Is There A Role for Abscisic Acid, A Proven Anti-Inflammatory Agent, in the Treatment of Ischemic Retinopathies?
Abstract
1. Ischemic Retinopathies (IRs)
1.1. Possible Therapies for IRs
1.2. Phytohormones are Cross-Kingdom Molecules
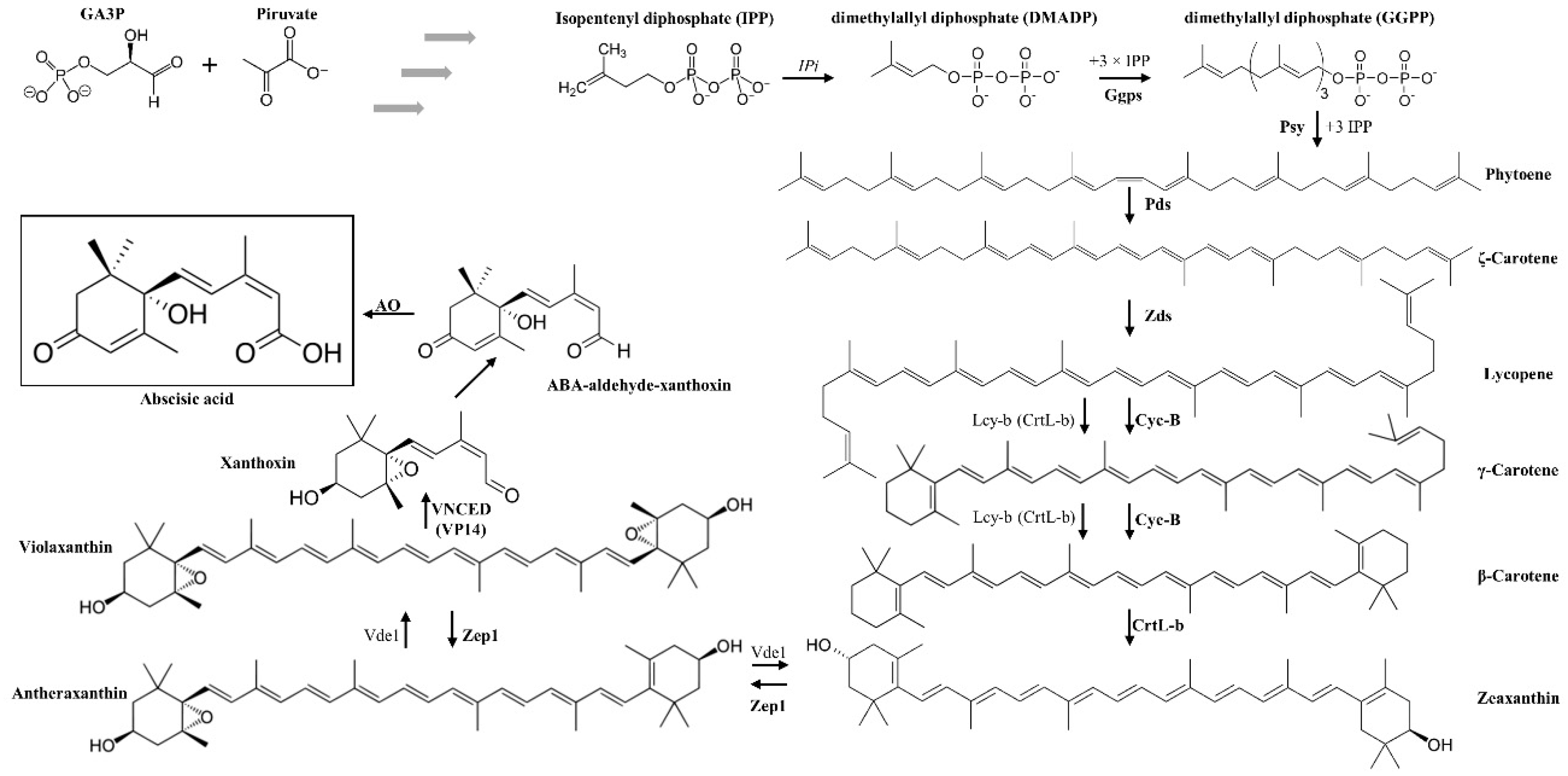
2. Abscisic Acid (ABA)
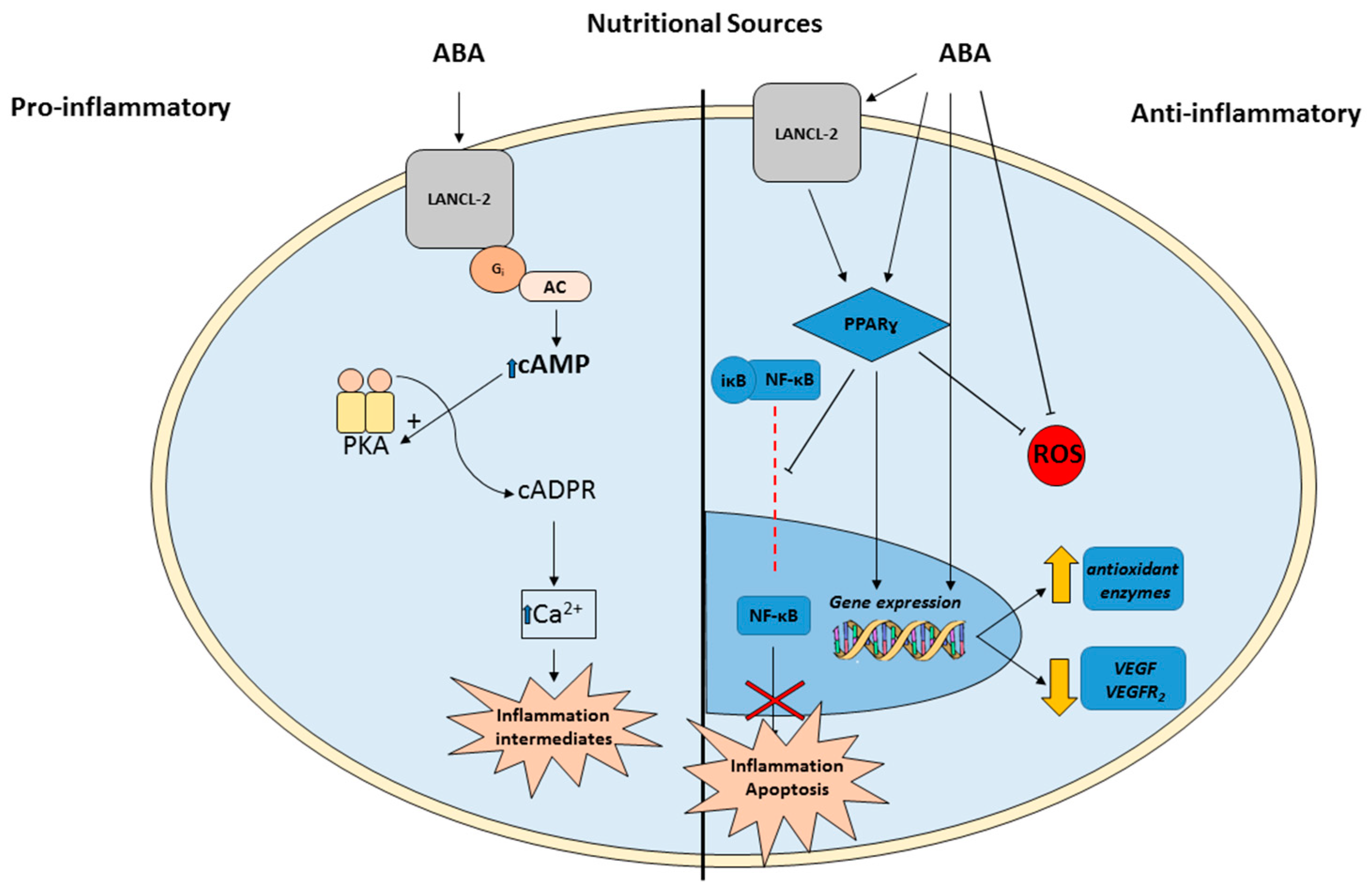
3. ABA Actions in Animals
4. ABA Presence in Food and Its Relationship with Lutein and Zeaxanthin
5. ABA as a Novel Therapy for IRs
6. Conclusions
Funding
Conflicts of Interest
References
- Kolb, H. How the retina works. Am. Sci. 2003, 91, 28–35. [Google Scholar] [CrossRef]
- Rivera, J.C.; Dabouz, R.; Noueihed, B.; Omri, S.; Tahiri, H.; Chemtob, S. Ischemic Retinopathies: Oxidative Stress and Inflammation. Oxid Med. Cell Longev. 2017, 2017, 3940241. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, E.; Mammadzada, P.; André, H. The Different Facades of Retinal and Choroidal endothelial Cells in Response to Hypoxia. Int. J. Mol. Sci. 2018, 19, 3846. [Google Scholar] [CrossRef] [PubMed]
- Moran, E.P.; Wang, Z.; Chen, J.; Sapieha, P.; Smith, L.E.; Ma, J.X. Neurovascular cross talk in diabetic retinopathy: Pathophysiological roles and therapeutic implications. Am. J. Physiol. Heart Circ. Physiol. 2016, 311, H738–H749. [Google Scholar] [CrossRef]
- Neely, K.A.; Gardner, T.W. Ocular neovascularization: Clarifying complex interactions. Am. J. Pathol. 1998, 153, 665–670. [Google Scholar] [CrossRef]
- Santiago, A.R.; Boia, R.; Aires, I.D.; Ambrósio, A.F.; Fernandes, R. Sweet Stress: Coping with Vascular Dysfunction in Diabetic Retinopathy. Front. Physiol. 2018, 13, 820. [Google Scholar] [CrossRef]
- Wu, L.; Fernandez-Loaiza, P.; Sauma, J.; Hernandez-Bogantes, E.; Masis, M. Classification of diabetic retinopathy and diabetic macular edema. World J. Diabetes 2013, 4, 290–294. [Google Scholar] [CrossRef]
- Stitt, A.W.; Curtis, T.M.; Chen, M.; Medina, R.J.; McKay, G.J.; Jenkins, A.; Gardiner, T.A.; Lyons, T.J.; Hammes, H.P.; Simó, R.; et al. The progress in understanding and treatment of diabetic retinopathy. Prog. Retin. Eye Res. 2016, 51, 156–186. [Google Scholar] [CrossRef] [PubMed]
- Stratton, I.M.; Kohner, E.M.; Aldington, S.J.; Turner, R.C.; Holman, R.R.; Manley, S.E.; Matthews, D.R. UKPDS 50: Risk factors for incidence and progression of retinopathy in Type II diabetes over 6 years from diagnosis. Diabetologia 2001, 44, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Chew, E.Y.; Davis, M.D.; Danis, R.P.; Lovato, J.F.; Perdue, L.H.; Greven, C.; Genuth, S.; Goff, D.C.; Leiter, L.A.; Ismail-Beigi, F.; et al. The effects of medical management on the progression of diabetic retinopathy in persons with type 2 diabetes: The Action to Control Cardiovascular Risk in Diabetes (ACCORD) Eye Study. Ophthalmology 2014, 121, 2443–2451. [Google Scholar] [CrossRef]
- Nowak, J.Z. Age-related macular degeneration (AMD): Pathogenesis and therapy. Pharmacol. Rep. 2006, 58, 353–363. [Google Scholar] [PubMed]
- Gehrs, K.M.; Anderson, D.H.; Johnson, L.V.; Hageman, G.S. Age-related macular degeneration—Emerging pathogenetic and therapeutic concepts. Ann. Med. 2006, 38, 450–471. [Google Scholar] [CrossRef]
- Robinson, C.J.; Stringer, S.E. The splice variants of vascular endothelial growth factor (VEGF) and their receptors. J. Cell Sci. 2001, 114, 853–865. [Google Scholar]
- Muriach, M.; Bosch-Morell, F.; Alexander, G.; Blomhoff, R.; Barcia, J.; Arnal, E.; Almansa, I.; Romero, F.J.; Miranda, M. Lutein effect on retina and hippocampus of diabetic mice. Free Radicbiol. Med. 2006, 41, 979–984. [Google Scholar] [CrossRef]
- Semeraro, F.; Cancarini, A.; dell’Omo, R.; Rezzola, S.; Romano, M.R.; Costagliola, C. Diabetic Retinopathy: Vascular and Inflammatory Disease. J. Diabetes Res. 2015, 2015, 582060. [Google Scholar] [CrossRef]
- Jarrett, S.G.; Boulton, M.E. Consequences of oxidative stress in age-related macular degeneration. Mol. Asp. Med. 2012, 33, 399–417. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, S.X.; Hartnett, M.E. Signaling pathways triggered by oxidative stress that mediate features of severe retinopathy of prematurity. JAMA Ophthalmol. 2013, 131, 80–85. [Google Scholar] [CrossRef]
- Brownlee, M. The pathobiology of diabetic complications: A unifying mechanism. Diabetes 2005, 54, 1615–1625. [Google Scholar] [CrossRef] [PubMed]
- Colavitti, R.; Pani, G.; Bedogni, B.; Anzevino, R.; Borrello, S.; Waltenberger, J.; Galeotti, T. Reactive oxygen species as downstream mediators of angiogenic signaling by vascular endothelial growth factor receptor-2/KDR. J. Biol. Chem. 2002, 277, 3101–3108. [Google Scholar] [CrossRef]
- Ferrara, N. Vascular endothelial growth factor: Basic science and clinical progress. Endocr. Rev. 2004, 25, 581–611. [Google Scholar] [CrossRef]
- Čolak, E.; Ignjatović, S.; Radosavljević, A.; Žorić, L. The association of enzymatic and non-enzymatic antioxidant defense parameters with inflammatory markers in patients with exudative form of age-related macular degeneration. J. Clin. Biochem. Nutr. 2017, 60, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.; Muriach, M.; Almansa, I.; Arnal, E.; Messeguer, A.; Díaz-Llopis, M.; Romero, F.J.; Bosch-Morell, F. CR-6 protects glutathione peroxidase activity in experimental diabetes. Free Radicbiol. Med. 2007, 43, 1494–1498. [Google Scholar] [CrossRef]
- Tangvarasittichai, O.; Tangvarasittichai, S. Oxidative Stress, Ocular Disease and Diabetes Retinopathy. Curr. Pharm. Des. 2019, 24, 4726–4741. [Google Scholar] [CrossRef] [PubMed]
- Beauchamp, M.H.; Martinez-Bermudez, A.K.; Gobeil, F., Jr.; Marrache, A.M.; Hou, X.; Speranza, G.; Abran, D.; Quiniou, C.; Lachapelle, P.; Roberts, J.; et al. Role of thromboxane in retinal microvascular degeneration in oxygen-induced retinopathy. J. App. Physiol. 2001, 90, 2279–2288. [Google Scholar] [CrossRef] [PubMed]
- Balazy, M.; Chemtob, S. Trans-arachidonic acids: New mediators of nitro-oxidative stress. Pharmacol. Ther. 2008, 119, 275–290. [Google Scholar] [CrossRef]
- Brault, S.; Gobeil, F.; Fortier, A.; Honoré, J.C.; Joyal, J.S.; Sapieha, P.S.; Kooli, A.; Martin, E.; Hardy, P.; Ribeiro-da-Silva, A.; et al. Lysophosphatidic acid induces endothelial cell death by modulating the redox environment. Am. J. Physiol. 2007, 292, R1174–R1183. [Google Scholar] [CrossRef]
- Chaqour, J.; Lee, S.; Ravichandra, A.; Chaqour, B. Abscisic acid—An anti-angiogenic phytohormone that modulates the phenotypical plasticity of endothelial cells and macrophages. J. Cell Sci. 2018, 131, jcs210492. [Google Scholar] [CrossRef]
- Brar, V.S.; Sharma, R.K.; Murthy, R.K.; Chalam, K.V. Bevacizumab neutralizes the protective effect of vascular endothelial growth factor on retinal ganglion cells. Mol. Vis. 2010, 16, 1848–1853. [Google Scholar]
- Piotrowska, A.; Bajguz, A. Conjugates of abscisic acid, brassinosteroids, ethylene, gibberellins, and jasmonates. Phytochemistry 2011, 72, 2097–2211. [Google Scholar] [CrossRef]
- Zandalinas, S.I.; Mittler, R.; Balfagón, D.; Arbona, V.; Gómez-Cadenas, A. Plant adaptations to the combination of drought and high temperatures. Physiol. Plant. 2018, 162, 2–12. [Google Scholar] [CrossRef]
- Chanclud, E.; Lacombe, B. Plant Hormones: Key Players in Gut Microbiota and Human Diseases? Trends Plant. Sci. 2017, 22, 754–758. [Google Scholar] [CrossRef]
- Lin, L.; Tan, R.X. Cross-kingdom actions of phytohormones: A functional scaffold exploration. Chem. Rev. 2011, 111, 2734–2760. [Google Scholar] [CrossRef] [PubMed]
- Cutler, S.R.; Rodriguez, P.L.; Finkelstein, R.R.; Abrams, S.R. Abscisic Acid: Emergence of a Core Signaling Network. Ann. Rev. Plant. Biol. 2010, 61, 651–679. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Alvero, M.; Yunta, C.; Gonzalez-Guzman, M.; Lozano-Juste, J.; Benavente, J.; Arbona, V.; Menendez, M.; Martinez-Ripoll, M.; Infantes, L.; Gómez-Cadenas, A.; et al. The structure of ligand-bound intermediates of crop ABA receptors highlights the role of the PP2C as necessary ABA co-receptor. Mol. Plant. 2017, 10, 1250–1253. [Google Scholar] [CrossRef] [PubMed]
- De Ollas, C.; Arbona, V.; Gómez-Cadenas, A. Jasmonoyl isoleucine accumulation is needed for abscisic acid build-up in roots of Arabidopsis under water stress conditions. Plant. Cell Environ. 2015, 38, 2157–2170. [Google Scholar] [CrossRef]
- Shatil-Cohen, A.; Attia, Z.; Moshelion, M. Bundle-sheath cell regulation of xylem-mesophyll water transport via aquaporins under drought stress: A target of xylem-borne ABA? Plant. J. 2011, 67, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, T.; Shinozaki, K. Perception and transduction of abscisic acid signals: Keys to the function of the versatile plant hormone ABA. Trends Plant. Sci. 2007, 12, 343–351. [Google Scholar] [CrossRef]
- Sreenivasulu, N.; Harshavardhan, V.T.; Govind, G.; Seiler, C.; Kohli, A. Contrapuntal role of ABA: Does it mediate stress tolerance or plant growth retardation under long-term drought stress? Gene 2012, 506, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Sturla, L.; Fresia, C.; Guida, L.; Bruzzone, S.; Scarfì, S.; Usai, C.; Fruscione, F.; Magnone, M.; Millo, E.; Basile, G.; et al. LANCL2 is necessary for abscisic acid binding and signaling in human granulocytes and in rat insulinoma cells. J. Biol. Chem. 2009, 284, 28045–28057. [Google Scholar] [CrossRef] [PubMed]
- Fresia, C.; Vigliarolo, T.; Guida, L.; Booz, V.; Bruzzone, S.; Sturla, L.; Di Bona, M.; Pesce, M.; Usai, C.; De Flora, A.; et al. G-protein coupling and nuclear translocation of the human abscisic acid receptor LANCL2. Sci. Rep. 2016, 25, 26658. [Google Scholar] [CrossRef]
- Li, H.H.; Hao, R.L.; Wu, S.S.; Guo, P.C.; Chen, C.J.; Pan, L.P.; Ni, H. Occurrence, function and potential medicinal applications of the phytohormone abscisic acid in animals and humans. Biochem. Pharm. 2011, 82, 701–712. [Google Scholar] [CrossRef]
- Lievens, L.; Pollier, J.; Goossens, A.; Beyaert, R.; Staal, J. Abscisic Acid as Pathogen Effector and Immune Regulator. Front. Plant. Sci. 2017, 8, 587. [Google Scholar] [CrossRef]
- Glennon, E.K.; Adams, L.G.; Hicks, D.R.; Dehesh, K.; Luckhart, S. Supplementation with Abscisic Acid Reduces Malaria Disease Severity and Parasite Transmission. Am. J. Trop Med. Hyg. 2016, 94, 1266–1275. [Google Scholar] [CrossRef]
- Rakic, B.; Clarke, J.; Tremblay, T.L.; Taylor, J.; Schreiber, K.; Nelson, K.M.; Abrams, S.R.; Pezacki, J.P. A small-molecule probe for hepatitis C virus replication that blocks protein folding. Chem. Biol. 2006, 13, 1051–1060. [Google Scholar] [CrossRef]
- Hontecillas, R.; Roberts, P.C.; Carbo, A.; Vives, C.; Horne, W.T.; Genis, S.; Velayudhan, B.; Bassaganya-Riera, J. Dietary abscisic acid ameliorates influenza-virus-associated disease and pulmonary immunopathology through a PPARγ-dependent mechanism. J. Nutrbiochem. 2013, 24, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- Zhou, N.; Yao, Y.; Ye, H.; Zhu, W.; Chen, L.; Mao, Y. Abscisic-acid-induced cellular apoptosis and differentiation in glioma via the retinoid acid signaling pathway. Int. J. Cancer 2016, 138, 1947–1958. [Google Scholar] [CrossRef]
- Bruzzone, S.; Magnone, M.; Mannino, E.; Sociali, G.; Sturla, L.; Fresia, C.; Booz, V.; Emionite, L.; De Flora, A.; Zocchi, E. Abscisic Acid Stimulates Glucagon-Like Peptide-1 Secretion from L-Cells and Its Oral Administration Increases Plasma Glucagon-Like Peptide-1 Levels in Rats. PLoS ONE 2015, 10, e0140588. [Google Scholar] [CrossRef]
- Magnone, M.; Leoncini, G.; Vigliarolo, T.; Emionite, L.; Sturla, L.; Zocchi, E.; Murialdo, G. Chronic Intake of Micrograms of Abscisic Acid Improves Glycemia and Lipidemia in a Human Study and in High-Glucose Fed Mice. Nutrients 2018, 10, 1495. [Google Scholar] [CrossRef]
- Sánchez-Sarasúa, S.; Moustafa, S.; García-Avilés, Á.; López-Climent, M.F.; Gómez-Cadenas, A.; Olucha-Bordonau, F.E.; Sánchez-Pérez, A.M. The effect of abscisic acid chronic treatment on neuroinflammatory markers and memory in a rat model of high-fat diet induced neuroinflammation. Nutrmetab (Lond.) 2016, 13, 73. [Google Scholar] [CrossRef] [PubMed]
- Ribes-Navarro, A.; Atef, M.; Sánchez-Sarasúa, S.; Beltrán-Bretones, M.T.; Olucha-Bordonau, F.; Sánchez-Pérez, A.M. Abscisic Acid Supplementation Rescues High Fat Diet-Induced Alterations in Hippocampal Inflammation and IRSs Expression. Mol. Neurobiol. 2019, 56, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Ye, R.D. Role of G protein-coupled receptors in inflammation. Actapharmacol. Sin. 2012, 33, 342–350. [Google Scholar] [CrossRef]
- Pascual, G.; Fong, A.L.; Ogawa, S.; Gamliel, A.; Li, A.C.; Perissi, V.; Rose, D.W.; Willson, T.M.; Rosenfeld, M.G.; Glass, C.K. A SUMOylation-dependent pathway mediates transrepression of inflammatory response genes by PPAR-gamma. Nature 2005, 437, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Glass, C.K.; Rosenfeld, M.G. The coregulator exchange in transcriptional functions of nuclear receptors. Genes Dev. 2000, 14, 121–141. [Google Scholar] [PubMed]
- Guan, L.; Zhao, J.; Scandalios, J.G. Cis-elements and trans-factors that regulate expression of the maize cat1 antioxidant gene in response to ABA and osmotic stress: H2O2 is the likely intermediary signaling molecule for the response. Plant. J. 2000, 22, 87–95. [Google Scholar] [CrossRef]
- Jiang, M.; Zhang, J. Effect of abscisic acid on active oxygen species, antioxidativedefence system and oxidative damage in leaves of maize seedlings. Plant. Cell Physiol. 2001, 42, 1265–1273. [Google Scholar] [CrossRef]
- Bellaire, B.A.; Carmody, J.; Braud, J.; Gossett, D.R.; Banks, S.W.; Lucas, M.C.; Fowler, T.E. Involvement of abscisic acid-dependent and -independent pathways in the upregulation of antioxidant enzyme activity during NaCl stress in cotton callus tissue. Free Radic. Res. 2000, 33, 531–545. [Google Scholar] [CrossRef]
- Rafiepour, K.; Esmaeili-Mahani, S.; Salehzadeh, A.; Sheibani, V. Phytohormone Abscisic Acid Protects Human Neuroblastoma SH-SY5Y Cells Against 6-Hydroxydopamine-Induced Neurotoxicity through Its Antioxidant and Antiapoptotic Properties. Rejuvenation Res. 2018. [Google Scholar] [CrossRef]
- Soti, M.; Abbasnejad, M.; Kooshki, R.; Esmaeili-Mahani, S. Central microinjection of phytohormone abscisic acid changes feeding behavior, decreases body weight, and reduces brain oxidative stress in rats. Nutr. Neurosci. 2018, 6, 1–10. [Google Scholar] [CrossRef]
- Ye, N.; Jia, L.; Zhang, J. ABA signal in rice under stress conditions. Rice 2012, 5, 1–9. [Google Scholar] [CrossRef]
- Endo, A.; Okamoto, M.; Koshiba, T. ABA Biosynthetic and Catabolic Pathways. In Abscisic Acid Metabolism Transport Signaling, 1st ed.; Zhang, D.-P., Ed.; Springer Science + Business Media: Dordrecht, The Netherlands, 2014; pp. 21–45. [Google Scholar]
- Liotenberg, S.; North, H.; Marion-Poll, A. Molecular biology and regulation of abscisic acid biosynthesis in plants. Plant. Physiol. Biochem. 1999, 37, 341–350. [Google Scholar] [CrossRef]
- Bernstein, P.S.; Li, B.; Vachali, P.P.; Gorusupudi, A.; Shyam, R.; Henriksen, B.S.; Nolan, J.M. Lutein, zeaxanthin, and meso-zeaxanthin: The basic and clinical science underlying carotenoid-based nutritional interventions against ocular disease. Progretin Eye Res. 2016, 50, 34–66. [Google Scholar] [CrossRef]
- Eisenhauer, B.; Natoli, S.; Liew, G.; Flood, V.M. Lutein and zeaxanthin—Food sources, bioavailability and dietary variety in age-related macular degeneration protection. Nutrients 2017, 9, 120. [Google Scholar] [CrossRef]
- Sommerburg, O.; Keunen, J.E.E.; Bird, A.C.; van Kuijk, F.J.G.M. Fruits and vegetables that are sources for lutein and zeaxanthin: The macular pigment in human eyes. Br. J. Opthalmol. 1998, 82, 907–991. [Google Scholar] [CrossRef]
- Hirschberg, J. Carotenoid biosynthesis in flowering plants. Curr. Opin. Plant Biol. 2001, 4, 210–218. [Google Scholar] [CrossRef]
- Wu, S.; Hu, C.; Tan, Q.; Zhao, X.; Xu, S.; Xia, Y.; Sun, X. Nitric oxide acts downstream of abscisic acid in molybdenum-induced oxidative tolerance in wheat. Plant. Cell Rep. 2018, 37, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Sakthivel, P.; Sharma, N.; Klahn, P.; Gereke, M.; Bruder, D. Abscisic acid: A phytohormone and mammalian cytokine as novel pharmacon with potential for future development into clinical applications. Curr. Med. Chem. 2016, 23, 1549–1570. [Google Scholar] [CrossRef] [PubMed]
- Bassaganya-Riera, J.; Guri, A.J.; Lu, P.; Climent, M.; Carbo, A.; Sobral, B.W.; Horne, W.T.; Lewis, S.N.; Bevan, D.R.; Hontecillas, R. Abscisic acid regulates inflammation via ligand-binding domain-independent activation of peroxisome proliferator-activated receptor gamma. J. Biol. Chem. 2011, 286, 2504–2516. [Google Scholar] [CrossRef]
- Li, Z.; He, T.; Du, K.; Xing, Y.Q.; Run, Y.M.; Yan, Y.; Shen, Y. Inhibition of oxygen-induced ischemic retinal neovascularization with adenoviral 15-lipoxygenase-1 gene transfer via up-regulation of PPAR-γ and down-regulation of VEGFR-2 expression. PLoS ONE 2014, 9, e85824. [Google Scholar] [CrossRef] [PubMed]
- Villapol, S. Roles of Peroxisome Proliferator-Activated Receptor Gamma on Brain and Peripheral Inflammation. Cell Mol. Neurobiol. 2018, 38, 121–132. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Z.; Yan, H. Simvastatin inhibits ischemia/reperfusion injury-induced apoptosis of retinal cells via downregulation of the tumor necrosis factor-α/nuclear factor-κB pathway. Int. J. Mol. Med. 2015, 36, 399–405. [Google Scholar] [CrossRef]
- DeNiro, M.; Al-Mohanna, F.A. Nuclear factor kappa-B signaling is integral to ocular neovascularization in ischemia-independent microenvironment. PLoS ONE 2014, 9, e101602. [Google Scholar] [CrossRef] [PubMed]
- Zocchi, E.; Hontecillas, R.; Leber, A.; Einerhand, A.; Carbo, A.; Bruzzone, S.; Tubau-Juni, N.; Philipson, N.; Zoccoli-Rodriguez, V.; Sturla, L.; et al. Abscisic Acid: A Novel Nutraceutical for Glycemic Control. Front. Nutr. 2017, 4, 1–13. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baliño, P.; Gómez-Cadenas, A.; López-Malo, D.; Romero, F.J.; Muriach, M. Is There A Role for Abscisic Acid, A Proven Anti-Inflammatory Agent, in the Treatment of Ischemic Retinopathies? Antioxidants 2019, 8, 104. https://doi.org/10.3390/antiox8040104
Baliño P, Gómez-Cadenas A, López-Malo D, Romero FJ, Muriach M. Is There A Role for Abscisic Acid, A Proven Anti-Inflammatory Agent, in the Treatment of Ischemic Retinopathies? Antioxidants. 2019; 8(4):104. https://doi.org/10.3390/antiox8040104
Chicago/Turabian StyleBaliño, Pablo, Aurelio Gómez-Cadenas, Daniel López-Malo, Francisco Javier Romero, and María Muriach. 2019. "Is There A Role for Abscisic Acid, A Proven Anti-Inflammatory Agent, in the Treatment of Ischemic Retinopathies?" Antioxidants 8, no. 4: 104. https://doi.org/10.3390/antiox8040104
APA StyleBaliño, P., Gómez-Cadenas, A., López-Malo, D., Romero, F. J., & Muriach, M. (2019). Is There A Role for Abscisic Acid, A Proven Anti-Inflammatory Agent, in the Treatment of Ischemic Retinopathies? Antioxidants, 8(4), 104. https://doi.org/10.3390/antiox8040104





