Caffeoylquinic Acids and Flavonoids of Fringed Sagewort (Artemisia frigida Willd.): HPLC-DAD-ESI-QQQ-MS Profile, HPLC-DAD Quantification, in Vitro Digestion Stability, and Antioxidant Capacity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials and Chemicals
2.2. Total Extract and Herbal Tea Preparation
2.3. High-Performance Liquid Chromatography with Diode Array Detection and Electrospray Ionization Triple Quadrupole Mass Spectrometric Detection (HPLC-DAD-ESI-QQQ-MS) Profiling Condition
2.4. HPLC-DAD Quantification Condition
2.5. Validation Analysis
2.6. Organoleptic Analysis and Crude Composition Analysis
2.7. General Phytochemical Analysis
2.8. Antioxidant Activity Analysis
2.8.1. DPPH• Radical Scavenging Assay
2.8.2. DPPH•-HPLC-DAD Procedure
2.8.3. Oxygen Radical Absorbance Capacity (ORAC) Assay
2.9. Simulated Gastrointestinal Digestion Assay
2.10. Statistical and Multivariative Analysis
3. Results and Discussion
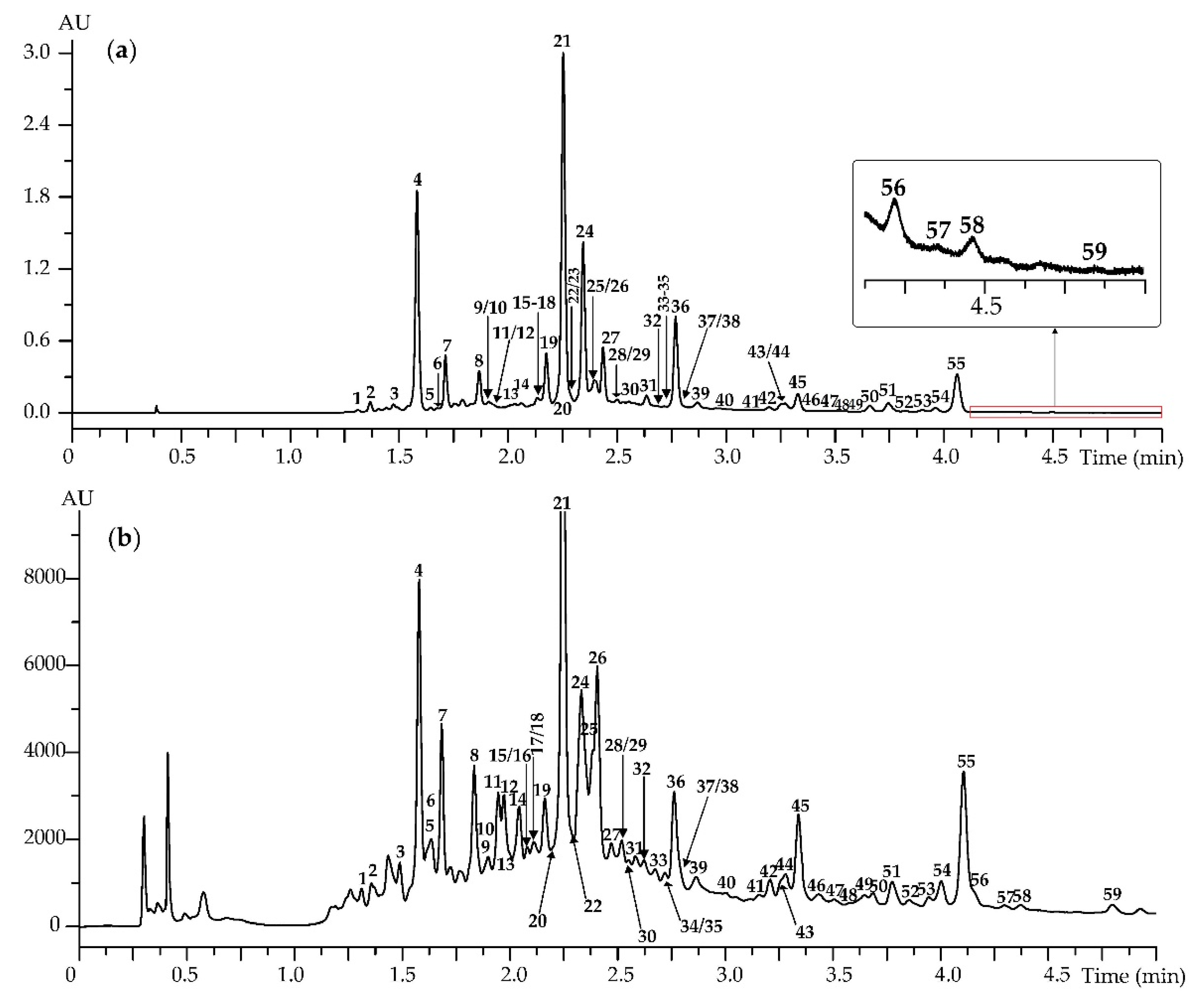
3.1. Caffeoylquinic Acids and Flavonoids of Artemisia frigida Herb: HPLC-DAD-ESI-QQQ-MS Profile, Organ Distribution and Chemotaxonomy
3.1.1. Caffeoylquinic Acids
3.1.2. Flavonoid Glycosides
3.1.3. Flavonoid Aglycones
3.1.4. Organ Distribution of Caffeoylquinic Acids and Flavonoids in A. frigida Plant
3.1.5. Chemotaxonomic Significance of Flavonoids and Caffeoylquinic Acids in the Subsection Frigidae of Artemisia Genus
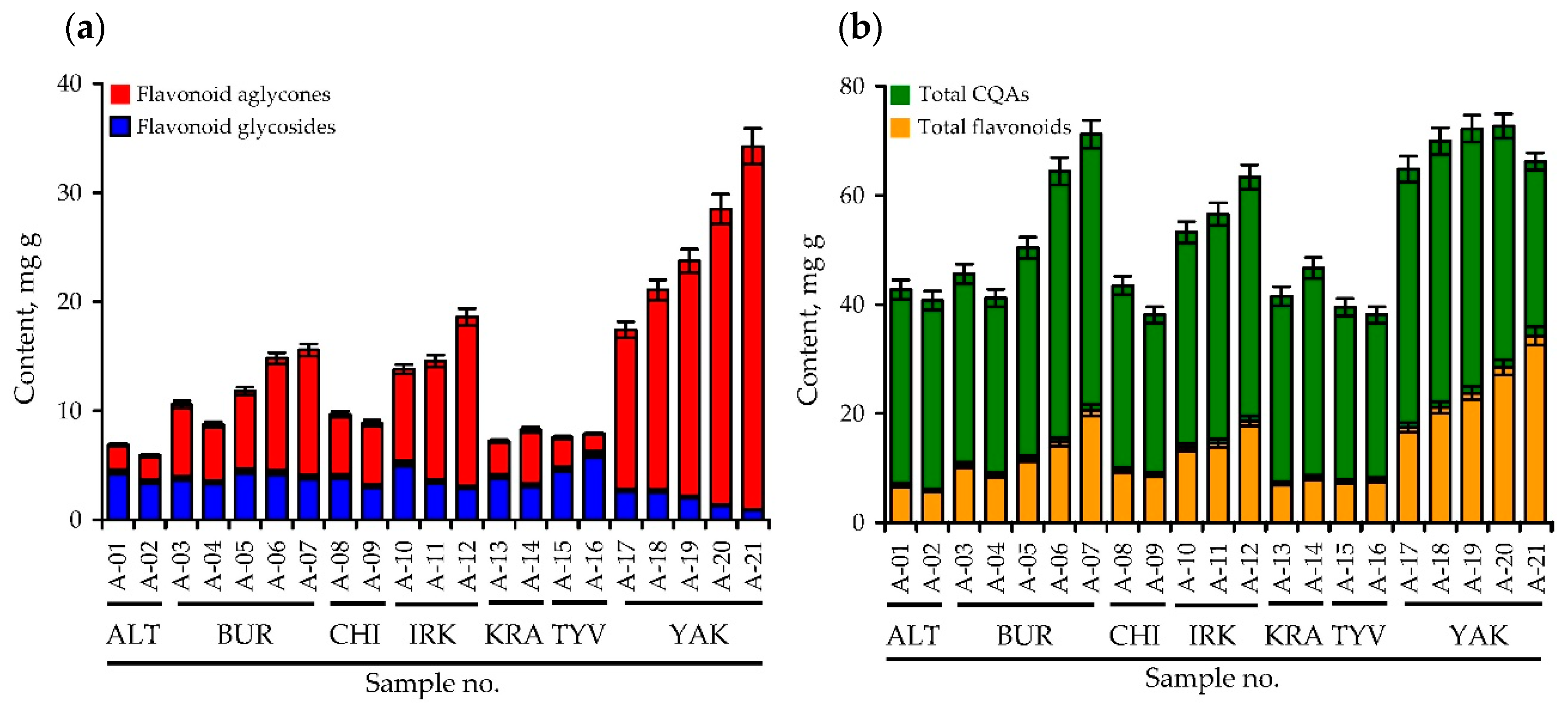
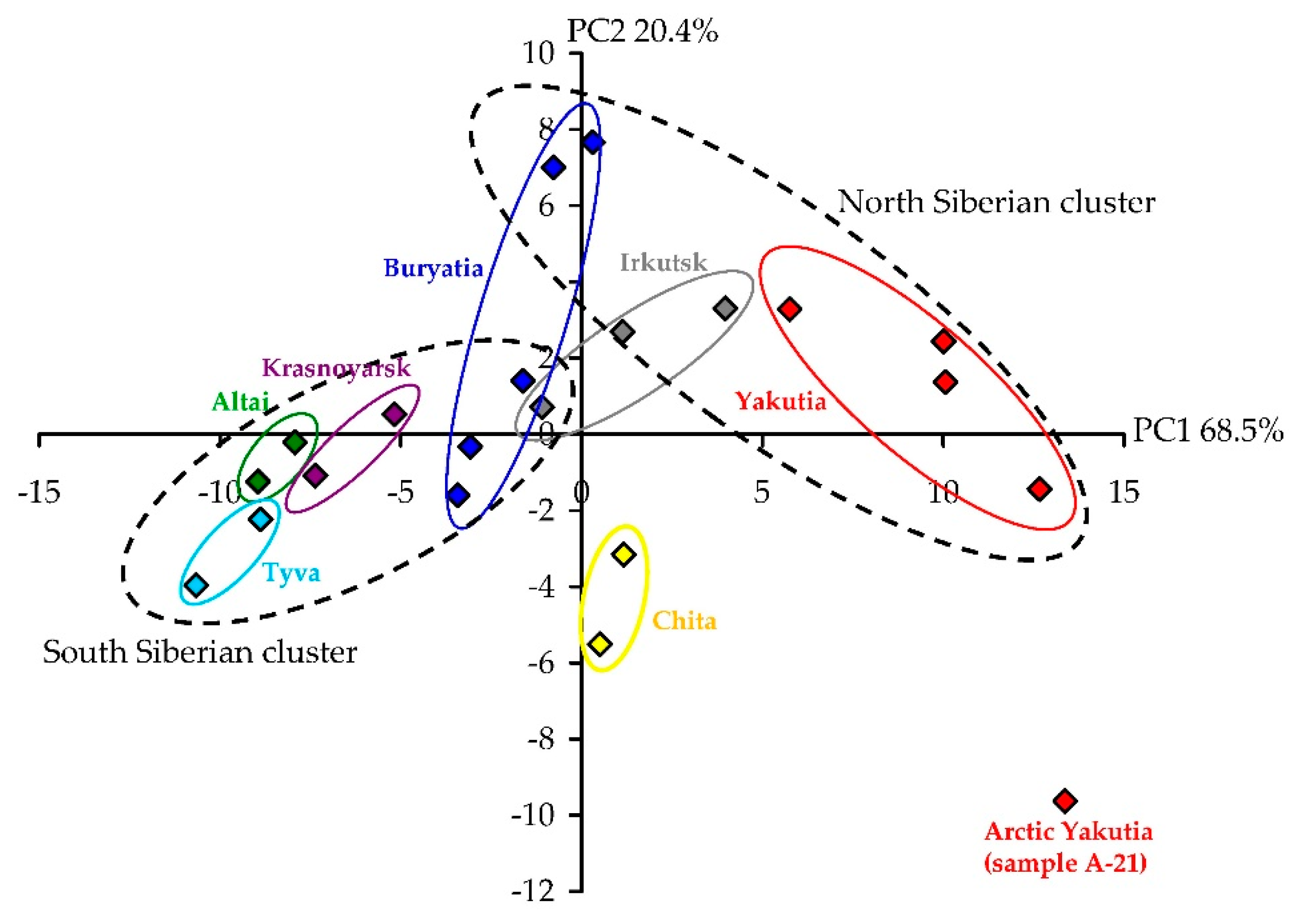
3.2. Variation of Phenolics in A. frigida Herb: HPLC Quantification and Principal Component Analysis Data of Twenty One Siberian Populations
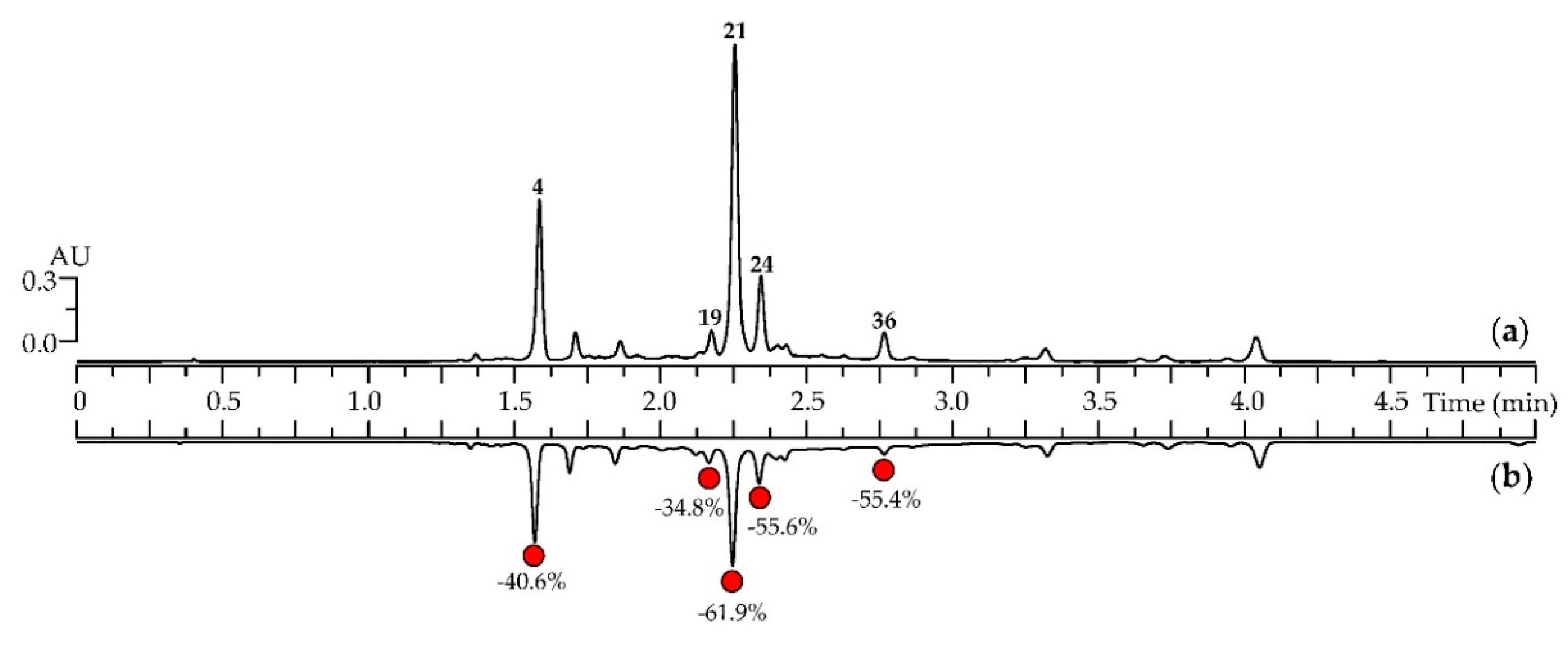
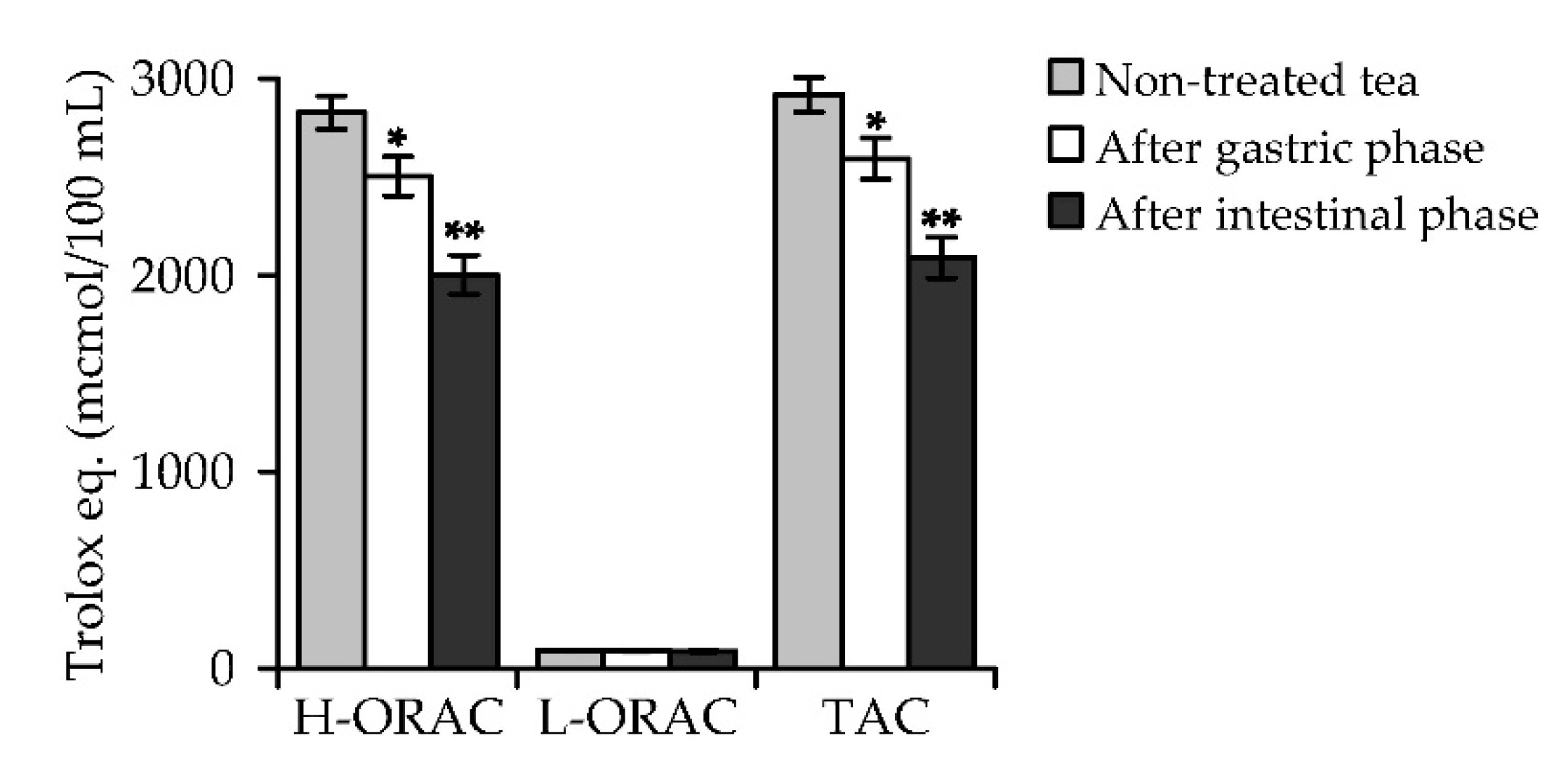
3.3. A. frigida Herbal Tea Phenolics: General Characteristics, HPLC Profile, in vitro Digestion Stability and Antioxidant Capacity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fabricant, D.S.; Farnsworth, N.R. The value of plants used in traditional medicine for drug discovery. Environ. Health Perspect. 2001, 10, 69–75. [Google Scholar] [CrossRef]
- Heinrich, M. Ethnopharmacology and drug discovery. In Comprehensive Natural Products II. Chemistry and Biology; Liu, H.W.B., Mander, L., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 3, pp. 351–377. [Google Scholar] [CrossRef]
- Aziz-Ul-Ikram; Zahra, N.B.; Shinwari, Z.K.; Muhammad, Q. Ethnomedicinal review of folklore medicinal plants belonging to family Apiaceae of Pakistan. Pak. J. Bot. 2015, 47, 1007–1014. [Google Scholar]
- Malyschev, L.I. (Ed.) Flora of Siberia. Asteraceae (Compositae); CRC Press: Boca Raton, FL, USA, 2007; Volume 13, pp. 90–141. [Google Scholar] [CrossRef]
- Danzin, P. Perennial medicinal plants. In Shel Pkxreng. Pure Crystal Necklace (Pharmacognosy of Tibetan Medicine); Aseeva, T.A., Ed.; Nauka: Moscow, Russia, 2017; pp. 201–255. ISBN 978-5-02-039772-9. [Google Scholar]
- Sumati, P. Decoctions. In Kunpan Dudzi. Useful for All Amrita Extract (Big Recipe Book of Aginsk Datsan); Aseeva, T.A., Ed.; Nauka: Vostochnaya Literatura, Russia, 2008; pp. 35–48. ISBN 978-5-02-036370-0. [Google Scholar]
- Desrid, S.C. Action of various medicinal remedies. In Vaidur’ya Onbo. Blue Beryl Garland (Commentary to Bdud Rtsi, a Decoration of the Doctrine of the King of Medicine); Aseeva, T.A., Ed.; Nauka: Vostochnaya Literatura, Russia, 2014; pp. 232–316. ISBN 978-5-02-036553-7. [Google Scholar]
- Batorova, S.M.; Yakovlev, G.P.; Aseeva, T.A. Reference-Book of Traditional Tibetan Medicine Herbs; Nauka: Novosibirsk, Russia, 2013; pp. 174–175. ISBN 978-5-02-019103-7. [Google Scholar]
- Chirikova, N.K. Phenolic and Terpenic Compounds of Plants from the Flora of Yakutia Republic. Ph.D. Thesis, Institute of General and Experimental Biology, Ulan-Ude, Russia, 13 December 2019. [Google Scholar]
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative stress: Harms and benefits for human health. Oxid. Med. Cell. Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef] [PubMed]
- Huyut, Z.; Beydemir, S.; Gülçin, İ. Antioxidant and antiradical properties of selected flavonoids and phenolic compounds. Biochem. Res. Int. 2017, 2017, 7616791. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-L.; Mabry, T.J. Two methylated flavones from Artemisia frigida. Phytochemistry 1981, 20, 309–311. [Google Scholar] [CrossRef]
- Liu, Y.-L.; Mabry, T.J. Flavonoids from Artemisia frigida. Phytochemistry 1981, 20, 1389–1395. [Google Scholar] [CrossRef]
- Wang, Q.-H.; Wang, J.-H.; Eerdunbagen; Tana. Chemical constituents of Artemisia frigida. Chin. Trad. Herb. Drugs 2009, 40, 1540–1543. [Google Scholar]
- Wang, Q.-H.; Ao, W.-L.-J.; Wang, X.-L.; Bao, X.-H.; Wang, J.-H. Two new flavonoid glycosides from Artemisia frigida Willd. J. Asian Nat. Prod. Res. 2010, 12, 950–954. [Google Scholar] [CrossRef]
- Wang, Q.-H.; Ao, W.-L.-J.; Tai, W.-Q. Simultaneous determination of seven flavonoids in aerial parts of Artemisia frigida by HPLC. Chin. Herb. Med. 2012, 4, 252–258. [Google Scholar]
- Wang, Q.-H.; Ao, W.-L.; Dai, N.-Y. Structural elucidation and HPLC analysis of six flavone glycosides from Artemisia frigida Willd. Chem. Res. Chin. Univ. 2013, 29, 439–444. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, J.; Wu, X.; Han, N.; Tai, W.; Dai, N.; Wu, R.; Ao, W. Anti-inflammatory effects and structure elucidation of flavonoid and biflavonoid glycosides from Artemisia frigida Willd. Monatsh. Chem. 2015, 146, 383–387. [Google Scholar] [CrossRef]
- Wang, Q.; Jin, J.; Dai, N.; Han, N.; Han, J.; Bao, B. Anti-inflammatory effects, nuclear magnetic resonance identification, and high-performance liquid chromatography isolation of the total flavonoids from Artemisia frigida. J. Food Drug Anal. 2016, 24, 385–391. [Google Scholar] [CrossRef]
- Belenovskaya, L.M.; Markova, L.P.; Kapranova, G.I. Phenolic compounds of Artemisia frigida. Chem. Nat. Comp. 1980, 16, 834. [Google Scholar]
- Olennikov, D.N.; Chirikova, N.K.; Kashchenko, N.I.; Nikolaev, V.M.; Kim, S.-W.; Vennos, C. Bioactive phenolics of the genus Artemisia (Asteraceae): HPLC-DAD-ESI-TQ-MS/MS profile of the Siberian species and their inhibitory potential against α-amylase and α-glucosidase. Front. Pharmacol. 2018, 9, 756. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Kashchenko, N.I. New flavonoids and turkesterone-2-O-cinnamate from leaves of Rhaponticum uniflorum. Chem. Nat. Comp. 2019, 55, 256–264. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Kashchenko, N.I. New isorhamnetin glucosides and other phenolic compounds from Calendula officinalis. Chem. Nat. Comp. 2013, 49, 833–840. [Google Scholar] [CrossRef]
- Chirikova, N.K.; Olennikov, D.N. Phenolic compounds from Siberian species Thymus baicalensis and T. sibiricus. Chem. Nat. Comp. 2018, 54, 572–576. [Google Scholar] [CrossRef]
- Anonymous. Organoleptic Analysis of Herbal Ingredients; American Herbal Products Association: Silver Spring, MD, USA, 2013. [Google Scholar]
- Anonymous. Quality Control Methods for Herbal Materials; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 76, 248–254. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K. Meadowsweet teas as new functional beverages: Comparative analysis of nutrients, phytochemicals and biological effects of four Filipendula species. Molecules 2017, 22, 16. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K.; Gornostai, T.G.; Selyutina, I.Y.; Zilfikarov, I.N. Effect of low temperature cultivation on the phytochemical profile and bioactivity of Arctic plants: A case of Dracocephalum palmatum. Int. J. Molec. Sci. 2017, 18, 2579. [Google Scholar] [CrossRef]
- Chirikova, N.K.; Olennikov, D.N.; Tankhaeva, L.M. Quantitative determination of flavonoid content in the aerial parts of Baikal skullcap (Scutellaria baicalensis Georgi). Russ. J. Bioorg. Chem. 2010, 36, 915–922. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Tankhaeva, L.M. Quantitative determination of phenolic compounds in Mentha piperita leaves. Chem. Nat. Comp. 2010, 46, 22–27. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Fedorov, I.A.; Kashchenko, N.I.; Chirikova, N.K.; Vennos, C. Khellactone derivatives and other phenolics of Phlojodicarpus sibiricus (Apiaceae): HPLC-DAD-ESI-QQQ-MS/MS and HPLC-UV profile, and antiobesity potential of dihydrosamidin. Molecules 2019, 24, 2286. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Kruglova, M.Y. A new quercetin glycoside and other phenolic compounds from the genus Filipendula. Chem. Nat. Comp. 2013, 49, 610–616. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K.; Tankhaeva, L.M. Iridoids and flavonoids of four Siberian gentians: Chemical profile and gastric stimulatory effect. Molecules 2015, 20, 19172–19188. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Tankhaeva, L.M.; Samuelsen, A.B. Quantitative analysis of polysaccharides from Plantago major using the Dreywood method. Chem. Nat. Comp. 2006, 42, 265–268. [Google Scholar] [CrossRef]
- Fadhil, S.; Reza, M.H.; Rouhollah, G.; Reza, V.R.M. Spectrophotometric determination of total alkaloids in Peganum harmala L. using bromocresol green. Res. J. Phytochem. 2007, 1, 79–82. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Chirikova, N.K.; Okhlopkova, Z.M.; Zulfugarov, I.S. Chemical composition and antioxidant activity of Tánara Ótó (Dracocephalum palmatum Stephan), a medicinal plant used by the North-Yakutian nomads. Molecules 2013, 18, 14105–14121. [Google Scholar] [CrossRef]
- Prior, R.L.; Hoang, H.; Gu, L.; Wu, X.; Bacchiocca, M.; Howard, L.; Hampsch-Woodill, M.; Huang, D.; Ou, B.; Jacob, R. Assays for hydrophilic and lipophilic antioxidant capacity (oxygen radical absorbance capacity (ORAC(FL)) of plasma and other biological and food samples. J. Agric. Food Chem. 2003, 51, 3273–3279. [Google Scholar] [CrossRef]
- Wu, X.; Gu, L.; Holden, J.; Haytowitz, D.B.; Gebhardt, S.E.; Beecher, G.; Prior, R.L. Development of a database for total antioxidant capacity in foods: A preliminary study. J. Food Comp. Anal. 2004, 17, 407–422. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K. In Vitro bioaccessibility, human gut microbiota metabolites and hepatoprotective potential of chebulic ellagitannins: A case of Padma Hepaten® formulation. Nutrients 2015, 7, 8735–8749. [Google Scholar] [CrossRef]
- Lin, L.-Z.; Hanley, J.M. Identification of hydroxycinnamoylquinic acids of arnica flowers and burdock roots using a standardized LC-DAD-ESI/MS profiling method. J. Agric. Food Chem. 2008, 56, 10105–10114. [Google Scholar] [CrossRef]
- Lu, Y.; Sun, Y.; Foo, L.Y.; McNabb, W.C.; Molan, A.L. Phenolic glycosides of forage legume Onobrychis viciifolia. Phytochemistry 2000, 55, 67–75. [Google Scholar] [CrossRef]
- Azimova, S.S.; Vinogradova, V.I. Natural Compounds. Flavonoids, Plant Sources, Structures and Properties; Springer Science: New York, NY, USA, 2008; pp. 84–215. [Google Scholar] [CrossRef]
- Li, Q.; Wang, L.; Dai, P.; Zeng, X.; Qi, X.; Zhu, L.; Yan, T.; Wang, Y.; Lu, L.; Hu, M.; et al. A combined strategy of mass fragmentation, post-column cobalt complexation and shift in ultraviolet absorption spectra to determine the uridine 5′-diphospho-glucuronosyltransferase metabolism profiling of flavones after oral administration of a flavone mixture in rats. J. Chromatogr. A 2015, 1395, 116–128. [Google Scholar] [CrossRef]
- Han, J.; Ye, M.; Qiao, X.; Xu, M.; Wang, B.; Guo, D. Characterization of phenolic compounds in the Chinese herbal drug Artemisia annua by liquid chromatography coupled to electrospray ionization mass spectrometry. J. Pharm. Biomed. Anal. 2008, 47, 516–525. [Google Scholar] [CrossRef]
- Alipieva, K.I.; Kostadinova, E.; Evstatieva, L.N.; Stefova, M.; Bankova, V.S. An iridoid and a flavonoid from Sideritis lanata. Fitoterapia 2009, 80, 51–53. [Google Scholar] [CrossRef]
- Ali, B.; Imran, M.; Hussain, R.; Ahmed, Z.; Malik, A. Structural determination of abutilins A and B, new flavonoids from Abutilon pakistanicum, by 1D and 2D NMR spectroscopy. Magn. Reson. Chem. 2010, 48, 159–163. [Google Scholar] [CrossRef]
- Iwashina, T.; Kamenosono, K.; Ueno, T. Hispidulin and nepetin 4′-glucosides from Cirsium oligophyllum. Phytochemistry 1999, 51, 1109–1111. [Google Scholar] [CrossRef]
- Yang, H.-B.; Wang, Y.-C.; Zhang, Z.-T.; Chang, Y. Synthesis and crystal structure of pilloin. Turk. J. Chem. 2018, 32, 87–95. [Google Scholar]
- Valant-Vetschera, K.M.; Wollenweber, E. Flavonoid aglycones from the leaf surfaces of some Artemisia spp. (Compositae-Anthemideae). Z. Naturforsch. 1995, 50, 353–357. [Google Scholar] [CrossRef]
- Luo, S.Q.; Yuan, L.; Wu, Y.K.; Huang, J.G. Effect of fertilization on phenolic components and antioxidant activities of Artemisia annua. China J. Chin. Mater. Med. 2013, 38, 1493–1499. [Google Scholar] [PubMed]
- Pereira, C.G.; Barreira, L.; Bijttebier, S.; Pieters, L.; Marques, C.; Santos, T.F.; Rodrigues, M.J.; Varela, J.; Custódio, L. Health promoting potential of herbal teas and tinctures from Artemisia campestris subsp. maritima: From traditional remedies to prospective products. Sci. Rep. 2018, 16, 4689. [Google Scholar] [CrossRef]
- Melikoglu, G.; Cubukcu, B.; Ozhatay, N. Flavonoids of Artemisia caucasica. J. Fac. Pharm. Istanbul Univ. 2003, 35, 145–147. [Google Scholar]
- Cubukcu, B.; Melikoglu, G. Flavonoids of Artemisia austriaca. Planta Med. 1995, 61, 488. [Google Scholar] [CrossRef]
- Belenovskaya, L.M.; Markova, L.P.; Kapranova, G.I. Phenolic compounds of Artemisia xerophytica. Chem. Nat. Comp. 1982, 18, 115. [Google Scholar] [CrossRef]
- Kikhanova, Z.S.; Iskakova, Z.B.; Dzhalmakhanbetova, R.I.; Seilkhanov, T.M.; Ross, S.A.; Suleimen, E.M. Constituents of Artemisia austriaca and their biological activity. Chem. Nat. Comp. 2013, 49, 967–968. [Google Scholar] [CrossRef]
- Al-Hazimi, H.M.G.; Basha, R.M.Y. Phenolic compounds from various Artemisia species. J. Chem. Soc. Pak. 1991, 13, 277–289. [Google Scholar]
- Rivero, R.M.; Ruiz, J.M.; García, P.C.; López-Lefebre, L.R.; Sánchez, E.; Romero, L. Resistance to cold and heat stress: Accumulation of phenolic compounds in tomato and watermelon plants. Plant Sci. 2001, 160, 315–321. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K. Spinacetin, a new caffeoylglucoside, and other phenolic compounds from Gnaphalium uliginosum. Chem. Nat. Comp. 2015, 51, 1085–1090. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Chirikova, N.K. Caffeoylglucaric acids and other phenylpropanoids of the Siberian Leonurus species. Chem. Nat. Comp. 2016, 52, 780–782. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K.; Koryakina, L.P.; Vladimirov, L.N. Bitter gentian teas: Nutritional and phytochemical profiles, polysaccharide characterisation and bioactivity. Molecules 2015, 20, 20014–20030. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Yu, F.; Qian, H.; Zhang, J.; Sun, J.; Ma, Z. Simultaneous quantification of eight organic acid components in Artemisia capillaris Thunb (Yinchen) extract using high-performance liquid chromatography coupled with diode array detection and high-resolution mass spectrometry. J. Food Drug Anal. 2018, 26, 788–795. [Google Scholar] [CrossRef]
- Farah, A.; Lima, J.D.P. Consumption of chlorogenic acids through coffee and health implications. Beverages 2019, 5, 11. [Google Scholar] [CrossRef]
- Li, X.; Li, K.; Xie, H.; Xie, Y.; Li, Y.; Zhao, X.; Jiang, X.; Chen, D. Antioxidant and cytoprotective effects of the di-O-caffeoylquinic acid family: The mechanism, structure—Activity relationship, and conformational effect. Molecules 2018, 23, 222. [Google Scholar] [CrossRef]
- Alminger, M.; Aura, A.M.; Bohn, T.; Dufour, C.; El, S.N.; Gomes, A.; Karakaya, S.; Martínez-Cuesta, M.C.; McDougall, G.J.; Requena, T.; et al. In Vitro models for studying secondary plant metabolite digestion and bioaccessibility. Compr. Rev. Food Sci. Food Saf. 2014, 13, 413–436. [Google Scholar] [CrossRef]
- Bohn, T. Dietary factors affecting polyphenol bioavailability. Nutr Rev. 2014, 72, 429–452. [Google Scholar] [CrossRef]
- Pandey, A.K.; Singh, P. The genus Artemisia: A 2012–2017 literature review on chemical composition, antimicrobial, insecticidal and antioxidant activities of essential oils. Medicines 2017, 4, 68. [Google Scholar] [CrossRef]
- Lima, K.; Silva, O.; Figueira, M.E.; Pires, C.; Cruz, D.; Gomes, S.; Maurício, E.M.; Duarte, M.P. Influence of the in vitro gastrointestinal digestion on the antioxidant activity of Artemisia gorgonum Webb and Hyptis pectinata (L.) Poit. infusions from Cape Verde. Food Res. Int. 2019, 115, 150–159. [Google Scholar] [CrossRef]

| No | Collection Place | Collection Date | Coordinates | Height (m a.s.l.) | Voucher Specimens No |
|---|---|---|---|---|---|
| A-01 | Altaiskii Krai, Smolenskii District, Belokurikha | 20.VII.2017 | 51°58′12.5′′N, 84°57′23.5′′E | 448 | BU/AK-As-ar/h-0717-027 |
| A-02 | Altaiskii Krai, Novichikhinskii District, Mel’nikovo | 27.VII.2017 | 52°10′47.0′′N, 81°12′34.1′′E | 230 | BU/AK-As-ar/h-0717-032 |
| A-03 | Buryatia Republic, Mukhorshibirskii District, Mukhorshibir’ | 21.VII.2018 | 51°01′45.3′′N, 107°47′49.9′′E | 765 | BU/BR-As-ar/h-0718-094 |
| A-04 | Buryatia Republic, Zakamenskii District, Tsakir | 19.VII.2016 | 50°24′59.2′′N, 103°34′02.6′′E | 1078 | BU/BR-As-ar/h-0716-041 |
| A-05 | Buryatia Republic, Okinskii District, Orlik | 22.VII.2017 | 52°29′35.3′′N, 99°47′08.2′′E | 1857 | BU/BR-As-ar/h-0717-026 |
| A-06 | Buryatia Republic, Bauntovskii District, Malovskii | 29.VII.2015 | 54°22′45.1′′N, 113°32′16.3′′E | 950 | BU/BR-As-ar/h-0715-116 |
| A-07 | Buryatia Republic, Severobaikal’skii District, Kumora | 31.VII.2017 | 55°52′02.7′′N, 111°10′58.4′′E | 578 | BU/BR-As-ar/h-0717-094 |
| A-08 | Chita Oblast, Akshinskii District, Ureisk | 21.VII.2017 | 50°16′59.0′′N, 113°10′42.5′′E | 902 | BU/CO-As-ar/h-0717-012 |
| A-09 | Chita Oblast, Alexandrovo Zavodskii District, Klichka | 24.VII.2017 | 50°28′07.9′′N, 118°06′30.0′′E | 1085 | BU/CO-As-ar/h-0717-029 |
| A-10 | Irkutsk Oblast, Shelokhovskii District, Olkha | 18.VII.2017 | 52°09′10.4′′N, 104°06′46.2′′E | 490 | BU/IO-As-ar/h-0717-006 |
| A-11 | Irkutsk Oblast, Bratskii District, Vikhorevka | 24.VII.2016 | 56°08′32.7′′N, 101°12′32.3′′E | 381 | BU/IO-As-ar/h-0716-011 |
| A-12 | Irkutsk Oblast, Katangskii District, Erbogachen | 28.VII.2016 | 61°15′50.3′′N 108°00′50.5′′E | 301 | BU/IO-As-ar/h-0716-027 |
| A-13 | Krasnoyarskii Krai, Berezovskii District, Lopatino | 17.VII.2015 | 55°56′39.1′′N, 93°17′33.1′′E | 375 | BU/KK-As-ar/h-0715-006 |
| A-14 | Krasnoyarskii Krai, Turukhanskii District, Kellog | 22.VII.2015 | 62°26′38.5′′N, 86°18′16.1′′E | 77 | BU/KK-As-ar/h-0715-014 |
| A-15 | Tyva Republic, Kaa-Khemskii District, Saryg-Sep | 25.VII.2018 | 51°28′48.5′′N, 95°29′26.5′′E | 778 | BU/TR-As-ar/h-0718-012 |
| A-16 | Tyva Republic, Barun-Khemchikskii District, Ak-Dovurak | 27.VII.2018 | 51°08′28.0′′N, 90°37′38.1′′E | 853 | BU/TR-As-ar/h-0718-018 |
| A-17 | Yakutia (Sakha) Republic, Mirninskii Ulus, Almaznyi | 25.VII.2017 | 62°28′01.9′′N, 113°50′58.1′′E | 388 | BU/YR-As-ar/h-0717-063 |
| A-18 | Yakutia (Sakha) Republic, Oimyakonskii Ulus, Ust’-Nera | 30.VII.2017 | 64°31′50.4′′N, 142°59′42.1′′E | 506 | BU/YR-As-ar/h-0717-085 |
| A-19 | Yakutia (Sakha) Republic, Verkhekolymskii Ulus, Ugol’noye | 1.VIII.2017 | 65°43′40.7′′N, 149°45′19.4′′E | 134 | BU/YR-As-ar/h-0817-106 |
| A-20 | Yakutia (Sakha) Republic, Srednekolymskii Ulus, Sylgy-Ytar | 21.VII.2018 | 67°50′00.4′′N, 154°48′01.6′′E | 31 | BU/YR-As-ar/h-0718-064 |
| A-21 | Yakutia (Sakha) Republic, Nizhnekolymskii Ulus, Tymkino | 27.VII.2018 | 68°28′46.8′′N, 160°07′56.7′′E | 9 | BU/YR-As-ar/h-0718-072 |
| No | tR, min | Compound | UV, λmax, nm | CE, eV | ESI-MS/MS, m/z | Ref. | |
|---|---|---|---|---|---|---|---|
| (M–H) − | MS/MS Fragment Ions | ||||||
| 1 | 1.285 | 1-O-Caffeoylquinic acid a | 327 | 30 | 353 | (353): 191, 179, 135 | [42] |
| 2 | 1.376 | 4-O-Caffeoylquinic acid a | 327 | 30 | 353 | (353): 191, 179, 135 | [42] |
| 3 | 1.485 | Friginoside B (Tent.) b | 270, 352 | 15 | 695 | (695): 519, 343; [343]: 329, 301 | [15] |
| 4 | 1.614 | 5-O-Caffeoylquinic acid a | 327 | 30 | 353 | (353): 191, 179, 135 | [42] |
| 5 | 1.627 | 3-O-Caffeoylquinic acid a | 327 | 30 | 353 | (353): 191, 179, 135 | [42] |
| 6 | 1.687 | 1,3-Di-O-caffeoylquinic acid a | 328 | 35 | 515 | (515): 353, 335, 191, 179 | [42] |
| 7 | 1.742 | Vicenin-2 (Api-6,8-di-C-Glc) a | 255, 329 | 30 | 593 | (593): 503, 473, 413; (473): 383, 353 | [43] |
| 8 | 1.871 | Isoorientin (Lut-6-C-Glc) a | 255, 267, 348 | 25 | 447 | (447): 357, 327 | [44] |
| 9 | 1.886 | Isoschaftoside (Api-6-C-Ara-8-C-Glc) a | 273, 329 | 30 | 563 | (563): 503, 473, 443, 413, 383, 353; (353): 325, 297 | [44] |
| 10 | 1.897 | Orientin (Lut-8-C-Glc) a | 255, 267, 348 | 25 | 447 | (447): 357, 327 | [44] |
| 11 | 1.938 | Schaftoside (Api-6-C-Glc-8-C-Ara) a | 273, 329 | 30 | 563 | (563): 503, 473, 443, 413, 383, 353; (353): 325, 297 | [44] |
| 12 | 1.942 | Chrysoeriol-O-Ac-Hex b | 269, 336 | 15 | 503 | (503): 461, 299 | [43] |
| 13 | 2.043 | Vitexin (Api-8-C-Glc) a | 272, 330 | 25 | 431 | (431): 341, 311 | [44] |
| 14 | 2.063 | Apigenin-C-Hex-O-Hex b | 273, 329 | 30 | 593 | (593): 431, 341, 311 | [44] |
| 15 | 1.121 | Isovitexin (Api-6-C-Glc) a | 272, 330 | 20 | 431 | (431): 341, 311 | [44] |
| 16 | 1.124 | 6-Hydroxyluteolin-7-O-Glc a | 251, 280, 345 | 10 | 463 | (463): 301 | [22] |
| 17 | 1.129 | Cynaroside (Lut-7-O-Glc) a | 256, 265, 347 | 10 | 447 | (447): 285 | [44] |
| 18 | 1.132 | Nepitrin (Nep-7-O-Glc) a | 271, 345 | 20 | 477 | (477): 315, 301 | [22] |
| 19 | 2.188 | 3,4-Di-O-caffeoylquinic acid a | 328 | 40 | 515 | (515): 353, 335, 191 | [42] |
| 20 | 2.210 | Diosmetin-7-O-Glc a | 251, 268, 345 | 15 | 461 | (461): 299, 285 | [22] |
| 21 | 2.251 | 3,5-Di-O-caffeoylquinic acid a | 327 | 40 | 515 | (515): 353, 191, 179, 135 | [42] |
| 22 | 2.312 | Thermopsoside (Chr-7-O-Glc) a | 253, 267, 346 | 15 | 461 | (461): 299, 285 | [44] |
| 23 | 3.316 | Rhaunoside F (Nep-3′-O-Glc) a | 268, 339 | 10 | 477 | (477): 315, 301 | [22] |
| 24 | 2.372 | 4,5-Di-O-caffeoylquinic acid a | 328 | 40 | 515 | (515): 353, 179 | [42] |
| 25 | 2.378 | 6-Hydroxyluteolin-dimethyl ether-O-Hex b | 253, 267, 343 | 25 | 491 | (491): 329, 301 | [22] |
| 26 | 2.380 | 1,5-Di-O-caffeoylquinic acid a | 328 | 35 | 515 | (515): 353, 191, 179, 135 | [42] |
| 27 | 2.437 | Luteolin-4′-O-Glc a | 267, 337 | 10 | 447 | (447): 285 | [44] |
| 28 | 2.497 | Chrysoeriol-O-Ac-Hex b | 269, 336 | 15 | 503 | (503): 461, 299 | [43] |
| 29 | 2.504 | Apigenin-7-O-glucoside (cosmosiin) a | 265, 334 | 10 | 431 | (431): 269 | [44] |
| 30 | 2.562 | 6-Hydroxyluteolin-3′-O-Glc (Tent.) b | 275, 339 | 10 | 463 | (463): 301 | [22] |
| 31 | 2.626 | 1,3,5-Tri-O-caffeoylquinic acid a | 326 | 45 | 677 | (677): 515, 353; (515): 353, 191, 179, 135 | [42] |
| 32 | 2.688 | 1,4,5-Tri-O-caffeoylquinic acid a | 326 | 45 | 677 | (677): 515, 353; (515): 353, 191, 179 | [42] |
| 33 | 2.714 | Chrysoeriol-4′-O-Glc a | 268, 337 | 15 | 461 | (461): 299, 285 | [44] |
| 34 | 2.718 | Nepetin-4′-O-Glc a | 268, 341 | 25 | 477 | (477): 315, 301 | [22] |
| 35 | 2.722 | 6-Hydroxyluteolin-dimethyl ether-O-Hex b | 253, 267, 344 | 25 | 491 | (491): 329, 301 | [22] |
| 36 | 2.756 | 3,4,5-Tri-O-caffeoylquinic acid a | 326 | 48 | 677 | (677): 515; (515): 353, 179 | [42] |
| 37 | 2.810 | Chrysoeriol-O-Ac-Hex b | 269, 336 | 15 | 503 | (503): 461, 299 | [43] |
| 38 | 2.819 | Rhaunoside C (6-hydroxyluteolin-4′-O-Glc) a | 286, 335 | 10 | 463 | (463): 301 | [22] |
| 39 | 2.875 | Chrysoeriol-5-O-Glc (Tent.) b | 261, 343 | 15 | 461 | (461): 299, 285 | [45] |
| 40 | 2.934 | 6-Hydroxyluteolin-dimethyl ether-O-Hex b | 252, 267, 342 | 25 | 491 | (491): 329, 301 | [22] |
| 41 | 3.120 | Tricin a | 270, 345 | 35 | 329 | (329): 315, 301 | [44] |
| 42 | 3.236 | Apigenin a | 267, 334 | 10 | 269 | [44] | |
| 43 | 3.251 | Hispidulin a | 273, 333 | 10 | 299 | (299): 285 | [44] |
| 44 | 3.259 | 5,7,3′-Trihydroxy-6,4′,5′-trimethoxyflavone b | 273, 331 | 35 | 359 | (359): 345, 331, 317 | [12] |
| 45 | 3.312 | Jaceosidine a | 273, 343 | 35 | 329 | (329): 315, 301 | [44] |
| 46 | 3.375 | 5,7,3′,4′,5′-Pentahydroxy-6,8-dimethoxyflavone b | 269, 370 | 40 | 361 | (361): 347, 333 | [17] |
| 47 | 3.482 | Chrysoeriol a | 252, 272, 345 | 35 | 299 | (299): 285 | [44] |
| 48 | 3.562 | 5,7,4′-Trihydroxy-6,3′,5′-trimethoxyflavone b | 272, 347 | 35 | 359 | (359): 345, 331, 317 | [12] |
| 49 | 3.621 | Desmethylcentaureidin b | 252, 273, 346 | 35 | 329 | (329): 315, 301 | [44] |
| 50 | 3.638 | Luteolin-3′,4′-dimethyl ether a | 251, 271, 342 | 40 | 313 | (313): 299, 285 | [44] |
| 51 | 3.752 | Eupatorin a | 252, 275, 343 | 40 | 343 | (343): 329, 315, 301 | [44] |
| 52 | 3.812 | 5,7,3′,4′-Tetrahydroxy-6,5′-dimethoxyflavone b | 272, 350 | 40 | 345 | (345): 331, 317 | [13] |
| 53 | 3.879 | Quercetagetin-3,6,3′,4′-tetramethyl ether b | 256, 272, 344 | 40 | 373 | (373): 359, 345, 331 | [44] |
| 54 | 3.894 | Acacetin a | 270, 327 | 20 | 283 | (283): 269 | [44] |
| 55 | 4.062 | Cirsimaritin a | 274, 332 | 35 | 313 | (313): 299, 285 | [44] |
| 56 | 4.124 | Cirsilineol a | 273, 345 | 35 | 343 | (343): 329, 315, 301 | [44] |
| 57 | 4.187 | Velutin a | 251, 344 | 35 | 313 | (313): 299, 285 | [44] |
| 58 | 4.251 | Pilloin b | 251, 275, 340 | 35 | 313 | (313): 299, 285 | [44] |
| 59 | 4.782 | Genkwanin a | 268, 335 | 20 | 283 | (283): 269 | [44] |
| Compounds | A. frigida Herbal Tea, mg/100 mL | ||
|---|---|---|---|
| Non-Treated | After Gastric Phase | After Intestinal Phase | |
| Caffeoylquinic acids | |||
| 4-O-Caffeoylquinic acid | 0.45 ± 0.01 | 0.44 ± 0.01 | 0.39 ± 0.01 |
| 5-O-Caffeoylquinic acid | 16.09 ± 0.45 | 15.12 ± 0.42 | 12.98 ± 0.33 |
| 3,4-Di-O-caffeoylquinic acid | 0.82 ± 0.02 | 0.76 ± 0.02 | 0.57 ± 0.02 |
| 3,5-Di-O-caffeoylquinic acid | 16.35 ± 0.43 | 14.02 ± 0.35 | 8.46 ± 0.22 |
| 4,5-Di-O-caffeoylquinic acid | 2.87 ± 0.08 | 2.33 ± 0.06 | 1.29 ± 0.03 |
| 3,4,5-Tri-O-caffeoylquinic acid | tr. | tr. | tr. |
| Subtotal caffeoylquinic acids | 36.58 | 32.67 | 23.69 |
| Flavonoid glycosides | |||
| Vicenin-2 | 1.34 ± 0.04 | 1.32 ± 0.04 | 1.31 ± 0.03 |
| Isoorientin | 2.41 ± 0.06 | 2.37 ± 0.06 | 2.04 ± 0.06 |
| Cynaroside | tr. | tr. | tr. |
| Subtotal flavonoid glycosides | 3.75 | 3.69 | 3.35 |
| Flavonoid aglycones | |||
| Apigenin | n.d. | n.d. | n.d. |
| Hispidulin | n.d. | n.d. | n.d. |
| Jaceosidine | tr. | tr. | tr. |
| Luteolin-3′,4′-dimethyl ether | n.d. | n.d. | n.d. |
| Eupatorin | n.d. | n.d. | n.d. |
| Acacetin | n.d. | n.d. | n.d. |
| Cirsimaritin | tr. | tr. | tr. |
| Subtotal flavonoid aglycones | tr. | tr. | tr. |
| Total flavonoids | 3.75 | 3.69 | 3.35 |
| Total phenolics | 40.33 | 36.36 | 27.04 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K.; Vasil’eva, A.G.; Gadimli, A.I.; Isaev, J.I.; Vennos, C. Caffeoylquinic Acids and Flavonoids of Fringed Sagewort (Artemisia frigida Willd.): HPLC-DAD-ESI-QQQ-MS Profile, HPLC-DAD Quantification, in Vitro Digestion Stability, and Antioxidant Capacity. Antioxidants 2019, 8, 307. https://doi.org/10.3390/antiox8080307
Olennikov DN, Kashchenko NI, Chirikova NK, Vasil’eva AG, Gadimli AI, Isaev JI, Vennos C. Caffeoylquinic Acids and Flavonoids of Fringed Sagewort (Artemisia frigida Willd.): HPLC-DAD-ESI-QQQ-MS Profile, HPLC-DAD Quantification, in Vitro Digestion Stability, and Antioxidant Capacity. Antioxidants. 2019; 8(8):307. https://doi.org/10.3390/antiox8080307
Chicago/Turabian StyleOlennikov, Daniil N., Nina I. Kashchenko, Nadezhda K. Chirikova, Aina G. Vasil’eva, Aydan I. Gadimli, Javanshir I. Isaev, and Cecile Vennos. 2019. "Caffeoylquinic Acids and Flavonoids of Fringed Sagewort (Artemisia frigida Willd.): HPLC-DAD-ESI-QQQ-MS Profile, HPLC-DAD Quantification, in Vitro Digestion Stability, and Antioxidant Capacity" Antioxidants 8, no. 8: 307. https://doi.org/10.3390/antiox8080307
APA StyleOlennikov, D. N., Kashchenko, N. I., Chirikova, N. K., Vasil’eva, A. G., Gadimli, A. I., Isaev, J. I., & Vennos, C. (2019). Caffeoylquinic Acids and Flavonoids of Fringed Sagewort (Artemisia frigida Willd.): HPLC-DAD-ESI-QQQ-MS Profile, HPLC-DAD Quantification, in Vitro Digestion Stability, and Antioxidant Capacity. Antioxidants, 8(8), 307. https://doi.org/10.3390/antiox8080307







