Safety and Immunogenicity of Adenovirus and Poxvirus Vectored Vaccines against a Mycobacterium Avium Complex Subspecies
Abstract
1. Introduction
2. Materials and Methods
2.1. ChAdOx2 HAV and MVA HAV Vaccines
2.2. Study Design and Participants
2.3. Procedures
2.4. IFN-γ ELISpot
2.5. Statistical Analysis
3. Results
3.1. Study Population
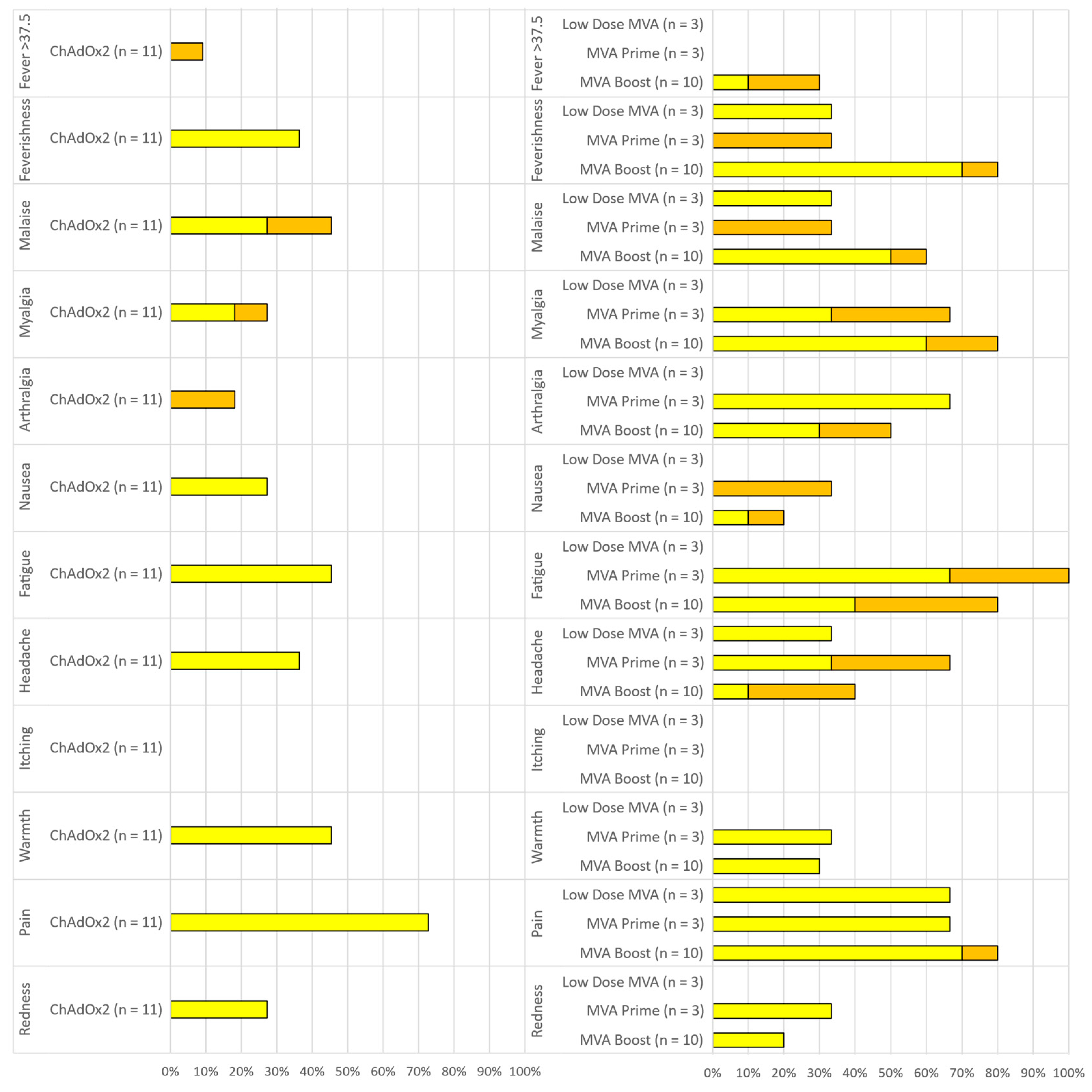
3.2. Vaccine Safety
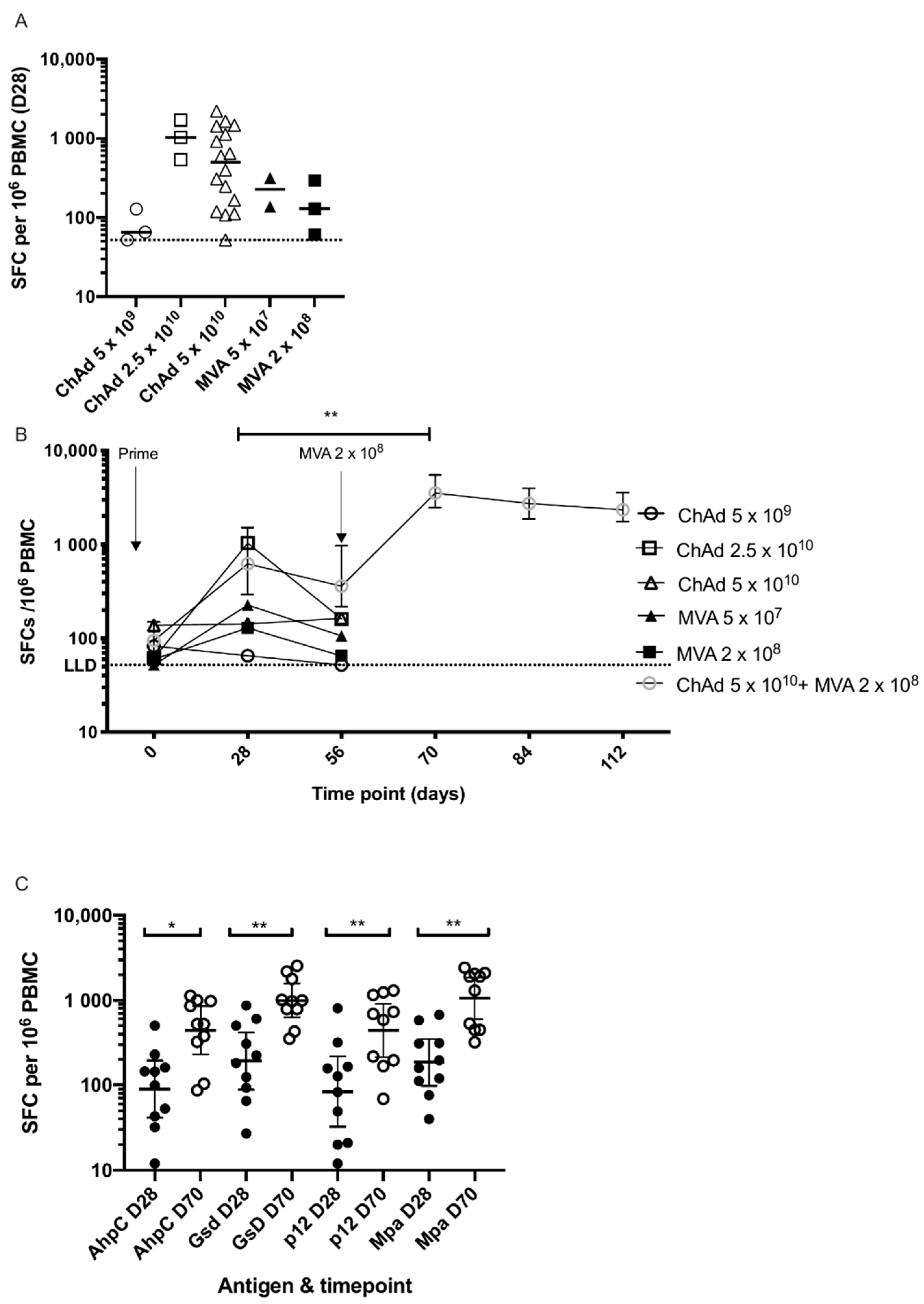
3.3. Cellular Immunogenicity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sechi, L.A.; Dow, C.T. Mycobacterium avium ss. paratuberculosis Zoonosis—The Hundred Year War—Beyond Crohn’s Disease. Front. Immunol. 2015, 6, 96. [Google Scholar] [CrossRef] [PubMed]
- Kardani, K.; Bolhassani, A.; Shahbazi, S. Prime-boost vaccine strategy against viral infections: Mechanisms and benefits. Vaccine 2016, 34, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Milligan, I.D.; Gibani, M.M.; Sewell, R.; Clutterbuck, E.A.; Campbell, D.; Plested, E.; Snape, M.D. Safety and Immunogenicity of Novel Adenovirus Type 26- and Modified Vaccinia Ankara-Vectored Ebola Vaccines: A Randomized Clinical Trial. JAMA 2016, 315, 1610–1623. [Google Scholar] [CrossRef] [PubMed]
- Barouch, D.H.; Tomaka, F.L.; Wegmann, F.; Stieh, D.J.; Alter, G.; Robb, M.L.; Schuitemaker, H. Evaluation of a mosaic HIV-1 vaccine in a multicentre, randomised, double-blind, placebo-controlled, phase 1/2a clinical trial (APPROACH) and in rhesus monkeys (NHP 13–19). Lancet 2018, 392, 232–243. [Google Scholar] [CrossRef]
- Milligan, I.D.; Gibani, M.M.; Sewell, R.; Clutterbuck, E.A.; Campbell, D.; Plested, E.; Snape, M.D. Safety and Immunogenicity of a Novel Recombinant Simian Adenovirus ChAdOx2 as a Vectored Vaccine. Vaccines 2019, 7, 1610–1623. [Google Scholar]
- Morris, S.J.; Sebastian, S.; Spencer, A.J.; Gilbert, S.C. Simian adenoviruses as vaccine vectors. Future Virol. 2016, 11, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Hochstein-Mintzel, V.; Hanichen, T.; Huber, H.C.; Stickl, H. An attenuated strain of vaccinia virus (MVA). Successful intramuscular immunization against vaccinia and variola (author’s transl). Zentralbl. Bakteriol. Orig. A 1975, 230, 283–297. [Google Scholar] [PubMed]
- Tiono, A.B.; Nébié, I.; Anagnostou, N.; Coulibaly, A.S.; Bowyer, G.; Lam, E.; Bougouma, E.C.; Ouedraogo, A.; Yaro, J.B.B.; Barry, A.; et al. First field efficacy trial of the ChAd63 MVA ME-TRAP vectored malaria vaccine candidate in 5–17 months old infants and children. PLoS ONE 2018, 13, e0208328. [Google Scholar] [CrossRef] [PubMed]
- Wilkie, M.; Satti, I.; Minhinnick, A.; Harris, S.; Riste, M.; Ramon, R.L.; McShane, H. A phase I trial evaluating the safety and immunogenicity of a candidate tuberculosis vaccination regimen, ChAdOx1 85A prime—MVA85A boost in healthy UK adults. Vaccine 2020, 38, 779–789. [Google Scholar] [CrossRef] [PubMed]
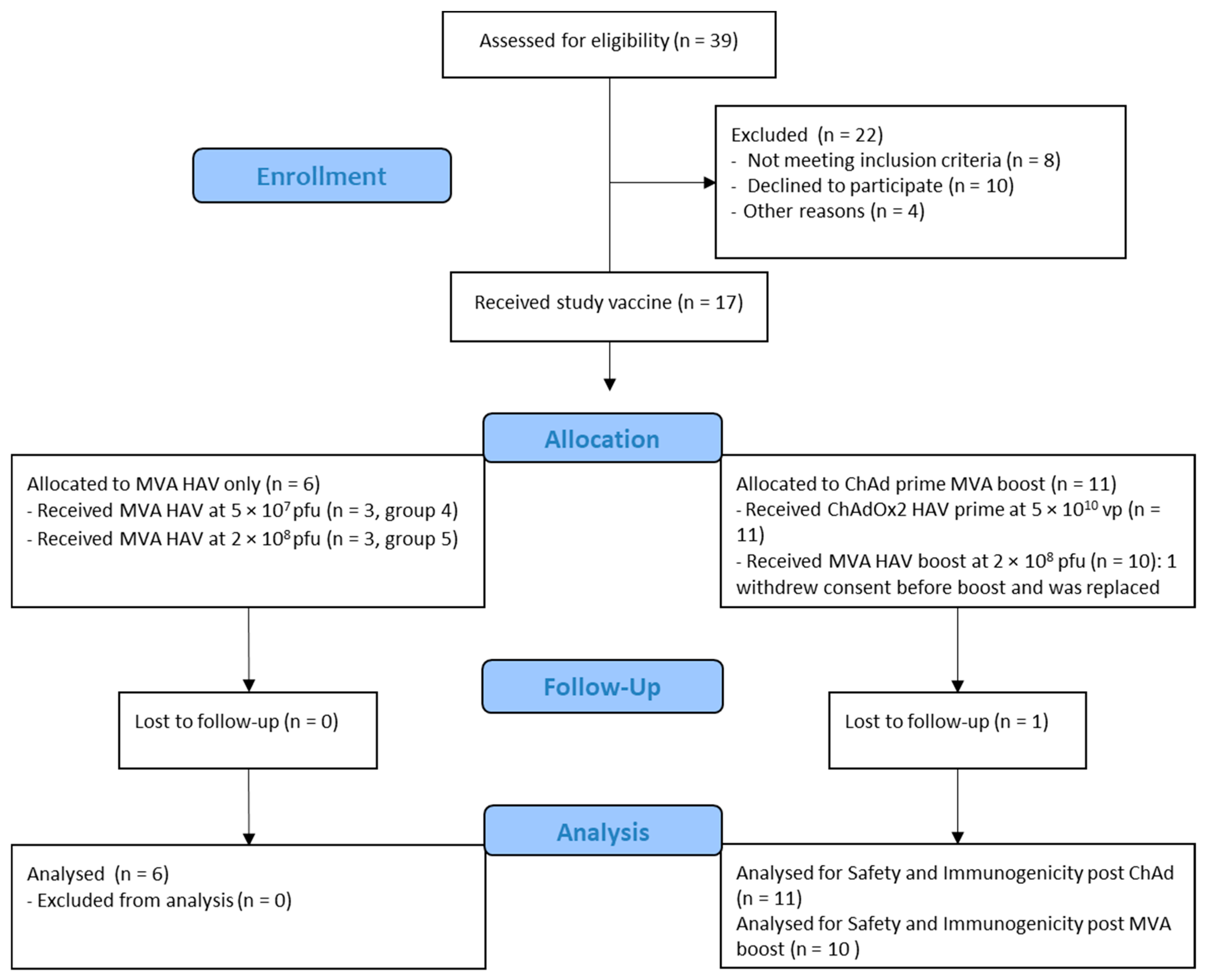
| Variable | Group 4 (MVA Only Low Dose) (n = 3) | Group 5 (MVA Only Standard Dose) (n = 3) | Group 6 (Heterologous Prime-Boost) (n = 11) | All Groups (n = 17) |
|---|---|---|---|---|
| Age | ||||
| Median | 22 | 24 | 27.5 | 27 |
| Range | 19–23 | 20–48 | 21–50 | 19–50 |
| Sex | ||||
| Male—n (%) | 0 | 2 (66.7) | 2 (18.2) | 4 (23.5) |
| Female—n (%) | 3 (100) | 1 (33.3) | 9 (81.8) | 13 (76.5) |
| Ethnicity | ||||
| White—n (%) | 3 (100) | 3 (100) | 11 (100) | 17 (100) |
| BMI (Kg/m2) | ||||
| Median | 19.7 | 24 | 25.7 | 25.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Folegatti, P.M.; Flaxman, A.; Jenkin, D.; Makinson, R.; Kingham-Page, L.; Bellamy, D.; Ramos Lopez, F.; Sheridan, J.; Poulton, I.; Aboagye, J.; et al. Safety and Immunogenicity of Adenovirus and Poxvirus Vectored Vaccines against a Mycobacterium Avium Complex Subspecies. Vaccines 2021, 9, 262. https://doi.org/10.3390/vaccines9030262
Folegatti PM, Flaxman A, Jenkin D, Makinson R, Kingham-Page L, Bellamy D, Ramos Lopez F, Sheridan J, Poulton I, Aboagye J, et al. Safety and Immunogenicity of Adenovirus and Poxvirus Vectored Vaccines against a Mycobacterium Avium Complex Subspecies. Vaccines. 2021; 9(3):262. https://doi.org/10.3390/vaccines9030262
Chicago/Turabian StyleFolegatti, Pedro M., Amy Flaxman, Daniel Jenkin, Rebecca Makinson, Lucy Kingham-Page, Duncan Bellamy, Fernando Ramos Lopez, Jonathan Sheridan, Ian Poulton, Jeremy Aboagye, and et al. 2021. "Safety and Immunogenicity of Adenovirus and Poxvirus Vectored Vaccines against a Mycobacterium Avium Complex Subspecies" Vaccines 9, no. 3: 262. https://doi.org/10.3390/vaccines9030262
APA StyleFolegatti, P. M., Flaxman, A., Jenkin, D., Makinson, R., Kingham-Page, L., Bellamy, D., Ramos Lopez, F., Sheridan, J., Poulton, I., Aboagye, J., Tran, N., Mitton, C., Roberts, R., Lawrie, A. M., Hill, A. V. S., Ewer, K. J., & Gilbert, S. (2021). Safety and Immunogenicity of Adenovirus and Poxvirus Vectored Vaccines against a Mycobacterium Avium Complex Subspecies. Vaccines, 9(3), 262. https://doi.org/10.3390/vaccines9030262






