Impact of Melatonin Supplementation in Women with Unexplained Infertility Undergoing Fertility Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Experimental Design
2.3. Ovarian Stimulation and Follicular Puncture
2.4. Collection of Samples
2.5. Embryo Classification
2.6. Urinary 6-Sulfatoxymelatonin (aMT6s) Determination
2.7. Quantification of Melatonin
2.8. Total Antioxidant Capacity (TAC) Assay
2.9. Estimation of Superoxide Dismutase (SOD) Activity
2.10. Measurement of Lipid Peroxidation (LPO)
2.11. DNA Oxidative Damage Assay
2.12. Statistical Analysis
3. Results
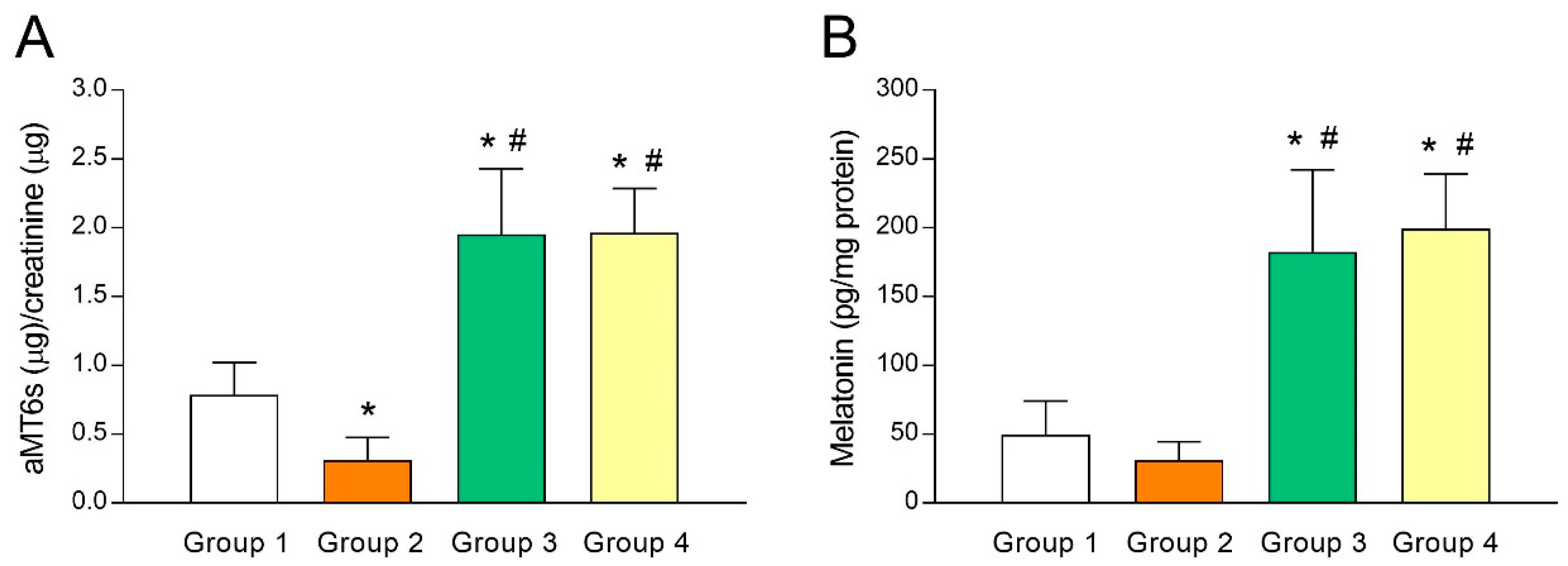
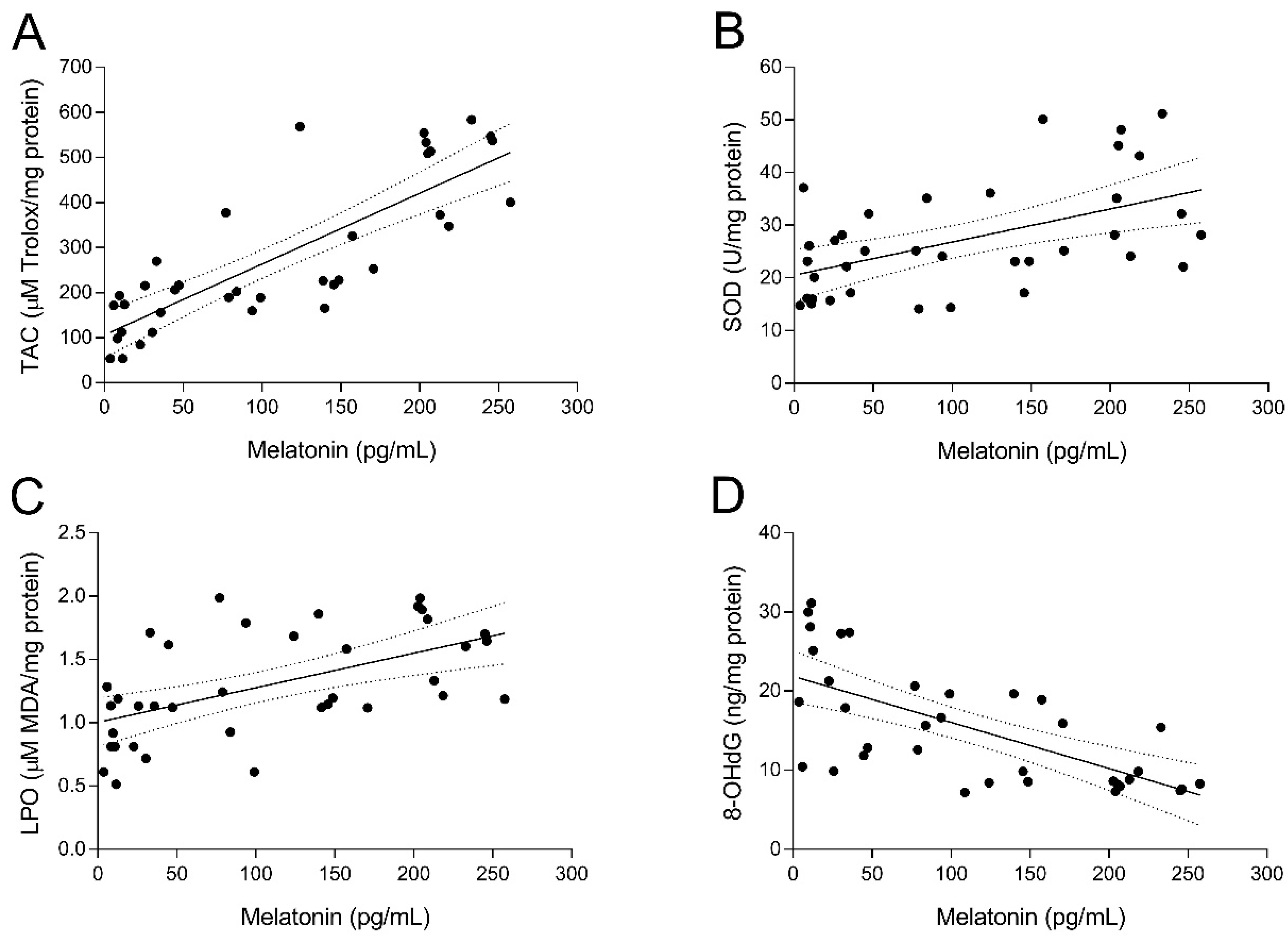
3.1. Analysis of Melatonin Concentrations in Urine and Follicular Fluid (FF)
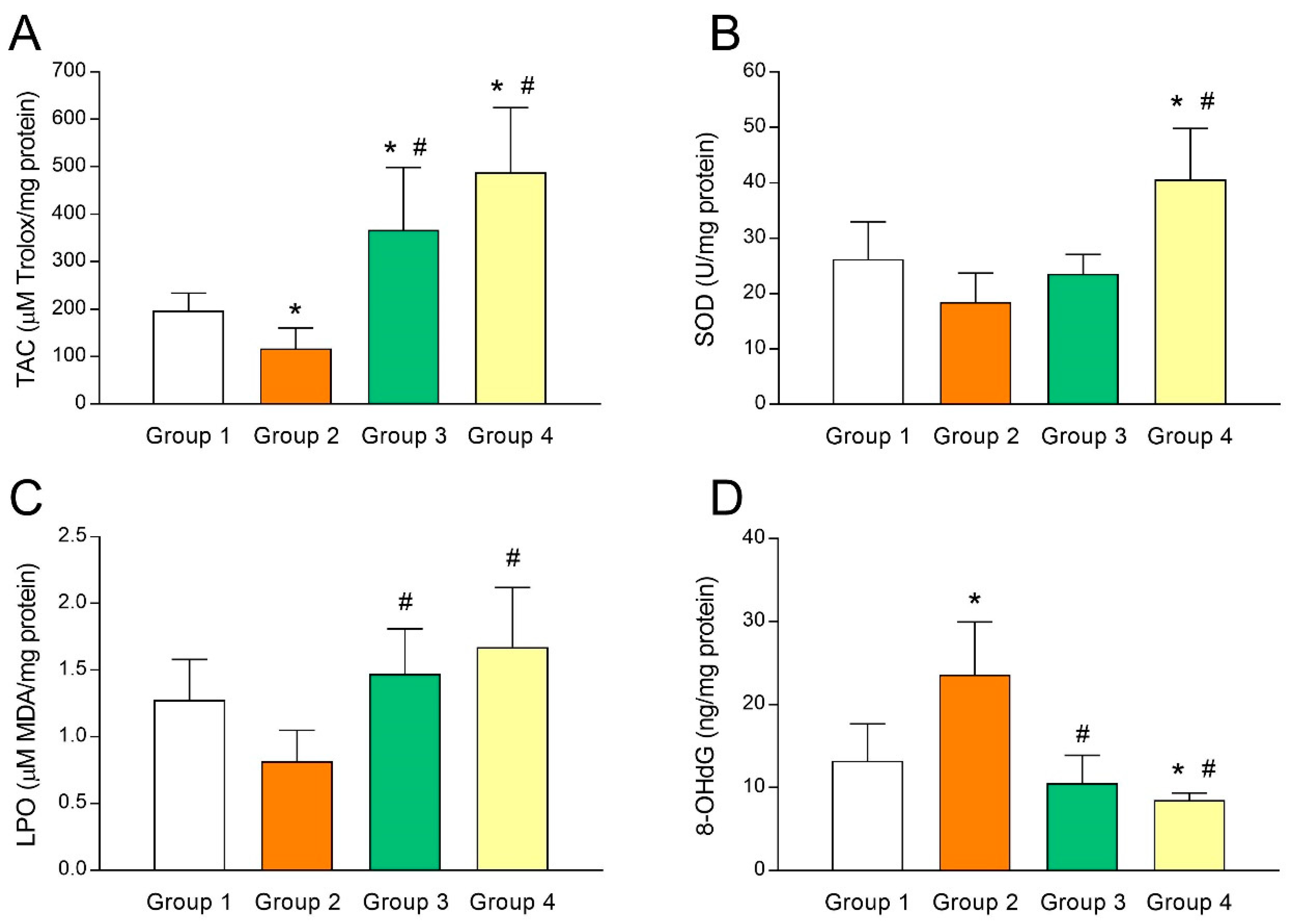
3.2. Intrafollicular Oxidative Balance in UI Patients and Effect of Melatonin Administration
3.3. Clinical Outcomes of Melatonin Supplementation in IVF-Embryo Transfer for UI Patients
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zegers-Hochschild, F.; Adamson, G.D.; de Mouzon, J.; Ishihara, O.; Mansour, R.; Nygren, K.; Sullivan, E.; Vanderpoel, S.; International Committee for Monitoring Assisted Reproductive Technology; World Health Organization. The International Committee for Monitoring Assisted Reproductive Technology (ICMART) glossary on ART terminology, 2009. Hum. Reprod. 2009, 24, 2683–2687. [Google Scholar] [CrossRef] [PubMed]
- Crosignani, P.G.; Collins, J.; Cooke, I.D.; Diczfalusy, E.; Rubin, B. Recommendations of the ESHRE workshop on “Unexplained Infertility”. Anacapri, August 28–29, 1992. Hum. Reprod. 1993, 8, 977–980. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.S.; Joo, B.S.; Na, Y.J.; Yoon, M.S.; Choi, O.H.; Kim, W.W. Relationships between concentrations of tumor necrosis factor-alpha and nitric oxide in follicular fluid and oocyte quality. J. Assist. Reprod. Genet. 2000, 17, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Polak, G.; Rola, R.; Gogacz, M.; Kozioł-Montewka, M.; Kotarski, J. Malonyldialdehyde and total antioxidant status in the peritoneal fluid of infertile women. Ginekol. Polska 1999, 70, 135–140. [Google Scholar]
- Tamura, H.; Takasaki, A.; Miwa, I.; Taniguchi, K.; Maekawa, R.; Asada, H.; Taketani, T.; Matsuoka, A.; Yamagata, Y.; Shimamura, K.; et al. Oxidative stress impairs oocyte quality and melatonin protects oocytes from free radical damage and improves fertilization rate. J. Pineal Res. 2008, 44, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H.; Takasaki, A.; Taketani, T.; Tanabe, M.; Kizuka, F.; Lee, L.; Tamura, I.; Maekawa, R.; Aasada, H.; Yamagata, Y.; et al. The role of melatonin as an antioxidant in the follicle. J. Ovarian Res. 2012, 5, 5. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Rosales-Corral, S.; Manchester, L.C. The Universal Nature, Unequal Distribution and Antioxidant Functions of Melatonin and Its Derivatives. Mini Rev. Med. Chem. 2013, 13, 373–384. [Google Scholar]
- Reiter, R.J.; Tan, D.X.; Tamura, H.; Cruz, M.H.C.; Fuentes-Broto, L. Clinical relevance of melatonin in ovarian and placental physiology: A review. Gynecol. Endocrinol. 2014, 30, 83–89. [Google Scholar] [CrossRef]
- Arendt, J.; Skene, D.J. Melatonin as a chronobiotic. Sleep Med. Rev. 2005, 9, 25–39. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Paredes, S.D.; Mayo, J.C.; Sainz, R.M. Melatonin and Reproduction Revisited. Biol. Reprod. 2009, 81, 445–456. [Google Scholar] [CrossRef]
- Guerrero, J.M.; Reiter, R.J. Melatonin-immune system relationships. Curr. Top. Med. Chem. 2002, 2, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Calvo, J.R.; González-Yanes, C.; Maldonado, M.D.; Gutiérrez, J.R.C.; Gonzalez-Yanes, C.; González-Yanes, C. The role of melatonin in the cells of the innate immunity: A review. J. Pineal Res. 2013, 55, 103–120. [Google Scholar] [CrossRef] [PubMed]
- Kubatka, P.; Bojková, B.; Ciková-Kalická, K.M.; Mníchová-Chamilová, M.; Adámeková, E.; Ahlers, I.; Ahlersová, E.; Cermáková, M. Effects of tamoxifen and melatonin on mammary gland cancer induced by N-methyl-N-nitrosourea and by 7, 12-dimethylbenz(a)anthracene, respectively, in female Sprague-Dawley rats. Folia Biol. 2001, 47, 5–10. [Google Scholar]
- Marková, M.; Adámeková, E.; Kubatka, P.; Bojková, B.; Ahlersová, E.; Ahlers, I.; Ahlersová, E. Effect of prolonged melatonin administration on metabolic parameters and organ weights in young male and female Sprague-Dawley rats. Acta Vet. Brno 2003, 72, 163–173. [Google Scholar] [CrossRef]
- Tan, D.X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Reiter, R.J. One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species? J. Pineal Res. 2007, 42, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.M.; Zhang, Y. Melatonin: A well-documented antioxidant with conditional pro-oxidant actions. J. Pineal Res. 2014, 57, 131–146. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.J.; Wang, H.; Kaya, A.; Memili, E.; Rodriguez-Osorio, N.; Rodríguez-Osorio, N.; Rodriguez-Osorio, N. Melatonin increases cleavage rate of porcine preimplantation embryos in vitro. J. Pineal Res. 2007, 43, 283–288. [Google Scholar]
- Kim, M.K.; Park, E.A.; Kim, H.J.; Choi, W.Y.; Cho, J.H.; Lee, W.S.; Cha, K.Y.; Kim, Y.S.; Lee, D.R.; Yoon, T.K. Does supplementation of in-vitro culture medium with melatonin improve IVF outcome in PCOS? Reprod. Biomed. Online 2013, 26, 22–29. [Google Scholar] [CrossRef]
- Ortiz, A.; Espino, J.; Bejarano, I.; Lozano, G.M.; Monllor, F.; García, J.F.; Pariente, J.A.; Rodríguez, A.B. High endogenous melatonin concentrations enhance sperm quality and short-term in vitro exposure to melatonin improves aspects of sperm motility. J. Pineal Res. 2011, 50, 132–139. [Google Scholar] [CrossRef]
- Espino, J.; Ortiz, Á.; Bejarano, I.; Lozano, G.M.; Monllor, F.; García, J.F.; Rodríguez, A.B.; Pariente, J.A. Melatonin protects human spermatozoa from apoptosis via melatonin receptor- and extracellular signal-regulated kinase-mediated pathways. Fertil. Steril. 2011, 95, 2290–2296. [Google Scholar] [CrossRef]
- Bejarano, I.; Monllor, F.; Marchena, A.M.; Ortiz, Á.; Lozano, G.; Jiménez, M.I.; Gaspar, P.; Garcia, J.F.; Pariente, J.A.; Rodríguez, A.B.; et al. Exogenous melatonin supplementation prevents oxidative stress-evoked DNA damage in human spermatozoa. J. Pineal Res. 2014, 57, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Lord, T.; Nixon, B.; Jones, K.T.; Aitken, R.J. Melatonin Prevents Postovulatory Oocyte Aging in the Mouse and Extends the Window for Optimal Fertilization In Vitro. Biol. Reprod. 2013, 88, 67. [Google Scholar] [CrossRef] [PubMed]
- Eryilmaz, O.G.; Devran, A.; Sarikaya, E.; Aksakal, F.N.; Mollamahmutoglu, L.; Cicek, N. Melatonin improves the oocyte and the embryo in IVF patients with sleep disturbances, but does not improve the sleeping problems. J. Assist. Reprod. Genet. 2011, 28, 815–820. [Google Scholar] [CrossRef] [PubMed]
- Batıoğlu, A.S.; Şahin, U.; Gürlek, B.; Öztürk, N.; Ünsal, E. The efficacy of melatonin administration on oocyte quality. Gynecol. Endocrinol. 2012, 28, 91–93. [Google Scholar] [CrossRef] [PubMed]
- Taketani, T.; Tamura, H.; Takasaki, A.; Lee, L.; Kizuka, F.; Tamura, I.; Taniguchi, K.; Maekawa, R.; Asada, H.; Shimamura, K.; et al. Protective role of melatonin in progesterone production by human luteal cells. J. Pineal Res. 2011, 51, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Manca, M.E.; Manunta, M.L.; Spezzigu, A.; Torres-Rovira, L.; Gonzalez-Bulnes, A.; Pasciu, V.; Piu, P.; Leoni, G.G.; Succu, S.; Chesneau, D.; et al. Melatonin deprival modifies follicular and corpus luteal growth dynamics in a sheep model. Reproduction 2014, 147, 885–895. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, P.; Raffone, E.; Benedetto, V. Effect of the treatment with myo-inositol plus folic acid plus melatonin in comparison with a treatment with myo-inositol plus folic acid on oocyte quality and pregnancy outcome in IVF cycles. A prospective, clinical trial. Eur. Rev. Med. Pharmacol. Sci. 2010, 14, 555–561. [Google Scholar]
- Unfer, V.; Raffone, E.; Rizzo, P.; Buffo, S. Effect of a supplementation with myo-inositol plus melatonin on oocyte quality in women who failed to conceive in previous in vitro fertilization cycles for poor oocyte quality: A prospective, longitudinal, cohort study. Gynecol. Endocrinol. 2011, 27, 857–861. [Google Scholar] [CrossRef]
- Torello, M.J.; Ardoy, M.; Calderon, G.; Cuadros, J.; Herrer, R.; Moreno, J.M.; Ortiz, A.; Prados, F.; Rodrguez, L.; Ten, J. Criterios ASEBIR de valoracion morfologica de Oocitos, Embriones tempranos y Blastocistos. In Proceedings of the 3rd ASEBIR Congress, Góbalo, Madrid, Spain, 17–18 November 2015; pp. 17–18. [Google Scholar]
- Zheng, M.; Tong, J.; Li, W.P.; Chen, Z.J.; Zhang, C. Melatonin concentration in follicular fluid is correlated with antral follicle count (AFC) and in vitro fertilization (IVF) outcomes in women undergoing assisted reproductive technology (ART) procedures. Gynecol. Endocrinol. 2018, 34, 446–450. [Google Scholar] [CrossRef]
- Tong, J.; Sheng, S.; Sun, Y.; Li, H.; Li, W.P.; Zhang, C.; Chen, Z.J. Melatonin levels in follicular fluid as markers for IVF outcomes and predicting ovarian reserve. Reproduction 2017, 153, 443–451. [Google Scholar] [CrossRef]
- Nakamura, Y.; Tamura, H.; Takayama, H.; Kato, H. Increased endogenous level of melatonin in preovulatory human follicles does not directly influence progesterone production. Fertil. Steril. 2003, 80, 1012–1016. [Google Scholar] [CrossRef]
- Artimani, T.; Karimi, J.; Mehdizadeh, M.; Yavangi, M.; Khanlarzadeh, E.; Ghorbani, M.; Asadi, S.; Kheiripour, N. Evaluation of pro-oxidant-antioxidant balance (PAB) and its association with inflammatory cytokines in polycystic ovary syndrome (PCOS). Gynecol. Endocrinol. 2018, 34, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Jana, S.K.; Narendra Babu, K.; Chattopadhyay, R.; Chakravarty, B.; Chaudhury, K.; Karuputhula, N. Upper control limit of reactive oxygen species in follicular fluid beyond which viable embryo formation is not favorable. Reprod. Toxicol. 2010, 29, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Seleem, A.K.; El Refaeey, A.A.; Shaalan, D.; Sherbiny, Y.; Badawy, A. Superoxide dismutase in polycystic ovary syndrome patients undergoing intracytoplasmic sperm injection. J. Assist. Reprod. Genet. 2014, 31, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Öztürk, G.; Coşkun, S.; Erbaş, D.; Hasanoğlu, E. The Effect of Melatonin on Liver Superoxide Dismutase Activity, Serum Nitrate and Thyroid Hormone Levels. Jpn. J. Physiol. 2000, 50, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Ng, T.B. Effect of pineal indoles on activities of the antioxidant defense enzymes superoxide dismutase, catalase, and glutathione reductase, and levels of reduced and oxidized glutathione in rat tissues. Biochem. Cell Biol. 2000, 78, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Allamaneni, S.S.R. Role of free radicals in female reproductive diseases and assisted reproduction. Reprod. Biomed. Online 2004, 9, 338–347. [Google Scholar] [CrossRef]
- Pasqualotto, E.B.; Agarwal, A.; Sharma, R.K.; Izzo, V.M.; Pinotti, J.A.; Joshi, N.J.; Rose, B.I. Effect of oxidative stress in follicular fluid on the outcome of assisted reproductive procedures. Fertil. Steril. 2004, 81, 973–976. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Betzendahl, I.; Cortvrindt, R.; Smitz, J.; Eichenlaub-Ritter, U. Effects of low O2 and ageing on spindles and chromosomes in mouse oocytes from pre-antral follicle culture. Hum. Reprod. 2001, 16, 737–748. [Google Scholar] [CrossRef]
- Ménézo, Y.; Dale, B.; Cohen, M. DNA damage and repair in human oocytes and embryos: A review. Zygote 2010, 18, 357–365. [Google Scholar] [CrossRef]
- Seino, T. Eight-hydroxy-2’-deoxyguanosine in granulosa cells is correlated with the quality of oocytes and embryos in an in vitro fertilization-embryo transfer program. Fertil. Steril. 2002, 77, 1184–1190. [Google Scholar] [CrossRef]
- Nishihara, T.; Hashimoto, S.; Ito, K.; Nakaoka, Y.; Matsumoto, K.; Hosoi, Y.; Morimoto, Y. Oral melatonin supplementation improves oocyte and embryo quality in women undergoing in vitro fertilization-embryo transfer. Gynecol. Endocrinol. 2014, 30, 359–362. [Google Scholar] [CrossRef] [PubMed]
| Variables | Group 1 | Group 2 | Group 3 | Group 4 | p Value |
|---|---|---|---|---|---|
| Age (years) | 36.27 ± 2.08 | 35.93 ± 3.20 | 34.73 ± 3.03 | 36.22 ± 2.71 | 0.577 |
| BMI (kg/m2) | 24.07 ± 3.29 | 23.83 ± 3.53 | 23.42 ± 2.88 | 23.66 ± 3.40 | 0.975 |
| bE2 (pg/mL) | 57.99 ± 22.04 | 59.21 ± 29.17 | 56.37 ± 22.19 | 56.50 ± 26.63 | 0.993 |
| bLH (IU/mL) | 6.49 ± 1.94 | 6.60 ± 3.29 | 7.32 ± 3.11 | 5.91 ± 2.85 | 0.744 |
| bFSH (IU/mL) | 9.42 ± 3.53 | 10.21 ± 4.68 | 9.28 ± 3.40 | 9.62 ± 3.33 | 0.949 |
| Variables | Group 1 | Group 2 | Group 3 | Group 4 |
|---|---|---|---|---|
| Average no. of oocytes retrieved (95% CI) | 8.7 (5.9–11.5) | 6.8 (4.4–9.2) | 10.0 # (6.7–13.3) | 9.5 # (5.8–13.5) |
| % of mature oocytes (k/n) | 81.9 (86/105) | 70.6 (48/68) | 83.6 # (92/110) | 76.2 (80/105) |
| % of fertilized oocytes (k/n) | 51.1 (44/86) | 47.9 (23/48) | 67.4 # (62/92) | 63.7 # (51/80) |
| Average no. of transferable embryos (95% CI) | 2.3 (0.5–4.0) | 2.0 (0.4–3.6) | 5.1 *# (2.8–7.4) | 4.6 *# (2.8–6.3) |
| % of clinical pregnancies/initiated cycle (k/n) | 50.0 (5/10) | 20.0 (2/10) | 30.0 (3/10) | 30.0 (3/10) |
| no. of full-term pregnancies | 5 | 2 | 3 | 3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Espino, J.; Macedo, M.; Lozano, G.; Ortiz, Á.; Rodríguez, C.; Rodríguez, A.B.; Bejarano, I. Impact of Melatonin Supplementation in Women with Unexplained Infertility Undergoing Fertility Treatment. Antioxidants 2019, 8, 338. https://doi.org/10.3390/antiox8090338
Espino J, Macedo M, Lozano G, Ortiz Á, Rodríguez C, Rodríguez AB, Bejarano I. Impact of Melatonin Supplementation in Women with Unexplained Infertility Undergoing Fertility Treatment. Antioxidants. 2019; 8(9):338. https://doi.org/10.3390/antiox8090338
Chicago/Turabian StyleEspino, Javier, María Macedo, Graciela Lozano, Águeda Ortiz, Carmina Rodríguez, Ana B. Rodríguez, and Ignacio Bejarano. 2019. "Impact of Melatonin Supplementation in Women with Unexplained Infertility Undergoing Fertility Treatment" Antioxidants 8, no. 9: 338. https://doi.org/10.3390/antiox8090338
APA StyleEspino, J., Macedo, M., Lozano, G., Ortiz, Á., Rodríguez, C., Rodríguez, A. B., & Bejarano, I. (2019). Impact of Melatonin Supplementation in Women with Unexplained Infertility Undergoing Fertility Treatment. Antioxidants, 8(9), 338. https://doi.org/10.3390/antiox8090338






