Assessing the Long-Term Role of Vaccination against HPV after Loop Electrosurgical Excision Procedure (LEEP): A Propensity-Score Matched Comparison
Abstract
1. Introduction
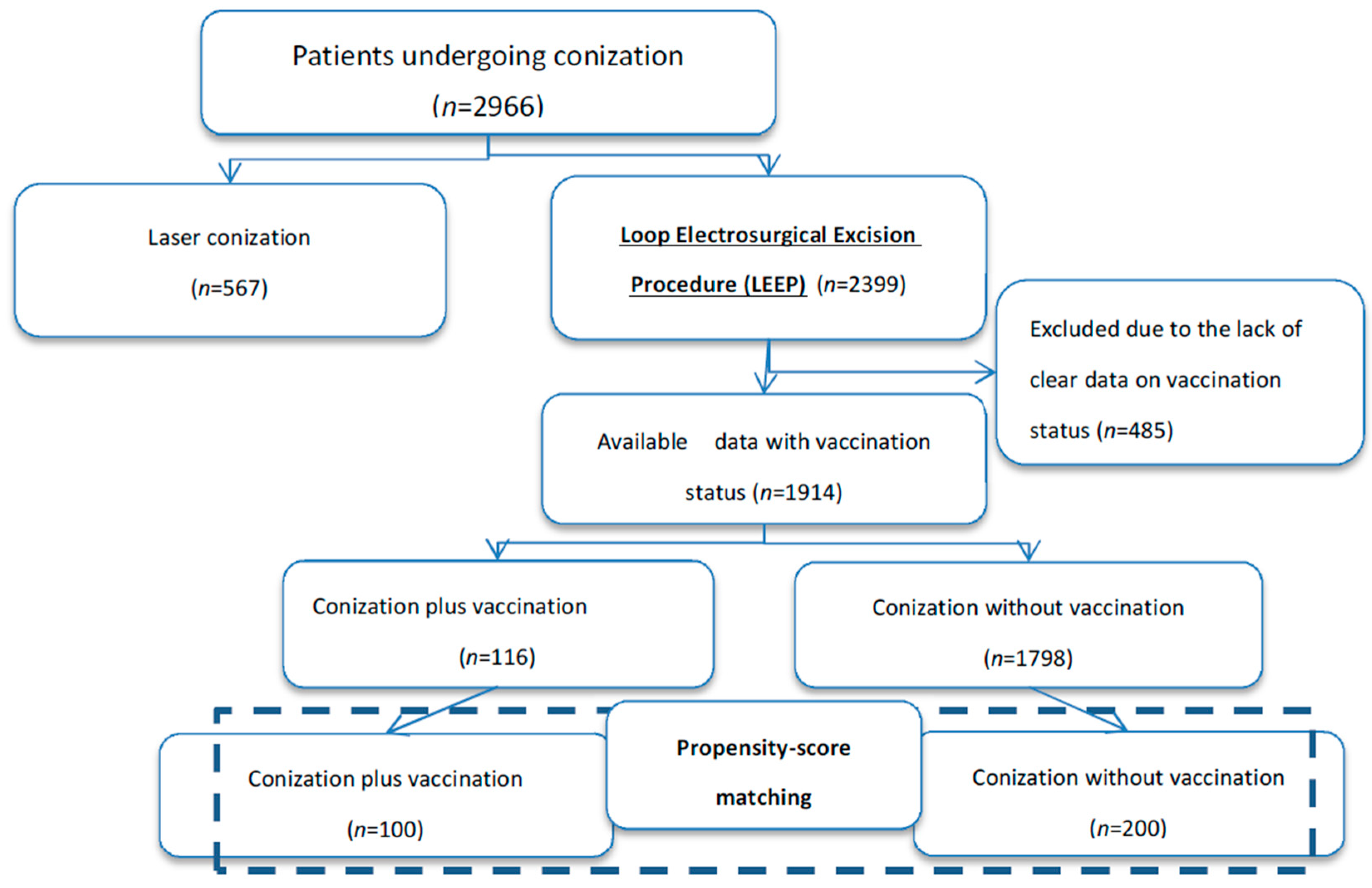
2. Materials and Methods
Statistical Methods
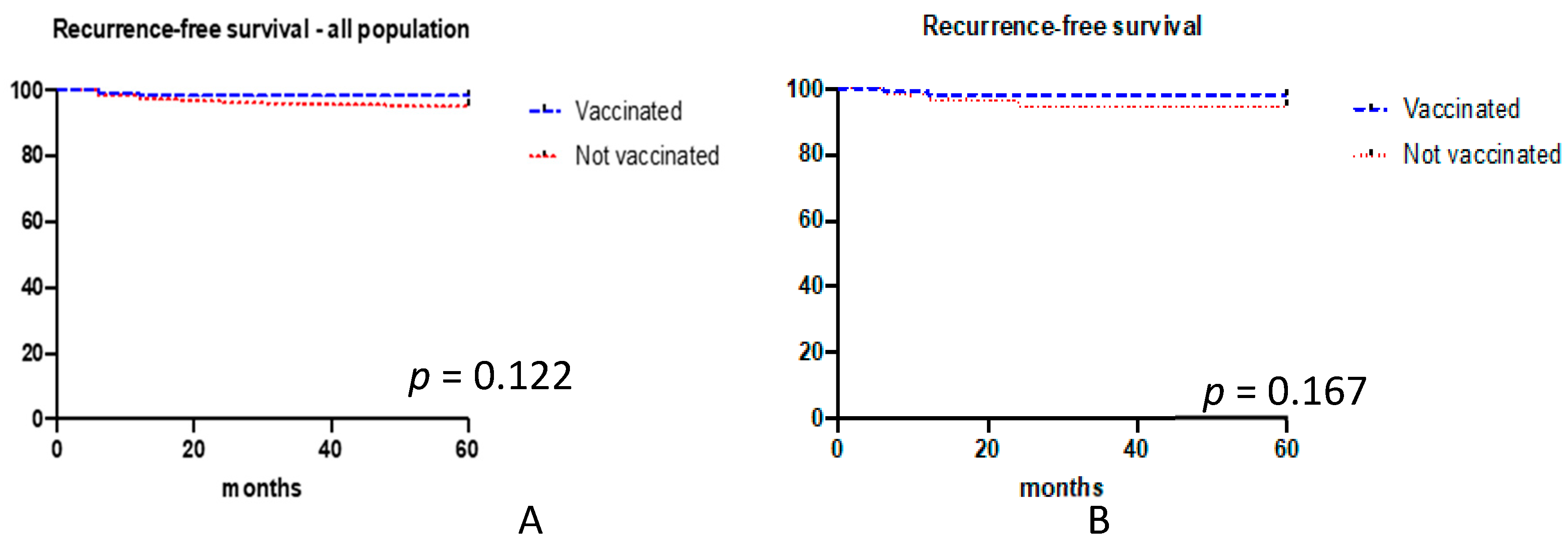
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Burger, E.A.; Portnoy, A.; Campos, N.G.; Sy, S.; Regan, C.; Kim, J.J. Choosing the optimal HPV vaccine: The health impact and economic value of the nonavalent and bivalent HPV vaccines in 48 Gavi-eligible countries. Int. J. Cancer 2020. [Google Scholar] [CrossRef] [PubMed]
- Olsson, S.-E.; Restrepo, J.A.; Reina, J.C.; Pitisuttithum, P.; Ulied, A.; Varman, M.; Van Damme, P.; Moreira, E.D.; Ferris, D.; Block, S.; et al. Long-term immunogenicity, effectiveness, and safety of nine-valent human papillomavirus vaccine in girls and boys 9 to 15 years of age: Interim analysis after 8 years of follow-up. Papillomavirus Res. 2020, 10, 100203. [Google Scholar] [CrossRef] [PubMed]
- Saslow, D.; Ba, K.S.A.; Manassaram-Baptiste, D.; Smith, R.A.; Fontham MPH on behalf of the American Cancer Society Guideline Development Group. Human papillomavirus vaccination 2020 guideline update: American Cancer Society guideline adaptation. CA Cancer J. Clin. 2020, 70, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Baussano, I.; Lazzarato, F.; Ronco, G.; Franceschi, S. Impacts of human papillomavirus vaccination for different populations: A modeling study. Int. J. Cancer 2018, 143, 1086–1092. [Google Scholar] [CrossRef] [PubMed]
- Kulasingam, S.L.; Connelly, L.B.; Conway, E.; Hocking, J.S.; Myers, E.; Regan, D.G.; Roder, D.; Ross, J.; Wain, G. A cost-effectiveness analysis of adding a human papillomavirus vaccine to the Australian National Cervical Cancer Screening Program. Sex. Heal. 2007, 4, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Kang, W.D.; Choi, H.S.; Kim, S.M. Is vaccination with quadrivalent HPV vaccine after loop electrosurgical excision procedure effective in preventing recurrence in patients with high-grade cervical intraepithelial neoplasia (CIN2–3)? Gynecol. Oncol. 2013, 130, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Ghelardi, A.; Parazzini, F.; Martella, F.; Pieralli, A.; Bay, P.; Tonetti, A.; Svelato, A.; Bertacca, G.; Lombardi, S.; Joura, E.A. SPERANZA project: HPV vaccination after treatment for CIN2+. Gynecol. Oncol. 2018, 151, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Petrillo, M.; Dessole, M.; Tinacci, E.; Saderi, L.; Muresu, N.; Capobianco, G.; Cossu, A.; Dessole, S.; Sotgiu, G.; Piana, A.F. Efficacy of HPV Vaccination in Women Receiving LEEP for Cervical Dysplasia: A Single Institution’s Experience. Vaccines 2020, 8, 45. [Google Scholar] [CrossRef] [PubMed]
- Sand, F.L.; Kjaer, S.K.; Frederiksen, K.; Dehlendorff, C. Risk of cervical intraepithelial neoplasia grade 2 or worse after conization in relation to HPV vaccination status. Int. J. Cancer 2020, 147, 641–647. [Google Scholar] [CrossRef]
- Paavonen, J.; Naud, P.; Salmerón, J.; Wheeler, C.M.; Chow, S.-N.; Apter, D.; Kitchener, H.; Castellsague, X.; Teixeira, J.C.; Skinner, S.R.; et al. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): Final analysis of a double-blind, randomised study in young women. Lancet 2009, 374, 301–314. [Google Scholar] [CrossRef]
- Bogani, G.; Di Donato, V.; Sopracordevole, F.; Ciavattini, A.; Ghelardi, A.; Lopez, S.; Simoncini, T.; Plotti, F.; Casarin, J.; Serati, M.; et al. Recurrence rate after loop electrosurgical excision procedure (LEEP) and laser Conization: A 5-year follow-up study. Gynecol. Oncol. 2020. [Google Scholar] [CrossRef]
- Bogani, G.; Tagliabue, E.; Ferla, S.; Martinelli, F.; Ditto, A.; Chiappa, V.; Maggiore, U.L.R.; Taverna, F.; Lombardo, C.; Lorusso, D.; et al. Nomogram-based prediction of cervical dysplasia persistence/recurrence. Eur. J. Cancer Prev. 2019, 28, 435–440. [Google Scholar] [CrossRef]
- Bogani, G.; Ditto, A.; Martinelli, F.; Signorelli, M.; Chiappa, V.; Maggiore, U.L.R.; Taverna, F.; Lombardo, C.; Borghi, C.; Scaffa, C.; et al. Artificial intelligence estimates the impact of human papillomavirus types in influencing the risk of cervical dysplasia recurrence. Eur. J. Cancer Prev. 2019, 28, 81–86. [Google Scholar] [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Human Papillomaviruses; International Agency for Research on Cancer: Lyon, France, 2007; Volume 90, pp. 1–636. [Google Scholar]
- Scheller, N.M.; Pasternak, B.; Mølgaard-Nielsen, D.; Svanström, H.; Hviid, A. Quadrivalent HPV Vaccination and the Risk of Adverse Pregnancy Outcomes. N. Engl. J. Med. 2017, 376, 1223–1233. [Google Scholar] [CrossRef] [PubMed]
- Del Pino, M.; Martí, C.; Torras, I.; Henere, C.; Munmany, M.; Marimon, L.; Saco, A.; Torné, A.; Ordi, J. HPV Vaccination as Adjuvant to Conization in Women with Cervical Intraepithelial Neoplasia: A Study under Real-Life Conditions. Vaccines 2020, 8, 245. [Google Scholar] [CrossRef]
- Villa, L.L.; Perez, G.; Kjaer, S.K.; Paavonen, J.; Lehtinen, M.; Munoz, N.; Sigurdsson, K.; Hernandez-Avila, M.; Skjeldestad, F.E.; Thoresen, S.; et al. Quadrivalent Vaccine against Human Papillomavirus to Prevent High-Grade Cervical Lesions. N. Engl. J. Med. 2007, 356, 1915–1927. [Google Scholar] [CrossRef]
- Lehtinen, M.; Paavonen, J.; Wheeler, C.M.; Jaisamrarn, U.; Garland, S.M.; Castellsagué, X.; Skinner, S.R.; Apter, D.; Naud, P.; Salmerón, J.; et al. Overall efficacy of HPV-16/18 AS04-adjuvanted vaccine against grade 3 or greater cervical intraepithelial neoplasia: 4-year end-of-study analysis of the randomised, double-blind PATRICIA trial. Lancet Oncol. 2012, 13, 89–99. [Google Scholar] [CrossRef]
- GISCi. Gruppo Italiano Screening del Cervicocarcinoma. Available online: http://www.gisci.it/ (accessed on 8 June 2020).

| Characteristics | Whole Study Population (n = 1914) | Conization Plus Vaccination (n = 116) | Conization Alone (n = 1798) | p Value (Vaccinated vs. Not Vaccinated) |
|---|---|---|---|---|
| Age, years | 39 (17–89) | 35 (24–45) | 39 (17–89) | <0.001 |
| Body mass index | 22.85 (14.4–44) | 21 (17–33) | 23 (14.4–44) | <0.001 |
| Menopause | ||||
| No | 1545 (80.7%) | 116 (100%) | 1429 (79.5%) | <0.001 |
| Yes | 369 (19.3%) | // | 369 (20.5%) | |
| Reason for conization | 0.066 | |||
| CIN2 | 827 (43.2%) | 60 (51.7%) | 767 (42.7%) | |
| CIN3 | 1087 (56.8%) | 56 (48.3%) | 1031 (57.3%) | |
| High-risk HPV involved * | <0.001 | |||
| No | 1026 (53.6%) | 41 (35.3%) | 985 (54.8%) | |
| Yes | 888 (46.4%) | 75 (64.7%) | 813 (45.2%) | |
| Positive margins | 189 (9.9%) | 25 (21.5%) | 164 (9.1%) | <0.001 |
| Endocervical | 135 (7%) | 19 (16.4%) | 116 (6.4%) | <0.001 |
| Esocervical | 55 (2.9%) | 6 (5.2%) | 49 (2.7%) | 0.142 |
| HPV persistence ** | ||||
| No | 1053 (55%) | 60 (51.7%) | 993 (55.2%) | 0.501 |
| Yes | 335 (17.5%) | 51 (44%) | 284 (15.8%) | <0.001 |
| Unknown | 526 (27.5%) | 5 (4.3%) | 521(29%) | <0.001 |
| Recurrence | 104 (5.4%) | 2 (1.7%) | 102 (5.7%) | 0.068 |
| Characteristics | Conization Plus Vaccination (n = 100) | Conization Alone (n = 200) | p Value |
|---|---|---|---|
| Age, years | 33.5 (24–43) | 33.3 (24–44) | 0.895 |
| BMI, kg/mq | 21 (17–33) | 21.1 (17–32.8) | 0.867 |
| Menopause | 1.00 | ||
| No | 100 (100%) | 200 (100%) | |
| Yes | 0 | 0 | |
| HR-HPV detected | 0.895 | ||
| No/Unknown | 31 (31%) | 64 (32%) | |
| Yes | 69 (69%) | 136 (68%) | |
| Not tested | |||
| Type of cervical dysplasia | 0.902 | ||
| CIN2 | 54 (54%) | 106 (53%) | |
| CIN3 | 46 (46%) | 94 (47%) | |
| Positive margins | 1.00 | ||
| No | 76 (76%) | 151 (75.5%) | |
| Yes | 24 (24%) | 49 (24.5%) ** | |
| Type of involved margins | |||
| Endocervical | 18 (18%) | 37 (18.5%) | 1.00 |
| Esocervical | 6 (6%) | 13 (6.5%) | 1.00 |
| HPV persistence * | 0.707 | ||
| No/Unknown | 62 (62%) | 118 (59%) | |
| Yes | 38 (38%) | 82 (41%) | |
| Recurrence/Persistence | |||
| No | 98 (98%) | 189 (94.5%) | 0.231 (yes vs. no) |
| Yes, persistence | 2 (2%) | 2 (1%) | 0.603 (persistence vs no) |
| Yes, recurrence | 0 | 9 (4.5%) | 0.031 (recurrence vs. no) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bogani, G.; Raspagliesi, F.; Sopracordevole, F.; Ciavattini, A.; Ghelardi, A.; Simoncini, T.; Petrillo, M.; Plotti, F.; Lopez, S.; Casarin, J.; et al. Assessing the Long-Term Role of Vaccination against HPV after Loop Electrosurgical Excision Procedure (LEEP): A Propensity-Score Matched Comparison. Vaccines 2020, 8, 717. https://doi.org/10.3390/vaccines8040717
Bogani G, Raspagliesi F, Sopracordevole F, Ciavattini A, Ghelardi A, Simoncini T, Petrillo M, Plotti F, Lopez S, Casarin J, et al. Assessing the Long-Term Role of Vaccination against HPV after Loop Electrosurgical Excision Procedure (LEEP): A Propensity-Score Matched Comparison. Vaccines. 2020; 8(4):717. https://doi.org/10.3390/vaccines8040717
Chicago/Turabian StyleBogani, Giorgio, Francesco Raspagliesi, Francesco Sopracordevole, Andrea Ciavattini, Alessandro Ghelardi, Tommaso Simoncini, Marco Petrillo, Francesco Plotti, Salvatore Lopez, Jvan Casarin, and et al. 2020. "Assessing the Long-Term Role of Vaccination against HPV after Loop Electrosurgical Excision Procedure (LEEP): A Propensity-Score Matched Comparison" Vaccines 8, no. 4: 717. https://doi.org/10.3390/vaccines8040717
APA StyleBogani, G., Raspagliesi, F., Sopracordevole, F., Ciavattini, A., Ghelardi, A., Simoncini, T., Petrillo, M., Plotti, F., Lopez, S., Casarin, J., Serati, M., Pinelli, C., Valenti, G., Bergamini, A., Giannella, B., Dell’Acqua, A., Monti, E., Vercellini, P., D’ippolito, G., ... Di Donato, V. (2020). Assessing the Long-Term Role of Vaccination against HPV after Loop Electrosurgical Excision Procedure (LEEP): A Propensity-Score Matched Comparison. Vaccines, 8(4), 717. https://doi.org/10.3390/vaccines8040717















