Antioxidant and Anti-Inflammatory Activities of the Crude Extracts of Moringa oleifera from Kenya and Their Correlations with Flavonoids
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Chemicals and Reagents
2.3. Preparation of Sample Extraction
2.4. Antioxidant Assays of M. oleifera
2.4.1. DPPH Assay
2.4.2. ABTS Assay
2.4.3. FRAP Assay
2.5. Determinations of Total Flavonoids Content (TFC)
2.6. Anti-Inflammatory Activities of M. oleifera
2.7. HPLC-UV/ESI-MS/MS Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Antioxidant Activity of M. oleifera Leaves, Seeds and Roots
3.2. Total Flavonoids Content (TFC)
3.3. Anti-Inflammatory Activity
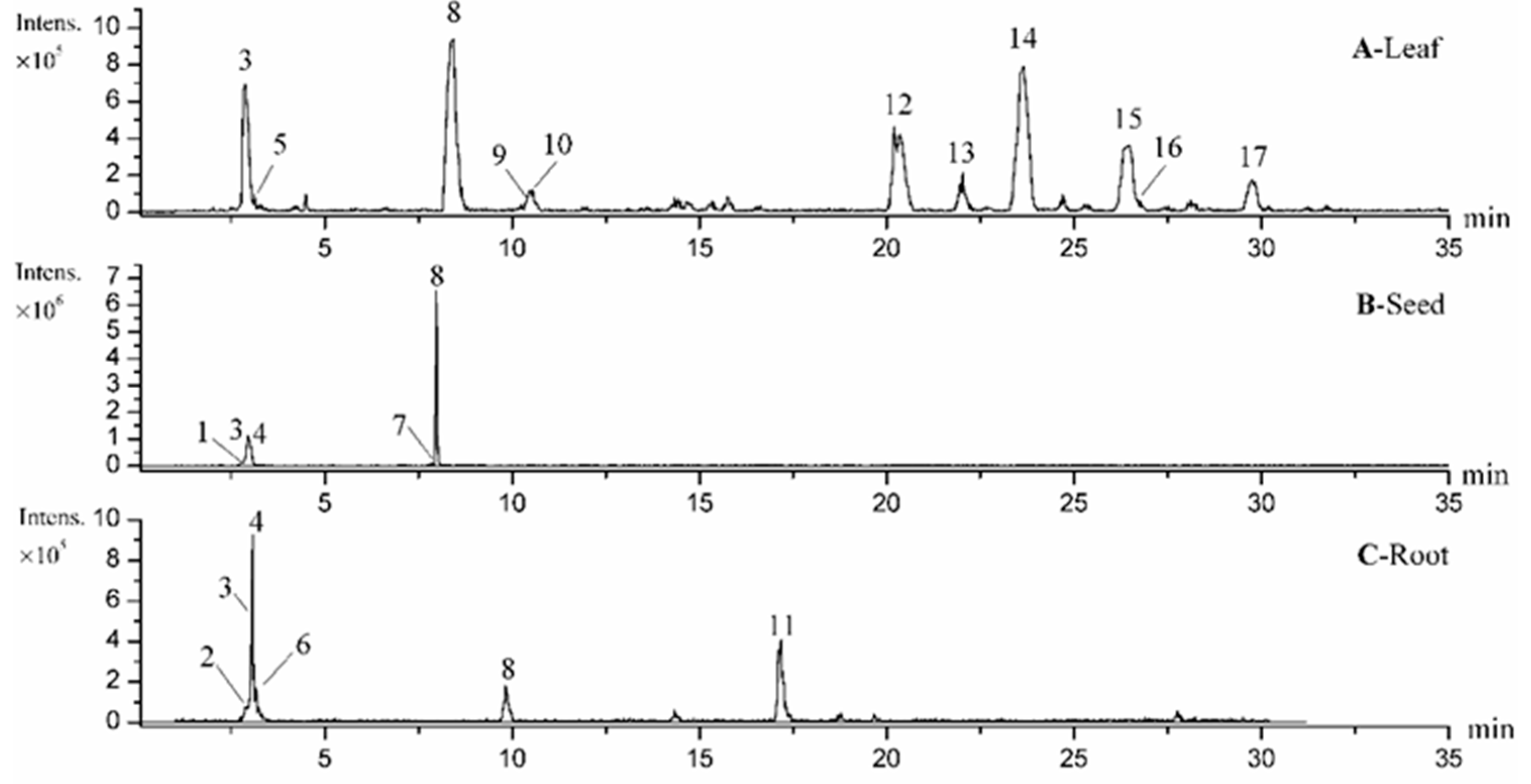
3.4. Analysis of M. oleifera Leaves, Seeds and Roots by HPLC-UV/ESI-MS/MS
3.4.1. Saccharides and Nitrile Glycosides
3.4.2. Organic Acids and Phenylpropanoids
3.4.3. Glucosinolates
3.4.4. Phenolic Acids and Flavonoids
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Leone, A.; Spada, A.; Battezzati, A.; Schiraldi, A.; Aristil, J.; Bertoli, S. Cultivation, genetic, ethnopharmacology, phytochemistry and pharmacology of Moringa oleifera leaves: An overview. Int. J. Mol. Sci. 2015, 16, 12791–12835. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Pérez, C.; Quirantes-Piné, R.; Fernández-Gutiérrez, A.; Carretero, A.S. Optimization of extraction method to obtain a phenolic compounds-rich extract from Moringa oleifera Lam leaves. Ind. Crop. Prod. 2015, 66, 246–254. [Google Scholar] [CrossRef]
- Gupta, S.; Jain, R.; Kachhwaha, S.; Kothari, S. Nutritional and medicinal applications of Moringa oleifera Lam.—Review of current status and future possibilities. J. Herb. Med. 2018, 11, 1–11. [Google Scholar] [CrossRef]
- Kasolo, J.N.; Bimenya, G.S.; Ojok, L.; Ochieng, J.; Ogwal-Okeng, J.W. Phytochemicals and uses of Moringa oleifera leaves in Ugandan rural communities. J. Med. Plants Res. 2010, 4, 753–757. [Google Scholar]
- Coppin, J.P.; Xu, Y.; Chen, H.; Pan, M.H.; Ho, C.T.; Juliani, R.; Simon, J.E.; Wu, Q. Determination of flavonoids by LC/MS and anti-inflammatory activity in Moringa oleifera. J. Funct. Foods 2013, 5, 1892–1899. [Google Scholar] [CrossRef]
- Panda, S.; Kar, A.; Sharma, P.; Sharma, A. Cardioprotective potential of N,α-l-rhamnopyranosyl vincosamide, an indole alkaloid, isolated from the leaves of Moringa oleifera in isoproterenol induced cardiotoxic rats: In vivo and in vitro studies. Bioorganic Med. Chem. Lett. 2013, 23, 959–962. [Google Scholar] [CrossRef] [PubMed]
- Mathur, M.; Yadav, S.; Katariya, P.K.; Kamal, R. In vitro propagation and biosynthesis of steroidal sapogenins from various morphogenetic stages of Moringa oleifera Lam., and their antioxidant potential. Acta Physiol. Plant 2014, 36, 1749–1762. [Google Scholar] [CrossRef]
- Roy, S.K.; Chandra, K.; Ghosh, K.; Mondal, S.; Maiti, D.; Ojha, A.K.; Das, D.; Mondal, S.; Chakraborty, I.; Islam, S.S. Structural investigation of a heteropolysaccharide isolated from the pods (fruits) of Moringa oleifera (Sajina). Carbohydr. Res. 2007, 342, 2380–2389. [Google Scholar] [CrossRef] [PubMed]
- Maldini, M.; Maksoud, S.A.; Natella, F.; Montoro, P.; Petretto, G.L.; Foddai, M.; De Nicola, G.R.; Chessa, M.; Pintore, G.A.M. Moringa oleifera: Study of phenolics and glucosinolates by mass spectrometry. J. Mass Spectrom. 2014, 49, 900–910. [Google Scholar] [CrossRef] [PubMed]
- Bhatta, R.; Saravanan, M.; Baruah, L.; Sampath, K.T. Nutrient content, in vitro ruminal fermentation characteristics and methane reduction potential of tropical tannin-containing leaves. J. Sci. Food Agric. 2012, 92, 2929–2935. [Google Scholar] [CrossRef] [PubMed]
- Coz-Bolaños, X.; Campos-Vega, R.; Reynoso-Camacho, R.; Ramos-Gómez, M.; Loarca-Piña, G.F.; Guzmán-Maldonado, S.H. Moringa infusion (Moringa oleifera) rich in phenolic compounds and high antioxidant capacity attenuate nitric oxide pro-inflammatory mediator in vitro. Ind. Crops Prod. 2018, 118, 95–101. [Google Scholar] [CrossRef]
- Sahakitpichan, P.; Mahidol, C.; Disadee, W.; Ruchirawat, S.; Kanchanapoom, T. Unusual glycosides of pyrrole alkaloid and 4′-hydroxyphenylethanamide from leaves of Moringa oleifera. Phytochemistry 2011, 72, 791–795. [Google Scholar] [CrossRef] [PubMed]
- Jung, I.L. Soluble extract from Moringa oleifera leaves with a new anticancer activity. PLoS ONE 2014, 9, e95492. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.R.; Vijayakumar, M.; Mathela, C.S.; Rao, C.V. In vitro and in vivo antioxidant properties of different fractions of Moringa oleifera leaves. Food Chem. Toxicol. 2009, 47, 2196–2201. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, J.R.O.; Silva, G.C.; Costa, R.A.; Fontenelle, J.R.L.D.S.; Vieira, G.H.F.; Filho, A.A.F.; Vieira, R.H.S.D.F. In vitro antibacterial effect of aqueous and ethanolic Moringa leaf extracts. Asian Pac. J. Trop. Med. 2011, 4, 201–204. [Google Scholar] [CrossRef]
- Atta, A.H.; Nasr, S.M.; Almaweri, A.H.; Sedky, D.; Mohamed, A.M.; Desouky, H.M.; Shalaby, M.A. Phytochemical, antioxidant and hepatoprotective effects of different fractions of Moringa oleifera leaves methanol extract against liver injury in animal model. Asian Pac. J. Trop. Med. 2018, 11, 423–429. [Google Scholar]
- Dangi, S.Y.; Jolly, C.I.; Narayanan, S. Antihypertensive activity of the total alkaloids from the leaves of Moringa oleifera. Pharm. Biol. 2008, 40, 144–148. [Google Scholar] [CrossRef]
- Helmy, S.A.; Morsy, N.F.; Elaby, S.M.; Ghaly, M.A. Hypolipidemic effect of Moringa oleifera Lam leaf powder and its extract in diet-induced hypercholesterolemic rats. J. Med. Food 2017, 20, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, D.; Rai, P.K.; Kumar, A.; Mehta, S.; Watal, G. Effect of Moringa oleifera Lam. leaves aqueous extract therapy on hyperglycemic rats. J. Ethnopharmacol. 2009, 123, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.G.; Negi, P.S.; Radha, C. Phenolic composition, antioxidant and antimicrobial activities of free and bound phenolic extracts of Moringa oleifera seed flour. J. Funct. Foods 2013, 5, 1883–1891. [Google Scholar] [CrossRef]
- Al-Malki, A.L.; El Rabey, H.A. The antidiabetic effect of low doses of Moringa oleifera Lam. seeds on streptozotocin induced diabetes and diabetic nephropathy in male rats. BioMed Res. Int. 2015, 2015, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Araújo, L.C.C.; Aguiar, J.S.; Napoleao, T.H.; Mota, F.V.B.; Barros, A.L.S.; Moura, M.C.; Coriolano, M.C.; Coelho, L.C.B.B.; Silva, T.G.; Paiva, P.M.G. Evaluation of cytotoxic and anti-Inflammatory activities of extracts and lectins from Moringa oleifera seeds. PLoS ONE 2013, 8, e81973. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.; Chen, S.; Wang, X.; Yuan, G.; Jiang, F.; Chen, X.; Wang, L. Characterization of Moringa oleifera roots polysaccharide MRP-1 with anti-inflammatory effect. Int. J. Biol. Macromol. 2019, 132, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.; Bhattacharya, R.; Pradhan, C.; Chaudhuri, K.; Mukhopadhyay, A.; Bose, C.K. Antiproliferative effect of Moringa oleifera root extract on ovarian carcinoma: An in vitro study. Ann. Oncol. 2016, 27, 318. [Google Scholar] [CrossRef]
- Choudhary, M.K.; Bodakhe, S.H.; Gupta, S.K. Assessment of the antiulcer potential of Moringa oleifera root-bark extract in rats. J. Acupunct. Meridian Stud. 2013, 6, 214–220. [Google Scholar] [CrossRef]
- Shukla, S.; Mathur, R.; Prakash, A.O. Antifertility profile of the aqueous extract of Moringa oleifera roots. J. Ethnopharmacol. 1988, 22, 51–62. [Google Scholar] [CrossRef]
- Karadi, R.V.; Gadge, N.B.; Alagawadi, K.R.; Savadi, R.V. Effect of Moringa oleifera Lam. root-wood on ethylene glycol induced urolithiasis in rats. J. Ethnopharmacol. 2006, 105, 306–311. [Google Scholar] [CrossRef]
- Ndhlala, A.R.; Mulaudzi, R.; Ncube, B.; Abdelgadir, H.A.; du Plooy, C.P.; Van Staden, J. Antioxidant, antimicrobial and phytochemical variations in thirteen Moringa oleifera Lam. cultivars. Molecules 2014, 19, 10480–10494. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef]
- Zhu, M.-Z.; Wu, W.; Jiao, L.L.; Yang, P.F.; Guo, M.Q. Analysis of flavonoids in lotus (Nelumbo nucifera) leaves and their antioxidant activity using macroporous resin chromatography coupled with LC-MS/MS and antioxidant biochemical assays. Molecules 2015, 20, 10553–10565. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Chang, S.K.C.; Gu, Y.; Qian, S.Y. Antioxidant activity and phenolic compositions of lentil (Lens culinaris var. Morton) extract and its fractions. J. Agric. Food Chem. 2011, 59, 2268–2276. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Li, W.; Zhang, Q.; Chen, L.; Chen, W.; Zhang, H.; Ni, Y. Anti-inflammatory activities of Guang-Pheretima extract in lipopolysaccharide-stimulated RAW 264.7 murine macrophages. BMC Complement. Altern. Med. 2018, 18, 46. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Liu, T.; Zhang, C.; Guo, M. Flavonoids of lotus (Nelumbo nucifera) seed embryos and their antioxidant potential. J. Food Sci. 2017, 82, 1834–1841. [Google Scholar] [CrossRef] [PubMed]
- Atawodi, S.E.; Atawodi, J.C.; Idakwo, G.A.; Pfundstein, B.; Haubner, R.; Wurtele, G.; Bartsch, H.; Owen, R.W. Evaluation of the polyphenol content and antioxidant properties of methanol extracts of the leaves, stem, and root barks of Moringa oleifera Lam. J. Med. Food 2010, 13, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Nwidu, L.L.; Elmorsy, E.; Aprioku, J.S.; Siminialayi, I.; Carter, W.G. In vitro anti-cholinesterase and antioxidant activity of extracts of Moringa oleifera plants from Rivers State, Niger Delta, Nigeria. Medicines (Basel) 2018, 5, 71. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, S.; Bhanger, M. Effect of season and production location on antioxidant activity of Moringa oleifera leaves grown in Pakistan. J. Food Compos. Anal. 2006, 19, 544–551. [Google Scholar] [CrossRef]
- Peng, H.; Lin, L.; Liu, Y.H.; Ruan, R.S.; Liu, C.M. Negative ion mode ESI-MS analysis of cellooligosaccharides. Food Sci. 2009, 30, 147–149. [Google Scholar]
- Jaja-Chimedza, A.; Graf, B.L.; Simmler, C.; Kim, Y.; Kühn, P.; Pauli, G.F.; Raskin, I. Biochemical characterization and anti-inflammatory properties of an isothiocyanate-enriched moringa (Moringa oleifera) seed extract. PLoS ONE 2017, 12, e0182658. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Li, X.; Saleri, F.; Guo, M. Analysis of flavonoids in Rhamnus davurica and its antiproliferative activities. Molecules 2016, 21, 1275. [Google Scholar] [CrossRef] [PubMed]
- Li, F.H.; Wang, H.Q.; Su, X.M.; Li, C.K.; Li, B.M.; Chen, R.Y.; Kang, J. Constituents isolated from n-butanol extract of leaves of Moringa oleifera. China J. Chin. Mater. Med. 2018, 43, 114–118. [Google Scholar]
- Matei, M.F.; Jaiswal, R.; Bassil, B.S.; Kortz, U.; Kuhnert, N.; Deshpande, S. Synthesis, structure, and tandem mass spectrometric characterization of the diastereomers of quinic acid. J. Agric. Food Chem. 2016, 64, 7298–7306. [Google Scholar]
- Clifford, M.N.; Knight, S.; Kuhnert, N. Discriminating between the six isomers of dicaffeoylquinic acid by LC-MSn. J. Agric. Food Chem. 2005, 53, 3821–3832. [Google Scholar] [CrossRef] [PubMed]

| Sample | IC50 of DPPH * (mg/mL) | IC50 of ABTS * (mg/mL) | FRAP * (mM Fe2+/g) | TFC * (mg RE/g) |
|---|---|---|---|---|
| Leaves | 1.87 ± 0.03 c | 1.36 ± 0.02 b | 0.99 ± 0.06 a | 192.36 ± 2.96 a |
| Seeds | >64 a | 40.35 ± 1.47 a | 0.02 ± 0.00 c | 5.89 ± 0.65 c |
| Roots | 3.33 ± 0.11 b | 1.24 ± 0.03 b | 0.20 ± 0.01 b | 106.79 ± 2.12 b |
| Trolox | 0.10 ± 0.01 d | NT * | NT | NT |
| Ascorbic acid | 0.05 ± 0.00 e | 0.11 ± 0.01 b | NT | NT |
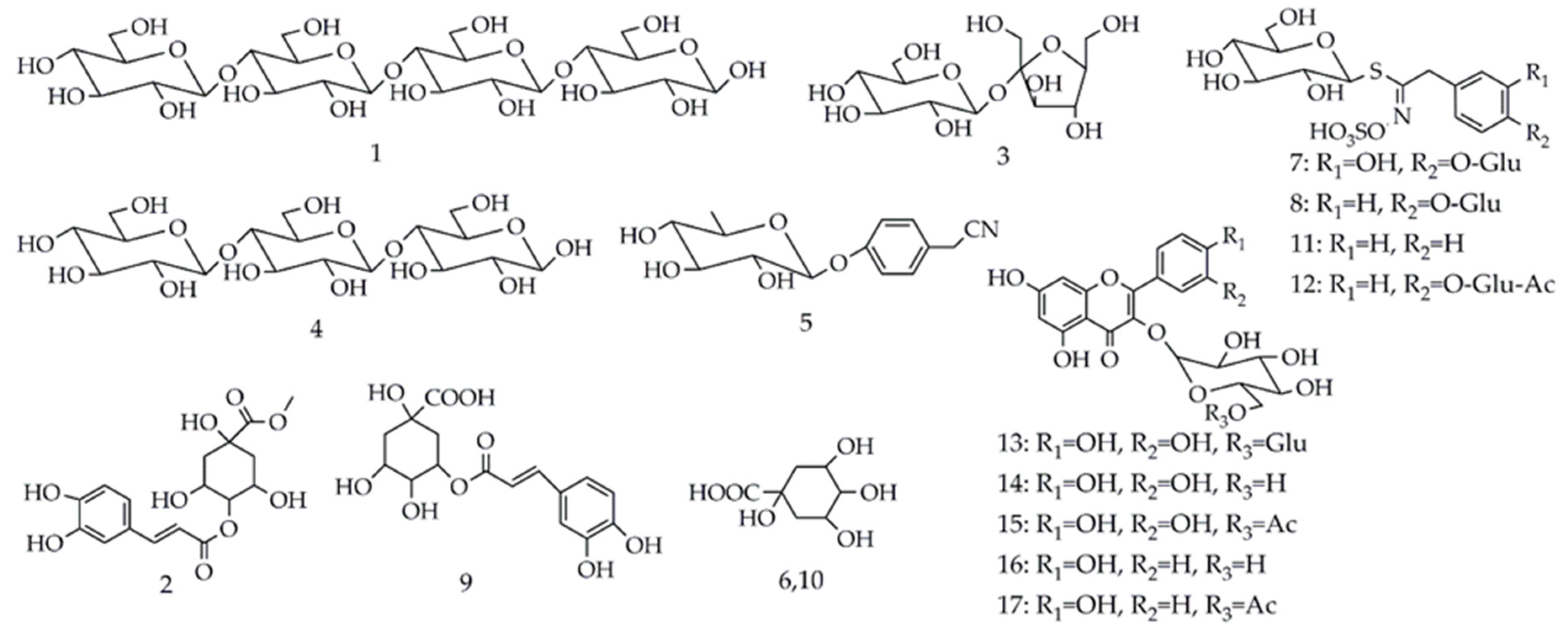
| Peak No. a | RT b (min) | [M−H]− | Molecule Formula | MS2 (m/z) | Identification | Part c | Reference |
|---|---|---|---|---|---|---|---|
| Saccharides | |||||||
| 1 | 2.83 | 665 | C24H42O21 | 485, 383, 341, 179 | Cellotetraose | S | [38] |
| 3 | 2.88 | 341 | C12H22O11 | 179, 161, 131, 119, 113, 101, 89 | Sucrose | L, S, R | [39] |
| 4 | 2.90 | 503 | C18H32O16 | 383, 323, 281, 221, 179, 119, 89 | Cellotriose | S, R | [38] |
| Phenylpropanoids | |||||||
| 2 | 2.87 | 367 | C17H20O9 | 277, 205, 187, 157, 113 | Methyl 4-caffeoylquinate | R | [41] |
| Nitrile Glycosides | |||||||
| 5 | 2.95 | 278 | C14H17NO5 | 212, 188, 158, 116, 101 | Niazirin | L | [41] |
| Organic acids | |||||||
| 6 | 3.20 | 191 | C7H12O6 | 173, 127, 111, 85 | Quinic acid isomer 1 | R | [42] |
| 10 | 10.55 | 191 | C7H12O6 | 173, 127, 87, 85 | Quinic acid isomer 2 | L | [42] |
| Glucosinolates | |||||||
| 7 | 7.94 | 586 | C20H29NO15S2 | 440, 390, 344, 259, 198, 164, 97 | 3-Hydroxy-4-(α-l-rhamnopyranosyloxy) benzyl glucosinolate | S | [9] |
| 8 | 8.44 | 570 | C20H29NO14S2 | 490, 328, 275, 259, 241, 97, 96 | Glucomoringin | L, S, R | [9] |
| 11 | 16.90 | 408 | C14H19NO9S2 | 259, 241, 215, 212, 195, 166, 97 | Glucotropaeolin | R | [9] |
| 12 | 20.20 | 612 | C22H31NO15S2 | 370, 275, 259, 241, 106, 97 | Acetyl-4-(α-l-rhamnopyranosyloxy) benzyl glucosinolate | L | [9] |
| Phenolic acids | |||||||
| 9 | 10.44 | 353 | C16H18O9 | 191, 179, 135 | 3-Caffeoylquinic acid | L | [43] |
| Flavonoids | |||||||
| 13 | 22.03 | 609 | C27H30O16 | 301, 300, 271 | Rutin | L | standard |
| 14 | 23.65 | 463 | C21H20O12 | 301, 300, 271 | Quercetin 3-O-glucoside | L | [2] |
| 15 | 26.47 | 505 | C23H22O13 | 301, 300, 271 | Quercetin-acetyl-glycoside | L | [2] |
| 16 | 26.61 | 447 | C21H20O11 | 285, 284, 255 | Kaempferol 3-O-glucoside | L | [2] |
| 17 | 29.75 | 489 | C23H22O12 | 285, 284, 255 | Kaempferol-acetyl-glycoside | L | [2] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Y.-B.; Chen, G.-L.; Guo, M.-Q. Antioxidant and Anti-Inflammatory Activities of the Crude Extracts of Moringa oleifera from Kenya and Their Correlations with Flavonoids. Antioxidants 2019, 8, 296. https://doi.org/10.3390/antiox8080296
Xu Y-B, Chen G-L, Guo M-Q. Antioxidant and Anti-Inflammatory Activities of the Crude Extracts of Moringa oleifera from Kenya and Their Correlations with Flavonoids. Antioxidants. 2019; 8(8):296. https://doi.org/10.3390/antiox8080296
Chicago/Turabian StyleXu, Yong-Bing, Gui-Lin Chen, and Ming-Quan Guo. 2019. "Antioxidant and Anti-Inflammatory Activities of the Crude Extracts of Moringa oleifera from Kenya and Their Correlations with Flavonoids" Antioxidants 8, no. 8: 296. https://doi.org/10.3390/antiox8080296
APA StyleXu, Y.-B., Chen, G.-L., & Guo, M.-Q. (2019). Antioxidant and Anti-Inflammatory Activities of the Crude Extracts of Moringa oleifera from Kenya and Their Correlations with Flavonoids. Antioxidants, 8(8), 296. https://doi.org/10.3390/antiox8080296





