Saffron: A Multitask Neuroprotective Agent for Retinal Degenerative Diseases
Abstract
1. Introduction
2. Materials and Methods
- -
- Healthy control (n = 10) that received neither retinal damage, nor treatment;
- -
- Light damaged no recovery group (n = 10) that received retinal damage but no treatment and were sacrificed immediately after light damage to perform SDS-page, western blotting, biochemical assay and MMP-3 immunostaining experiments;
- -
- Light damaged with recovery (n = 10) that received retinal damage but no treatment and were sacrificed 7 days after the exposure to the light damage;
- -
- Saffron treated + Light damaged no recovery group (n = 10) that received retinal damage and saffron treatment and were sacrificed immediately after light damage to perform SDS-page, western blotting, biochemical assay and MMP3 immunostaining experiments;
- -
- Saffron treated + Light damaged with recovery (n = 10) that received retinal damage and saffron treatment and were sacrificed 7 days after the exposure to the light damage;
3. Results
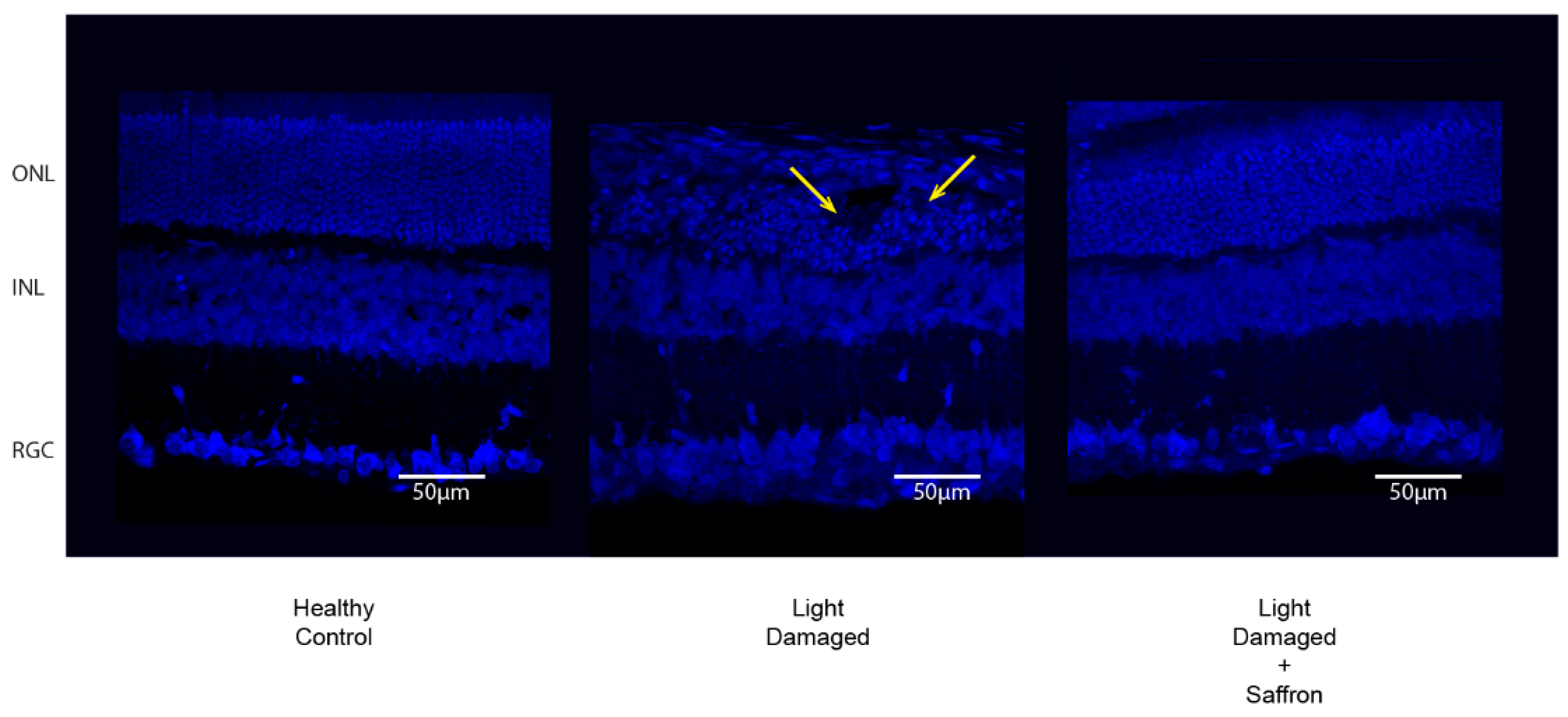
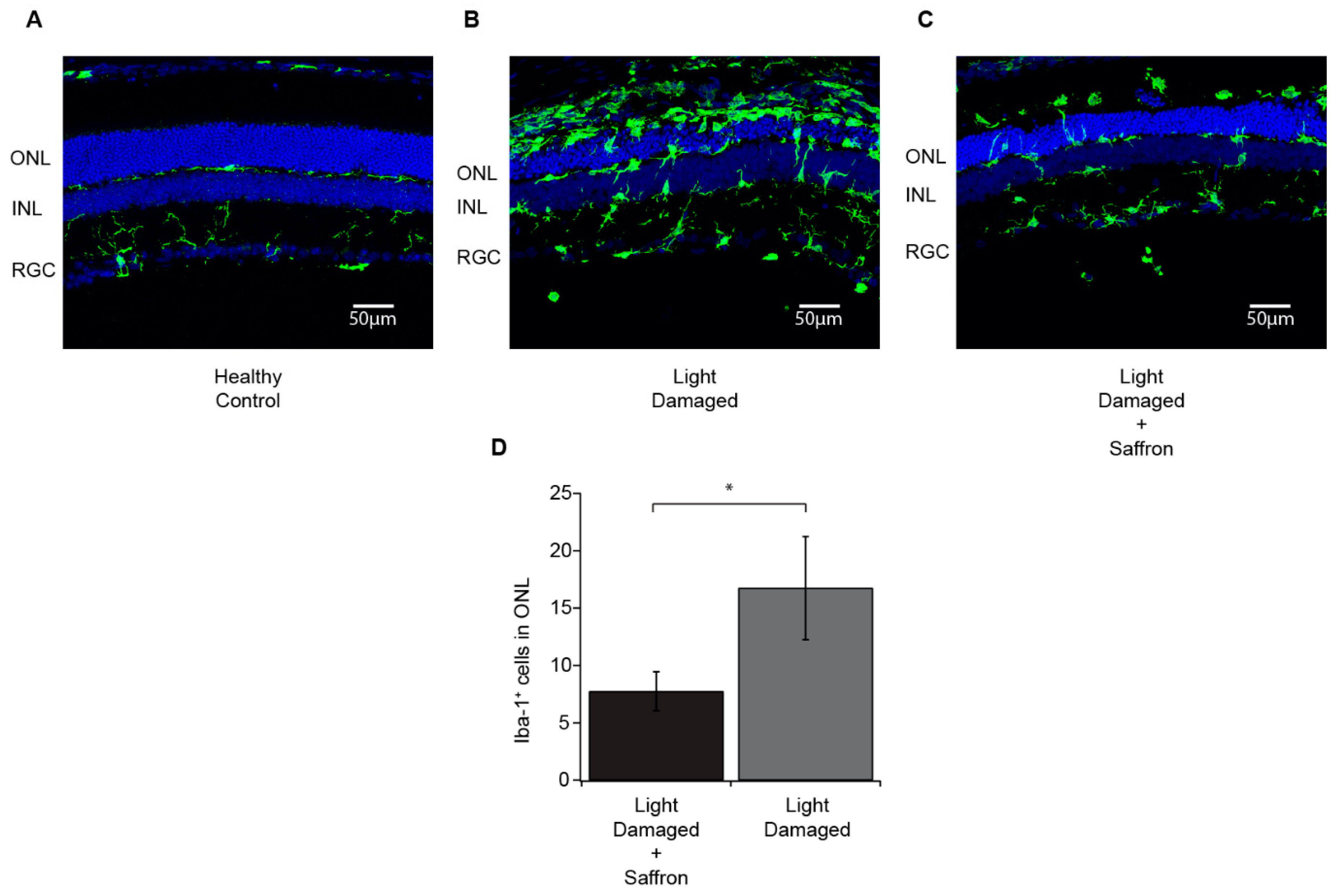
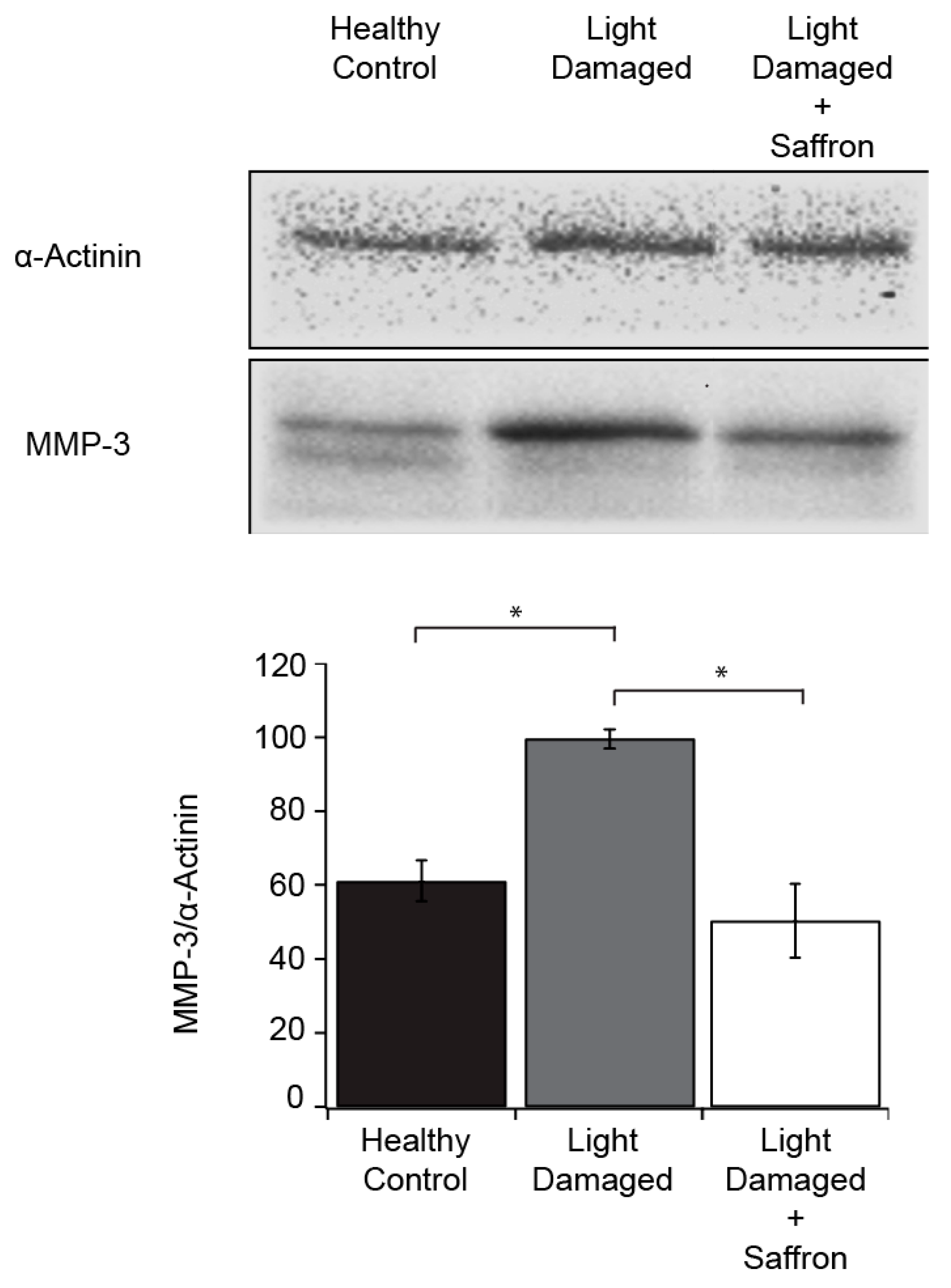
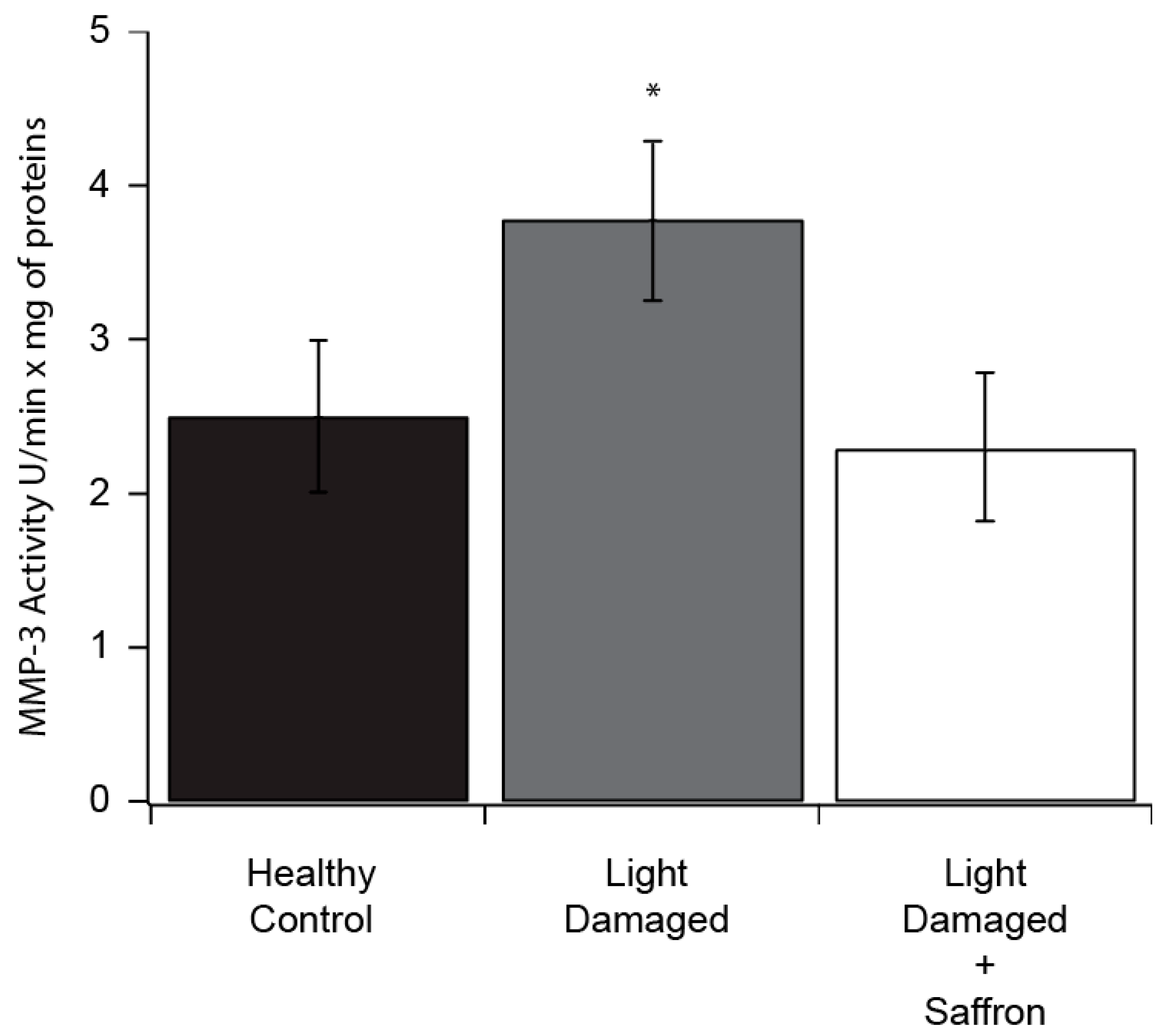
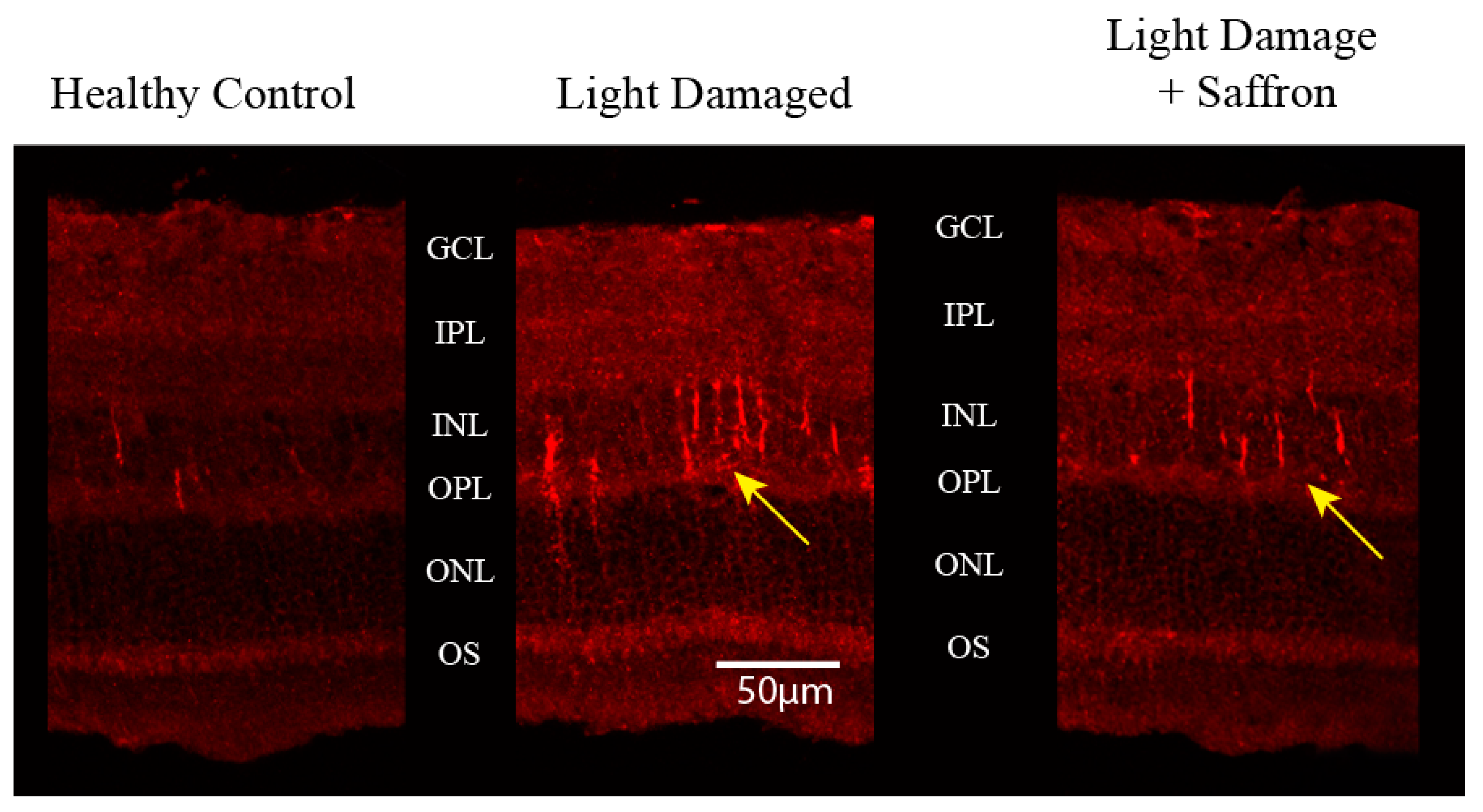
3.1. Light Induced Retinal Degeneration
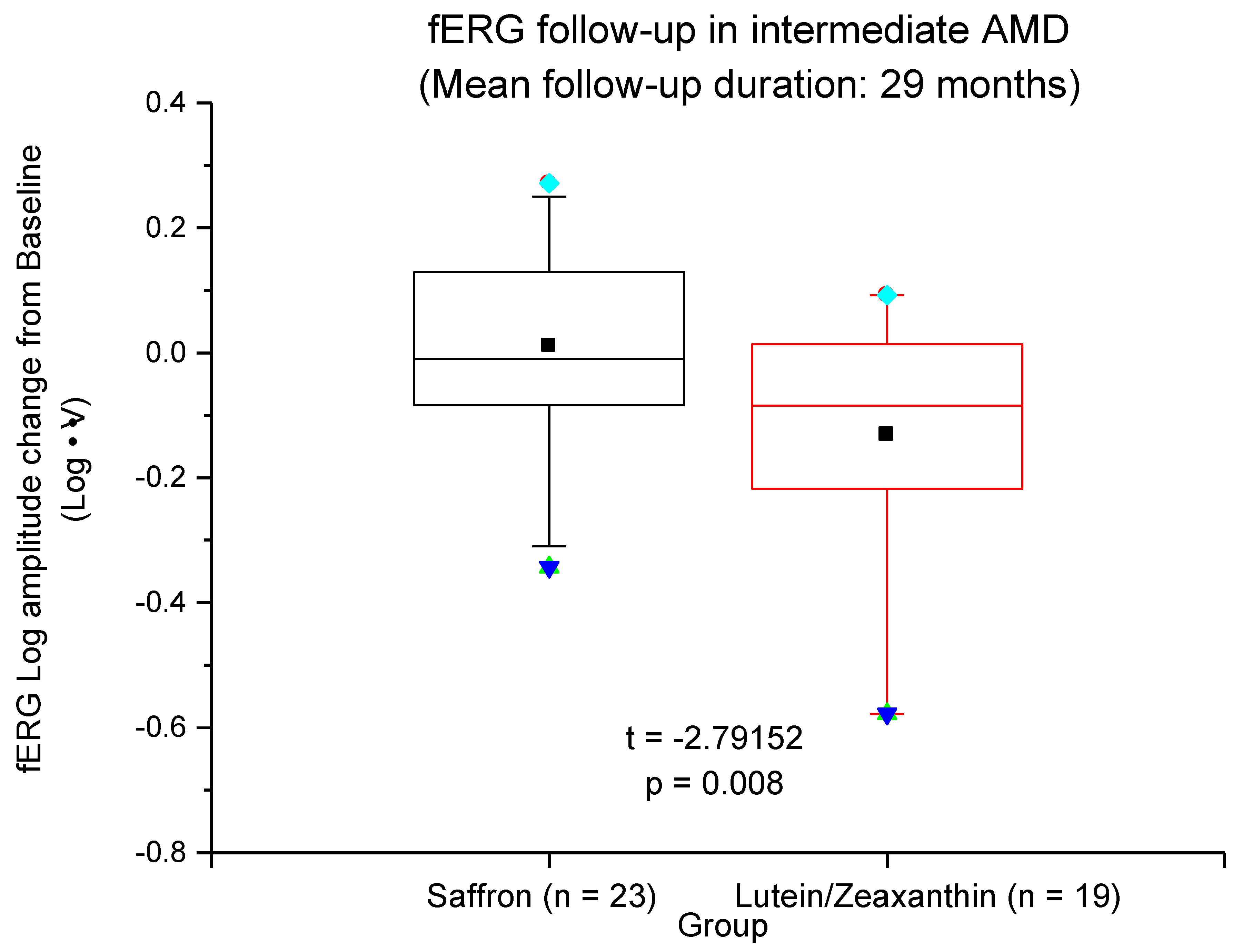
3.2. Long-Term Treatment in AMD Patients (AREDS and Saffron)
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ROS | reactive Oxygen Species |
| MMP-3 | matrix metalloproteinase-3 |
| Mt2A | metallothionein 2A |
| TIMP-1 | TIMP metalloproteinase inhibitor 1 |
| AMD | age related macular degeneration |
| RPE | retinal pigmented epithelium |
| AREDS | Age-Related Eye Disease Study |
| fERG | focal electroretinogram |
| ONL | outer nuclear layer |
| LD | light damaged |
| LMW | Low molecular weight |
References
- Rohowetz, L.; Kraus, J.; Koulen, P. Reactive Oxygen Species-Mediated Damage of Retinal Neurons: Drug Development Targets for Therapies of Chronic Neurodegeneration of the Retina. Int. J. Mol. Sci. 2018, 19, 3362. [Google Scholar] [CrossRef] [PubMed]
- Moreno, J.A.; Gomez-Guerrero, C.; Mas, S.; Sanz, A.B.; Lorenzo, O.; Ruiz-Ortega, M.; Opazo, L.; Mezzano, S.; Egido, J. Targeting inflammation in diabetic nephropathy: A tale of hope. Expert Opin. Investig. Drugs 2018, 27, 917–930. [Google Scholar] [CrossRef] [PubMed]
- Léveillard, T.; Philp, N.J.; Sennlaub, F. Is Retinal Metabolic Dysfunction at the Center of the Pathogenesis of Age-related Macular Degeneration? Int. J. Mol. Sci. 2019, 20, 762. [Google Scholar] [CrossRef] [PubMed]
- Datta, S.; Cano, M.; Ebrahimi, K.; Wang, L.; Handa, J.T. The impact of oxidative stress and inflammation on RPE degeneration in non-neovascular AMD. Prog. Retin. Eye Res. 2017, 60, 201–218. [Google Scholar] [CrossRef] [PubMed]
- Rashid, K.; Wolf, A.; Langmann, T. Microglia Activation and Immunomodulatory Therapies for Retinal Degenerations. Front. Cell. Neurosci. 2018, 12, 176. [Google Scholar] [CrossRef] [PubMed]
- Bringmann, A.; Pannicke, T.; Grosche, J.; Francke, M.; Wiedemann, P.; Skatchkov, S.N.; Osborne, N.N.; Reichenbach, A. Müller cells in the healthy and diseased retina. Prog. Retin. Eye Res. 2006, 25, 397–424. [Google Scholar] [CrossRef] [PubMed]
- Vecino, E.; Rodriguez, F.D.D.; Ruzafa, N.; Pereiro, X.; Sharma, S.C. Glia-neuron interactions in the mammalian retina. Prog. Retin. Eye Res. 2016, 51, 1–40. [Google Scholar] [CrossRef]
- Li, Q.; Barres, B.A. Microglia and macrophages in brain homeostasis and disease. Nat. Rev. Immunol. 2018, 18, 225–242. [Google Scholar] [CrossRef]
- Guillot-Sestier, M.-V.; Town, T. Let’s make microglia great again in neurodegenerative disorders. J. Neural Transm. 2018, 125, 751–770. [Google Scholar] [CrossRef]
- Di Marco, F.; Di Paolo, M.; Romeo, S.; Colecchi, L.; Fiorani, L.; Spana, S.; Stone, J.; Bisti, S. Combining neuroprotectants in a model of retinal degeneration: No additive benefit. PLoS ONE 2014, 9, e100389. [Google Scholar] [CrossRef]
- Marco, F.D.; Romeo, S.; Nandasena, C.; Purushothuman, S.; Adams, C.; Bisti, S.; Stone, J. The time course of action of two neuroprotectants, dietary saffron and photobiomodulation, assessed in the rat retina. Am. J. Neurodegener. Dis. 2013, 2, 208–220. [Google Scholar] [PubMed]
- Fiorani, L.; Passacantando, M.; Santucci, S.; Di Marco, S.; Bisti, S.; Maccarone, R. Cerium Oxide Nanoparticles Reduce Microglial Activation and Neurodegenerative Events in Light Damaged Retina. PLoS ONE 2015, 10, e0140387. [Google Scholar] [CrossRef] [PubMed]
- Maccarone, R.; Di Marco, S.; Bisti, S. Saffron supplement maintains morphology and function after exposure to damaging light in mammalian retina. Invest. Ophthalmol. Vis. Sci. 2008, 49, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Rutar, M.; Provis, J.M.; Valter, K. Brief exposure to damaging light causes focal recruitment of macrophages, and long-term destabilization of photoreceptors in the albino rat retina. Curr. Eye Res. 2010, 35, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Organisciak, D.T.; Vaughan, D.K. Retinal light damage: Mechanisms and protection. Prog. Retin. Eye Res. 2010, 29, 113–134. [Google Scholar] [CrossRef]
- Demontis, G.C.; Longoni, B.; Marchiafava, P.L. Molecular steps involved in light-induced oxidative damage to retinal rods. Invest. Ophthalmol. Vis. Sci. 2002, 43, 2421–2427. [Google Scholar] [PubMed]
- Karlstetter, M.; Scholz, R.; Rutar, M.; Wong, W.T.; Provis, J.M.; Langmann, T. Retinal microglia: Just bystander or target for therapy? Prog. Retin. Eye Res. 2015, 45, 30–57. [Google Scholar] [CrossRef]
- Natoli, R.; Zhu, Y.; Valter, K.; Bisti, S.; Eells, J.; Stone, J. Gene and noncoding RNA regulation underlying photoreceptor protection: Microarray study of dietary antioxidant saffron and photobiomodulation in rat retina. Mol. Vis. 2010, 16, 1801–1822. [Google Scholar]
- Maeda, A.; Maeda, T.; Golczak, M.; Chou, S.; Desai, A.; Hoppel, C.L.; Matsuyama, S.; Palczewski, K. Involvement of all-trans-retinal in acute light-induced retinopathy of mice. J. Biol. Chem. 2009, 284, 15173–15183. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study Research Group. The Age-Related Eye Disease Study (AREDS): Design implications. AREDS report no. 1. Control. Clin. Trials 1999, 20, 573–600. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study 2 (AREDS2) Research Group; Chew, E.Y.; SanGiovanni, J.P.; Ferris, F.L.; Wong, W.T.; Agron, E.; Clemons, T.E.; Sperduto, R.; Danis, R.; Chandra, S.R.; et al. Lutein/zeaxanthin for the treatment of age-related cataract: AREDS2 randomized trial report no. 4. JAMA Ophthalmol. 2013, 131, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Chew, E.Y.; Clemons, T.; SanGiovanni, J.P.; Danis, R.; Domalpally, A.; McBee, W.; Sperduto, R.; Ferris, F.L.; Ferris, F.L. The Age-related Eye Disease Study 2 (AREDS2). Ophthalmology 2012, 119, 2282–2289. [Google Scholar] [CrossRef] [PubMed]
- Falsini, B.; Piccardi, M.; Minnella, A.; Savastano, C.; Capoluongo, E.; Fadda, A.; Balestrazzi, E.; Maccarone, R.; Bisti, S. Influence of Saffron Supplementation on Retinal Flicker Sensitivity in Early Age-Related Macular Degeneration. Invest. Ophthalmol. Vis. Sci. 2010, 51, 6118–6124. [Google Scholar] [CrossRef] [PubMed]
- Piccardi, M.; Marangoni, D.; Minnella, A.M.; Savastano, M.C.; Valentini, P.; Ambrosio, L.; Capoluongo, E.; Maccarone, R.; Bisti, S.; Falsini, B. A Longitudinal Follow-Up Study of Saffron Supplementation in Early Age-Related Macular Degeneration: Sustained Benefits to Central Retinal Function. Evidence-Based Complement. Altern. Med. 2012, 2012, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, D.; Falsini, B.; Piccardi, M.; Ambrosio, L.; Minnella, A.M.; Savastano, M.C.; Bisti, S.; Maccarone, R.; Fadda, A.; Mello, E.; et al. Functional effect of Saffron supplementation and risk genotypes in early age-related macular degeneration: A preliminary report. J. Transl. Med. 2013, 11, 228. [Google Scholar] [CrossRef] [PubMed]
- Bisti, S.; Maccarone, R.; Falsini, B. Saffron and retina: Neuroprotection and pharmacokinetics. Vis. Neurosci. 2014, 31, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Riazi, A.; Panahi, Y.; Alishiri, A.A.; Hosseini, M.A.; Karimi Zarchi, A.A.; Sahebkar, A. The impact of saffron (Crocus sativus) supplementation on visual function in patients with dry age-related macular degeneration. Ital. J. Med. 2016, 10, 196–201. [Google Scholar] [CrossRef]
- Broadhead, G.K.; Grigg, J.R.; McCluskey, P.; Hong, T.; Schlub, T.E.; Chang, A.A. Saffron therapy for the treatment of mild/moderate age-related macular degeneration: A randomised clinical trial. Graefe’s Arch. Clin. Exp. Ophthalmol. 2019, 257, 31–40. [Google Scholar] [CrossRef]
- Corso, L.; Cavallero, A.; Baroni, D.; Garbati, P.; Prestipino, G.; Bisti, S.; Nobile, M.; Picco, C. Saffron reduces ATP-induced retinal cytotoxicity by targeting P2X7 receptors. Purinergic Signal. 2016, 12, 161–174. [Google Scholar] [CrossRef]
- Maccarone, R.; Rapino, C.; Zerti, D.; di Tommaso, M.; Battista, N.; Di Marco, S.; Bisti, S.; Maccarrone, M. Modulation of Type-1 and Type-2 Cannabinoid Receptors by Saffron in a Rat Model of Retinal Neurodegeneration. PLoS ONE 2016, 11, e0166827. [Google Scholar] [CrossRef]
- Stone, J.; Mitrofanis, J.; Johnstone, D.M.; Falsini, B.; Bisti, S.; Adam, P.; Nuevo, A.B.; George-Weinstein, M.; Mason, R.; Eells, J. Acquired Resilience: An Evolved System of Tissue Protection in Mammals. Dose-Response 2018, 16, 1559325818803428. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Chew, E.Y.; Clemons, T.E.; SanGiovanni, J.P.; Danis, R.P.; Ferris, F.L.; Elman, M.J.; Antoszyk, A.N.; Ruby, A.J.; Orth, D.; Bressler, S.B.; et al. Secondary Analyses of the Effects of Lutein/Zeaxanthin on Age-Related Macular Degeneration Progression. JAMA Ophthalmol. 2014, 132, 142. [Google Scholar] [CrossRef] [PubMed]
- Karlstetter, M.; Ebert, S.; Langmann, T. Microglia in the healthy and degenerating retina: Insights from novel mouse models. Immunobiology 2010, 215, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Liutkeviciene, R.; Liutkevicius, V.; Giedraitiene, A.; Kriauciuniene, L.; Asmoniene, V. Influence of Matrix Metalloproteinases MMP-2, -3 and on Age- Related Macular Degeneration Development. In The Role of Matrix Metalloproteinase in Human Body Pathologies; Liutkevicius, V., Ed.; InTech: Rijeka, Croatia, 2017; Chapter 4; ISBN 978-953-51-3718-4. [Google Scholar]
- Karimi, E.; Oskoueian, E.; Hendra, R.; Jaafar, H.Z.E.E. Evaluation of Crocus sativus L. Stigma Phenolic and Flavonoid Compounds and Its Antioxidant Activity. Molecules 2010, 15, 6244–6256. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Marco, S.; Carnicelli, V.; Franceschini, N.; Di Paolo, M.; Piccardi, M.; Bisti, S.; Falsini, B. Saffron: A Multitask Neuroprotective Agent for Retinal Degenerative Diseases. Antioxidants 2019, 8, 224. https://doi.org/10.3390/antiox8070224
Di Marco S, Carnicelli V, Franceschini N, Di Paolo M, Piccardi M, Bisti S, Falsini B. Saffron: A Multitask Neuroprotective Agent for Retinal Degenerative Diseases. Antioxidants. 2019; 8(7):224. https://doi.org/10.3390/antiox8070224
Chicago/Turabian StyleDi Marco, Stefano, Veronica Carnicelli, Nicola Franceschini, Mattia Di Paolo, Marco Piccardi, Silvia Bisti, and Benedetto Falsini. 2019. "Saffron: A Multitask Neuroprotective Agent for Retinal Degenerative Diseases" Antioxidants 8, no. 7: 224. https://doi.org/10.3390/antiox8070224
APA StyleDi Marco, S., Carnicelli, V., Franceschini, N., Di Paolo, M., Piccardi, M., Bisti, S., & Falsini, B. (2019). Saffron: A Multitask Neuroprotective Agent for Retinal Degenerative Diseases. Antioxidants, 8(7), 224. https://doi.org/10.3390/antiox8070224





