scholar] [CrossRef]
- Mecerreyes, D. Polymeric ionic liquids: Broadening the properties and applications of polyelectrolytes. Prog. Polym. Sci. 2011, 36, 1629–1648. [Google Scholar] [CrossRef]
- Yuan, J.; Antonietti, M. Poly(ionic liquid)s: Polymers expanding classical property profiles. Polymer 2011, 52, 1469–1482. [Google Scholar] [CrossRef]
- Sasikumar, B.; Arthanareeswaran, G.; Ismail, A.F. Recent progress in ionic liquid membranes for gas separation. J. Mol. Liq. 2018, 266, 330–341. [Google Scholar] [CrossRef]
- Torresi, R.M.; Corrêa, C.M.; Benedetti, T.M.; Martins, V.L. CHAPTER 13 Tailoring Transport Properties Aiming for Versatile Ionic Liquids and Poly(Ionic Liquids) for Electrochromic and Gas Capture Applications. In Polymerized Ionic Liquids; The Royal Society of Chemistry: London, UK, 2018; pp. 342–380. ISBN 978-1-78262-960-3. [Google Scholar]
- Eftekhari, A.; Saito, T. Synthesis and properties of polymerized ionic liquids. Eur. Polym. J. 2017, 90, 245–272. [Google Scholar] [CrossRef]
- Ding, S.; Tang, H.; Radosz, M.; Shen, Y. Atom transfer radical polymerization of ionic liquid 2-(1-butylimidazolium- 3-yl)ethyl methacrylate tetrafluoroborate. J. Polym. Sci. Part A Polym. Chem. 2004, 42, 5794–5801. [Google Scholar] [CrossRef]
- Tang, H.; Tang, J.; Ding, S.; Radosz, M.; Shen, Y. Atom transfer radical polymerization of styrenic ionic liquid monomers and carbon dioxide absorption of the polymerized ionic liquids. J. Polym. Sci. Part A Polym. Chem. 2005, 43, 1432–1443. [Google Scholar] [CrossRef]
- Marcilla, R.; Blazquez, J.A.; Fernandez, R.; Grande, H.; Pomposo, J.A.; Mecerreyes, D. Synthesis of novel polycations using the chemistry of ionic liquids. Macromol. Chem. Phys. 2005, 206, 299–304. [Google Scholar] [CrossRef]
- Green, M.D.; Long, T.E. Designing Imidazole-Based Ionic Liquids and Ionic Liquid Monomers for Emerging Technologies. Polym. Rev. 2009, 49, 291–314. [Google Scholar] [CrossRef]
- Marcilla, R.; Blazquez, J.A.; Rodriguez, J.; Pomposo, J.A.; Mecerreyes, D. Tuning the Solubility of Polymerized Ionic Liquids by Simple Anion-Exchange Reactions. J. Polym. Sci. Part A Polym. Chem. 2003, 1, 208–212. [Google Scholar] [CrossRef]
- Snedden, P.; Cooper, A.I.; Scott, K.; Winterton, N. Cross-linked polymer-ionic liquid composite materials. Macromolecules 2003, 36, 4549–4556. [Google Scholar] [CrossRef]
- Appetecchi, G.B.; Kim, G.T.; Montanino, M.; Carewska, M.; Marcilla, R.; Mecerreyes, D.; De Meatza, I. Ternary polymer electrolytes containing pyrrolidinium-based polymeric ionic liquids for lithium batteries. J. Power Sources 2010, 195, 3668–3675. [Google Scholar] [CrossRef]
- Nikolaeva, D.; Azcune, I.; Tanczyk, M.; Warmuzinski, K.; Jaschik, M.; Sandru, M.; Dahl, P.I.; Genua, A.; Loïs, S.; Sheridan, E.; et al. The performance of affordable and stable cellulose-based poly-ionic membranes in CO2/N2 and CO2/CH4 gas separation. J. Membr. Sci. 2018, 564, 552–561. [Google Scholar] [CrossRef]
- Xu, X.; Wang, J.; Dong, J.; Li, H.B.; Zhang, Q.; Zhao, X. Ionic polyimide membranes containing Tröger’s base: Synthesis, microstructure and potential application in CO2 separation. J. Membr. Sci. 2020, 602, 117967. [Google Scholar] [CrossRef]
- Bernard, F.L.; dos Santos, L.M.; Schwab, M.B.; Polesso, B.B.; do Nascimento, J.F.; Einloft, S. Polyurethane-based poly (ionic liquid)s for CO2 removal from natural gas. J. Appl. Polym. Sci. 2019, 136, 4–11. [Google Scholar] [CrossRef]
- Kujawa, J.; Rynkowska, E.; Fatyeyeva, K.; Knozowska, K.; Wolan, A.; Dzieszkowski, K.; Li, G.; Kujawski, W. Preparation and Characterization of Cellulose Acetate Propionate Films Functionalized with Reactive Ionic Liquids. Polymers 2019, 11, 1217. [Google Scholar] [CrossRef]
- Guiver, M.D.; Yahia, M.; Dal-Cin, M.M.; Robertson, G.P.; Saeedi Garakani, S.; Du, N.; Tavajohi, N. Gas Transport in a Polymer of Intrinsic Microporosity (PIM-1) Substituted with Pseudo-Ionic Liquid Tetrazole-Type Structures. Macromolecules 2020, 53, 8951–8959. [Google Scholar] [CrossRef] [PubMed]
- Nikolaeva, D.; Verachtert, K.; Azcune, I.; Jansen, J.C.; Vankelecom, I.F.J. Influence of ionic liquid-like cationic pendants composition in cellulose based polyelectrolytes on membrane-based CO2 separation. Carbohydr. Polym. 2021, 255, 117375. [Google Scholar] [CrossRef]
- Paquin, F.; Rivnay, J.; Salleo, A.; Stingelin, N.; Silva, C. Multi-phase semicrystalline microstructures drive exciton dissociation in neat plastic semiconductors. J. Mater. Chem. C 2015, 3, 10715–10722. [Google Scholar] [CrossRef]
- Nikolaeva, D.; Luis, P. Top-Down Polyelectrolytes for Membrane-Based Post-Combustion CO2 Capture. Molecules 2020, 25, 323. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Texter, J.; Yan, F. Frontiers in poly(ionic liquid)s: Syntheses and applications. Chem. Soc. Rev. 2017, 46, 1124–1159. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Ledin, P.A.; Shevchenko, V.V.; Tsukruk, V.V. Architecture, Assembly, and Emerging Applications of Branched Functional Polyelectrolytes and Poly(ionic liquid)s. ACS Appl. Mater. Interfaces 2015, 7, 12570–12596. [Google Scholar] [CrossRef] [PubMed]
- Ohno, H.; Yoshizawa, M. Preparation and properties of polymerized ionic liquids as film electrolytes. ACS Symp. Ser. 2005, 902, 159–170. [Google Scholar] [CrossRef]
- Brown, R.H.; Duncan, A.J.; Choi, J.H.; Park, J.K.; Wu, T.; Leo, D.J.; Winey, K.I.; Moore, R.B.; Long, T.E. Effect of ionic liquid on mechanical properties and morphology of zwitterionic copolymer membranes. Macromolecules 2010, 43, 790–796. [Google Scholar] [CrossRef]
- Soll, S.; Zhang, P.; Zhao, Q.; Wang, Y.; Yuan, J. Mesoporous zwitterionic poly(ionic liquid)s: Intrinsic complexation and efficient catalytic fixation of CO2. Polym. Chem. 2013, 4, 5048–5051. [Google Scholar] [CrossRef]
- Biswas, Y.; Mandal, T.K. Structural Variation in Homopolymers Bearing Zwitterionic and Ionic Liquid Pendants for Achieving Tunable Multi-Stimuli Responsiveness and Hierarchical Nanoaggregates. Macromolecules 2017, 50, 9807–9820. [Google Scholar] [CrossRef]
- Duong, P.H.H.; Daumann, K.; Hong, P.Y.; Ulbricht, M.; Nunes, S.P. Interfacial Polymerization of Zwitterionic Building Blocks for High-Flux Nanofiltration Membranes. Langmuir 2019, 35, 1284–1293. [Google Scholar] [CrossRef]
- Petroff, M.G.; Garcia, E.A.; Herrera-Alonso, M.; Bevan, M.A. Ionic Strength-Dependent Interactions and Dimensions of Adsorbed Zwitterionic Copolymers. Langmuir 2019, 35, 4976–4985. [Google Scholar] [CrossRef]
- Soll, S.; Zhao, Q.; Weber, J.; Yuan, J. Activated CO2 sorption in mesoporous imidazolium-type poly(ionic liquid)-based polyampholytes. Chem. Mater. 2013, 25, 3003–3010. [Google Scholar] [CrossRef]
- Laschewsky, A. Structures and synthesis of zwitterionic polymers. Polymers 2014, 6, 1544–1601. [Google Scholar] [CrossRef]
- Nguyen, P.T.; Wiesenauer, E.F.; Gin, D.L.; Noble, R.D. Effect of composition and nanostructure on CO2/N2 transport properties of supported alkyl-imidazolium block copolymer membranes. J. Membr. Sci. 2013, 430, 312–320. [Google Scholar] [CrossRef]
- Ajjan, F.N.; Ambrogi, M.; Tiruye, G.A.; Cordella, D.; Fernandes, A.M.; Grygiel, K.; Isik, M.; Patil, N.; Porcarelli, L.; Rocasalbas, G.; et al. Innovative polyelectrolytes/poly(ionic liquid)s for energy and the environment. Polym. Int. 2017, 66, 1119–1128. [Google Scholar] [CrossRef]
- Zhou, X.; Weber, J.; Yuan, J. Poly(ionic liquid)s: Platform for CO2 capture and catalysis. Curr. Opin. Green Sustain. Chem. 2019, 16, 39–46. [Google Scholar] [CrossRef]
- Dunn, C.A.; Shi, Z.; Zhou, R.; Gin, D.L.; Noble, R.D. (Cross-Linked Poly(Ionic Liquid)-Ionic Liquid-Zeolite) Mixed-Matrix Membranes for CO2/CH4 Gas Separations Based on Curable Ionic Liquid Prepolymers. Ind. Eng. Chem. Res. 2019, 58, 4704–4708. [Google Scholar] [CrossRef]
- Morozova, S.M.; Lozinskaya, E.I.; Sardon, H.; Suárez-García, F.; Vlasov, P.S.; Vaudemont, R.; Vygodskii, Y.S.; Shaplov, A.S. Ionic Polyureas—A Novel Subclass of Poly(Ionic Liquid)s for CO2 Capture. Membranes 2020, 10, 240. [Google Scholar] [CrossRef]
- Gouveia, A.S.L.; Malcaitè, E.; Lozinskaya, E.I.; Shaplov, A.S.; Tomé, L.C.; Marrucho, I.M. Poly(ionic liquid)–Ionic Liquid Membranes with Fluorosulfonyl-Derived Anions: Characterization and Biohydrogen Separation. ACS Sustain. Chem. Eng. 2020, 8, 7087–7096. [Google Scholar] [CrossRef]
- Mazzei, I.R.; Nikolaeva, D.; Fuoco, A.; Loïs, S.; Fantini, S.; Monteleone, M.; Esposito, E.; Ashtiani, S.J.; Lanč, M.; Vopička, O.; et al. Poly[3 -ethyl-1-vinyl-imidazolium] diethyl phosphate/Pebax® 1657 Composite Membranes and Their Gas Separation Performance. Membranes 2020, 10, 224. [Google Scholar] [CrossRef]
- Dani, A.; Crocellà, V.; Magistris, C.; Santoro, V.; Yuan, J.; Bordiga, S. Click-based porous cationic polymers for enhanced carbon dioxide capture. J. Mater. Chem. A 2017, 5, 372–383. [Google Scholar] [CrossRef]
- McDanel, W.M.; Cowan, M.G.; Carlisle, T.K.; Swanson, A.K.; Noble, R.D.; Gin, D.L. Cross-linked ionic resins and gels from epoxide-functionalized imidazolium ionic liquid monomers. Polymer 2014, 55, 3305–3313. [Google Scholar] [CrossRef]
- Erwin, A.J.; Lee, H.; Ge, S.; Zhao, S.; Korolovych, V.F.; He, H.; Matyjaszewski, K.; Sokolov, A.P.; Tsukruk, V.V. Viscoelastic properties and ion dynamics in star-shaped polymerized ionic liquids. Eur. Polym. J. 2018, 109, 326–335. [Google Scholar] [CrossRef]
- Tang, Q.; Zhang, W.; Yuan, J.; Zhao, Q. Templated synthesis of cyclic poly(ionic liquid)s. React. Funct. Polym. 2019, 138, 1–8. [Google Scholar] [CrossRef]
- Cardiano, P.; Mineo, P.G.; Neri, F.; Lo Schiavo, S.; Piraino, P. A new application of ionic liquids: Hydrophobic properties of tetraalkylammonium-based poly(ionic liquid)s. J. Mater. Chem. 2008, 18, 1253–1260. [Google Scholar] [CrossRef]
- Arora, S.; Liang, J.; Fullerton-Shirey, S.K.; Laaser, J.E. Triggerable Ion Release in Polymerized Ionic Liquids Containing Thermally Labile Diels-Alder Linkages. ACS Mater. Lett. 2020, 2, 331–335. [Google Scholar] [CrossRef]
- Gu, Y.; Lodge, T.P. Synthesis and Gas Separation Performance of Triblock Copolymer Ion Gels with a Polymerized Ionic Liquid Mid-Block. Macromolecules 2011, 44, 1732–1736. [Google Scholar] [CrossRef]
- Adzima, B.J.; Venna, S.R.; Klara, S.S.; He, H.; Zhong, M.; Luebke, D.R.; Mauter, M.S.; Matyjaszewski, K.; Nulwala, H.B. Modular polymerized ionic liquid block copolymer membranes for CO2/N2 separation. J. Mater. Chem. A 2014, 2, 7967–7972. [Google Scholar] [CrossRef]
- Sun, X.; Zhang, M.; Guo, R.; Luo, J.; Li, J. CO2-facilitated transport performance of poly(ionic liquids) in supported liquid membranes. J. Mater. Sci. 2015, 50, 104–111. [Google Scholar] [CrossRef]
- Isik, M.; Zulfiqar, S.; Edhaim, F.; Ruiperez, F.; Rothenberger, A.; Mecerreyes, D. Sustainable Poly(Ionic Liquids) for CO2 Capture Based on Deep Eutectic Monomers. ACS Sustain. Chem. Eng. 2016, 4, 7200–7208. [Google Scholar] [CrossRef]
- Nikolaeva, D.; Azcune, I.; Sheridan, E.; Sandru, M.; Genua, A.; Tanczyk, M.; Jaschik, M.; Warmuzinski, K.; Jansen, J.C.; Vankelecom, I.F.J. Poly(vinylbenzyl chloride)-based poly(ionic liquids) as membranes for CO2 capture from flue gas. J. Mater. Chem. A 2017, 5, 19808–19818. [Google Scholar] [CrossRef]
- Kamio, E.; Tanaka, M.; Shirono, Y.; Keun, Y.; Moghadam, F.; Yoshioka, T.; Nakagawa, K.; Matsuyama, H. Hollow Fiber-Type Facilitated Transport Membrane Composed of a Polymerized Ionic Liquid-Based Gel Layer with Amino Acidate as the CO2 Carrier. Ind. Eng. Chem. Res. 2020, 59, 2083–2092. [Google Scholar] [CrossRef]
- Wang, J.; He, X.; Zhu, H.; Chen, D. Preparation of a ROMP-type imidazolium-functionalized norbornene ionic liquid block copolymer and the electrochemical property for lithium-ion batteries polyelectrolyte membranes. RSC Adv. 2015, 5, 43581–43588. [Google Scholar] [CrossRef]
- Choi, U.H.; Price, T.L.; Schoonover, D.V.; Gibson, H.W.; Colby, R.H. The Effect of Oligo(oxyethylene) Moieties on Ion Conduction and Dielectric Properties of Norbornene-Based Imidazolium Tf2N Ionic Liquid Monomers. Macromolecules 2020, 53, 4990–5000. [Google Scholar] [CrossRef]
- Malecha, J.J.; Biller, J.R.; Lama, B.; Gin, D.L. System for Living ROMP of a Paramagnetic FeCl4—Based Ionic Liquid Monomer: Direct Synthesis of Magnetically Responsive Block Copolymers. ACS Macro Lett. 2020, 9, 140–145. [Google Scholar] [CrossRef]
- Bara, J.E.; Lessmann, S.; Gabriel, C.J.; Hatakeyama, E.S.; Noble, R.D.; Gin, D.L. Synthesis and Performance of Polymerizable Room-Temperature Ionic Liquids as Gas Separation Membranes. Ind. Eng. Chem. Res. 2007, 46, 5397–5404. [Google Scholar] [CrossRef]
- Mecerreyes, D. Applications of Ionic Liquids in Polymer Science and Technology; Springer: Berlin/Heidelberg, Germany, 2015; ISBN 9783662449028. [Google Scholar]
- Jangu, C.; Long, T.E. Phosphonium cation-containing polymers: From ionic liquids to polyelectrolytes. Polymer 2014, 55, 3298–3304. [Google Scholar] [CrossRef]
- Bhaskar Reddy, A.V.; Moniruzzaman, M.; Bustam, M.A.; Goto, M.; Saha, B.B.; Janiak, C. Ionic liquid polymer materials with tunable nanopores controlled by surfactant aggregates: A novel approach for CO2capture. J. Mater. Chem. A 2020, 8, 15034–15041. [Google Scholar] [CrossRef]
- Hudiono, Y.C.; Carlisle, T.K.; LaFrate, A.L.; Gin, D.L.; Noble, R.D. Novel mixed matrix membranes based on polymerizable room-temperature ionic liquids and SAPO-34 particles to improve CO2 separation. J. Membr. Sci. 2011, 370, 141–148. [Google Scholar] [CrossRef]
- Lee, C.S.; Kim, N.U.; Park, J.T.; Kim, J.H. Imidazole-functionalized hydrophilic rubbery comb copolymers: Microphase-separation and good gas separation properties. Sep. Purif. Technol. 2020, 242, 116780. [Google Scholar] [CrossRef]
- Hirao, M.; Ito, K.; Ohno, H. Preparation and polymerization of new organic molten salts; N-alkylimidazolium salt derivatives. Electrochim. Acta 2000, 45, 1291–1294. [Google Scholar] [CrossRef]
- Vygodskii, Y.S.; Sapozhnikov, D.A.; Shaplov, A.S.; Lozinskaya, E.I.; Ignat’Ev, N.V.; Schulte, M.; Vlasov, P.S.; Malyshkina, I.A. New ionic liquids with hydrolytically stable anions as alternatives to hexafluorophosphate and tetrafluoroborate salts in the free radical polymerization and preparation of ion-conducting composites. Polym. J. 2011, 43, 126–135. [Google Scholar] [CrossRef][Green Version]
- Arshady, R. Suspension, emulsion, and dispersion polymerization: A methodological survey. Colloid Polym. Sci. 1992, 270, 717–732. [Google Scholar] [CrossRef]
- Asua, J.M. Challenges and Opportunities in Continuous Production of Emulsion Polymers: A Review. Macromol. React. Eng. 2016, 10, 311–323. [Google Scholar] [CrossRef]
- Chern, C.S. Emulsion polymerization mechanisms and kinetics. Prog. Polym. Sci. 2006, 31, 443–486. [Google Scholar] [CrossRef]
- Hejazifar, M.; Lanaridi, O.; Bica-Schröder, K. Ionic liquid based microemulsions: A review. J. Mol. Liq. 2020, 303, 112264. [Google Scholar] [CrossRef]
- Landfester, K. Miniemulsion polymerization and the structure of polymer and hybrid nanoparticles. Angew. Chem. Int. Ed. 2009, 48, 4488–4507. [Google Scholar] [CrossRef]
- Yang, L.; Ma, H.; Han, L.; Hao, X.; Liu, P.; Shen, H.; Li, Y. Synthesis of a sequence-controlled in-chain alkynyl/tertiary amino dual-functionalized terpolymer: Via living anionic polymerization. Polym. Chem. 2018, 9, 108–120. [Google Scholar] [CrossRef]
- Peng, Y.; Yu, H.; Chen, H.; Huang, Z.; Li, H. Cross-linking and de-cross-linking of triarylimidazole-based polymer. Polymer 2016, 99, 529–535. [Google Scholar] [CrossRef]
- Zhou, Y.N.; Lei, L.; Luo, Z.H.; Zhu, S. CO2/N2-Switchable Thermoresponsive Ionic Liquid Copolymer. Macromolecules 2017, 50, 8378–8389. [Google Scholar] [CrossRef]
- Venkidasubramonian, G.; Kratzer, D.; Trouillet, V.; Zydziak, N.; Franzreb, M.; Barner, L.; Lahann, J. Surface-initiated RAFT polymerization from vapor-based polymer coatings. Polymer 2018, 150, 26–34. [Google Scholar] [CrossRef]
- Chi, W.S.; Hong, S.U.; Jung, B.; Kang, S.W.; Kang, Y.S.; Kim, J.H. Synthesis, structure and gas permeation of polymerized ionic liquid graft copolymer membranes. J. Membr. Sci. 2013, 443, 54–61. [Google Scholar] [CrossRef]
- Mori, D.I.; Martin, R.M.; Noble, R.D.; Gin, D.L. Cross-linked, polyurethane-based, ammonium poly(ionic liquid)/ionic liquid composite films for organic vapor suppression and ion conduction. Polymer 2017, 112, 435–446. [Google Scholar] [CrossRef]
- Morozova, S.M.; Shaplov, A.S.; Lozinskaya, E.I.; Mecerreyes, D.; Sardon, H.; Zulfiqar, S.; Suárez-García, F.; Vygodskii, Y.S. Ionic Polyurethanes as a New Family of Poly(ionic liquid)s for Efficient CO2Capture. Macromolecules 2017, 50, 2814–2824. [Google Scholar] [CrossRef]
- Bernard, F.L.; Polesso, B.B.; Cobalchini, F.W.; Donato, A.J.; Seferin, M.; Ligabue, R.; Chaban, V.V.; do Nascimento, J.F.; Dalla Vecchia, F.; Einloft, S. CO2 capture: Tuning cation-anion interaction in urethane based poly(ionic liquids). Polymer 2016, 102, 199–208. [Google Scholar] [CrossRef]
- Bernard, F.L.; Polesso, B.B.; Cobalchini, F.W.; Chaban, V.V.; Do Nascimento, J.F.; Dalla Vecchia, F.; Einloft, S. Hybrid Alkoxysilane-Functionalized Urethane-Imide-Based Poly(ionic liquids) as a New Platform for Carbon Dioxide Capture. Energy Fuels 2017, 31, 9840–9849. [Google Scholar] [CrossRef]
- Matsumoto, K.; Endo, T. Confinement of ionic liquid by networked polymers based on multifunctional epoxy resins. Macromolecules 2008, 41, 6981–6986. [Google Scholar] [CrossRef]
- Maksym, P.; Tarnacka, M.; Dzienia, A.; Matuszek, K.; Chrobok, A.; Kaminski, K.; Paluch, M. Enhanced Polymerization Rate and Conductivity of Ionic Liquid-Based Epoxy Resin. Macromolecules 2017, 50, 3262–3272. [Google Scholar] [CrossRef]
- Radchenko, A.V.; Chabane, H.; Demir, B.; Searles, D.J.; Duchet-Rumeau, J.; Gérard, J.F.; Baudoux, J.; Livi, S. New Epoxy Thermosets Derived from a Bisimidazolium Ionic Liquid Monomer: An Experimental and Modeling Investigation. ACS Sustain. Chem. Eng. 2020, 8, 12208–12221. [Google Scholar] [CrossRef]
- Borodin, O.; Smith, G.D. Molecular dynamics simulations of comb-branched poly(epoxide ether)-based polymer electrolytes. Macromolecules 2007, 40, 1252–1258. [Google Scholar] [CrossRef]
- Livi, S.; Chardin, C.; Lins, L.C.; Halawani, N.; Pruvost, S.; Duchet-Rumeau, J.; Gérard, J.F.; Baudoux, J. From Ionic Liquid Epoxy Monomer to Tunable Epoxy-Amine Network: Reaction Mechanism and Final Properties. ACS Sustain. Chem. Eng. 2019, 7, 3602–3613. [Google Scholar] [CrossRef]
- Zhang, J.; Li, X.; Zhu, Z.; Chang, T.; Fu, X.; Hao, Y.; Meng, X.; Panchal, B.; Qin, S. Hydroxylamino-Anchored Poly(Ionic Liquid)s for CO2 Fixation into Cyclic Carbonates at Mild Conditions. Adv. Sustain. Syst. 2020, 2000133, 1–8. [Google Scholar] [CrossRef]
- McDanel, W.M.; Cowan, M.G.; Barton, J.A.; Gin, D.L.; Noble, R.D. Effect of monomer structure on curing behavior, CO2 solubility, and gas permeability of ionic liquid-based epoxy-amine resins and ion-gels. Ind. Eng. Chem. Res. 2015, 54, 4396–4406. [Google Scholar] [CrossRef]
- McDanel, W.M.; Cowan, M.G.; Chisholm, N.O.; Gin, D.L.; Noble, R.D. Fixed-site-carrier facilitated transport of carbon dioxide through ionic-liquid-based epoxy-amine ion gel membranes. J. Membr. Sci. 2015, 492, 303–311. [Google Scholar] [CrossRef]
- Friess, K.; Lanč, M.; Pilnáček, K.; Fíla, V.; Vopička, O.; Sedláková, Z.; Cowan, M.G.; McDanel, W.M.; Noble, R.D.; Gin, D.L.; et al. CO2/CH4 separation performance of ionic-liquid-based epoxy-amine ion gel membranes under mixed feed conditions relevant to biogas processing. J. Membr. Sci. 2017, 528, 64–71. [Google Scholar] [CrossRef]
- Dimitrov-Raytchev, P.; Beghdadi, S.; Serghei, A.; Drockenmuller, E. Main-chain 1,2,3-triazolium-based poly(ionic liquid)s issued from AB + AB click chemistry polyaddition. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 34–38. [Google Scholar] [CrossRef]
- Cheng, S.; Wojnarowska, Z.; Musiał, M.; Flachard, D.; Drockenmuller, E.; Paluch, M. Access to Thermodynamic and Viscoelastic Properties of Poly(ionic liquid)s Using High-Pressure Conductivity Measurements. ACS Macro Lett. 2019, 8, 996–1001. [Google Scholar] [CrossRef]
- Wojnarowska, Z.; Musiał, M.; Cheng, S.; Drockenmuller, E.; Paluch, M. Fast secondary dynamics for enhanced charge transport in polymerized ionic liquids. Phys. Rev. E 2020, 101, 32606. [Google Scholar] [CrossRef]
- Colliat-Dangus, G.; Obadia, M.M.; Vygodskii, Y.S.; Serghei, A.; Shaplov, A.S.; Drockenmuller, E. Unconventional poly(ionic liquid)s combining motionless main chain 1,2,3-triazolium cations and high ionic conductivity. Polym. Chem. 2015, 6, 4299–4308. [Google Scholar] [CrossRef]
- Obadia, M.M.; Colliat-Dangus, G.; Debuigne, A.; Serghei, A.; Detrembleur, C.; Drockenmuller, E. Poly(vinyl ester 1,2,3-triazolium)s: A new member of the poly(ionic liquid)s family. Chem. Commun. 2015, 51, 3332–3335. [Google Scholar] [CrossRef]
- Flachard, D.; Serghei, A.; Fumagalli, M.; Drockenmuller, E. Main-chain poly(1,2,3-triazolium hydroxide)s obtained through AA+BB click polyaddition as anion exchange membranes. Polym. Int. 2019, 68, 1591–1598. [Google Scholar] [CrossRef]
- Suckow, M.; Roy, M.; Sahre, K.; Häußler, L.; Singha, N.; Voit, B.; Böhme, F. Synthesis of polymeric ionic liquids with unidirectional chain topology by AB step growth polymerization. Polymer 2017, 111, 123–129. [Google Scholar] [CrossRef]
- Zhou, X.; Obadia, M.M.; Venna, S.R.; Roth, E.A.; Serghei, A.; Luebke, D.R.; Myers, C.; Chang, Z.; Enick, R.; Drockenmuller, E.; et al. Highly cross-linked polyether-based 1,2,3-triazolium ion conducting membranes with enhanced gas separation properties. Eur. Polym. J. 2016, 84, 65–76. [Google Scholar] [CrossRef]
- Mortimer, D.A. Synthetic polyelectrolytes—A review. Polym. Int. 1991, 25, 29–41. [Google Scholar] [CrossRef]
- Laschewsky, A. Recent trends in the synthesis of polyelectrolytes. Curr. Opin. Colloid Interface Sci. 2012, 17, 56–63. [Google Scholar] [CrossRef]
- Krasemann, L.; Tieke, B. Composite membranes with ultrathin separation layer prepared by self-assembly of polyelectrolytes. Mater. Sci. Eng. C 1999, 8–9, 513–518. [Google Scholar] [CrossRef]
- Song, Y.; Lugo, E.L.; Powell, S.; Tzeng, P.; Wilhite, B.A.; Grunlan, J.C. Highly selective multilayer polymer thin films for CO2/N2 separation. J. Polym. Sci. Part B Polym. Phys. 2017, 55, 1730–1737. [Google Scholar] [CrossRef]
- Pramanik, N.B.; Regen, S.L. Layer-by-layer assembly of a polymer of intrinsic microporosity: Targeting the CO 2 /N 2 separation problem. Chem. Commun. 2019, 55, 4347–4350. [Google Scholar] [CrossRef]
- Van Ackern, F.; Krasemann, L.; Tieke, B. Ultrathin membranes for gas separation and pervaporation prepared upon electrostatic self-assembly of polyelectrolytes. Thin Solid Films 1998, 327–329, 762–766. [Google Scholar] [CrossRef]
- Bernard, F.L.; Duczinski, R.B.; Rojas, M.F.; Fialho, M.C.C.; Carreño, L.Á.; Chaban, V.V.; Vecchia, F.D.; Einloft, S. Cellulose based poly(ionic liquids): Tuning cation-anion interaction to improve carbon dioxide sorption. Fuel 2018, 211, 76–86. [Google Scholar] [CrossRef]
- Wang, M.; Janout, V.; Regen, S.L. Gas transport across hyperthin membranes. Acc. Chem. Res. 2013, 46, 2743–2754. [Google Scholar] [CrossRef]
- Pramanik, N.B.; Regen, S.L. Hyperthin Membranes for Gas Separations via Layer-by-Layer Assembly. Chem. Rec. 2020, 20, 163–173. [Google Scholar] [CrossRef]
- Yi, S.; Leon, W.; Vezenov, D.; Regen, S.L. Tightening Polyelectrolyte Multilayers with Oligo Pendant Ions. ACS Macro Lett. 2016, 5, 915–918. [Google Scholar] [CrossRef]
- Lin, C.; Yi, S.; Regen, S.L. Consequences of Tacticity on the Growth and Permeability of Hyperthin Polyelectrolyte Multilayers. Langmuir 2016, 32, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Scholes, C.A.; Kentish, S.E.; Stevens, G.W.; Jin, J.; DeMontigny, D. Thin-film composite membrane contactors for desorption of CO2from Monoethanolamine at elevated temperatures. Sep. Purif. Technol. 2015, 156, 841–847. [Google Scholar] [CrossRef]
- Kaiser, E.T. Reactions of Sulfonate and Sulfate Esters. Org. Chem. Sulfur 1977, 649–679. [Google Scholar] [CrossRef]
- Izumrudov, V.A.; Mussabayeva, B.K.; Murzagulova, K.B. Polyelectrolyte multilayers: Preparation and applications. Russ. Chem. Rev. 2018, 87, 192–200. [Google Scholar] [CrossRef]
- Paulson, M.H. Sulfation and sulfonation. J. Am. Oil Chem. Soc. 1952, 29, 556–560. [Google Scholar] [CrossRef]
- Lohr, J.W. Sulfation and sulfonation. Practical aspects. J. Am. Oil Chem. Soc. 1958, 35, 532–537. [Google Scholar] [CrossRef]
- Dado, G.; Bernhardt, R. Sulfonation and Sulfation. In Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2017; pp. 1–30. ISBN 0471238961. [Google Scholar]
- Matsuyama, H.; Teramoto, M.; Sakakura, H. Selective permeation of CO2 through poly{2-(N,N-dimethyl) aminoethyl methacrylate} membrane prepared by plasma-graft polymerization technique. J. Membr. Sci. 1996, 114, 193–200. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Fujimoto, K.; Kinugawa, H.; Kitao, T.; Ogata, N. Selective Permeation of Carbon Dioxide through Synthetic Polymeric Membranes Having Amine Moiety. Chem. Lett. 1994, 23, 243–246. [Google Scholar] [CrossRef]
- Du, R.; Feng, X.; Chakma, A. Poly(N,N-dimethylaminoethyl methacrylate)/polysulfone composite membranes for gas separations. J. Membr. Sci. 2006, 279, 76–85. [Google Scholar] [CrossRef]
- Risangud, N.; Congdon, T.R.; Keddie, D.J.; Wilson, P.; Kempe, K.; Haddleton, D.M. Polyurea microcapsules from isocyanatoethyl methacrylate copolymers. J. Polym. Sci. Part A Polym. Chem. 2016, 54, 2698–2705. [Google Scholar] [CrossRef][Green Version]
- Sood, R.; Donnadio, A.; Giancola, S.; Kreisz, A.; Jones, D.J.; Cavaliere, S. 1,2,3-Triazole-Functionalized Polysulfone Synthesis through Microwave-Assisted Copper-Catalyzed Click Chemistry: A Highly Proton Conducting High Temperature Membrane. ACS Appl. Mater. Interfaces 2016, 8, 16897–16906. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, W.; Gao, H.; Bai, Y.; Sun, Y.; Chen, Y. Synthesis and gas transport properties of poly(ionic liquid) based semi-interpenetrating polymer network membranes for CO2/N2 separation. J. Membr. Sci. 2017, 528, 72–81. [Google Scholar] [CrossRef]
- Yan, X.; Anguille, S.; Bendahan, M.; Moulin, P. Ionic liquids combined with membrane separation processes: A review. Sep. Purif. Technol. 2019, 222, 230–253. [Google Scholar] [CrossRef]
- Li, P.; Paul, D.R.; Chung, T.-S. High performance membranes based on ionic liquid polymers for CO2 separation from the flue gas. Green Chem. 2012, 14, 1052. [Google Scholar] [CrossRef]
- Carlisle, T.K.; Wiesenauer, E.F.; Nicodemus, G.D.; Gin, D.L.; Noble, R.D. Ideal CO2/Light Gas Separation Performance of Poly(vinylimidazolium) Membranes and Poly(vinylimidazolium)-Ionic Liquid Composite Films. Ind. Eng. Chem. Res. 2013, 52, 1023–1032. [Google Scholar] [CrossRef]
- Jeffrey Horne, W.; Andrews, M.A.; Shannon, M.S.; Terrill, K.L.; Moon, J.D.; Hayward, S.S.; Bara, J.E. Effect of branched and cycloalkyl functionalities on CO2 separation performance of poly(IL) membranes. Sep. Purif. Technol. 2015, 155, 89–95. [Google Scholar] [CrossRef]
- Ansaloni, L.; Nykaza, J.R.; Ye, Y.; Elabd, Y.A.; Giacinti Baschetti, M. Influence of water vapor on the gas permeability of polymerized ionic liquids membranes. J. Membr. Sci. 2015, 487, 199–208. [Google Scholar] [CrossRef]
- Sakaguchi, T.; Ito, H.; Masuda, T.; Hashimoto, T. Highly CO2-permeable and -permselective poly(diphenylacetylene)s having imidazolium salts: Synthesis, characterization, gas permeation properties, and effects of counter anion. Polymer 2013, 54, 6709–6715. [Google Scholar] [CrossRef]
- Gye, B.; Kammakakam, I.; You, H.; Nam, S.; Kim, T.-H. PEG-imidazolium-incorporated polyimides as high-performance CO2-selective polymer membranes: The effects of PEG-imidazolium content. Sep. Purif. Technol. 2017, 179, 283–290. [Google Scholar] [CrossRef]
- Carlisle, T.K.; Bara, J.E.; Lafrate, A.L.; Gin, D.L.; Noble, R.D. Main-chain imidazolium polymer membranes for CO2 separations: An initial study of a new ionic liquid-inspired platform. J. Membr. Sci. 2010, 359, 37–43. [Google Scholar] [CrossRef]
- Bara, J.E.; Hatakeyama, E.S.; Gabriel, C.J.; Zeng, X.; Lessmann, S.; Gin, D.L.; Noble, R.D. Synthesis and light gas separations in cross-linked gemini room temperature ionic liquid polymer membranes. J. Membr. Sci. 2008, 316, 186–191. [Google Scholar] [CrossRef]
- Mittenthal, M.S.; Flowers, B.S.; Bara, J.E.; Whitley, J.W.; Spear, S.K.; Roveda, J.D.; Wallace, D.A.; Shannon, M.S.; Holler, R.; Martens, R.; et al. Ionic Polyimides: Hybrid Polymer Architectures and Composites with Ionic Liquids for Advanced Gas Separation Membranes. Ind. Eng. Chem. Res. 2017, 56, 5055–5069. [Google Scholar] [CrossRef]
- Kumbharkar, S.C.; Bhavsar, R.S.; Kharul, U.K. Film forming polymeric ionic liquids (PILs) based on polybenzimidazoles for CO2 separation. RSC Adv. 2014, 4, 4500–4503. [Google Scholar] [CrossRef]
- Shaligram, S.V.; Rewar, A.S.; Wadgaonkar, P.P.; Kharul, U.K. Incorporation of rigid polyaromatic groups in polybenzimidazole-based polymeric ionic liquids: Assertive effects on gas permeation properties. Polymer 2016, 93, 30–36. [Google Scholar] [CrossRef]
- Shaplov, A.S.; Morozova, S.M.; Lozinskaya, E.I.; Vlasov, P.S.; Gouveia, A.S.L.; Tomé, L.C.; Marrucho, I.M.; Vygodskii, Y.S. Turning into poly(ionic liquid)s as a tool for polyimide modification: Synthesis, characterization and CO2 separation properties. Polym. Chem. 2016, 7, 580–591. [Google Scholar] [CrossRef]
- Jourdain, A.; Antoniuk, I.; Serghei, A.; Espuche, E.; Drockenmuller, E. 1,2,3-Triazolium-based linear ionic polyurethanes. Polym. Chem. 2017, 8, 5148–5156. [Google Scholar] [CrossRef]
- Tomé, L.C.; Mecerreyes, D.; Freire, C.S.R.; Rebelo, L.P.N.; Marrucho, I.M. Pyrrolidinium-based polymeric ionic liquid materials: New perspectives for CO2 separation membranes. J. Membr. Sci. 2013, 428, 260–266. [Google Scholar] [CrossRef]
- Bara, J.E.; Gabriel, C.J.; Hatakeyama, E.S.; Carlisle, T.K.; Lessmann, S.; Noble, R.D.; Gin, D.L. Improving CO2 selectivity in polymerized room-temperature ionic liquid gas separation membranes through incorporation of polar substituents. J. Membr. Sci. 2008, 321, 3–7. [Google Scholar] [CrossRef]
- Deimede, V.; Vroulias, D.; Kallitsis, J.; Ioannides, T. Pyridinium based Poly(Ionic Liquids) membranes with exceptional high water vapor permeability and selectivity. Sep. Purif. Technol. 2020, 251, 117412. [Google Scholar] [CrossRef]
- Robeson, L.M. Correlation of separation factor versus permeability for polymeric membranes. J. Membr. Sci. 1991, 62, 165–185. [Google Scholar] [CrossRef]
- Robeson, L.M. The upper bound revisited. J. Membr. Sci. 2008, 320, 390–400. [Google Scholar] [CrossRef]
- Comesaña-Gándara, B.; Chen, J.; Bezzu, C.G.; Carta, M.; Rose, I.; Ferrari, M.-C.; Esposito, E.; Fuoco, A.; Jansen, J.C.; McKeown, N.B. Redefining the Robeson upper bounds for CO2/CH4 and CO2/N2 separations using a series of ultrapermeable benzotriptycene-based polymers of intrinsic microporosity. Energy Environ. Sci. 2019, 12, 2733–2740. [Google Scholar] [CrossRef]
- Kuzmina, O. CHAPTER 3 Cationic and Anionic Polymerized Ionic Liquids: Properties for Applications. In Polymerized Ionic Liquids; The Royal Society of Chemistry: London, UK, 2018; pp. 83–116. ISBN 978-1-78262-960-3. [Google Scholar]
- Einloft, S.; Bernard, F.L.; Dalla Vecchia, F. CHAPTER 17 Capturing CO2 with Poly(Ionic Liquid)s. In Polymerized Ionic Liquids; The Royal Society of Chemistry: London, UK, 2018; pp. 489–514. ISBN 978-1-78262-960-3. [Google Scholar]
- Zulfiqar, S.; Sarwar, M.I.; Mecerreyes, D. Polymeric ionic liquids for CO2 capture and separation: Potential, progress and challenges. Polym. Chem. 2015, 6, 6435–6451. [Google Scholar] [CrossRef]
- Tang, J.; Tang, H.; Sun, W.; Plancher, H.; Radosz, M.; Shen, Y. Poly(ionic liquid)s: A new material with enhanced and fast CO2 absorption. Chem. Commun. 2005, 3325–3327. [Google Scholar] [CrossRef]
- Tang, J.; Sun, W.; Tang, H.; Radosz, M.; Shen, Y. Enhanced CO2 Absorption of Poly(ionic liquid)s. Macromolecules 2005, 38, 2037–2039. [Google Scholar] [CrossRef]
- Radosz, M.; Shen, Y. Poly(Ionic Liquid)s as New Materials for CO2 Separation and Other Applications. Patent Application No. US2009/0266230A1, 29 November 2009. [Google Scholar]
- Tang, J.; Shen, Y.; Radosz, M.; Sun, W. Isothermal Carbon Dioxide Sorption in Poly(ionic liquid)s. Ind. Eng. Chem. Res. 2009, 48, 9113–9118. [Google Scholar] [CrossRef]
- Tang, J.; Tang, H.; Sun, W.; Radosz, M.; Shen, Y. Poly(ionic liquid)s as new materials for CO2 absorption. J. Polym. Sci. Part A Polym. Chem. 2005, 43, 5477–5489. [Google Scholar] [CrossRef]
- Supasitmongkol, S.; Styring, P. High CO2 solubility in ionic liquids and a tetraalkylammonium-based poly(ionic liquid). Energy Environ. Sci. 2010, 3, 1961–1972. [Google Scholar] [CrossRef]
- Simons, K.; Nijmeijer, K.; Bara, J.E.; Noble, R.D.; Wessling, M. How do polymerized room-temperature ionic liquid membranes plasticize during high pressure CO2 permeation? J. Membr. Sci. 2010, 360, 202–209. [Google Scholar] [CrossRef]
- Tien Nguyen, P.; Voss, B.A.; Wiesenauer, E.F.; Gin, D.L.; Noble, R.D. Physically Gelled Room-Temperature Ionic Liquid-Based Composite Membranes for CO2/N2 Separation: Effect of Composition and Thickness on Membrane Properties and Performance. Ind. Eng. Chem. Res. 2012, 52, 8812–8821. [Google Scholar] [CrossRef]
- Lodge, T.P. A Unique Platform for Materials Design. Science 2008, 321, 50–51. [Google Scholar] [CrossRef] [PubMed]
- Izák, P.; Hovorka, Š.; Bartovský, T.; Bartovská, L.; Crespo, J.G. Swelling of polymeric membranes in room temperature ionic liquids. J. Membr. Sci. 2007, 296, 131–138. [Google Scholar] [CrossRef]
- Jansen, J.C.; Friess, K.; Clarizia, G.; Schauer, J.; Izák, P. High Ionic Liquid Content Polymeric Gel Membranes: Preparation and Performance. Macromolecules 2011, 44, 39–45. [Google Scholar] [CrossRef]
- Uk Hong, S.; Park, D.; Ko, Y.; Baek, I. Polymer-ionic liquid gels for enhanced gas transport. Chem. Commun. 2009, 7227–7229. [Google Scholar] [CrossRef]
- Friess, K.; Jansen, J.C.; Bazzarelli, F.; Izák, P.; Jarmarová, V.; Kačírková, M.; Schauer, J.; Clarizia, G.; Bernardo, P. High ionic liquid content polymeric gel membranes: Correlation of membrane structure with gas and vapour transport properties. J. Membr. Sci. 2012, 415–416, 801–809. [Google Scholar] [CrossRef]
- Bernardo, P.; Jansen, J.C.; Bazzarelli, F.; Tasselli, F.; Fuoco, A.; Friess, K.; Izák, P.; Jarmarová, V.; Kačírková, M.; Clarizia, G. Gas transport properties of Pebax®/room temperature ionic liquid gel membranes. Sep. Purif. Technol. 2012, 97, 73–82. [Google Scholar] [CrossRef]
- Couto, R.M.; Carvalho, T.; Neves, L.A.; Ruivo, R.M.; Vidinha, P.; Paiva, A.; Coelhoso, I.M.; Barreiros, S.; Simões, P.C. Development of Ion-Jelly® membranes. Sep. Purif. Technol. 2013, 106, 22–31. [Google Scholar] [CrossRef]
- Lam, B.; Wei, M.; Zhu, L.; Luo, S.; Guo, R.; Morisato, A.; Alexandridis, P.; Lin, H. Cellulose triacetate doped with ionic liquids for membrane gas separation. Polymer 2016, 89, 1–11. [Google Scholar] [CrossRef]
- Mannan, H.A.; Mohshim, D.F.; Mukhtar, H.; Murugesan, T.; Man, Z.; Bustam, M.A. Synthesis, characterization, and CO2 separation performance of polyether sulfone/[EMIM][Tf2N] ionic liquid-polymeric membranes (ILPMs). J. Ind. Eng. Chem. 2017, 54, 98–106. [Google Scholar] [CrossRef]
- Abdollahi, S.; Mortaheb, H.R.; Ghadimi, A.; Esmaeili, M. Improvement in separation performance of Matrimid®5218 with encapsulated [Emim][Tf2N] in a heterogeneous structure: CO2/CH4 separation. J. Membr. Sci. 2018, 557, 38–48. [Google Scholar] [CrossRef]
- Kanehashi, S.; Kishida, M.; Kidesaki, T.; Shindo, R.; Sato, S.; Miyakoshi, T.; Nagai, K. CO2 separation properties of a glassy aromatic polyimide composite membranes containing high-content 1-butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ionic liquid. J. Membr. Sci. 2013, 430, 211–222. [Google Scholar] [CrossRef]
- Liang, L.; Gan, Q.; Nancarrow, P. Composite ionic liquid and polymer membranes for gas separation at elevated temperatures. J. Membr. Sci. 2014, 450, 407–417. [Google Scholar] [CrossRef]
- Ito, A.; Yasuda, T.; Ma, X.; Watanabe, M. Sulfonated polyimide/ionic liquid composite membranes for carbon dioxide separation. Polym. J. 2017, 49, 671–676. [Google Scholar] [CrossRef]
- Budd, P.M.; Ghanem, B.S.; Makhseed, S.; McKeown, N.B.; Msayib, K.J.; Tattershall, C.E. Polymers of intrinsic microporosity (PIMs): Robust, solution-processable, organic nanoporous materials. Chem. Commun. 2004, 4, 230–231. [Google Scholar] [CrossRef]
- Budd, P.M.; Elabas, E.S.; Ghanem, B.S.; Makhseed, S.; McKeown, N.B.; Msayib, K.J.; Tattershall, C.E.; Wang, D. Solution-Processed, Organophilic Membrane Derived from a Polymer of Intrinsic Microporosity. Adv. Mater. 2004, 16, 456–459. [Google Scholar] [CrossRef]
- Halder, K.; Khan, M.M.; Grünauer, J.; Shishatskiy, S.; Abetz, C.; Filiz, V.; Abetz, V. Blend membranes of ionic liquid and polymers of intrinsic microporosity with improved gas separation characteristics. J. Membr. Sci. 2017, 539, 368–382. [Google Scholar] [CrossRef]
- Han, J.; Bai, L.; Luo, S.; Yang, B.; Bai, Y.; Zeng, S.; Zhang, X. Ionic liquid cobalt complex as O2 carrier in the PIM-1 membrane for O2/N2 separation. Sep. Purif. Technol. 2020, 248, 117041. [Google Scholar] [CrossRef]
- Chen, H.Z.; Li, P.; Chung, T.-S. PVDF/ionic liquid polymer blends with superior separation performance for removing CO2 from hydrogen and flue gas. Int. J. Hydrogen Energy 2012, 37, 11796–11804. [Google Scholar] [CrossRef]
- Song, T.; Zhang, X.; Li, Y.; Jiang, K.; Zhang, S.; Cui, X.; Bai, L. Separation Efficiency of CO2 in Ionic Liquids/Poly(vinylidene fluoride) Composite Membrane: A Molecular Dynamics Study. Ind. Eng. Chem. Res. 2019, 58, 6887–6898. [Google Scholar] [CrossRef]
- Uchytil, P.; Schauer, J.; Petrychkovych, R.; Setnickova, K.; Suen, S.Y. Ionic liquid membranes for carbon dioxide–methane separation. J. Membr. Sci. 2011, 383, 262–271. [Google Scholar] [CrossRef]
- Jansen, J.C.; Clarizia, G.; Bernardo, P.; Bazzarelli, F.; Friess, K.; Randová, A.; Schauer, J.; Kubicka, D.; Kacirková, M.; Izak, P. Gas transport properties and pervaporation performance of fluoropolymer gel membranes based on pure and mixed ionic liquids. Sep. Purif. Technol. 2013, 109, 87–97. [Google Scholar] [CrossRef]
- Vopička, O.; Morávková, L.; Vejražka, J.; Sedláková, Z.; Friess, K.; Izák, P. Ethanol sorption and permeation in fluoropolymer gel membrane containing 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ionic liquid. Chem. Eng. Process. Process Intensif. 2015, 94, 72–77. [Google Scholar] [CrossRef]
- Morávková, L.; Vopička, O.; Vejražka, J.; Vychodilová, H.; Sedláková, Z.; Friess, K.; Izák, P. Vapour permeation and sorption in fluoropolymer gel membrane based on ionic liquid 1-ethyl-3-methylimidazolium bis(trifluoromethylsulphonyl)imide. Chem. Pap. 2014, 68, 1739–1746. [Google Scholar] [CrossRef]
- Dai, Z.; Bai, L.; Hval, K.N.; Zhang, X.; Zhang, S.; Deng, L. Pebax®/TSIL blend thin film composite membranes for CO2 separation. Sci. China Chem. 2016, 59, 538–546. [Google Scholar] [CrossRef]
- Bhattacharya, M.; Mandal, M.K. Synthesis and characterization of ionic liquid based mixed matrix membrane for acid gas separation. J. Clean. Prod. 2017, 156, 174–183. [Google Scholar] [CrossRef]
- Rabiee, H.; Ghadimi, A.; Mohammadi, T. Gas transport properties of reverse-selective poly(ether-b-amide6)/[Emim][BF4] gel membranes for CO2/light gases separation. J. Membr. Sci. 2015, 476, 286–302. [Google Scholar] [CrossRef]
- Fam, W.; Mansouri, J.; Li, H.; Chen, V. Improving CO2 separation performance of thin film composite hollow fiber with Pebax®1657/ionic liquid gel membranes. J. Membr. Sci. 2017, 537, 54–68. [Google Scholar] [CrossRef]
- Qiu, Y.; Ren, J.; Zhao, D.; Li, H.; Deng, M. Poly(amide-6-b-ethylene oxide)/[Bmim][Tf2N] blend membranes for carbon dioxide separation. J. Energy Chem. 2016, 25, 122–130. [Google Scholar] [CrossRef]
- Longo, M.; De Santo, M.P.; Esposito, E.; Fuoco, A.; Monteleone, M.; Giorno, L.; Jansen, J.C. Force spectroscopy determination of Young’s modulus in mixed matrix membranes. Polymer 2018, 156, 22–29. [Google Scholar] [CrossRef]
- Ghasemi Estahbanati, E.; Omidkhah, M.; Ebadi Amooghin, A. Preparation and characterization of novel Ionic liquid/Pebax membranes for efficient CO 2 /light gases separation. J. Ind. Eng. Chem. 2017, 51, 77–89. [Google Scholar] [CrossRef]
- Pardo, F.; Zarca, G.; Urtiaga, A. Effect of feed pressure and long-term separation performance of Pebax-ionic liquid membranes for the recovery of difluoromethane (R32) from refrigerant mixture R410A. J. Membr. Sci. 2021, 618, 118744. [Google Scholar] [CrossRef]
- Klepić, M.; Fuoco, A.; Monteleone, M.; Esposito, E.; Friess, K.; Petrusová, Z.; Izák, P.; Jansen, J.C. Tailoring the Thermal and Mechanical Properties of PolyActiveTM Poly(Ether-Ester) Multiblock Copolymers Via Blending with CO2-Phylic Ionic Liquid. Polymers 2020, 12, 890. [Google Scholar] [CrossRef] [PubMed]
- Klepić, M.; Fuoco, A.; Monteleone, M.; Esposito, E.; Friess, K.; Izák, P.; Jansen, J.C. Effect of the CO2-philic ionic liquid [BMIM][Tf2N] on the single and mixed gas transport in PolyActiveTM membranes. Sep. Purif. Technol. 2021, 256, 117813. [Google Scholar] [CrossRef]
- Yoon, I.-N.; Yoo, S.; Park, S.-J.; Won, J. CO2 separation membranes using ion gels by self-assembly of a triblock copolymer in ionic liquids. Chem. Eng. J. 2011, 172, 237–242. [Google Scholar] [CrossRef]
- Hwang, Y.; Cho, E.; Jung, J.; Kang, Y.S.; Won, J. Alkylmethylimidazolium tricyanomethanide based supported ion gel membranes for CO2 separation. RSC Adv. 2017, 7, 51257–51263. [Google Scholar] [CrossRef]
- Gu, Y.; Cussler, E.L.; Lodge, T.P. ABA-triblock copolymer ion gels for CO2 separation applications. J. Membr. Sci. 2012, 423–424, 20–26. [Google Scholar] [CrossRef]
- Wijayasekara, D.B.; Cowan, M.G.; Lewis, J.T.; Gin, D.L.; Noble, R.D.; Bailey, T.S. Elastic free-standing RTIL composite membranes for CO2/N2 separation based on sphere-forming triblock/diblock copolymer blends. J. Membr. Sci. 2016, 511, 170–179. [Google Scholar] [CrossRef]
- Dai, Z.; Ansaloni, L.; Ryan, J.J.; Spontak, R.J.; Deng, L. Incorporation of an ionic liquid into a midblock-sulfonated multiblock polymer for CO2 capture. J. Membr. Sci. 2019, 588, 117193. [Google Scholar] [CrossRef]
- Kasahara, S.; Kamio, E.; Yoshizumi, A.; Matsuyama, H. Polymeric ion-gels containing an amino acid ionic liquid for facilitated CO2 transport media. Chem. Commun. 2014, 50, 2996–2999. [Google Scholar] [CrossRef]
- Moghadam, F.; Kamio, E.; Yoshizumi, A.; Matsuyama, H. An amino acid ionic liquid-based tough ion gel membrane for CO2 capture. Chem. Commun. 2015, 51, 13658–13661. [Google Scholar] [CrossRef] [PubMed]
- Moghadam, F.; Kamio, E.; Matsuyama, H. High CO2 separation performance of amino acid ionic liquid-based double network ion gel membranes in low CO2 concentration gas mixtures under humid conditions. J. Membr. Sci. 2017, 525, 290–297. [Google Scholar] [CrossRef]
- Moghadam, F.; Kamio, E.; Yoshioka, T.; Matsuyama, H. New approach for the fabrication of double-network ion-gel membranes with high CO2/N2 separation performance based on facilitated transport. J. Membr. Sci. 2017, 530, 166–175. [Google Scholar] [CrossRef]
- Ranjbaran, F.; Kamio, E.; Matsuyama, H. Ion Gel Membrane with Tunable Inorganic/Organic Composite Network for CO2 Separation. Ind. Eng. Chem. Res. 2017, 56, 12763–12772. [Google Scholar] [CrossRef]
- Ranjbaran, F.; Kamio, E.; Matsuyama, H. Inorganic/organic composite ion gel membrane with high mechanical strength and high CO2 separation performance. J. Membr. Sci. 2017, 544, 252–260. [Google Scholar] [CrossRef]
- Gharedaghi, M.; Omidkhah, M.R.; Abdollahi, S.; Ghadimi, A. An Investigation on Gas Transport Properties of Elvaloy4170/[Emim][Tf2N] Hybrid Membranes for Efficient CO2/CH4 Separation. J. Membr. Sci. Res. 2020. [Google Scholar] [CrossRef]
- Kusuma, V.A.; Macala, M.K.; Baker, J.S.; Hopkinson, D. Cross-Linked Poly(ethylene oxide) Ion Gels Containing Functionalized Imidazolium Ionic Liquids as Carbon Dioxide Separation Membranes. Ind. Eng. Chem. Res. 2018, 57, 11658–11667. [Google Scholar] [CrossRef]
- Kusuma, V.A.; Chen, C.; Baker, J.S.; Macala, M.K.; Hopkinson, D. The effect of Poly(Ethylene oxide) cross-linking structure on the mechanical properties and CO2 separation performance of an ion gel membrane. Polymer 2019, 180. [Google Scholar] [CrossRef]
- Kusuma, V.A.; Macala, M.K.; Liu, J.; Marti, A.M.; Hirsch, R.J.; Hill, L.J.; Hopkinson, D. Ionic liquid compatibility in polyethylene oxide/siloxane ion gel membranes. J. Membr. Sci. 2018, 545, 292–300. [Google Scholar] [CrossRef]
- Zarca, G.; Horne, W.J.; Ortiz, I.; Urtiaga, A.; Bara, J.E. Synthesis and gas separation properties of poly(ionic liquid)-ionic liquid composite membranes containing a copper salt. J. Membr. Sci. 2016, 515, 109–114. [Google Scholar] [CrossRef]
- Kasahara, S.; Kamio, E.; Minami, R.; Matsuyama, H. A facilitated transport ion-gel membrane for propylene/propane separation using silver ion as a carrier. J. Membr. Sci. 2013, 431, 121–130. [Google Scholar] [CrossRef]
- Li, X.; Ding, S.; Zhang, J.; Wei, Z. Optimizing microstructure of polymer composite membranes by tailoring different ionic liquids to accelerate CO2 transport. Int. J. Greenh. Gas Control 2020, 101, 103136. [Google Scholar] [CrossRef]
- Mahdavi, H.R.; Azizi, N.; Arzani, M.; Mohammadi, T. Improved CO2/CH4 separation using a nanocomposite ionic liquid gel membrane. J. Nat. Gas Sci. Eng. 2017, 46, 275–288. [Google Scholar] [CrossRef]
- Bara, J.E.; Hatakeyama, E.S.; Gin, D.L.; Noble, R.D. Improving CO2 permeability in polymerized room-temperature ionic liquid gas separation membranes through the formation of a solid composite with a room-temperature ionic liquid. Polym. Adv. Technol. 2008, 19, 1415–1420. [Google Scholar] [CrossRef]
- Bara, J.E.; Gin, D.L.; Noble, R.D. Effect of Anion on Gas Separation Performance of Polymer−Room-Temperature Ionic Liquid Composite Membranes. Ind. Eng. Chem. Res. 2008, 47, 9919–9924. [Google Scholar] [CrossRef]
- Bara, J.E.; Noble, R.D.; Gin, D.L. Effect of “Free” Cation Substituent on Gas Separation Performance of Polymer−Room-Temperature Ionic Liquid Composite Membranes. Ind. Eng. Chem. Res. 2009, 48, 4607–4610. [Google Scholar] [CrossRef]
- Carlisle, T.K.; Nicodemus, G.D.; Gin, D.L.; Noble, R.D. CO2/light gas separation performance of cross-linked poly(vinylimidazolium) gel membranes as a function of ionic liquid loading and cross-linker content. J. Membr. Sci. 2012, 397–398, 24–37. [Google Scholar] [CrossRef]
- Cowan, M.G.; Gin, D.L.; Noble, R.D. Poly(ionic liquid)/Ionic Liquid Ion-Gels with High “Free” Ionic Liquid Content: Platform Membrane Materials for CO2 /Light Gas Separations. Acc. Chem. Res. 2016, 49, 724–732. [Google Scholar] [CrossRef]
- Singh, Z.V.; Cowan, M.G.; McDanel, W.M.; Luo, Y.; Zhou, R.; Gin, D.L.; Noble, R.D. Determination and optimization of factors affecting CO2/CH4 separation performance in poly(ionic liquid)-ionic liquid-zeolite mixed-matrix membranes. J. Membr. Sci. 2016, 509, 149–155. [Google Scholar] [CrossRef]
- Zhou, J.; Mok, M.M.; Cowan, M.G.; McDanel, W.M.; Carlisle, T.K.; Gin, D.L.; Noble, R.D. High-Permeance Room-Temperature Ionic-Liquid-Based Membranes for CO2/N2 Separation. Ind. Eng. Chem. Res. 2014, 53, 20064–20067. [Google Scholar] [CrossRef]
- Zanatta, M.; Lopes, M.; Cabrita, E.J.; Bernardes, C.E.S.; Corvo, M.C. Handling CO2 sorption mechanism in PIL@IL composites. J. CO2 Util. 2020, 41, 101225. [Google Scholar] [CrossRef]
- Teodoro, R.M.; Tomé, L.C.; Mantione, D.; Mecerreyes, D.; Marrucho, I.M. Mixing poly(ionic liquid)s and ionic liquids with different cyano anions: Membrane forming ability and CO2/N2 separation properties. J. Membr. Sci. 2018, 552, 341–348. [Google Scholar] [CrossRef]
- Tomé, L.C.; Guerreiro, D.C.; Teodoro, R.M.; Alves, V.D.; Marrucho, I.M. Effect of polymer molecular weight on the physical properties and CO2/N2 separation of pyrrolidinium-based poly(ionic liquid) membranes. J. Membr. Sci. 2018, 549, 267–274. [Google Scholar] [CrossRef]
- Lopez, A.M.; Cowan, M.G.; Gin, D.L.; Noble, R.D. Phosphonium-Based Poly(ionic liquid)/Ionic Liquid Ion Gel Membranes: Influence of Structure and Ionic Liquid Loading on Ion Conductivity and Light Gas Separation Performance. J. Chem. Eng. Data 2018, 63, 1154–1162. [Google Scholar] [CrossRef]
- Gouveia, A.S.L.; Ventaja, L.; Tomé, L.C.; Marrucho, I.M. Towards biohydrogen separation using poly(ionic liquid)/ionic liquid composite membranes. Membranes 2018, 8, 124. [Google Scholar] [CrossRef]
- Gouveia, A.S.L.; Yáñez, M.; Alves, V.D.; Palomar, J.; Moya, C.; Gorri, D.; Tomé, L.C.; Marrucho, I.M. CO2/H2 separation through poly(ionic liquid)–ionic liquid membranes: The effect of multicomponent gas mixtures, temperature and gas feed pressure. Sep. Purif. Technol. 2020, 118113. [Google Scholar] [CrossRef]
- Kammakakam, I.; O’Harra, K.E.; Jackson, E.M.; Bara, J.E. Synthesis of Imidazolium-Mediated Poly(benzoxazole) Ionene and Composites with Ionic Liquids as Advanced Gas Separation Membranes. Polymer 2020, 123239. [Google Scholar] [CrossRef]
- Mohshim, D.F.; Mukhtar, H.; Man, Z. The effect of incorporating ionic liquid into polyethersulfone-SAPO34 based mixed matrix membrane on CO2 gas separation performance. Sep. Purif. Technol. 2014, 135, 252–258. [Google Scholar] [CrossRef]
- Mohshim, D.F.; Mukhtar, H.; Man, Z. A study on carbon dioxide removal by blending the ionic liquid in membrane synthesis. Sep. Purif. Technol. 2018, 196, 20–26. [Google Scholar] [CrossRef]
- Hudiono, Y.C.; Carlisle, T.K.; Bara, J.E.; Zhang, Y.; Gin, D.L.; Noble, R.D. A three-component mixed-matrix membrane with enhanced CO2 separation properties based on zeolites and ionic liquid materials. J. Membr. Sci. 2010, 350, 117–123. [Google Scholar] [CrossRef]
- Shindo, R.; Kishida, M.; Sawa, H.; Kidesaki, T.; Sato, S.; Kanehashi, S.; Nagai, K. Characterization and gas permeation properties of polyimide/ZSM-5 zeolite composite membranes containing ionic liquid. J. Membr. Sci. 2014, 454, 330–338. [Google Scholar] [CrossRef]
- Ilyas, A.; Muhammad, N.; Gilani, M.A.; Vankelecom, I.F.J.; Khan, A.L. Effect of zeolite surface modification with ionic liquid [APTMS][Ac] on gas separation performance of mixed matrix membranes. Sep. Purif. Technol. 2018, 205, 176–183. [Google Scholar] [CrossRef]
- Li, H.; Tuo, L.; Yang, K.; Jeong, H.-K.; Dai, Y.; He, G.; Zhao, W. Simultaneous enhancement of mechanical properties and CO2 selectivity of ZIF-8 mixed matrix membranes: Interfacial toughening effect of ionic liquid. J. Membr. Sci. 2016, 511, 130–142. [Google Scholar] [CrossRef]
- Hao, L.; Li, P.; Yang, T.; Chung, T.-S. Room temperature ionic liquid/ZIF-8 mixed-matrix membranes for natural gas sweetening and post-combustion CO2 capture. J. Membr. Sci. 2013, 436, 221–231. [Google Scholar] [CrossRef]
- Vu, M.-T.; Lin, R.; Diao, H.; Zhu, Z.; Bhatia, S.K.; Smart, S. Effect of ionic liquids (ILs) on MOFs/polymer interfacial enhancement in mixed matrix membranes. J. Membr. Sci. 2019, 587, 117157. [Google Scholar] [CrossRef]
- Huang, G.; Isfahani, A.P.; Muchtar, A.; Sakurai, K.; Shrestha, B.B.; Qin, D.; Yamaguchi, D.; Sivaniah, E.; Ghalei, B. Pebax/ionic liquid modified graphene oxide mixed matrix membranes for enhanced CO2 capture. J. Membr. Sci. 2018, 565, 370–379. [Google Scholar] [CrossRef]
- Casado-Coterillo, C.; Del Mar López-Guerrero, M.; Irabien, A. Synthesis and Characterisation of ETS-10/Acetate-based Ionic Liquid/Chitosan Mixed Matrix Membranes for CO2/N2 Permeation. Membranes 2014, 4, 287–301. [Google Scholar] [CrossRef]
- Dani, A.; Crocellà, V.; Latini, G.; Bordiga, S. CHAPTER 2 Porous Ionic Liquid Materials. In Polymerized Ionic Liquids; The Royal Society of Chemistry: London, UK, 2008; pp. 23–82. ISBN 978-1-78262-960-3. [Google Scholar]
- Bhavsar, R.S.; Kumbharkar, S.C.; Kharul, U.K. Polymeric ionic liquids (PILs): Effect of anion variation on their CO2 sorption. J. Membr. Sci. 2012, 389, 305–315. [Google Scholar] [CrossRef]
- Magalhães, T.O.; Aquino, A.S.; Vecchia, F.D.; Bernard, F.L.; Seferin, M.; Menezes, S.C.; Ligabue, R.; Einloft, S. Syntheses and characterization of new poly(ionic liquid)s designed for CO2 capture. RSC Adv. 2014, 4, 18164–18170. [Google Scholar] [CrossRef]
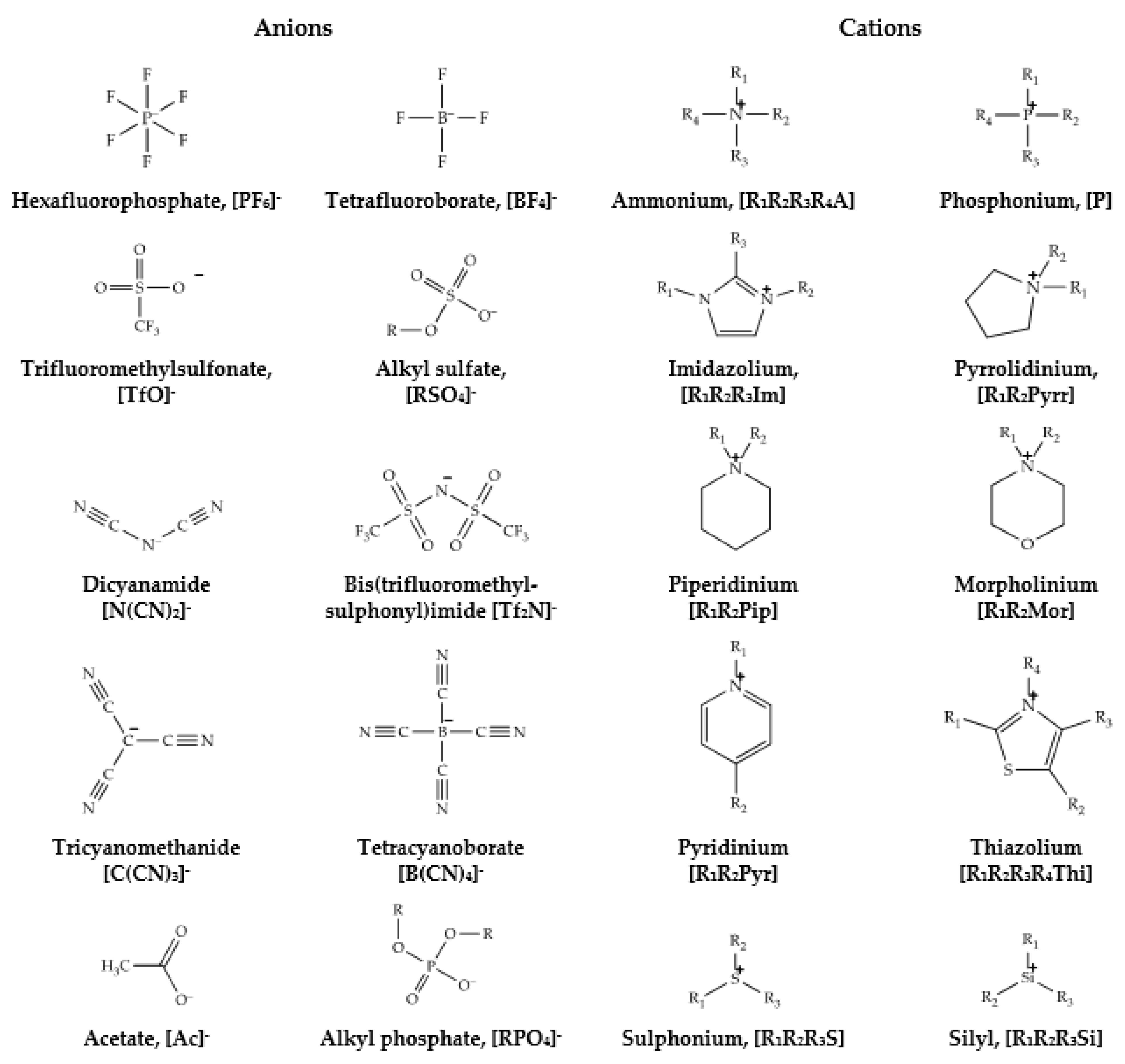
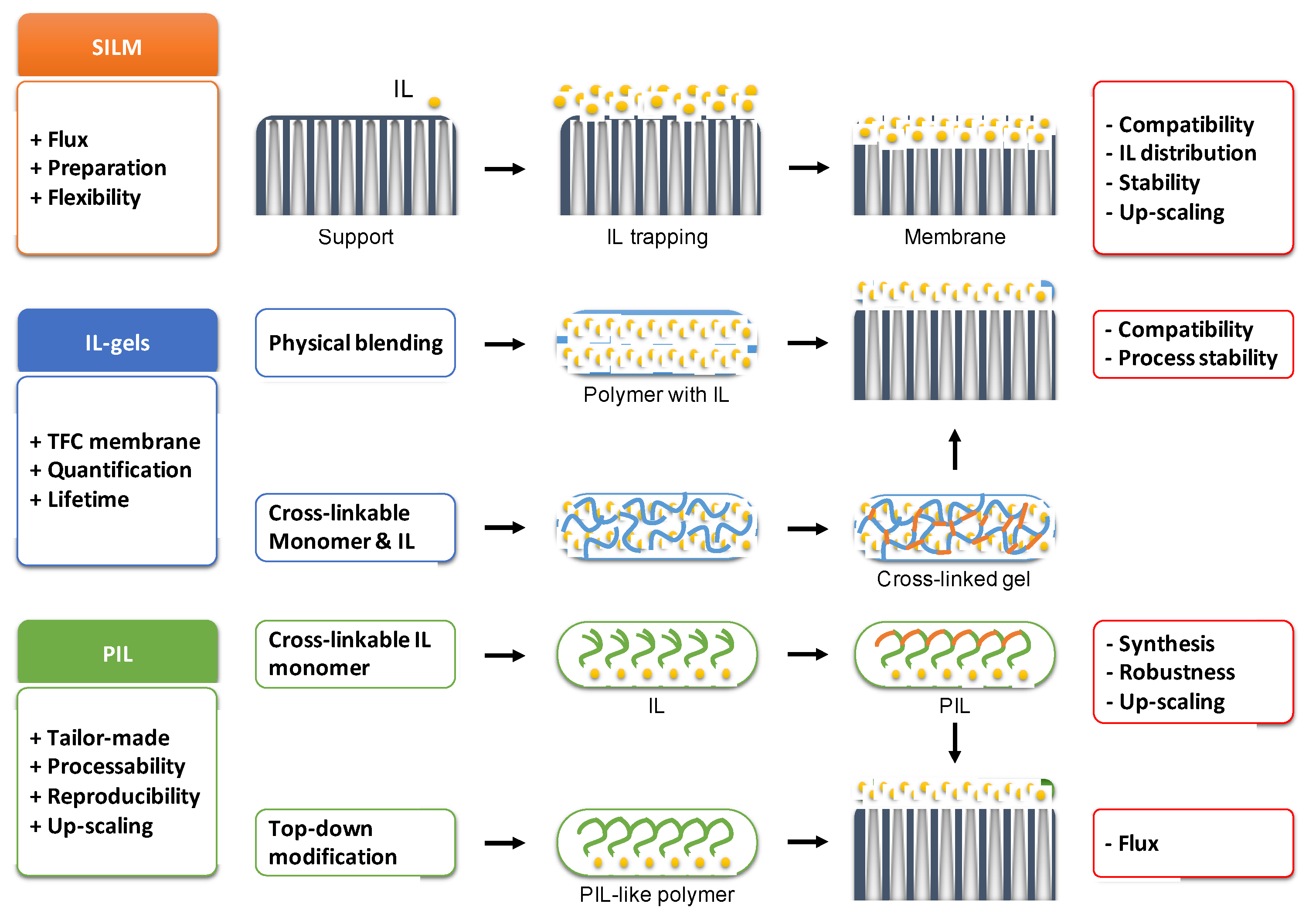
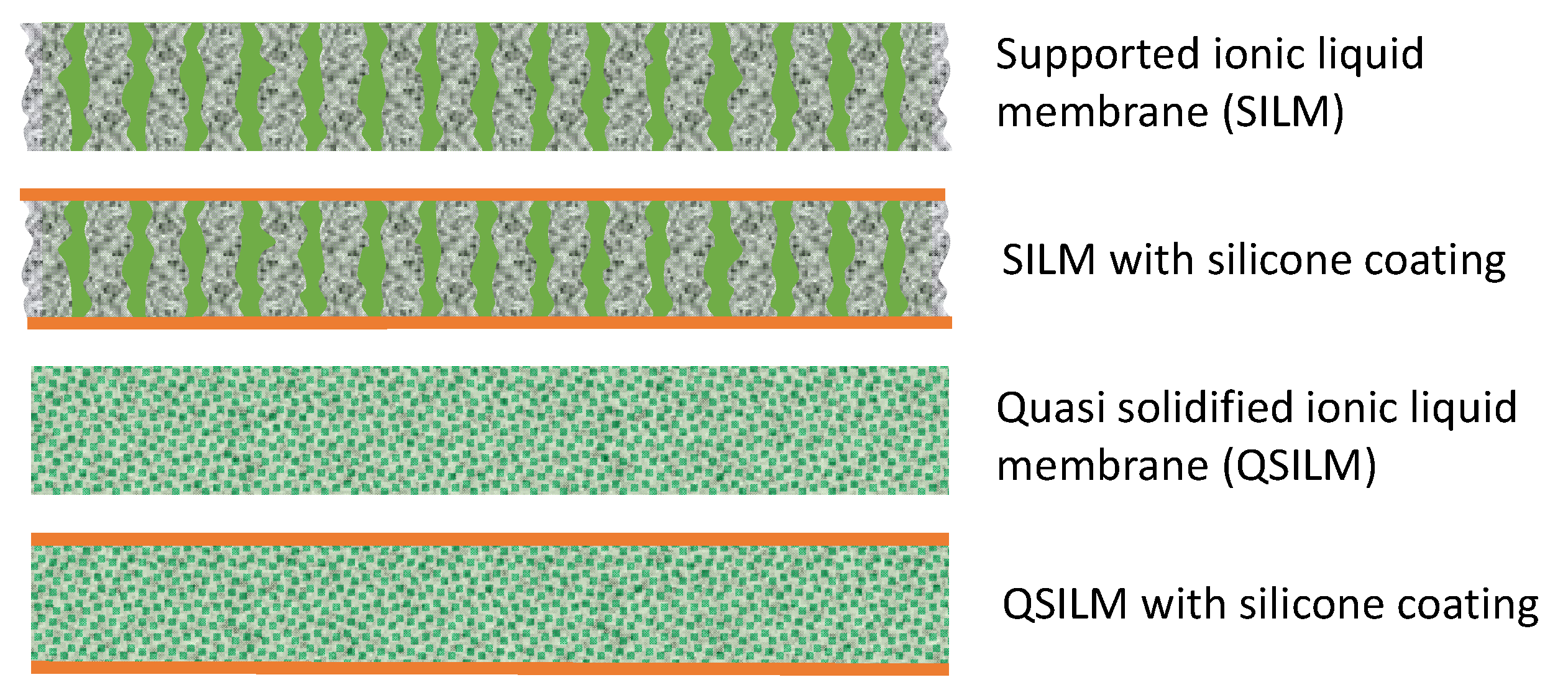
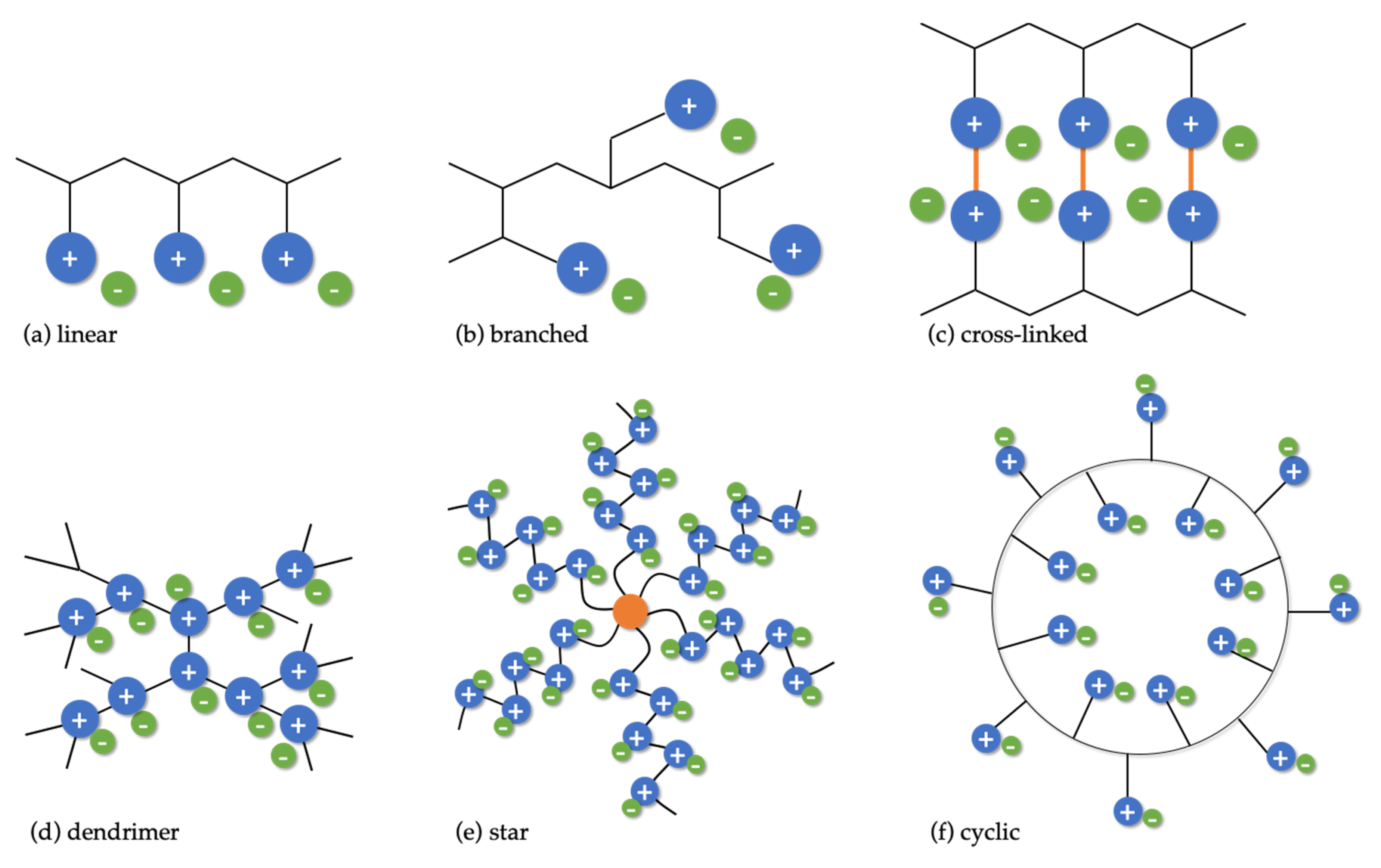
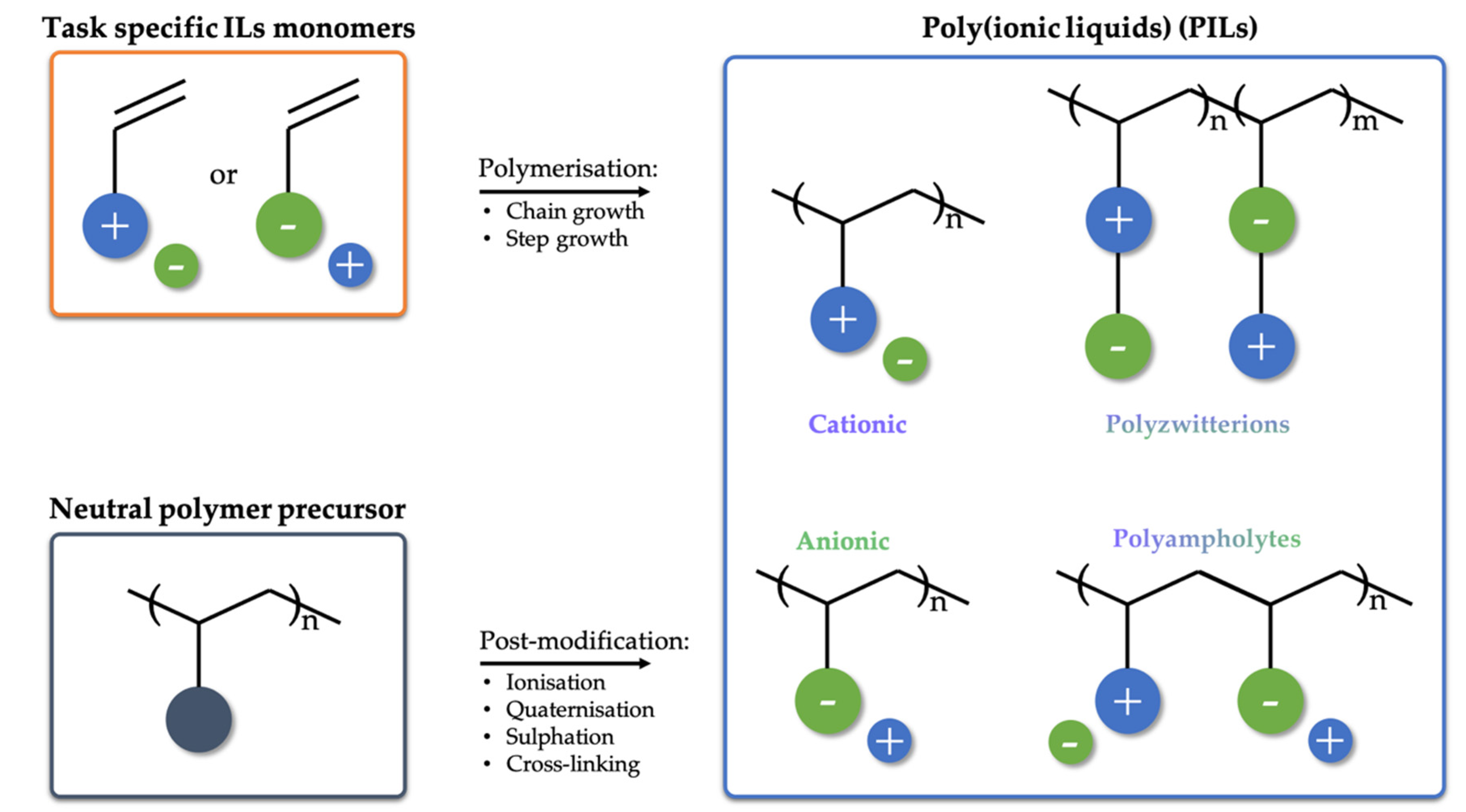
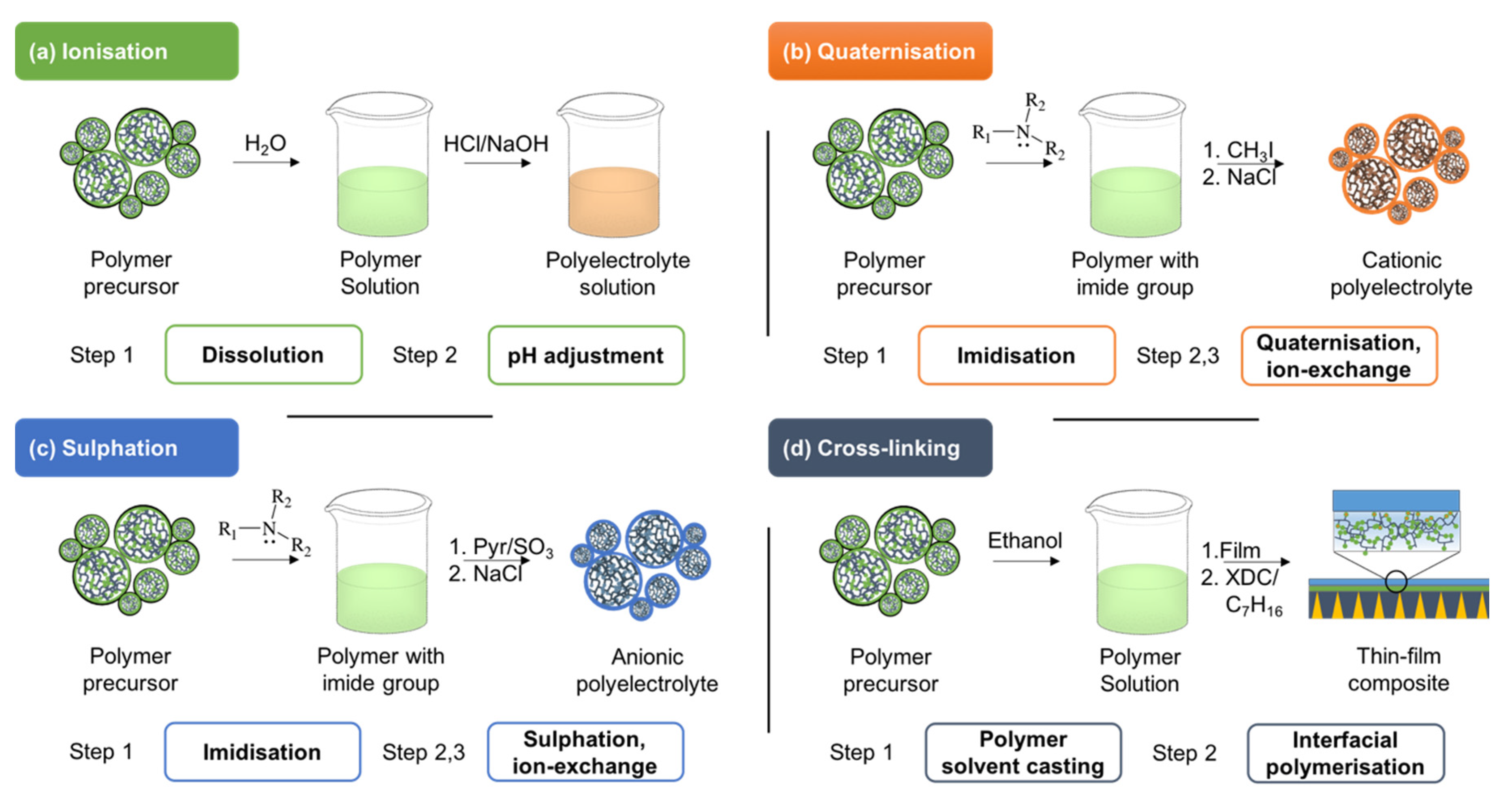


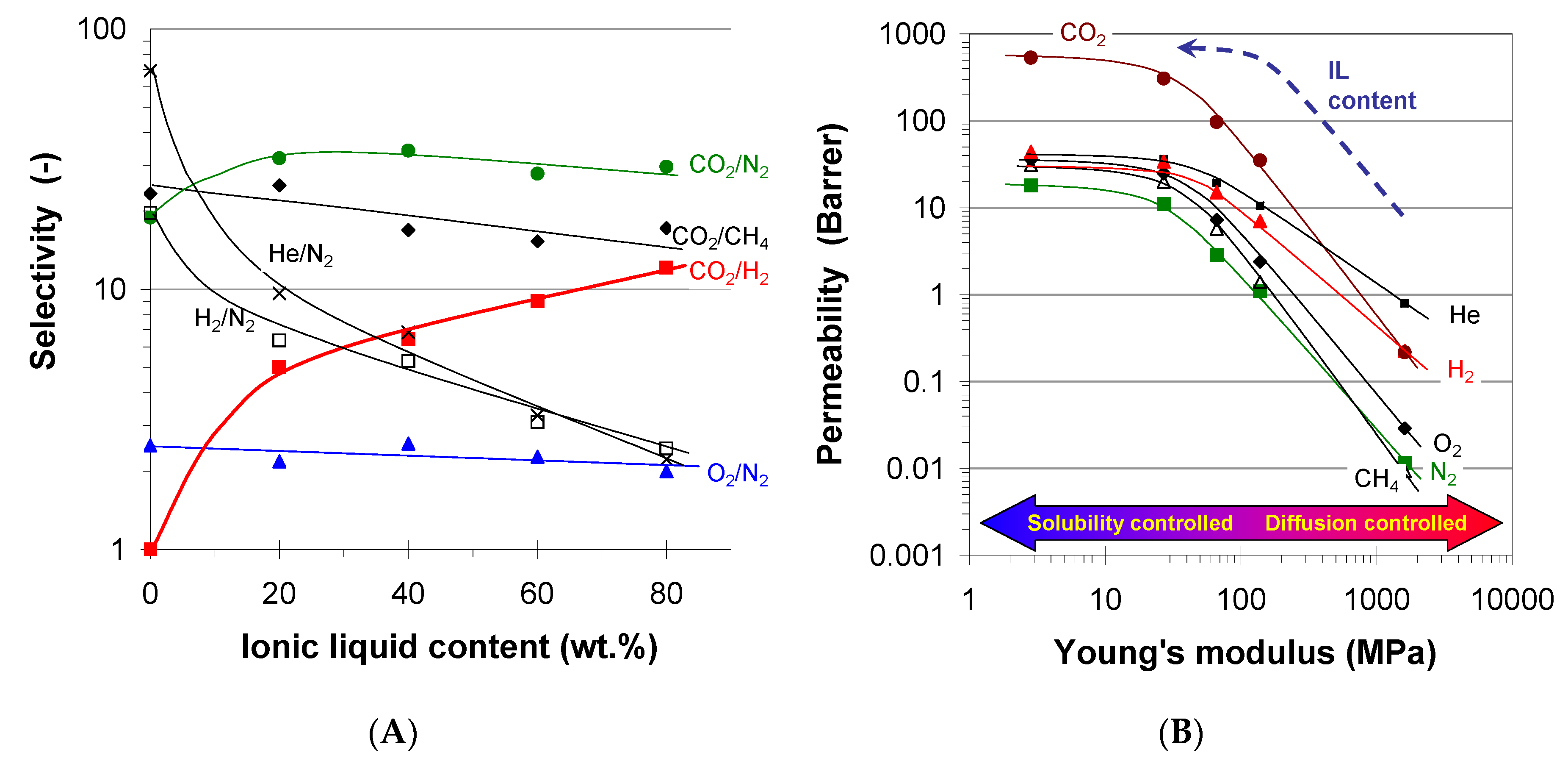
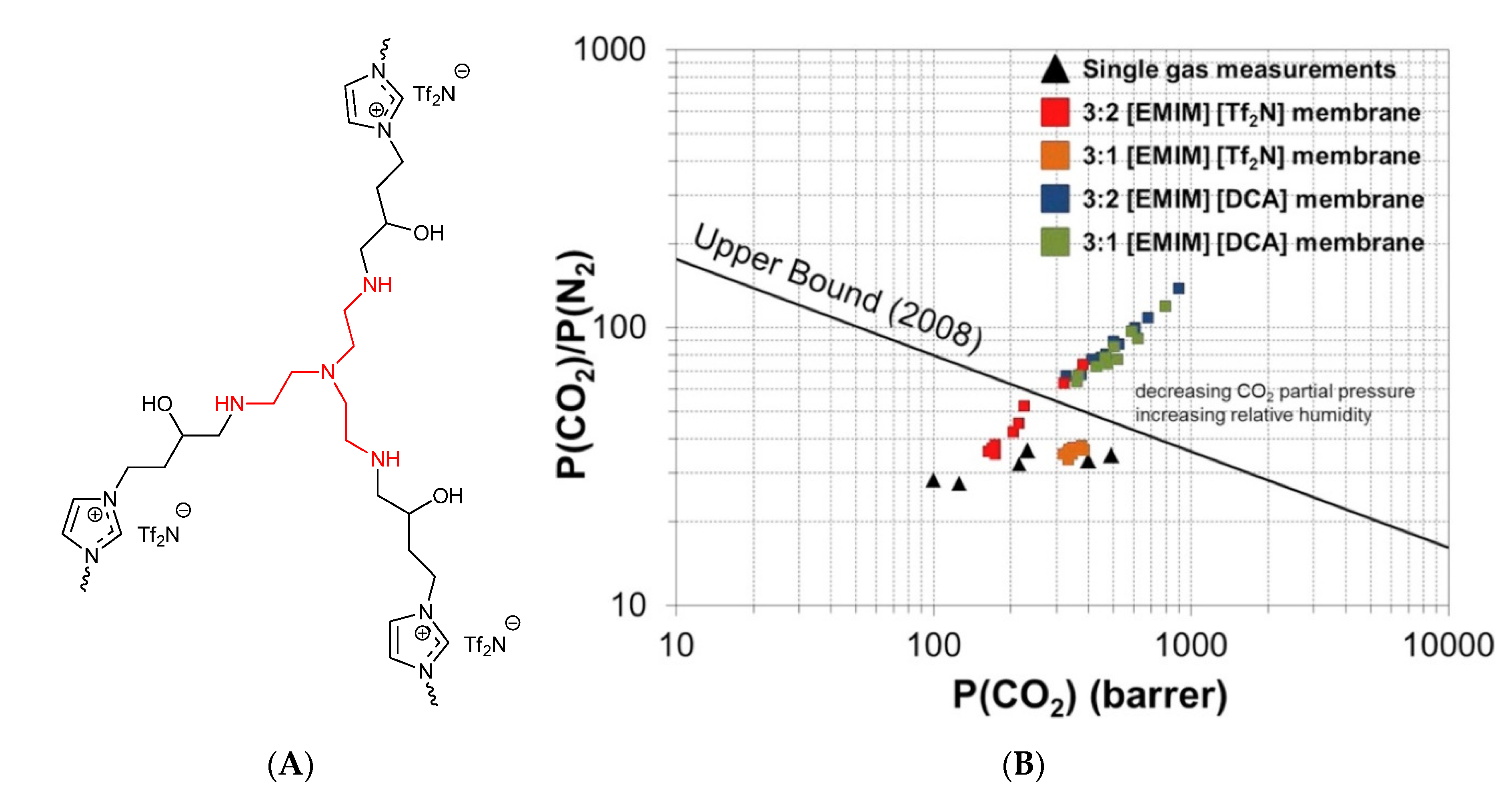
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Friess, K.; Izák, P.; Kárászová, M.; Pasichnyk, M.; Lanč, M.; Nikolaeva, D.; Luis, P.; Jansen, J.C. A Review on Ionic Liquid Gas Separation Membranes. Membranes 2021, 11, 97. https://doi.org/10.3390/membranes11020097
Friess K, Izák P, Kárászová M, Pasichnyk M, Lanč M, Nikolaeva D, Luis P, Jansen JC. A Review on Ionic Liquid Gas Separation Membranes. Membranes. 2021; 11(2):97. https://doi.org/10.3390/membranes11020097
Chicago/Turabian StyleFriess, Karel, Pavel Izák, Magda Kárászová, Mariia Pasichnyk, Marek Lanč, Daria Nikolaeva, Patricia Luis, and Johannes Carolus Jansen. 2021. "A Review on Ionic Liquid Gas Separation Membranes" Membranes 11, no. 2: 97. https://doi.org/10.3390/membranes11020097
APA StyleFriess, K., Izák, P., Kárászová, M., Pasichnyk, M., Lanč, M., Nikolaeva, D., Luis, P., & Jansen, J. C. (2021). A Review on Ionic Liquid Gas Separation Membranes. Membranes, 11(2), 97. https://doi.org/10.3390/membranes11020097











