A Comparison of the Level of Acceptance and Hesitancy towards the Influenza Vaccine and the Forthcoming COVID-19 Vaccine in the Medical Community
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample and Settings
2.2. Procedures
2.3. Data Collection
2.4. Analyses
3. Results
3.1. Study Group
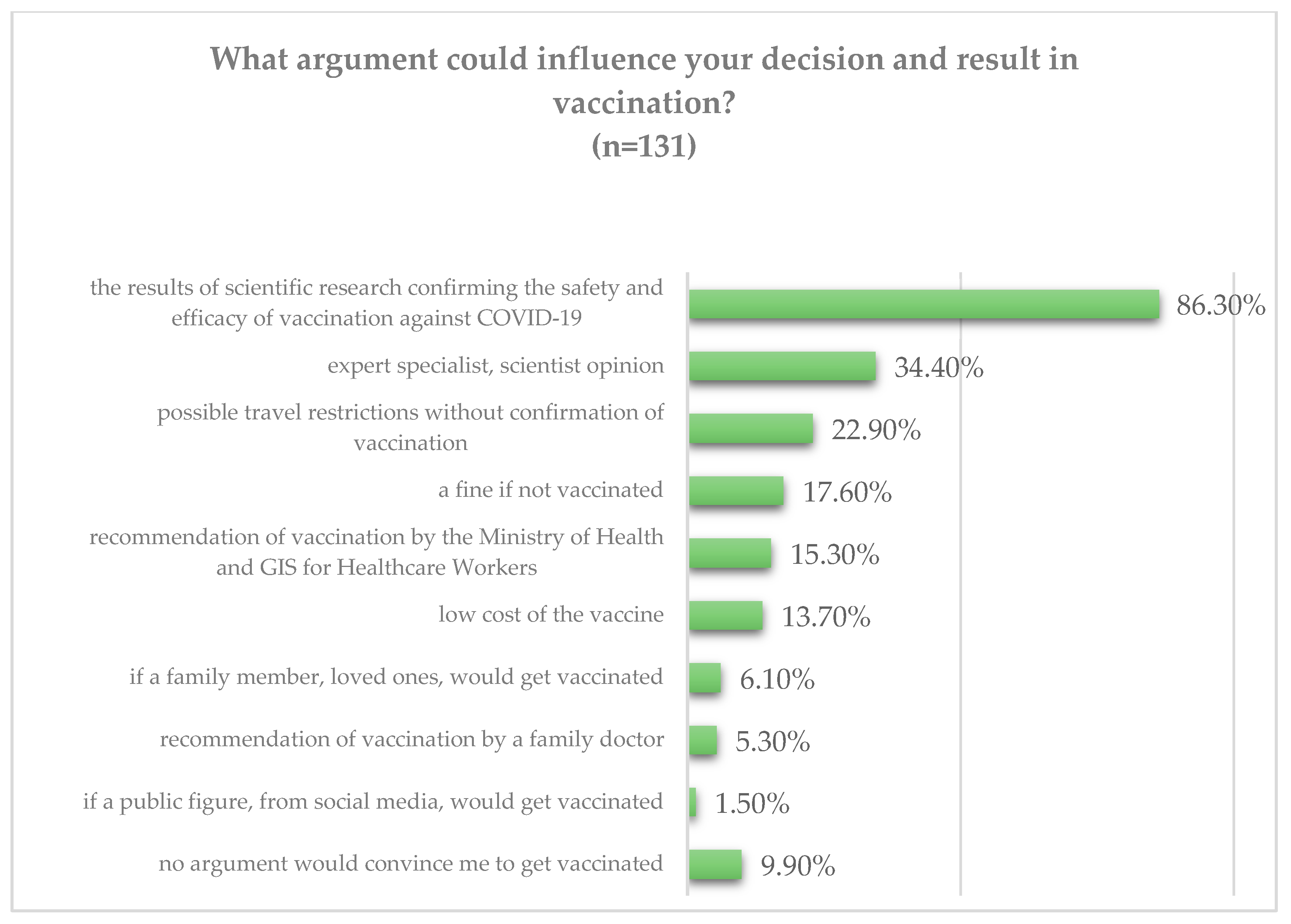
3.2. Evidence-Based Data on the Safety and Efficacy of COVID-19 Vaccination Is the Primary Convincing Argument to Get Vaccinated among HCWs and MS
3.3. Acceptance of Flu Vaccination among HCWs and MS Is a Strong Predictor for Attitude towards the Current COVID-19 Vaccination
3.4. Lower Age Is Associated with Higher COVID-19 Vaccine Acceptance
3.5. A Discrepancy in Vaccination Willingness between Physicians and Nurses
3.6. Almost 50% of Respondents Not Vaccinated against the Flu during the 2019/2020 Season Declared an Intent to Get the Flu Vaccine in the 2020/2021
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 324, 782–793. [Google Scholar] [CrossRef]
- Yang, J.; Zheng, Y.; Gou, X.; Pu, K.; Chen, Z.; Guo, Q.; Ji, R.; Wang, H.; Wang, Y.; Zhou, Y. Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: A systematic review and meta-analysis. Int. J. Infect. Dis. 2020, 94, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Jain, V.; Yuan, J.M. Predictive symptoms and comorbidities for severe COVID-19 and intensive care unit admission: A systematic review and meta-analysis. Int. J. Public Health 2020, 65, 533–546. [Google Scholar] [CrossRef] [PubMed]
- Docherty, A.B.; Harrison, E.M.; Green, C.A.; Hardwick, H.E.; Pius, R.; Norman, L.; Holden, K.A.; Read, J.M.; Dondelinger, F.; Carson, G.; et al. Features of 20,133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: Prospective observational cohort study. BMJ 2020, 369, m1985. [Google Scholar] [CrossRef] [PubMed]
- Sahu, A.K.; Amrithanand, V.T.; Mathew, R.; Aggarwal, P.; Nayer, J.; Bhoi, S. COVID-19 in health care workers—A systematic review and meta-analysis. Am. J. Emerg. Med. 2020, 38, 1727–1731. [Google Scholar] [CrossRef]
- WHO. COVID-19 Vaccines. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/covid-19-vaccines (accessed on 24 March 2021).
- Feleszko, W.; Lewulis, P.; Czarnecki, A.; Waszkiewicz, P. Flattening the Curve of COVID-19 Vaccine Rejection-An International Overview. Vaccines 2021, 9, 44. [Google Scholar] [CrossRef]
- Paules, C.; Subbarao, K. Influenza. Lancet 2017, 390, 697–708. [Google Scholar] [CrossRef]
- Houser, K.; Subbarao, K. Influenza vaccines: Challenges and solutions. Cell Host Microbe 2015, 17, 295–300. [Google Scholar] [CrossRef]
- Piroth, L.; Cottenet, J.; Mariet, A.-S.; Bonniaud, P.; Blot, M.; Tubert-Bitter, P.; Quantin, C. Comparison of the characteristics, morbidity, and mortality of COVID-19 and seasonal influenza: A nationwide, population-based retrospective cohort study. Lancet Respir. Med. 2021, 9, 251–259. [Google Scholar] [CrossRef]
- Paget, J.; Spreeuwenberg, P.; Charu, V.; Taylor, R.J.; Iuliano, A.D.; Bresee, J.; Simonsen, L.; Viboud, C. Global mortality associated with seasonal influenza epidemics: New burden estimates and predictors from the GLaMOR Project. J. Glob. Health 2019, 9, 020421. [Google Scholar] [CrossRef] [PubMed]
- OECD. Influenza Vaccination Rates (Indicator). Available online: https://www.oecd-ilibrary.org/social-issues-migration-health/influenza-vaccination-rates/indicator/english_e452582e-en (accessed on 1 April 2021).
- Dini, G.; Toletone, A.; Sticchi, L.; Orsi, A.; Bragazzi, N.L.; Durando, P. Influenza vaccination in healthcare workers: A comprehensive critical appraisal of the literature. Hum. Vaccines Immunother. 2018, 14, 772–789. [Google Scholar] [CrossRef]
- Paterson, P.; Meurice, F.; Stanberry, L.R.; Glismann, S.; Rosenthal, S.L.; Larson, H.J. Vaccine hesitancy and healthcare providers. Vaccine 2016, 34, 6700–6706. [Google Scholar] [CrossRef]
- Aveni, E.; Bauer, B.; Ramelet, A.S.; Decosterd, I.; Ballabeni, P.; Bonvin, E.; Rodondi, P.Y. Healthcare professionals’ sources of knowledge of complementary medicine in an academic center. PLoS ONE 2017, 12, e0184979. [Google Scholar] [CrossRef]
- OECD. Science Advice in Times of COVID-19. Available online: https://www.oecd.org/sti/science-technology-innovation-outlook/Science-advice-COVID/ (accessed on 18 April 2021).
- Brenner, P.S.; De Lamater, J. Lies, Damned Lies, and Survey Self-Reports? Identity as a Cause of Measurement Bias. Soc. Psychol. Q. 2016, 79, 333–354. [Google Scholar] [CrossRef]
- Antczak, A.; Ciszewski, A.; Jahnz-Różyk, K.; Mastalerz-Migas, A.; Nitsch-Osuch ASzenborn, L. Szczepienia Przeciw Grypie Wśród Pracowników Ochrony Zdrowia w Polsce-Teraźniejszość i Perspektywy; Instytut Oświaty Zdrowotnej Fundacja Haliny Osińskiej: Warsaw, Poland, 2013; pp. 8–9. ISBN 978-83-937893-2-0. [Google Scholar]
- PZH (National Institute of Public Health-National Institute of Hygiene). Przekonujemy, że w Sezonie 2014/2015 Warto Zaszczepić Się Przeciw Grypie. Available online: https://szczepienia.pzh.gov.pl/przekonujemy-ze-sezonie-20142015-warto-zaszczepic-sie-przeciw-grypie/ (accessed on 18 April 2021).
- WHO. Ten Threats to Global Health in 2019. Available online: https://www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019 (accessed on 23 March 2021).
- Lazarus, J.V.; Ratzan, S.C.; Palayew, A.; Gostin, L.O.; Larson, H.J.; Rabin, K.; Kimball, S.; El-Mohandes, A. A global survey of potential acceptance of a COVID-19 vaccine. Nat. Med. 2021, 27, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Szmyd, B.; Karuga, F.F.; Bartoszek, A.; Staniecka, K.; Siwecka, N.; Bartoszek, A.; Błaszczyk, M.; Radek, M. Attitude and Behaviors towards SARS-CoV-2 Vaccination among Healthcare Workers: A Cross-Sectional Study from Poland. Vaccines 2021, 9, 218. [Google Scholar] [CrossRef]
- Detoc, M.; Bruel, S.; Frappe, P.; Tardy, B.; Botelho-Nevers, E.; Gagneux-Brunon, A. Intention to participate in a COVID-19 vaccine clinical trial and to get vaccinated against COVID-19 in France during the pandemic. Vaccine 2020, 38, 7002–7006. [Google Scholar] [CrossRef] [PubMed]
- Shekhar, R.; Sheikh, A.B.; Upadhyay, S.; Singh, M.; Kottewar, S.; Mir, H.; Barrett, E.; Pal, S. COVID-19 Vaccine Acceptance among Health Care Workers in the United States. Vaccines 2021, 9, 119. [Google Scholar] [CrossRef]
- Verger, P.; Scronias, D.; Dauby, N.; Adedzi, K.A.; Gobert, C.; Bergeat, M.; Gagneur, A.; Dubé, E. Attitudes of healthcare workers towards COVID-19 vaccination: A survey in France and French-speaking parts of Belgium and Canada, 2020. Eurosurveillance 2021, 26. [Google Scholar] [CrossRef]
- Grech, V.; Gauci, C.; Agius, S. Withdrawn: Vaccine hesitancy among Maltese Healthcare workers toward influenza and novel COVID-19 vaccination. Early Hum. Dev. 2020, 105213. [Google Scholar] [CrossRef]
- Dror, A.A.; Eisenbach, N.; Taiber, S.; Morozov, N.G.; Mizrachi, M.; Zigron, A.; Srouji, S.; Sela, E. Vaccine hesitancy: The next challenge in the fight against COVID-19. Eur. J. Epidemiol. 2020, 35, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Tamang, N.; Rai, P.; Dhungana, S.; Sherchan, B.; Shah, B.; Pyakurel, P.; Rai, S. COVID-19: A National Survey on perceived level of knowledge, attitude and practice among frontline healthcare Workers in Nepal. BMC Public Health 2020, 20, 1905. [Google Scholar] [CrossRef]
- Kadoya, Y.; Zen, K.; Wakana, N.; Yanishi, K.; Senoo, K.; Nakanishi, N.; Yamano, T.; Nakamura, T.; Matoba, S. Knowledge, perception, and level of confidence regarding COVID-19 care among healthcare workers involved in cardiovascular medicine: A web-based cross-sectional survey in Japan. J. Cardiol. 2021, 77, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Pelullo, C.P.; Della Polla, G.; Napolitano, F.; Di Giuseppe, G.; Angelillo, I.F. Healthcare Workers’ Knowledge, Attitudes, and Practices about Vaccinations: A Cross-Sectional Study in Italy. Vaccines 2020, 8, 148. [Google Scholar] [CrossRef]
- Kwok, K.O.; Li, K.K.; Wei, W.I.; Tang, A.; Wong, S.Y.S.; Lee, S.S. Editor’s Choice: Influenza vaccine uptake, COVID-19 vaccination intention and vaccine hesitancy among nurses: A survey. Int. J. Nurs. Stud. 2021, 114, 103854. [Google Scholar] [CrossRef]
- Szmyd, B.; Bartoszek, A.; Karuga, F.F.; Staniecka, K.; Błaszczyk, M.; Radek, M. Medical Students and SARS-CoV-2 Vaccination: Attitude and Behaviors. Vaccines 2021, 9, 128. [Google Scholar] [CrossRef]
- Barello, S.; Nania, T.; Dellafiore, F.; Graffigna, G.; Caruso, R. ‘Vaccine hesitancy’ among university students in Italy during the COVID-19 pandemic. Eur. J. Epidemiol. 2020, 35, 781–783. [Google Scholar] [CrossRef]
- Gagneux-Brunon, A.; Detoc, M.; Bruel, S.; Tardy, B.; Rozaire, O.; Frappe, P.; Botelho-Nevers, E. Intention to get vaccinations against COVID-19 in French healthcare workers during the first pandemic wave: A cross-sectional survey. J. Hosp. Infect. 2021, 108, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Kose, S.; Mandiracioglu, A.; Sahin, S.; Kaynar, T.; Karbus, O.; Ozbel, Y. Vaccine hesitancy of the COVID-19 by health care personnel. Int. J. Clin. Pract. 2020, e13917. [Google Scholar] [CrossRef]
- Di Pumpo, M.; Vetrugno, G.; Pascucci, D.; Carini, E.; Beccia, V.; Sguera, A.; Zega, M.; Pani, M.; Cambieri, A.; Nurchis, M.C.; et al. Is COVID-19 a Real Incentive for Flu Vaccination? Let the Numbers Speak for Themselves. Vaccines 2021, 9, 276. [Google Scholar] [CrossRef]
- Dooling, K.; McClung, N.; Chamberland, M.; Marin, M.; Wallace, M.; Bell, B.P.; Lee, G.M.; Talbot, H.K.; Romero, J.R.; Oliver, S.E. The Advisory Committee on Immunization Practices’ Interim Recommendation for Allocating Initial Supplies of COVID-19 Vaccine—United States, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1857–1859. [Google Scholar] [CrossRef]
- Gov.pl, P. Pierwszy Miesiąc Szczepień Przeciw Covid-19 za Nami—Szczepienie Przeciwko COVID-19. Available online: https://www.gov.pl/web/szczepimysie/pierwszy-miesiac-szczepien-przeciw-covid-19-za-nami?fbclid=IwAR0C77xsqDP9nLJeAOSdDmehlJFq0wKBZBBTRXm3gBcM0VVfCLl7T7mIhrU (accessed on 23 March 2021).
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
| Variables | Total | Profession Groups; n (%) | |||
|---|---|---|---|---|---|
| Medical Students | Medical Doctors | Nurses | Other AHP * | ||
| Total; n (%) | 419 (100) | 239 (57.0) | 156 (37.2) | 18 (4.3) | 6 (1.4) |
| Male; n (%) | 88 (21) | 50 (56.8) | 38 (43.2) | 0 | 0 |
| Mean age (range) | 27.47 (19–78) | 23 (19–31) | 34 (24–78) | 46 (22–57) | 28 (25–32) |
| Age groups | |||||
| 19–25 | 253 | 220 (87) | 28 (11.1) | 2 (0.8) | 3 (1.2) |
| 26–30 | 96 | 17 (17.7) | 77 (80.2) | 0 | 2 (2.1) |
| 31–40 | 34 | 2 (5.9) | 28 (82.4) | 3 (8.8) | 1 (2.9) |
| 41–50 | 20 | 0 | 13 (65) | 7 (35.0) | 0 |
| over 50 | 16 | 0 | 10 (62.5) | 6 (37.5) | 0 |
| Variables | Total; n (%) | Do You Intend to Get a Flu Vaccination in the 2020/2021 Season?; n (%) | ||
|---|---|---|---|---|
| Yes | Not Sure | No | ||
| Total; n (%) | 419 (100) | 258 (61.6) | 90 (21.5) | 71 (16.9) |
| Do you intend to get a COVID-19 vaccination if an effective and safe vaccine is developed? | ||||
| Yes | 288 (68.7) | 215 (83.3) | 53 (58.9) | 20 (28.2) |
| Not sure | 88 (21) | 36 (14) | 29 (32.2) | 23 (32.4) |
| No | 43 (10.3) | 7 (2.7) | 8 (8.9) | 28 (39.4) |
| Did you get a flu vaccine during the previous 2019/2020 season? | ||||
| Yes | 138 (32.9) | 129 (93.5) | 8 (5.8) | 1 (0.7) |
| No | 281 (67.1) | 129 (45.9) | 82 (29.2) | 70 (24.9) |
| Variables | Total; n | Do You Intend to Get a COVID-19 Vaccination If an Effective and Safe Vaccine Is Developed?; % | Do You Intend to Get a Flu Vaccination in the 2020/2021 Season?; % | ||||
|---|---|---|---|---|---|---|---|
| Yes | Not Sure | No | Yes | Not Sure | No | ||
| Total; n (%) | 419 (100) | 288 (68.7) | 88 (21) | 43 (10.3) | 258 (61.6) | 71 (16.9) | 90 (21.5) |
| Gender | |||||||
| Male | 88 | 76.1 | 15.9 | 8 | 64.8 | 23.9 | 11.4 |
| Female | 331 | 66.8 | 22.4 | 10.9 | 60.7 | 20.8 | 18.4 |
| Profession | |||||||
| Medical students | 239 | 70.7 | 19.7 | 9.6 | 54 | 28.5 | 17.6 |
| Medical Doctors | 156 | 73.1 | 19.2 | 7.7 | 76.9 | 10.3 | 12.8 |
| Nurses | 18 | 22.2 | 50 | 27.8 | 33.3 | 33.3 | 33.3 |
| other AHP * | 6 | 33.3 | 33.3 | 33.3 | 33.3 | 33.3 | 33.3 |
| Age groups | |||||||
| 19–25 | 253 | 70.8 | 19 | 10.3 | 56.1 | 25.3 | 18.6 |
| 26–30 | 96 | 72.9 | 17.7 | 9.4 | 75 | 14.6 | 10.4 |
| 31–40 | 34 | 67.6 | 29.4 | 2.9 | 70.6 | 14.7 | 14.7 |
| 41–50 | 20 | 50 | 35 | 15 | 65 | 20 | 15 |
| over 50 | 16 | 37.5 | 37.5 | 25 | 43.8 | 18.8 | 37.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grochowska, M.; Ratajczak, A.; Zdunek, G.; Adamiec, A.; Waszkiewicz, P.; Feleszko, W. A Comparison of the Level of Acceptance and Hesitancy towards the Influenza Vaccine and the Forthcoming COVID-19 Vaccine in the Medical Community. Vaccines 2021, 9, 475. https://doi.org/10.3390/vaccines9050475
Grochowska M, Ratajczak A, Zdunek G, Adamiec A, Waszkiewicz P, Feleszko W. A Comparison of the Level of Acceptance and Hesitancy towards the Influenza Vaccine and the Forthcoming COVID-19 Vaccine in the Medical Community. Vaccines. 2021; 9(5):475. https://doi.org/10.3390/vaccines9050475
Chicago/Turabian StyleGrochowska, Magdalena, Aleksandra Ratajczak, Gabriela Zdunek, Aleksander Adamiec, Paweł Waszkiewicz, and Wojciech Feleszko. 2021. "A Comparison of the Level of Acceptance and Hesitancy towards the Influenza Vaccine and the Forthcoming COVID-19 Vaccine in the Medical Community" Vaccines 9, no. 5: 475. https://doi.org/10.3390/vaccines9050475
APA StyleGrochowska, M., Ratajczak, A., Zdunek, G., Adamiec, A., Waszkiewicz, P., & Feleszko, W. (2021). A Comparison of the Level of Acceptance and Hesitancy towards the Influenza Vaccine and the Forthcoming COVID-19 Vaccine in the Medical Community. Vaccines, 9(5), 475. https://doi.org/10.3390/vaccines9050475









