Targeting COVID-19 Vaccine Hesitancy in Minority Populations in the US: Implications for Herd Immunity
Abstract
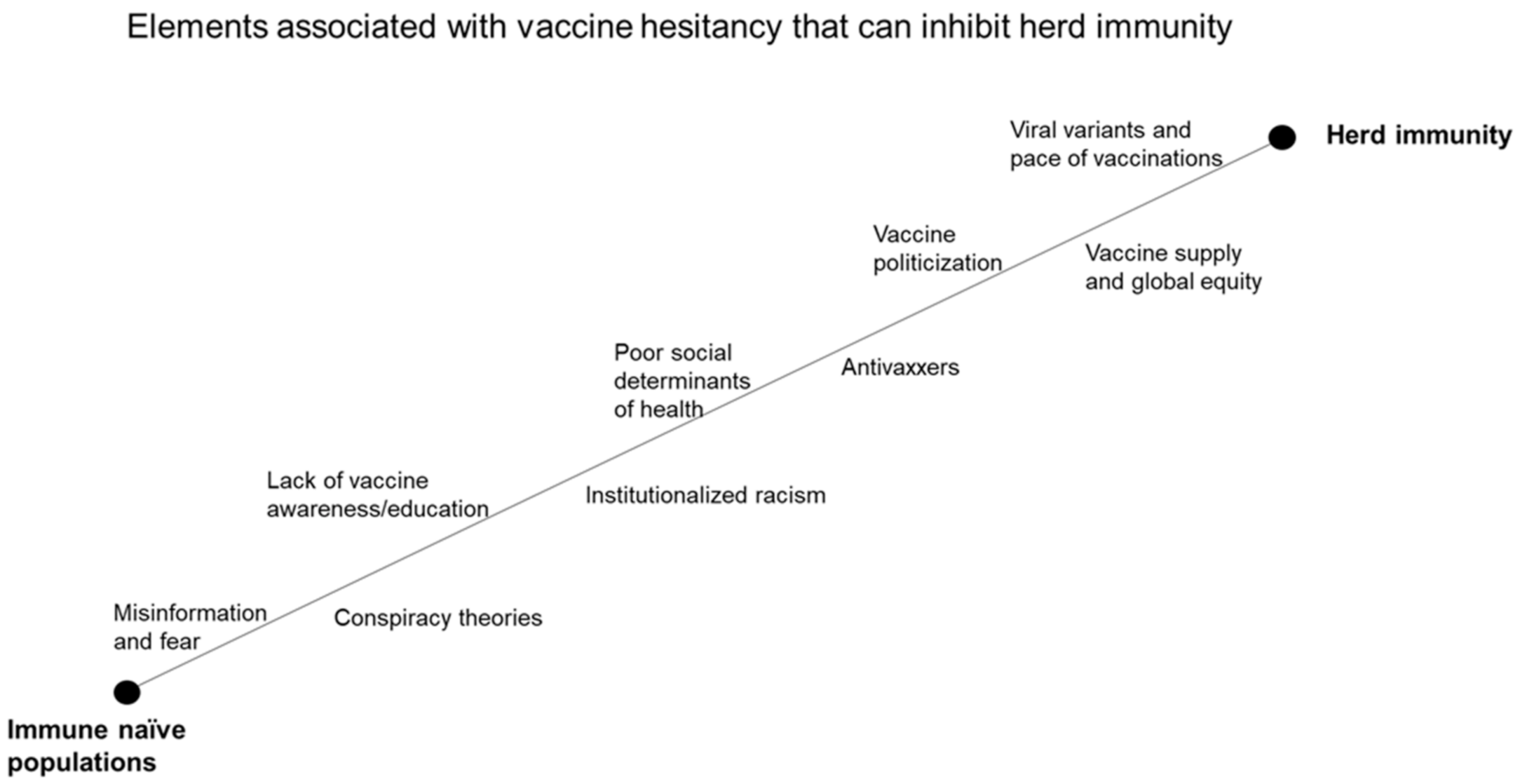
1. Introduction
2. The Safety and Efficacy of COVID-19 Vaccines
2.1. The Pfizer/BioNtech COVID-19 Vaccine (BNT162b2)
2.2. The Moderna COVID-19 Vaccine (mRNA-1273)
2.3. The J&J COVID-19 Vaccine (JNJ-78436735/Ad26.COV2.S)
2.4. Vaccine Safety
3. Vaccine Hesitancy
4. COVID-19 Vaccine Hesitancy among Major Ethnic Groups in the US
4.1. Non-Hispanic-Whites
4.2. African American
4.3. Hispanic/Latinx
5. Hesitancy Associated with Vaccine Supply and Global Inequities
6. Vaccine Hesitancy among Healthcare Providers
7. The Role of Antivaxxers in Promoting Vaccine Hesitancy
8. Strategies Targeting Vaccine Hesitancy to Achieve Equity in COVID-19 Vaccination
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alcendor, D.J. Racial Disparities-Associated COVID-19 Mortality among Minority Populations in the US. J. Clin. Med. 2020, 8, 2442. [Google Scholar] [CrossRef] [PubMed]
- Webb Hooper, M.; Nápoles, A.M.; Pérez-Stable, E.J. COVID-19 and Racial/Ethnic Disparities. JAMA 2020, 24, 2466–2467. [Google Scholar] [CrossRef]
- Laurencin, C.T.; McClinton, A. The COVID-19 Pandemic: A Call to Action to Identify and Address Racial and Ethnic Disparities. J. Racial Ethn. Health Disparities 2020, 3, 398–402. [Google Scholar] [CrossRef]
- Chowkwanyun, M.; Reed, A.L., Jr. Racial Health Disparities and Covid-19—Caution and Context. N. Engl. J. Med. 2020, 3, 201–203. [Google Scholar] [CrossRef]
- Bambra, C.; Riordan, R.; Ford, J.; Matthews, F. The COVID-19 pandemic and health inequalities. J. Epidemiol. Community Health 2020, 11, 964–968. [Google Scholar] [CrossRef]
- Dorn, A.V.; Cooney, R.E.; Sabin, M.L. COVID-19 exacerbating inequalities in the US. Lancet 2020, 10232, 1243–1244. [Google Scholar] [CrossRef]
- Raifman, M.A.; Raifman, J.R. Disparities in the Population at Risk of Severe Illness From COVID-19 by Race/Ethnicity and Income. Am. J. Prev. Med. 2020, 1, 137–139. [Google Scholar] [CrossRef]
- Abrams, E.M.; Szefler, S.J. COVID-19 and the impact of social determinants of health. Lancet Respir. Med. 2020, 7, 659–661. [Google Scholar] [CrossRef]
- Raine, S.; Liu, A.; Mintz, J.; Wahood, W.; Huntley, K.; Haffizulla, F. Racial and Ethnic Disparities in COVID-19 Outcomes: Social Determination of Health. Int. J. Environ. Res. Public Health 2020, 21, 8115. [Google Scholar] [CrossRef]
- Coustasse, A.; Kimble, C.; Maxik, K. COVID-19 and Vaccine Hesitancy: A Challenge the United States Must Overcome. J. Ambul. Care Manag. 2021, 1, 71–75. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Ten Threats to Global Health in 2019. 2019. Available online: https://www.who.int/emergencies/ten-threats-to-global-health-in-2019 (accessed on 25 February 2019).
- Oliver, S.E.; Gargano, J.W.; Marin, M.; Wallace, M.; Curran, K.G.; Chamberland, M.; McClung, N.; Campos-Outcalt, D.; Morgan, R.L.; Mbaeyi, S.; et al. The Advisory Committee on Immunization Practices’ Interim Recommendation for Use of Pfizer-BioNTech COVID-19 Vaccine—United States, December 2020. MMWR. Morb. Mortal. Wkly. Rep. 2020, 50, 1922–1924. [Google Scholar] [CrossRef]
- Oliver, S.E.; Gargano, J.W.; Marin, M.; Wallace, M.; Curran, K.G.; Chamberland, M.; McClung, N.; Campos-Outcalt, D.; Morgan, R.L.; Mbaeyi, S. The Advisory Committee on Immunization Practices’ Interim Recommendation for Use of Moderna COVID-19 Vaccine-United States, December 2020. MMWR. Morb. Mortal. Wkly. Rep. 2021, 5152, 1653–1656. [Google Scholar] [CrossRef]
- Livingston, E.H.; Malani, P.N.; Creech, C.B. The Johnson & Johnson Vaccine for COVID-19. JAMA 2021. [Google Scholar] [CrossRef]
- Neergaard, L.; Fingerhut, H. AP-NORC Poll: Half of Americans Would Get a COVID-19 Vaccine. University of Maryland School of Medicine, Associated Press: 27 May 2020. Available online: https://apnews.com/dacdc8bc428dd4df6511bfa259cfec44 (accessed on 10 September 2020).
- Momplaisir, F.; Haynes, N.; Nkwihoreze, H.; Nelson, M.; Werner, R.M.; Jemmott, J. Understanding Drivers of COVID-19 Vaccine Hesitancy Among Blacks. Clin. Infect Dis. 2021, ciab102. [Google Scholar] [CrossRef] [PubMed]
- Boodoosingh, R.; Olayemi, L.O.; Sam, F.A. COVID-19 vaccines: Getting Anti-vaxxers involved in the discussion. World Dev. 2020, 136, 105177. [Google Scholar] [CrossRef] [PubMed]
- Ashton, J. COVID-19 and the anti-vaxxers. J. R. Soc. Med. 2021, 1, 42–43. [Google Scholar] [CrossRef] [PubMed]
- Slaoui, M.; Hepburn, M. Developing Safe and Effective Covid Vaccines-Operation Warp Speed’s Strategy and Approach. N. Engl. J. Med. 2020, 18, 1701–1703. [Google Scholar] [CrossRef] [PubMed]
- Mullard, A. COVID-19 vaccine development pipeline gears up. Lancet 2020, 10239, 1751–1752. [Google Scholar] [CrossRef]
- Zimmer, C.; Corum, J.; Wee, S. Coronavirus Vaccine Tracker. The New York Times. 2020. Available online: https://www.nytimes.com/interactive/2020/science/coronavirus-vaccinetracker.html (accessed on 27 December 2020).
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Marc, G.P.; Moreira, E.D.; Zerbini, C.; et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Walsh, E.E.; Frenck, R.; Falsey, A.R.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Mulligan, M.J.; Bailey, R.; et al. RNA-Based COVID-19 Vaccine BNT162b2 Selected for a Pivotal Efficacy Study. medRxiv 2020. [Google Scholar] [CrossRef]
- Anderson, E.J.; Rouphael, N.G.; Widge, A.T. Safety and Immunogenicity of SARS-CoV-2 mRNA-1273 Vaccine in Older Adults. N. Engl. J. Med. 2020, 25, 2427–2438. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Keech, C.; Albert, G.; Cho, I.; Robertson, A.; Reed, P.; Neal, S.; Plested, J.S.; Zhu, M.; Cloney-Clark, S.; Zhou, H. Phase 1-2 Trial of a SARS-CoV-2 Recombinant Spike Protein Nanoparticle Vaccine. Clinical Trial. N. Engl. J. Med. 2020, 383, 2320–2332. [Google Scholar] [CrossRef]
- Sadoff, J.; Le Gars, M.; Shukarev, G.; Heerwegh, D.; Truyers, C.; de Groot, A.M.; Stoop, J.; Tete, S.; Van Damme, W.; Leroux-Roels, I. Interim Results of a Phase 1-2a Trial of Ad26.COV2.S Covid-19 Vaccine. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Mercado, N.B.; Zahn, R.; Wegmann, F.; Loos, C.; Chandrashekar, A.; Yu, J.; Liu, J.; Peter, L.; McMahan, K.; Tostanoski, L.H.; et al. Single-shot Ad26 vaccine protects against SARS-CoV-2 in rhesus macaques. Nature 2020, 7830, 583–588. [Google Scholar] [CrossRef]
- Banerji, A.; Wickner, P.G.; Saff, R.; Stone, C.A., Jr.; Robinson, L.B.; Long, A.A.; Wolfson, A.R.; Williams, P.; Khan, D.A.; Phillips, E.; et al. mRNA Vaccines to Prevent COVID-19 Disease and Reported Allergic Reactions: Current Evidence and Suggested Approach. J. Allergy Clin. Immunol. Pract. 2021, 4, 1423–1437. [Google Scholar] [CrossRef]
- Meo, S.A.; Bukhari, A.I.; Akram, J.; Meo, A.S.; Klonoff, D.C. COVID-19 vaccines: Comparison of biological, pharmacological characteristics and adverse effects of Pfizer/BioNTech and Moderna Vaccines. Eur. Rev. Med. Pharmacol. Sci. 2021, 3, 1663–1669. [Google Scholar]
- Shimabukuro, T.T.; Kim, S.Y.; Myers, T.R.; Moro, P.L.; Oduyebo, T.; Panagiotakopoulos, L.; Marquez, P.L.; Olson, C.K.; Liu, R.; Chang, K.T.; et al. Preliminary Findings of mRNA Covid-19 Vaccine Safety in Pregnant Persons. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention COVID-19 Response Team. Allergic Reactions Including Anaphylaxis after Receipt of the First Dose of Pfizer-BioNTech COVID-19 Vaccine—United States, December 14–23, 2020. MMWR 2021, 70, 46–51. [Google Scholar]
- MacDonald, N.E. SAGE Working Group on Vaccine Hesitancy. Vaccine hesitancy: Definition, scope and determinants. Vaccine 2015, 34, 4161–4164. [Google Scholar] [CrossRef]
- Dubé, E.; Laberge, C.; Guay, M.; Bramadat, P.; Roy, R.; Bettinger, J. Vaccine hesitancy: An overview. Hum. Vaccin. Immunother. 2013, 8, 1763–1773. [Google Scholar] [CrossRef]
- Khubchandani, J.; Sharma, S.; Price, J.H.; Wiblishauser, M.J.; Sharma, M.; Webb, F.J. COVID-19 Vaccination Hesitancy in the United States: A Rapid National Assessment. J. Comm. Health 2021, 2, 270–277. [Google Scholar] [CrossRef]
- Freeman, D.; Loe, B.S.; Chadwick, A.; Vaccari, C.; Waite, F.; Rosebrock, L.; Jenner, L.; Petit, A.; Lewandowsk, S.; Vanderslott, S.; et al. COVID-19 vaccine hesitancy in the UK: The Oxford coronavirus explanations, attitudes, and narratives survey (Oceans) II. Psychol. Med. 2020, 11, 1–15. [Google Scholar]
- Fisher, K.A.; Bloomstone, S.J.; Walder, J.; Crawford, S.; Fouayzi, H.; Mazor, K.M. Attitudes Toward a Potential SARS-CoV-2 Vaccine: A Survey of U.S. Adults. Ann. Intern. Med. 2020, 12, 964–973. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Wong, E.L.; Ho, K.F.; Cheung, A.W.; Yau, P.S.; Dong, D.; Wong, S.Y.; Yeoh, E.K. Change of Willingness to Accept COVID-19 Vaccine and Reasons of Vaccine Hesitancy of Working People at Different Waves of Local Epidemic in Hong Kong, China: Repeated Cross-Sectional Surveys. Vaccines 2021, 1, 62. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.H.; Srivastav, A.; Razzaghi, H.; Williams, W.; Lindley, M.C.; Jorgensen, C.; Abad, N.; Singleton, J.A. COVID-19 Vaccination Intent, Perceptions, and Reasons for Not Vaccinating Among Groups Prioritized for Early Vaccination-United States, September and December 2020. MMWR. Morb. Mortal Wkly. Rep. 2021, 6, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Latest Data on COVID-19 Vaccinations Race/Ethnicity Nambi Ndugga, Olivia Pham, Latoya Hill, Samantha Artiga, Raisa Alam, and Noah Parker Published: 17 March 2021. Available online: https://www.kff.org/coronavirus-covid-19/issue-brief/latest-data-on-covid-19-vaccinations-race-ethnicity/ (accessed on 5 May 2021).
- Vaccine Monitor: Nearly Half of the Public Wants to Get a COVID-19 Vaccine as Soon as They Can or Has Already Been Vaccinated, Up across Racial and Ethnic Groups Since December. People Who Know Someone Who Has Been Vaccinated Are More Eager to Do So Themselves. Published: 27 January 2021. Available online: https://www.kff.org/coronavirus-covid-19/press-release/vaccine-monitor-nearly-half-of-the-public-wants-to-get-covid-19-vaccine-as-soon-as-they-can-or-has-already-been-vaccinated-up-across-racial-and-ethnic-groups-since-december/ (accessed on 26 February 2021).
- Doherty, I.A.; Pilkington, W.; Brown, L.; Billings, V.; Hoffler, U.; Paulin, L.; Kimbro, K.S.; Baker, B.; Zhang, T.; Locklear, T.; et al. COVID-19 Vaccine Hesitancy in Underserved Communities of North Carolina. medRxiv 2021. [Google Scholar] [CrossRef]
- KFF COVID-19 Vaccine Monitor: December 2020 Liz Hamel Follow @lizhamel on Twitter, Ashley Kirzinger Follow @AshleyKirzinger on Twitter, Cailey Muñana, and Mollyann Brodie Follow @Mollybrodie on Twitter Published: 15 December 2020. Available online: https://www.kff.org/coronavirus-covid-19/report/kff-covid-19-vaccine-monitor-december-2020/ (accessed on 8 May 2021).
- Oxner, R. White Republicans Are Refusing to Get the COVID-19 Vaccine More Than Any Other Demographic Group in Texas. Available online: https://www.texastribune.org/2021/03/23/covid-vaccine-hesitancy-white-republicans/ (accessed on 24 March 2021).
- Price-Haygood, E.G.; Burton, J.; Fort, D.; Seoane, L. Hospitalization and Mortality among Black Patients and White Patients with Covid-19. N. Engl. J. Med. 2020, 26, 2534–2543. [Google Scholar] [CrossRef]
- Yehia, B.R.; Winegar, A.; Fogel, R.; Fakih, M.; Ottenbacher, A.; Jesser, C.; Bufalino, A.; Huang, R.; Cacchione, J. Association of Race With Mortality Among Patients Hospitalized With Coronavirus Disease 2019 (COVID-19) at 92 US Hospitals. JAMA Netw. Open 2020, 8, e2018039. [Google Scholar] [CrossRef]
- Garg, S.; Kim, L.; Whitaker, M.; O’Halloran, A.; Cummings, C.; Holstein, R.; Prill, M.; Chai, S.J.; Kirley, P.D.; Alden, N.B. Hospitalization Rates and Characteristics of Patients Hospitalized with Laboratory-Confirmed Coronavirus Disease 2019-COVID-NET, 14 States, March 1-30, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 15, 458–464. [Google Scholar] [CrossRef] [PubMed]
- Millett, G.A.; Jones, A.T.; Benkeser, D.; Baral, S.; Mercer, L.; Beyrer, C.; Honermann, B.; Lankiewicz, E.; Mena, L.; Crowley, J.S. Assessing Differential Impacts of COVID-19 on Black Communities. Ann. Epidemiol. 2020, 47, 37–44. [Google Scholar] [CrossRef]
- Kertscher, T. Melanin Doesn’t Protect Against Coronavirus. Available online: https://www.politifact.com/factchecks/2020/mar/10/facebook-posts/melanin-doesnt-protect-against-coronavirus/ (accessed on 10 March 2020).
- U.S. Centers for Disease Control and Prevention, “Risk for COVID-19 Infection, Hospitalization, and Death by Race/Ethnicity”. Available online: https://www.cdc.gov/coronavirus/2019-ncov/covid-data/investigations-discovery/hospitalization-death-by-race-ethnicity.html (accessed on 23 April 2021).
- Porter, D.; Porter, R. The politics of prevention: Anti-vaccinationism and public health in nineteenth-century England. Med. Hist. 1988, 32, 231–252. [Google Scholar] [CrossRef] [PubMed]
- GAVI. How South Africa Is Preparing for its COVID-19 Vaccine Introduction. Available online: https://www.gavi.org/vaccineswork/how-south-africa-preparing-its-covid-19-vaccine-introduction (accessed on 7 February 2021).
- Tozivepi, S.N.; Mundagowa, P.; Tirivavi, M.; Maponga, B.; Mugwagwa, N.; Magande, P.; Mutseyekwa, F.; Makurumidze, R. Covid-19 Vaccine Hesistancy Survey Preliminary Report; Zimbabwe College of Public Health Physicians: Harare, Zimbabwe, 2020. [Google Scholar]
- WHO. Managing the COVID-19 Infodemic: Promoting Healthy Behaviours and Mitigating the Harm from Misinformation and Disinformation. 2020. Available online: https://www.who.int/news/item/23-09-2020-managing-the-covid-19-infodemic-promoting-healthy-behaviours-and-mitigating-the-harm-from-misinformation-and-disinformation (accessed on 11 February 2021).
- Karlsson, L.C.; Lewandowsky, S.; Antfolk, J.; Salo, P.; Lindfelt, M.; Oksanen, T.; Kivimäki, M.; Soveri, A. The association between vaccination confidence, vaccination behavior, and willingness to recommend vaccines among Finnish healthcare workers. PLoS ONE 2019, 10, e0224330. [Google Scholar]
- Lucia, V.C.; Kelekar, A.; Afonso, N.M. COVID-19 vaccine hesitancy among medical students. J. Public Health 2020, 26, fdaa230. [Google Scholar] [CrossRef] [PubMed]
- Shekhar, R.; Sheikh, A.B.; Upadhyay, S.; Singh, M.; Kottewar, S.; Mir, H.; Barrett, E.; Pal, S. COVID-19 Vaccine Acceptance among Health Care Workers in the United States. Vaccines 2021, 2, 119. [Google Scholar] [CrossRef] [PubMed]
- Verger, P.; Scronias, D.; Dauby, N.; Adedzi, K.A.; Gobert, C.; Bergeat, M.; Gagneur, A.; Dubé, E. Attitudes of healthcare workers towards COVID-19 vaccination: A survey in France and French-speaking parts of Belgium and Canada, 2020. Eurosurveill 2021, 3, 2002047. [Google Scholar]
- Davies, P.; Chapman, S.; Leask, J. Antivaccination activists on the world wide web. Arch. Dis. Child. 2002, 87, 22–25. [Google Scholar] [CrossRef]
- Wolfe, R.M.; Sharp, L.K.; Lipsky, M.S. Content and design attributes of antivaccination web sites. JAMA 2002, 287, 245–248. [Google Scholar] [CrossRef]
- Hotez, P. COVID vaccines: Time to confront anti-vax aggression. Nature. 2021, 592, 661. [Google Scholar] [CrossRef]
- Brennan, J. A libertarian case for mandatory vaccination. J. Med. Ethics 2018, 1, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R.M.; Sharp, L.K. Anti-vaccinationists past and present. BMJ 2002, 7361, 430–432. [Google Scholar] [CrossRef] [PubMed]
- Durbach, N. ‘They might as well brand us’: Working-class resistance to compulsory vaccination in Victorian England. Soc. Hist. Med. 2000, 1, 45–62. [Google Scholar] [CrossRef]
- Baker, J.P. The pertussis vaccine controversy in Great Britain, 1974–1986. Vaccine 2003, 21, 4003–4010. [Google Scholar] [CrossRef]
- Omer, S.B.; Salmon, D.A.; Orenstein, W.A.; DeHart, M.P.; Halsey, N. Vaccine refusal, mandatory immunization, and the risks of vaccine preventable diseases. N. Eng. J. Med. 2009, 19, 1981–1988. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, I. Dismantling the anti-vaxx industry. Nat. Med. 2021, 3, 366. [Google Scholar] [CrossRef] [PubMed]
- The Changing Child Population of the United States, An Analysis of the U.S. Population Under 18 Using Data from the 2010 Census by the Annie E. Casey Foundation. 1 January 2011. Available online: https://www.census.gov/newsroom/releases/archives/2010_census/cb11-cn147.html (accessed on 26 May 2011).
| Developer | Type | Doses | Participants/P3 | Study Location | Efficacy/US | EUA Date |
|---|---|---|---|---|---|---|
| Pfizer-BioNTech | mRNA | 2 | 43,548 | International | 95% | 11 December 2020 |
| Moderna-NIAID | mRNA | 2 | 34,420 | United States | 94% | 18 December 2020 |
| Johnson&Johnson-Janssen | Viral vector | 1 | 43,783 | International | 72% | 27 February 2021 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hildreth, J.E.K.; Alcendor, D.J. Targeting COVID-19 Vaccine Hesitancy in Minority Populations in the US: Implications for Herd Immunity. Vaccines 2021, 9, 489. https://doi.org/10.3390/vaccines9050489
Hildreth JEK, Alcendor DJ. Targeting COVID-19 Vaccine Hesitancy in Minority Populations in the US: Implications for Herd Immunity. Vaccines. 2021; 9(5):489. https://doi.org/10.3390/vaccines9050489
Chicago/Turabian StyleHildreth, James E. K., and Donald J. Alcendor. 2021. "Targeting COVID-19 Vaccine Hesitancy in Minority Populations in the US: Implications for Herd Immunity" Vaccines 9, no. 5: 489. https://doi.org/10.3390/vaccines9050489
APA StyleHildreth, J. E. K., & Alcendor, D. J. (2021). Targeting COVID-19 Vaccine Hesitancy in Minority Populations in the US: Implications for Herd Immunity. Vaccines, 9(5), 489. https://doi.org/10.3390/vaccines9050489






