Optimization of Ultrasound-Assisted Extraction of Phenolic Compounds from Black Locust (Robiniae Pseudoacaciae) Flowers and Comparison with Conventional Methods
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Reagents
2.3. Extraction of Phenolic Compounds from Black Locust Flowers
2.3.1. Ultrasound-Assisted Extraction
2.3.2. Maceration
2.3.3. Soxhlet Extraction
2.4. Total Phenolic Compounds
2.5. DPPH Assay
2.6. HPLC Analysis
2.7. Morphological Analysis of Black Locust Flowers
2.8. Experimental Design
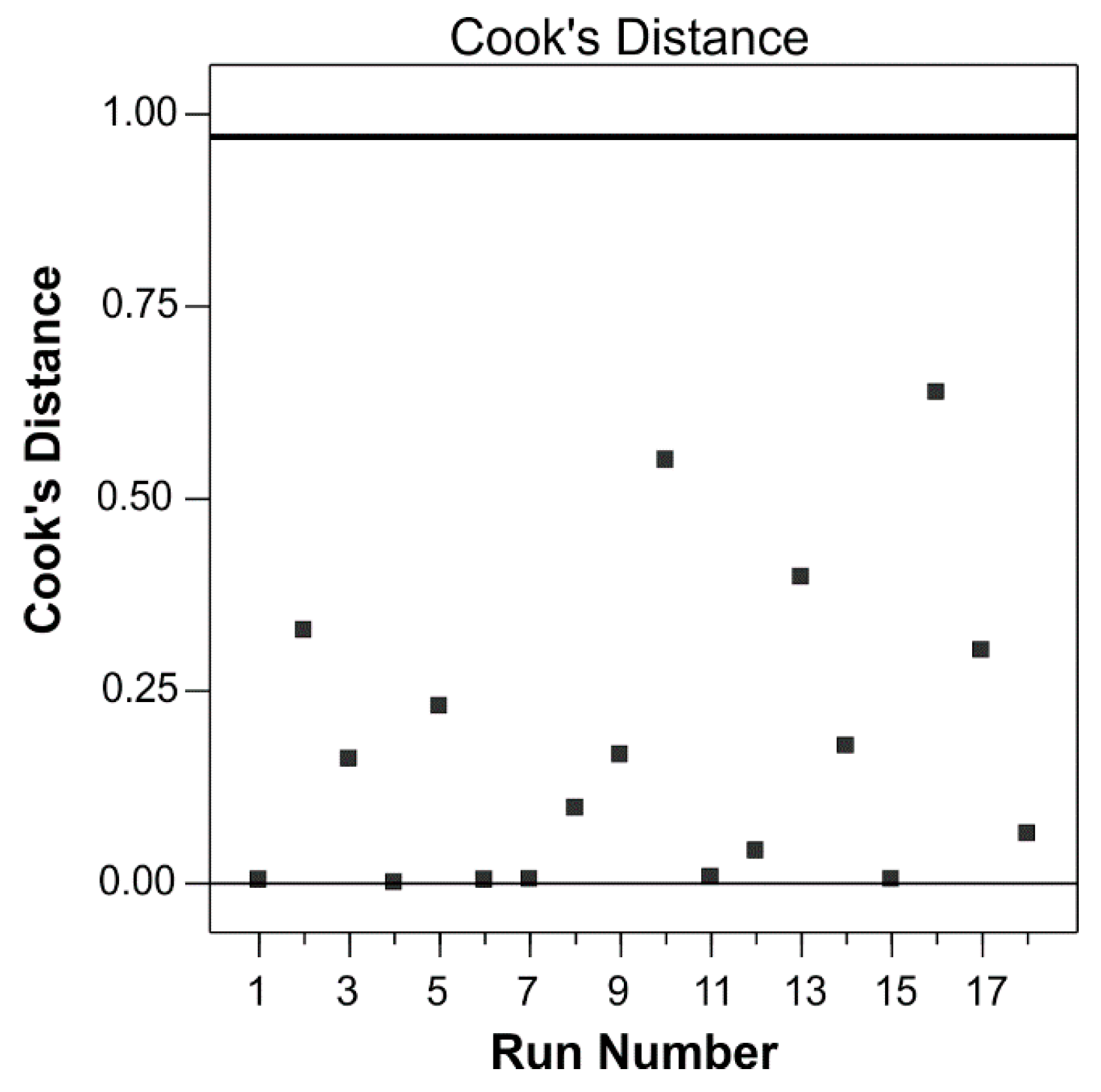
2.8.1. Statistical Analysis of the Regression Model
2.8.2. Optimization of Phenolic Compounds’ Extraction
3. Results and Discussion
3.1. Modeling of Phenolic Compounds’ Extraction Using CCD
3.2. The Impacts of Factors on the Response Surface
3.3. Optimization of the Extraction
3.4. HPLC Analysis
3.5. Comparison of Ultrasound-Assisted Extraction with Maceration and Soxhlet Extraction
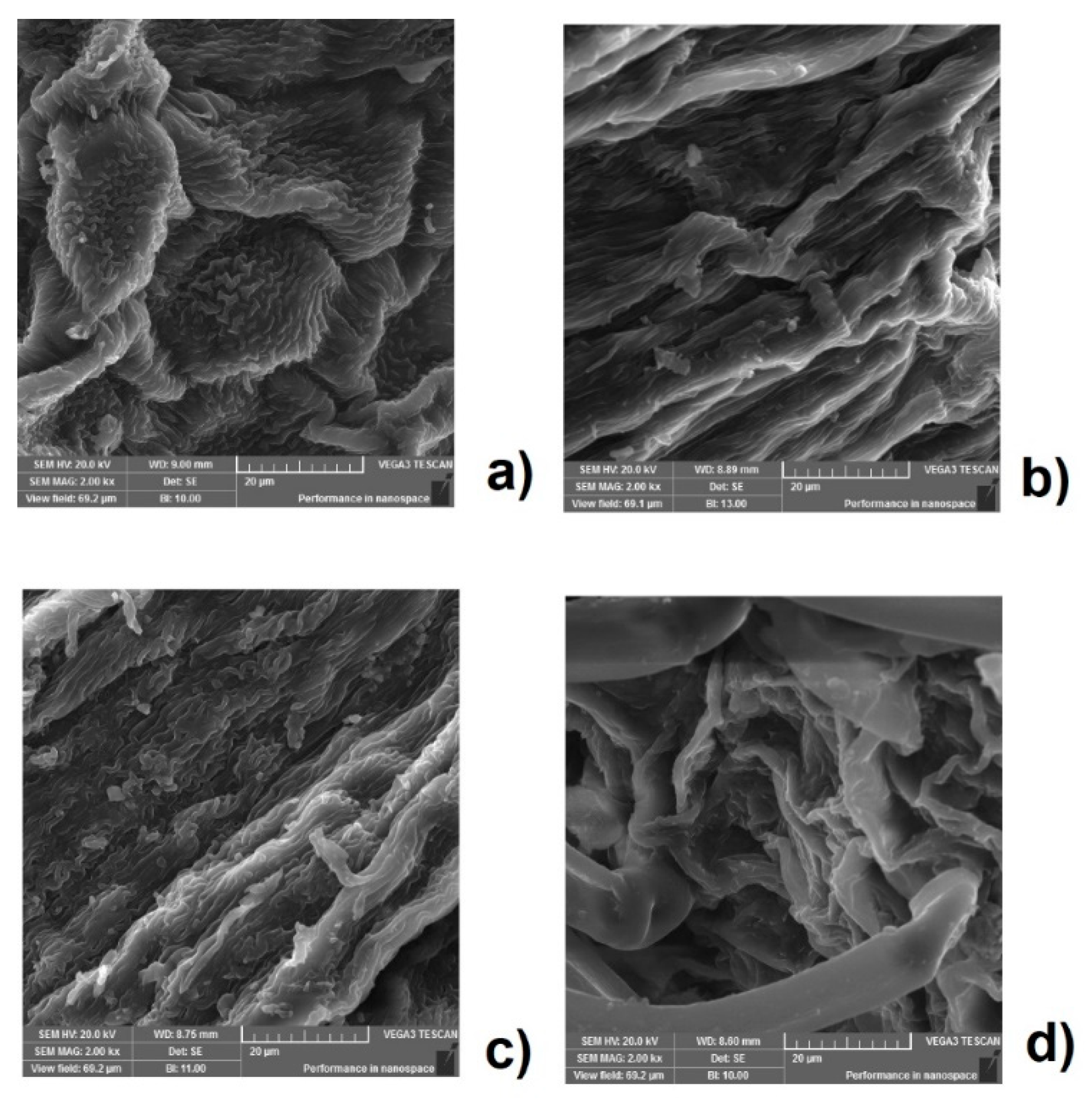
3.6. Morphological Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lavin, M.; Sousa, M. Phylogenetic systematics and biogeography of the tribe Robinieae (Leguminosae). Syst. Bot. Monogr. 1995, 45, 1–165. [Google Scholar] [CrossRef]
- Katiki, L.M.; Ferreira, J.F.; Gonzalez, J.M.; Zajac, A.M.; Lindsay, D.S.; Chagas, A.C.S.; Amarante, A.F. Anthelmintic effect of plant extracts containing condensed and hydrolyzable tannins on Caenorhabditis elegans, and their antioxidant capacity. Vet. Parasitol. 2013, 192, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Buzhdygan, O.Y.; Rudenko, S.S.; Kazanci, C.; Patten, B.C. Effect of invasive black locust (Robinia pseudoacacia L.) on nitrogen cycle in floodplain ecosystem. Ecol. Model. 2016, 319, 170–177. [Google Scholar] [CrossRef]
- Truchado, P.; Ferreres, F.; Bortolotti, L.; Sabatini, A.G.; Tomaas-Barberaan, F.A. Nectar flavonol rhamnosides are floral markers of acacia (Robinia pseudacacia) honey. J. Agric. Food Chem. 2008, 56, 8815–8824. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Y.; Tang, L.; Zhao, L.; Luan, Y.N.; Zhang, Z.Y. Determination of polyphenols in flowers of R. pseudoacacia L. by Folin-Ciocaileu method. Food Drug 2010, 12, 332–334. [Google Scholar]
- Veitch, N.C.; Elliott, P.C.; Kite, G.C.; Lewis, G.P. Flavonoid glycosides of the black locust tree, Robinia pseudoacacia (Leguminosae). Phytochemistry 2010, 71, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Sun, B.; Yu, M. Constituents of top fragrance from fresh flowers of Robinia pseudoacacia L. occurring in China. Flavour Fragr. J. 2006, 21, 798–800. [Google Scholar] [CrossRef]
- Sarikurkcu, C.; Kocak, M.S.; Tepe, B.; Uren, M.C. An alternative antioxidative and enzyme inhibitory agent from Turkey: Robinia pseudoacacia L. Ind. Crops Prod. 2015, 78, 110–115. [Google Scholar] [CrossRef]
- Tian, F.; McLaughlin, J.L. Bioactive flavonoids from the black locust tree, Robinia pseudoacacia. Pharm. Biol. 2000, 38, 229–234. [Google Scholar] [CrossRef]
- Stefova, M.; Kulevanova, S.; Stafilov, T. Assay of flavonols and quantification of quercetin in medicinal plants by HPLC with UV-diode array detection. J. Liq. Chromatogr. Relat. Technol. 2001, 24, 2283–2292. [Google Scholar] [CrossRef]
- Ji, H.F.; Du, A.L.; Zhang, L.W.; Xu, C.Y.; Yang, M.D.; Li, F.F. Effects of drying methods on antioxidant properties in Robinia pseudoacacia L. flowers. J. Med. Plants Res. 2012, 6, 3233–3239. [Google Scholar]
- Funk, D.T.; Roach, B.A. Black Locust, a Bibliography; US Deparment of Agriculture, Forest Service, Central States Forest Experiment Station: Columbus, OH, USA, 1961.
- Marinas, I.C.; Oprea, E.; Geana, E.I.; Chifiriuc, C.; Lazar, V. Antimicrobial and antioxidant activity of the vegetative and reproductive organs of Robinia pseudoacacia. J. Serb. Chem. Soc. 2014, 79, 1363–1378. [Google Scholar] [CrossRef]
- Rosu, A.F.; Bita, A.; Calina, D.; Rosu, L.; Zlatian, O.; Calina, V. Synergic antifungal and antibacterial activity of alcoholic extract of the species Robinia pseudoacacia L. (Fabaceae). Eur. J. Hosp. Pharm. Sci. Pract. 2012, 19, 216. [Google Scholar] [CrossRef]
- Bhalla, P.; Bajpai, V.K. Chemical composition and antibacterial action of Robinia pseudoacacia L. flower essential oil on membrane permeability of foodborne pathogens. J. Essent. Oil Bear. Plants 2017, 20, 632–645. [Google Scholar] [CrossRef]
- Xiang, C.G.; Xiang, N.; Huang, C.L.; Sun, H.B.; Li, W.F. Optimization of microwave extraction of flavonoids from Robinia pseudoacacia L. flowers using response surface methodology. Food Sci. 2011, 22, 006. [Google Scholar]
- He, B.; Zhang, L.L.; Yue, X.Y.; Liang, J.; Jiang, J.; Gao, X.L.; Yue, P.X. Optimization of ultrasound-assisted extraction of phenolic compounds and anthocyanins from blueberry (Vaccinium ashei) wine pomace. Food Chem. 2016, 204, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Guerra, E.; Gude, V.G.; Mondala, A.; Holmes, W.; Hernandez, R. Microwave and ultrasound enhanced extractive-transesterification of algal lipids. Appl. Energy 2014, 129, 354–363. [Google Scholar] [CrossRef]
- Mokrani, A.; Madani, K. Effect of solvent, time and temperature on the extraction of phenolic compounds and antioxidant capacity of peach (Prunus persica L.) fruit. Sep. Purif. Technol. 2016, 162, 68–76. [Google Scholar] [CrossRef]
- Bartnik, D.D.; Mohler, C.M.; Houlihan, M. Methods for the Production of Food Grade Extracts. U.S. Patent 20060088627, 27 April 2006. [Google Scholar]
- d’Alessandro, L.G.; Kriaa, K.; Nikov, I.; Dimitrov, K. Ultrasound assisted extraction of polyphenols from black chokeberry. Sep. Purif. Technol. 2012, 93, 42–47. [Google Scholar] [CrossRef]
- Drosou, C.; Kyriakopoulou, K.; Bimpilas, A.; Tsimogiannis, D.; Krokida, M. A comparative study on different extraction techniques to recover red grape pomace polyphenols from vinification byproducts. Ind. Crop. Prod. 2015, 75, 141–149. [Google Scholar] [CrossRef]
- Quiroz, J.Q.; Torres, A.C.; Ramirez, L.M.; Garcia, M.S.; Gomez, G.C.; Rojas, J. Optimization of the microwave-assisted extraction process of bioactive compounds from annatto seeds (Bixa orellana L.). Antioxidants 2019, 8, 37. [Google Scholar] [CrossRef] [PubMed]
- Le, B.; Golokhvast, K.S.; Yang, S.H.; Sun, S. Optimization of microwave-assisted extraction of polysaccharides from Ulva pertusa and evaluation of their antioxidant activity. Antioxidants 2019, 8, 129. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.Q.; Gan, R.Y.; Ge, Y.Y.; Zhang, D. Ultrasonic treatment increases extraction rate of common bean (Phaseolus vulgaris L.) antioxidants. Antioxidants 2019, 8, 83. [Google Scholar] [CrossRef] [PubMed]
- Izadiyan, P.; Hemmateenejad, B. Multi-response optimization of factors affecting ultrasonic assisted extraction from Iranian basil using central composite design. Food Chem. 2016, 190, 864–870. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Jia, J.; Wang, T.; Wang, C. Optimization of natural anthocyanin efficient extracting from purple sweet potato for silk fabric dyeing. J. Clean. Prod. 2017, 149, 673–679. [Google Scholar] [CrossRef]
- Živković, J.; Šavikin, K.; Janković, T.; Ćujić, N.; Menković, N. Optimization of ultrasound-assisted extraction of polyphenolic compounds from pomegranate peel using response surface methodology. Sep. Purif. Technol. 2018, 194, 40–47. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Molyneux, P. The use of the stable free radical diphenylpicrylhydrazyl (DPPH) for estimating antioxidant activity. Songklanakarin J. Sci. Technol. 2004, 26, 211–219. [Google Scholar]
- Xu, D.P.; Zheng, J.; Zhou, Y.; Li, Y.; Li, S.; Li, H.B. Ultrasound-assisted extraction of natural antioxidants from the flower of Limonium sinuatum: Optimization and comparison with conventional methods. Food Chem. 2017, 217, 552–559. [Google Scholar] [CrossRef]
- Chemat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef]
- Chavan, Y.; Singhal, R.S. Ultrasound-assisted extraction (UAE) of bioactives from arecanut (Areca catechu L.) and optimization study using response surface methodology. Innov. Food Sci. Emerg. Technol. 2013, 17, 106–113. [Google Scholar] [CrossRef]
- Asfaram, A.; Ghaedi, M.; Yousefi, F.; Dastkhoon, M. Experimental design and modeling of ultrasound assisted simultaneous adsorption of cationic dyes onto ZnS: Mn-NPs-AC from binary mixture. Ultrason. Sonochem. 2016, 33, 77–89. [Google Scholar] [CrossRef] [PubMed]
- Maran, J.P.; Manikandan, S.; Nivetha, C.V.; Dinesh, R. Ultrasound assisted extraction of bioactive compounds from Nephelium lappaceum L. fruit peel using central composite face centered response surface design. Arab. J. Chem. 2017, 10, S1145–S1157. [Google Scholar] [CrossRef]
- Khan, M.K.; Abert-Vian, M.; Fabiano-Tixier, A.S.; Dangles, O.; Chemat, F. Ultrasound-assisted extraction of polyphenols (flavanone glycosides) from orange (Citrus sinensis L.) peel. Food Chem. 2010, 119, 851–858. [Google Scholar] [CrossRef]
- Zoumpoulakis, P.; Sinanoglou, V.; Siapi, E.; Heropoulos, G.; Proestos, C. Evaluating modern techniques for the extraction and characterisation of sunflower (Hellianthus annus L.) seeds phenolics. Antioxidants 2017, 6, 46. [Google Scholar] [CrossRef] [PubMed]
- Vajic, U.J.; Grujic-Milanovic, J.; Zivkovic, J.; Savikin, K.; Godevac, D.; Miloradovic, Z.; Bugarski, B.; Mihajlovic-Stanojevic, N. Optimization of extraction of stinging nettle leaf phenolic compounds using response surface methodology. Ind. Crop. Prod. 2015, 74, 912–917. [Google Scholar] [CrossRef]
- Ali, A.; Lim, X.Y.; Chong, C.H.; Mah, S.H.; Chua, B.L. Optimization of ultrasound-assisted extraction of natural antioxidants from Piper betle using response surface methodology. LWT 2018, 89, 681–688. [Google Scholar] [CrossRef]
- Jovanović, A.A.; Đorđević, V.B.; Zdunić, G.M.; Pljevljakušić, D.S.; Šavikin, K.P.; Gođevac, D.M.; Bugarski, B.M. Optimization of the extraction process of polyphenols from Thymus serpyllum L. herb using maceration, heat-and ultrasound-assisted techniques. Sep. Purif. Technol. 2017, 179, 369–380. [Google Scholar] [CrossRef]


| Extraction Technique | Solvent | Temperature (°C) | Extraction Time (min) | Optimization of Factors | Ref. |
|---|---|---|---|---|---|
| Magnetic stirrer | Ethanol | Room | 720 | No | [11] |
| Reflux | Acetone | - | 30 | No | [10] |
| Maceration | Methanol | Room | - | No | [6] |
| Maceration | Water | 100 | 15 | No | [8] |
| Soxhlet | Ethyl acetate, acetone, methanol | - | 300 | No | [8] |
| Soxhlet | Ethanol | - | - | No | [14] |
| Microwave | Ethanol | - | 2 | Box–Behnken design | [16] |
| Factors | Coded | Levels | ||||
|---|---|---|---|---|---|---|
| −1.68 | −1 | 0 | +1 | +1.68 | ||
| ethanol concentration [%] | x1 | 33 | 40 | 50 | 60 | 67 |
| extraction temperature [°C] | x2 | 33 | 40 | 50 | 60 | 67 |
| extraction time [min] | x3 | 17 | 20 | 25 | 30 | 33 |
| Standard Order | Ru Order | X1, Ethanol Concentration [%] | X2, Extraction Temperature [°C] | X3, Extraction Time [min] | Y, Total Phenolic Content [gGAE 100 g−1 d.p.m.] | |
|---|---|---|---|---|---|---|
| Experimental | Predicted | |||||
| 11 | 1 | 50 (0) | 33 (−1.68) | 25 (0) | 2.71 | 2.72 |
| 8 | 2 | 60 (+1) | 60 (+1) | 30 (+1) | 3.15 | 3.19 |
| 9 | 3 | 33 (−1.68) | 50 (0) | 25 (0) | 2.65 | 2.73 |
| 10 | 4 | 67 (+1.68) | 50 (0) | 25 (0) | 3.02 | 3.02 |
| 5 | 5 | 40 (−1) | 40 (−1) | 30 (+1) | 2.58 | 2.55 |
| 17 | 6 | 50 (0) | 50 (0) | 25 (0) | 2.85 | 2.88 |
| 16 | 7 | 50 (0) | 50 (0) | 25 (0) | 2.91 | 2.88 |
| 3 | 8 | 40 (−1) | 60 (+1) | 20 (−1) | 2.71 | 2.69 |
| 12 | 9 | 50 (0) | 67 (+1.68) | 25 (0) | 3.12 | 3.04 |
| 7 | 10 | 40 (−1) | 60 (+1) | 30 (+1) | 3.11 | 3.16 |
| 18 | 11 | 50 (0) | 50 (0) | 25 (0) | 2.92 | 2.88 |
| 13 | 12 | 50 (0) | 50 (0) | 17 (−1.68) | 2.33 | 2.35 |
| 6 | 13 | 60 (+1) | 40 (−1) | 30 (+1) | 3.00 | 3.04 |
| 2 | 14 | 60 (+1) | 40 (−1) | 20 (−1) | 2.81 | 2.78 |
| 15 | 15 | 50 (0) | 50 (0) | 25 (0) | 2.91 | 2.88 |
| 4 | 16 | 60 (+1) | 60 (+1) | 20 (−1) | 2.48 | 2.54 |
| 14 | 17 | 50 (0) | 50 (0) | 33 (+1.68) | 3.02 | 2.97 |
| 1 | 18 | 40 (−1) | 40 (−1) | 20 (−1) | 2.48 | 2.46 |
| Source | SS | df | MS | F-Value | p-Value Prob > F |
|---|---|---|---|---|---|
| Model | 0.9795 | 9 | 0.1088 | 34.64 | <0.0001 * |
| X1 | 0.1023 | 1 | 0.1023 | 32.57 | 0.0005 * |
| X2 | 0.1180 | 1 | 0.1180 | 37.56 | 0.0003 * |
| X3 | 0.4652 | 1 | 0.4652 | 148.04 | <0.0001 * |
| X1X2 | 0.1105 | 1 | 0.1105 | 35.15 | 0.0004 * |
| X1X3 | 0.0162 | 1 | 0.0162 | 5.16 | 0.0528 ** |
| X2X3 | 0.0761 | 1 | 0.0761 | 24.20 | 0.0012 * |
| X12 | 0.0078 | 1 | 0.0078 | 2.50 | 0.1529 ** |
| X22 | 0.0001 | 1 | 0.0001 | 0.05 | 0.8351 ** |
| X32 | 0.0839 | 1 | 0.0839 | 26.72 | 0.0009 * |
| Residual | 0.0251 | 8 | 0.0031 | ||
| Lack-of-fit | 0.0221 | 5 | 0.0044 | 4.30 | 0.1296 ** |
| Pure error | 0.0031 | 3 | 0.0010 | ||
| Corrected total | 1.0046 | 17 | |||
| Std. Dev. | 0.06 | R2 | 0.975 | ||
| Mean | 2.82 | Adj-R2 | 0.947 | ||
| C.V. % | 1.99 | Pred-R2 | 0.821 | ||
| PRESS | 0.18 | Adequate precision | 19.8 | ||
| Coefficient | Parameter Estimate | Standard Error |
|---|---|---|
| Intercept | 2.898 | 0.028 |
| X1 | 0.087 | 0.015 |
| X2 | 0.093 | 0.015 |
| X3 | 0.185 | 0.015 |
| X1X2 | −0.118 | 0.020 |
| X1X3 | 0.045 | 0.020 |
| X2X3 | 0.098 | 0.020 |
| X12 | −0.025 | 0.016 |
| X22 | 0.003 | 0.016 |
| X32 | −0.082 | 0.016 |
| Extraction Technique | Extraction Temperature [°C] | Ethanol Concentration [%] | Extraction Time [h] | TPC [gGAE 100 g−1 d.p.m.] | IC50 [µg cm−3] |
|---|---|---|---|---|---|
| UAE | 59 | 60 | 0.5 | 3.12 | 120.5 |
| Maceration | 25 | 60 | 24 | 2.54 | 150.6 |
| Soxhlet Extraction | 90 | 60 | 6 | 3.22 | 204.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savic Gajic, I.; Savic, I.; Boskov, I.; Žerajić, S.; Markovic, I.; Gajic, D. Optimization of Ultrasound-Assisted Extraction of Phenolic Compounds from Black Locust (Robiniae Pseudoacaciae) Flowers and Comparison with Conventional Methods. Antioxidants 2019, 8, 248. https://doi.org/10.3390/antiox8080248
Savic Gajic I, Savic I, Boskov I, Žerajić S, Markovic I, Gajic D. Optimization of Ultrasound-Assisted Extraction of Phenolic Compounds from Black Locust (Robiniae Pseudoacaciae) Flowers and Comparison with Conventional Methods. Antioxidants. 2019; 8(8):248. https://doi.org/10.3390/antiox8080248
Chicago/Turabian StyleSavic Gajic, Ivana, Ivan Savic, Ivana Boskov, Stanko Žerajić, Ivana Markovic, and Dragoljub Gajic. 2019. "Optimization of Ultrasound-Assisted Extraction of Phenolic Compounds from Black Locust (Robiniae Pseudoacaciae) Flowers and Comparison with Conventional Methods" Antioxidants 8, no. 8: 248. https://doi.org/10.3390/antiox8080248
APA StyleSavic Gajic, I., Savic, I., Boskov, I., Žerajić, S., Markovic, I., & Gajic, D. (2019). Optimization of Ultrasound-Assisted Extraction of Phenolic Compounds from Black Locust (Robiniae Pseudoacaciae) Flowers and Comparison with Conventional Methods. Antioxidants, 8(8), 248. https://doi.org/10.3390/antiox8080248







