A Single-Dose of a Polyphenol-Rich Fucus Vesiculosus Extract is Insufficient to Blunt the Elevated Postprandial Blood Glucose Responses Exhibited by Healthy Adults in the Evening: A Randomised Crossover Trial
Abstract
1. Introduction
2. Materials and Methods
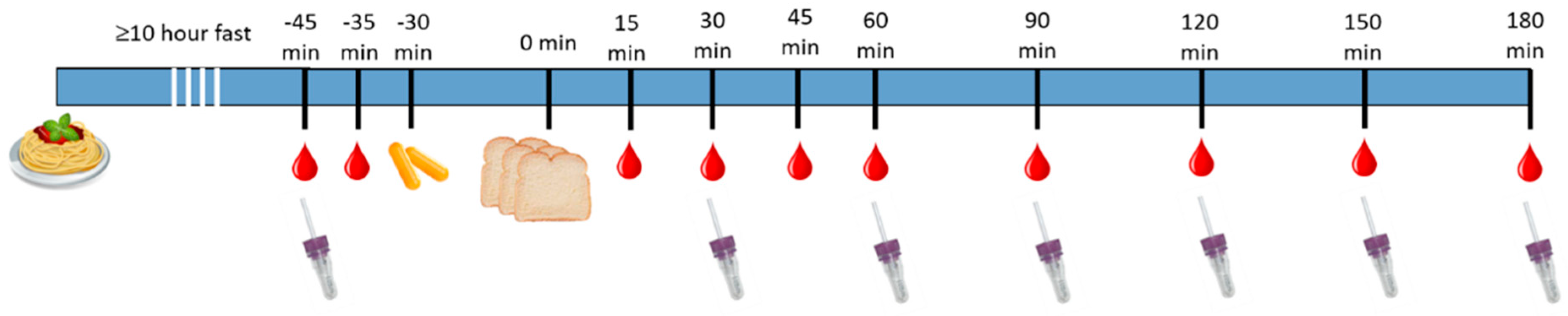
2.1. Trial Design
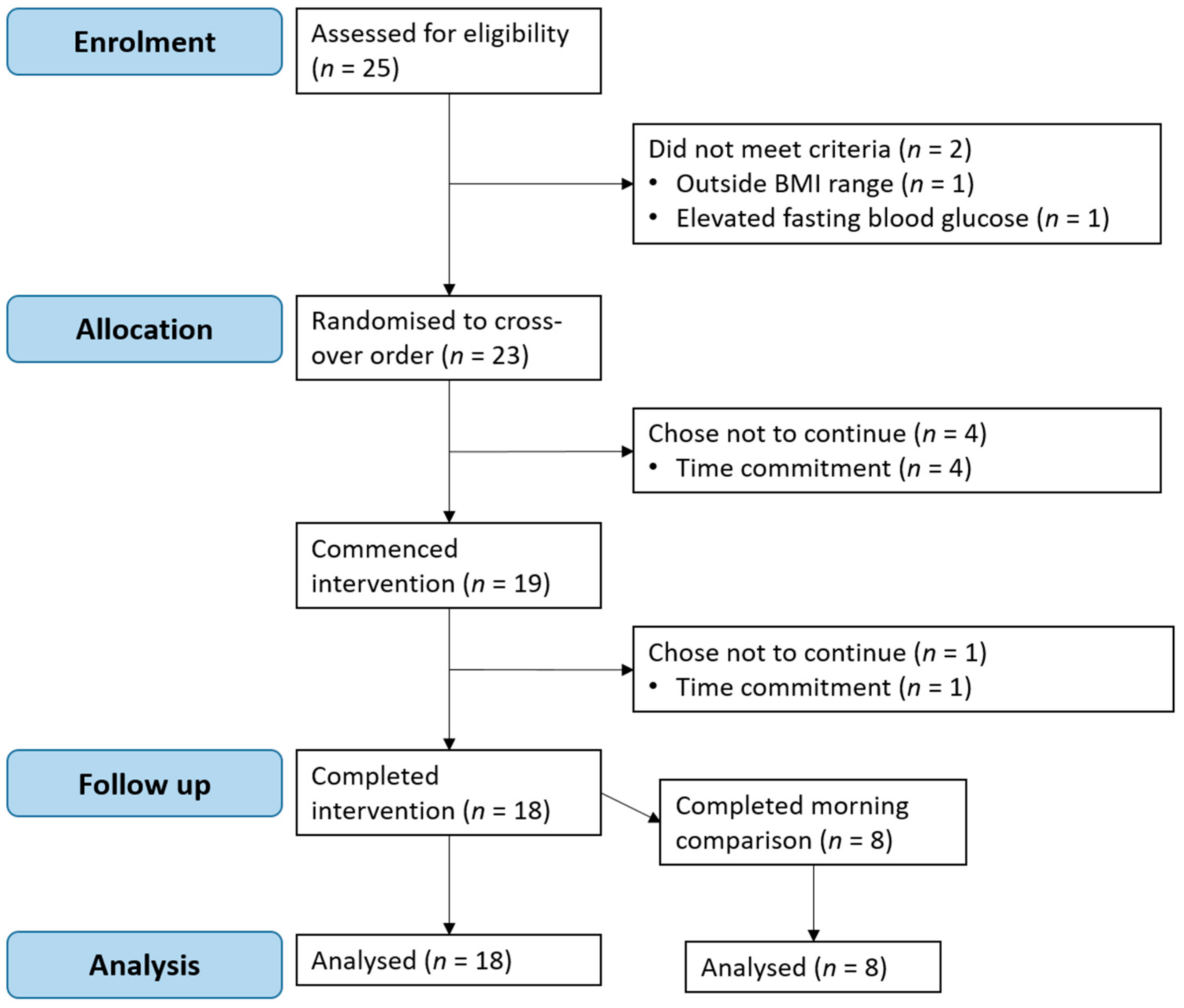
2.2. Participants
2.3. Test Products
2.4. Randomisation and Blinding
2.5. Procedure
2.6. Outcome Measures
2.6.1. Blood Glucose and Plasma Insulin
2.6.2. Anthropometric Data
2.6.3. Intolerance Symptoms
2.6.4. Diaries and Questionnaires
2.7. Quantification of Soluble Polyphenols
2.8. Statistical Analysis
3. Results
3.1. Participants
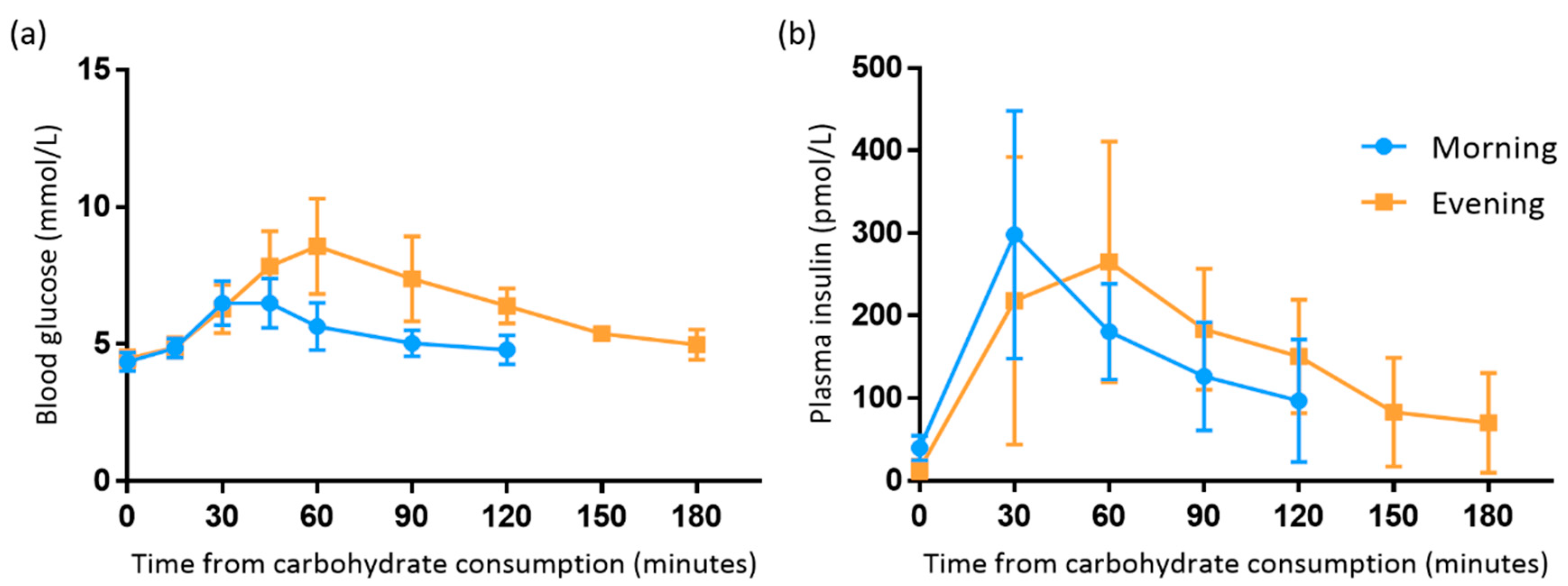
3.2. Comparison between Morning and Evening
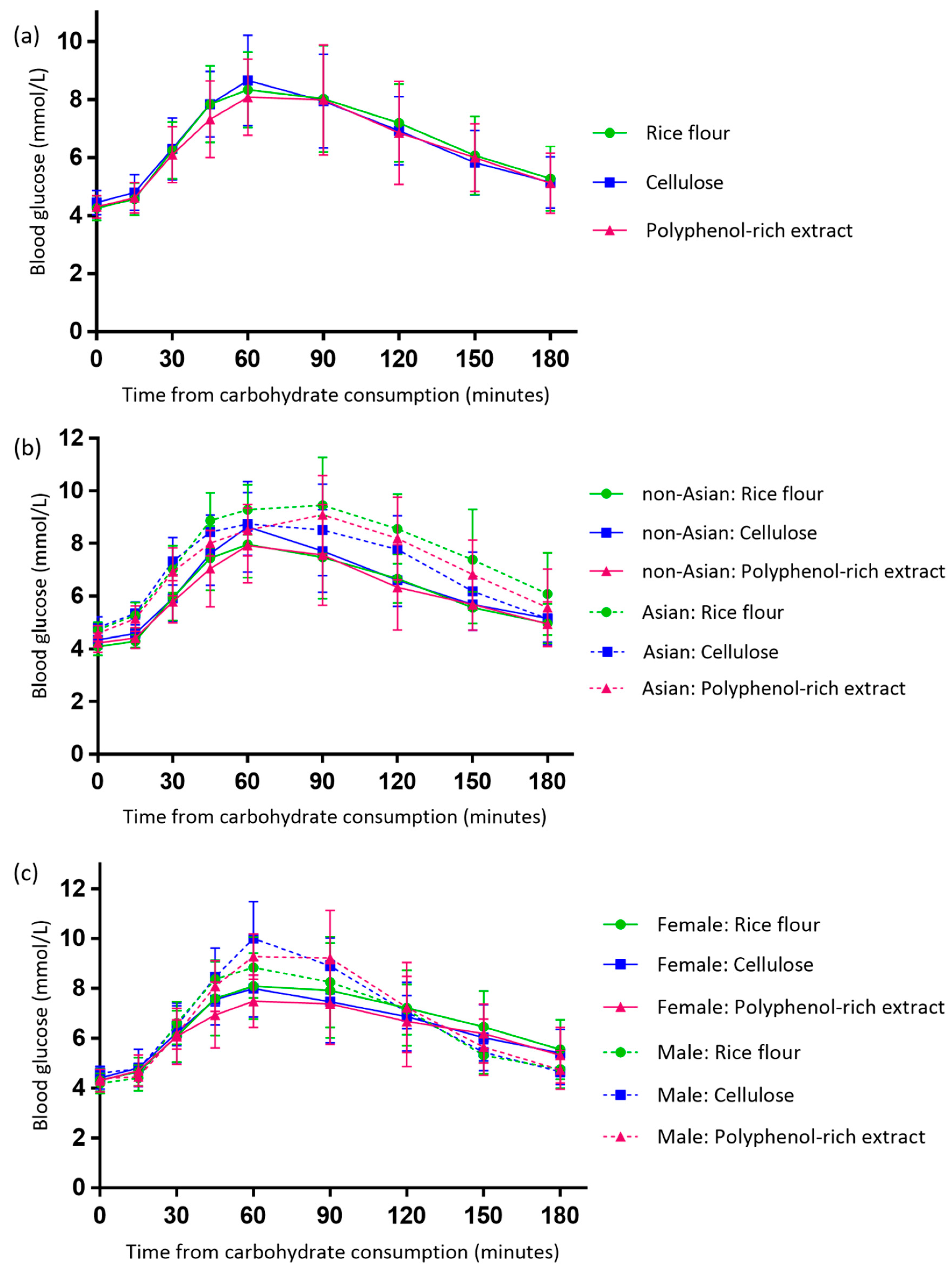
3.3. Effect of the Polyphenol-Rich Extract on Evening Postprandial Blood Glucose
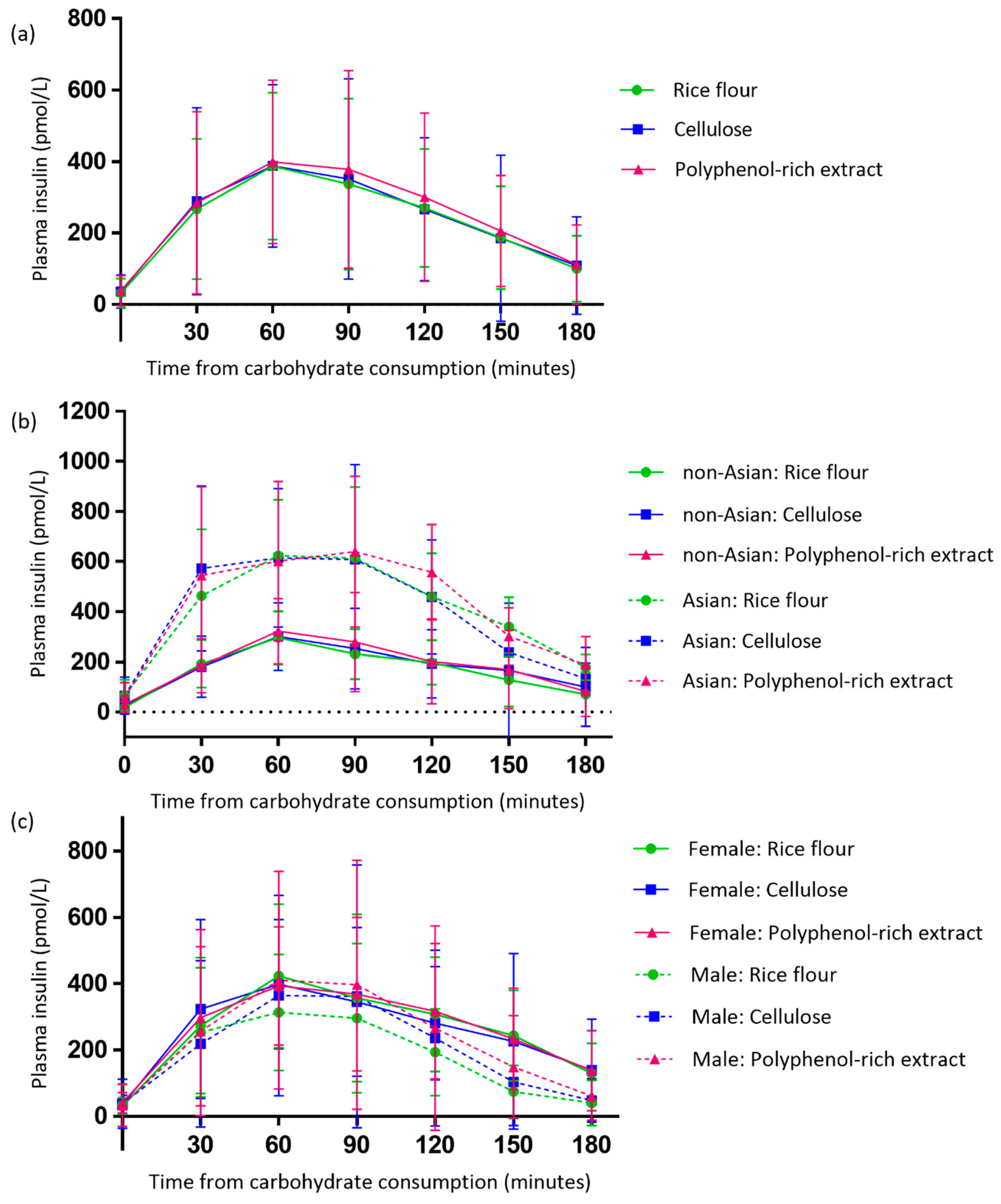
3.4. Effect of the Polyphenol-Rich Extract on Evening Postprandial Plasma Insulin
3.5. Intolerance Symptoms
3.6. Polyphenol Content of Extract
4. Discussion
4.1. A Reduction in Peak Postprandial Blood Glucose Concentration Was Observed in Females but Not Males
4.2. The Polyphenol-Rich Extract Had No Effect on Evening Postprandial Glycaemia Overall
4.3. Asian Participants Experienced Exaggerated Postprandial Plasma Insulin Responses
4.4. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ipp, E. Impaired glucose tolerance. Diabetes Care 2000, 23, 569. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Pre-diabetes, metabolic syndrome, and cardiovascular risk. J. Am. Coll. Cardiol. 2012, 59, 635–643. [Google Scholar] [CrossRef] [PubMed]
- General Practice Management of Type 2 Diabetes—2014-15; The Royal Australian College of General Practitioners and Diabetes Australia: Melbourne, Australia, 2014.
- Murray, M.; Dordevic, A.; Ryan, L.; Bonham, M. An emerging trend in functional foods for the prevention of cardiovascular disease and diabetes: Marine algal polyphenols. Crit. Rev. Food Sci. Nutr. 2018, 58, 1342–1358. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Dordevic, A.; Ryan, L.; Bonham, M. Phlorotannins and macroalgal polyphenols: Potential as functional food ingredients and role in health promotion. In Functional Food in Human Health; Rani, V., Yadav, U., Eds.; Springer: Chennai, India, 2018. [Google Scholar]
- Murugan, A.C.; Karim, M.R.; Yusoff, M.B.M.; Tan, S.H.; Asras, M.F.B.F.; Rashid, S.S. New insights into seaweed polyphenols on glucose homeostasis. Pharm. Biol. 2015, 53, 1087–1097. [Google Scholar] [CrossRef] [PubMed]
- Min, K.-H.; Kim, H.-J.; Jeon, Y.-J.; Han, J.-S. Ishige okamurae ameliorates hyperglycemia and insulin resistance in C57BL/KsJ-db/db mice. Diabetes Res. Clin. Pract. 2011, 93, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.; Jin, Y.B.; Lee, H.; Cha, M.; Sohn, E.-T.; Moon, J.; Park, C.; Chun, S.; Jung, E.S.; Hong, J.S.; et al. Brown alga Ecklonia cava attenuates type 1 diabetes by activating AMPK and Akt signaling pathways. Food Chem. Toxicol. 2010, 48, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Ko, S.-C.; Kang, M.-C.; Lee, D.H.; Jeon, Y.-J. Octaphlorethol A, a marine algae product, exhibits antidiabetic effects in type 2 diabetic mice by activating AMP-activated protein kinase and upregulating the expression of glucose transporter 4. Food Chem. Toxicol. 2016, 91, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Castro-Acosta, M.L.; Smith, L.; Miller, R.J.; McCarthy, D.I.; Farrimond, J.A.; Hall, W.L. Drinks containing anthocyanin-rich blackcurrant extract decrease postprandial blood glucose, insulin and incretin concentrations. J. Nutr. Biochem. 2016, 38, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Castro-Acosta, M.L.; Stone, S.G.; Mok, J.E.; Mhajan, R.K.; Fu, C.-I.; Lenihan-Geels, G.N.; Corpe, C.P.; Hall, W.L. Apple and blackcurrant polyphenol-rich drinks decrease postprandial glucose, insulin and incretin response to a high-carbohydrate meal in healthy men and women. J. Nutr. Biochem. 2017, 49, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Nyambe-Silavwe, H.; Williamson, G. Polyphenol- and fibre-rich dried fruits with green tea attenuate starch-derived postprandial blood glucose and insulin: A randomised, controlled, single-blind, cross-over intervention. Br. J. Nutr. 2016, 116, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Torronen, R.; Sarkkinen, E.; Tapola, N.; Hautaniemi, E.; Kilpi, K.; Niskanen, L. Berries modify the postprandial plasma glucose response to sucrose in healthy subjects. Br. J. Nutr. 2010, 103, 1094–1097. [Google Scholar] [PubMed]
- Coe, S.; Ryan, L. Impact of polyphenol-rich sources on acute postprandial glycaemia: A systematic review. J. Nutr. Sci. 2016, 5, e24. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Jeon, Y.-J. Efficacy and safety of a dieckol-rich extract (AG-dieckol) of brown algae, Ecklonia cava, in pre-diabetic individuals: A double-blind, randomized, placebo-controlled clinical trial. J. Funct. Foods 2015, 6, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, N.J.; Puchulu, F.E.; Gagliardino, J.J.; Ruiz, M.; Llorens, J.M.; Ruiz, J.; Lamas, A.; De Miguel, R. Circadian Variation of the Blood Glucose, Plasma Insulin and Human Growth Hormone Levels in Response to an Oral Glucose Load in Normal Subjects. Diabetes 1974, 23, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Leung, G.K.W.; Huggins, C.E.; Bonham, M.P. Effect of meal timing on postprandial glucose responses to a low glycemic index meal: A crossover trial in healthy volunteers. Clin. Nutr. 2019, 38, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.D. Physiological responses to food intake throughout the day. Nutr. Res. Rev. 2014, 27, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Challet, E. Circadian clocks, food intake, and metabolism. Prog. Mol. Biol. Transl. Sci. 2013, 119, 105–135. [Google Scholar] [PubMed]
- Kalsbeek, A.; Fleur, S.L.; Fliers, E. Circadian control of glucose metabolism. Mol. Metab. 2014, 3, 372–383. [Google Scholar] [CrossRef] [PubMed]
- Al-Naimi, S.; Hampton, S.M.; Richard, P.; Tzung, C.; Morgan, L.M. Postprandial metabolic profiles following meals and snacks eaten during simulated night and day shift work. Chronobiol. Int. 2004, 21, 937–947. [Google Scholar] [CrossRef] [PubMed]
- Pan, A.; Schernhammer, E.; Sun, Q.; Hu, F. Rotating night shift work and risk of type 2 diabetes: Two prospective cohort studies in women. PLoS Med. 2011, 8, e1001141. [Google Scholar] [CrossRef] [PubMed]
- Laakso, M. Hyperglycemia and cardiovascular disease in type 2 diabetes. Diabetes 1999, 48, 937–942. [Google Scholar] [CrossRef] [PubMed]
- Hermida, R.C.; Ayala, D.E.; Mojon, A.; Fernandez, J.R. Influence of Time of Day ofBlood Pressure–Lowering Treatment on Cardiovascular Risk in Hypertensive PatientsWith Type 2 Diabetes. Diabetes Care 2011, 34, 1270–1276. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, M.; Harrington, D.; Starkey, S.; Williams, P.; Hampton, S. Diurnal postprandial responses to low and high glycaemic index mixed meals. Clin. Nutr. 2014, 33, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A. A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Dordevic, A.; Ryan, L.; Bonham, M. The impact of a single dose of a polyphenol-rich seaweed extract on postprandial glycaemic control in healthy adults: A randomised cross-over trial. Nutrients 2018, 10, 270. [Google Scholar] [CrossRef] [PubMed]
- Wootton-Beard, P.C.; Brandt, K.; Fell, D.; Warner, S.; Ryan, L. Effects of a beetroot juice with high neobetanin content on the early-phase insulin response in healthy volunteers. J. Nutr. Sci. 2014, 3, e9. [Google Scholar] [CrossRef] [PubMed]
- Phenol-Explorer: An Online Comprehensive Database on Polyphenol Contents in Foods. Available online: http://phenol-explorer.eu/ (accessed on 15 February 2017).
- USDA Database for the Flavonoid Content of Selected Foods, Release 3.1. Available online: https://www.ars.usda.gov/northeast-area/beltsville-md/beltsville-human-nutrition-research-center/nutrient-data-laboratory/docs/usda-database-for-the-flavonoid-content-of-selected-foods-release-31-december-2013/ (accessed on 15 February 2017).
- Craig, C.; Marshall, A.; Sjöström, M.; Bauman, A.; Booth, M.; Ainsworth, B.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, J.; Shen, J.; Silva, A.; Dennis, D.A.; Barrow, C.J. A simple 96-well microplate method for estimation of total polyphenol content in seaweeds. J. Appl. Phycol. 2006, 18, 445–450. [Google Scholar] [CrossRef]
- Vilsbøll, T.; Knop, F.K.; Krarup, T.; Johansen, A.; Madsbad, S.; Larsen, S.; Hansen, T.; Pedersen, O.; Holst, J.J. The pathophysiology of diabetes involves a defective amplification of the late-phase insulin response to glucose by glucose-dependent insulinotropic polypeptide—Regardless of etiology and phenotype. J. Clin. Endocrinol. Metab. 2003, 88, 4897–4903. [Google Scholar] [CrossRef] [PubMed]
- Campesi, I.; Marino, M.; Cipolletti, M.; Romani, A.; Franconi, F. Put “gender glasses” on the effects of phenolic compounds on cardiovascular function and diseases. Eur. J. Nutr. 2018, 57, 2677–2691. [Google Scholar] [CrossRef] [PubMed]
- Campesi, I.; Romani, A.; Marino, M.; Franconi, F. Phenolic Compounds from a Sex-Gender Perspective. In Recent Advances in Polyphenol Research; Romani, A., Lattanzio, V., Quideau, S., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2014. [Google Scholar]
- Varlamov, O.; Bethea, C.L.; Roberts, C.T., Jr. Sex-specific differences in lipid and glucose metabolism. Front. Endocrinol. 2015, 5, 241. [Google Scholar] [CrossRef] [PubMed]
- Gesensway, D. Reasons for sex-specific and gender-specific study of health topics. Ann. Intern. Med. 2001, 135, 935–938. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.L.; Greaves, L.; Repta, R. Better science with sex and gender: Facilitating the use of a sex and gender-based analysis in health research. Int. J. Equity Health 2009, 8, 14. [Google Scholar] [CrossRef] [PubMed]
- Wizemann, T.M.; Pardue, M.-L. Exploring the Biological Contributions to Human Health: Does Sex Matter? National Academy Press: Washington, DC, USA, 2001. [Google Scholar]
- Mauvais-Jarvis, F. Sex differences in metabolic homeostasis, diabetes, and obesity. Biol. Sex Differ. 2015, 6, 14. [Google Scholar] [CrossRef] [PubMed]
- Paradis, M.-E.; Couture, P.; Lamarche, B. A randomised crossover placebo-controlled trial investigating the effect of brown seaweed (Ascophyllum nodosum and Fucus vesiculosus) on postchallenge plasma glucose and insulin levels in men and women. Appl. Physiol. Nutr. Metab. 2011, 36, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Bonora, E. Postprandial peaks as a risk factor for cardiovascular disease: Epidemiological perspectives. Int. J. Clin. Pract. Suppl. 2002, 129, 5–11. [Google Scholar]
- Gabbia, D.; Dall’Acqua, S.; Gangi, I.M.D.; Bogialli, S.; Caputi, V.; Albertoni, L.; Marsilio, I.; Paccagnella, N.; Carrara, M.; Giron, M.C.; et al. The phytocomplex from Fucus vesiculosus and Ascophyllum nodosum controls postprandial plasma glucose levels: An in vitro and in vivo study in a mouse model of NASH. Mar. Drugs 2017, 15, 41. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.-J.; Hwang, J.-Y.; Choi, J.-I.; Han, J.-S.; Kim, H.-J.; Jeon, Y.-J. Diphlorethohydroxycarmalol isolated from Ishige okamurae, a brown algae, a potent α-glucosidase and α-amylase inhibitor, alleviates postprandial hyperglycemia in diabetic mice. Eur. J. Pharmacol. 2009, 615, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Iwai, K. Antidiabetic and antioxidant effects of polyphenols in brown alga Ecklonia stolonifera in genetically diabetic KK-Ay mice. Plant Foods Hum. Nutr. 2008, 63, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Horie, I.; Abiru, N.; Eto, M.; Sako, A.; Akeshima, J.; Nakao, T.; Nakashima, Y.; Nir, T.; Ito, A.; Nozaki, A.; et al. Sex differences in insulin and glucagon responses for glucose homeostasis in young healthy Japanese adults. J. Diabetes Investig. 2018, 9, 1283–1287. [Google Scholar] [CrossRef] [PubMed]
- Sicree, R.A.; Zimmet, P.Z.; Dunstan, D.W.; Cameron, A.J.; Welborn, T.A.; Shaw, J.E. Differences in height explain gender differences in the response to the oral glucose tolerance test—The ausdiab study. Diabet. Med. 2017, 25, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, G. Insulin and insulin resistance. Clin. Biochem. Rev. 2005, 26, 19–39. [Google Scholar] [PubMed]
- Taylor, R.H.; Jenkins, D.J.; Barker, H.M.; Fielden, H.; Goff, D.V.; Misiewicz, J.; Lee, D.A.; Allen, H.B.; MacDonald, G.; Wallrabe, H. Effect of acarbose on the 24-hour blood glucose profile and pattern of carbohydrate absorption. Diabetes Care 1982, 5, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Saad, A.; Dalla Man, C.; Nandy, D.K.; Levine, J.A.; Bharucha, A.E.; Rizza, R.A.; Basu, R.; Carter, R.E.; Cobelli, C.; Kudva, Y.C.; et al. Diurnal pattern to insulin secretion and insulin action in healthy individuals. Diabetes 2012, 61, 2691–2700. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Dordevic, A.; Bonham, M.; Ryan, L. Do marine algal polyphenols have antidiabetic, anti-hyperlipidaemic or anti-inflammatory effects in humans? A systematic review. Crit. Rev. Food Sci. Nutr. 2018, 58, 2039–2054. [Google Scholar] [CrossRef] [PubMed]
- Holdenson, Z.; Catanzariti, L.; Phillips, G.; Waters, A. A Picture of Diabetes in Overseas-Born Australians; Bulletin No. 9; Australian Institute of Health and Welfare: Canberra, Australia, 2003. [Google Scholar]
- Frayn, K.N. Visceral fat and insulin resistance—Causative or correlative? Br. J. Nutr. 2000, 83 (Suppl. 1), S71–S77. [Google Scholar] [CrossRef]
- McBean, A.M.; Li, S.; Gilbertson, D.T.; Collins, A.J. Differences in diabetes prevalence, incidence, and mortality among the elderly of four racial/ethnic groups: Whites, Blacks, Hispanics, and Asians. Diabetes Care 2004, 27, 2317–2324. [Google Scholar] [CrossRef] [PubMed]
- Beck-Nielsen, H.; Pedersen, O. Diurnal variation in insulin binding to human monocytes. J. Clin. Endocrinol. Metab. 1978, 47, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Gibson, T.; Jarrett, R.J. Diurnal variation in insulin sensitivity. Lancet 1972, 300, 947–948. [Google Scholar] [CrossRef]
| Characteristic | Mean (SD) |
|---|---|
| Age (years) | 25.5 (19) * |
| BMI (kg/m2) | 23.8 (2.6) |
| Waist circumference (cm) | 76.3 (4.4) |
| Fat mass (%)—females | 32.2 (5.8) |
| Fat mass (%)—males | 15.0 (5.3) |
| Estimated daily polyphenol intake (mg) | 1559 (1676) * |
| Estimated daily energy intake (kJ) | 8978 (5508) * |
| Systolic blood pressure (mmHg) | 121 (9) |
| Diastolic blood pressure (mmHg) | 77 (5) |
| n(frequency) | |
| Female | 12 (67%) |
| Male | 6 (33%) |
| Ethnicity | |
| Caucasian background | 13 (72%) |
| Asian background | 5 (28%) |
| Physical Activity level | |
| Moderate | 9 (50%) |
| High | 9 (50%) |
| Characteristic | Mean (SD) |
|---|---|
| Age (years) | 24 (20) * |
| BMI (kg/m2) | 22.5 (2.4) |
| Waist circumference (cm) | 74.9 (5.4) |
| Percent fat mass—males (%) | 12.5 (1.9) |
| Percent fat mass—females (%) | 28.8 (3.0) |
| Systolic blood pressure (mmHg) | 123.8 (8.4) |
| Diastolic blood pressure (mmHg) | 75.8 (4.3) |
| Male:Female (n) | 4:4 |
| Caucasian:Asian (n) | 8:0 |
| Outcome | Group | n | Rice Flour | Cellulose | Polyphenol-Rich Extract | p-Value 1 | p-Value 2 |
|---|---|---|---|---|---|---|---|
| Blood glucose for whole group | |||||||
| Fasting (mmol/L) | Total | 18 | 4.3 (0.4) | 4.5 (0.4) | 4.3 (0.4) | 0.077 | NA |
| iAUC (mmol/L·3 h) | Total | 18 | 411 (152) | 410 (127) | 414 (169) | 0.881 | NA |
| Peak concentration (mmol/L) | Total | 18 | 8.7 (1.5) | 8.9 (1.5) | 8.5 (1.6) | 0.459 | NA |
| Blood glucose by ethnicity | |||||||
| Fasting (mmol/L) | Caucasian | 13 | 4.1 (0.3) | 4.3 (0.3) | 4.2 (0.4) | 0.106 | 0.007 |
| Asian | 5 | 4.7 (0.3) | 4.8 (0.4) | 4.6 (0.3) | 0.084 | ||
| iAUC (mmol/L·3 h) | Caucasian | 13 | 368 (130) | 399 (140) | 376 (177) | 0.607 | 0.118 |
| Asian | 5 | 521 (162) | 439 (93) | 515 (105) | 0.408 | ||
| Peak concentration (mmol/L) | Caucasian | 13 | 8.3 (1.3) | 8.8 (1.7) | 8.2 (1.7) | 0.052 | 0.177 |
| Asian | 5 | 9.7 (1.6) | 9.3 (1.0) | 9.3 (1.2) | 0.820 | ||
| Blood glucose by sex | |||||||
| Fasting (mmol/L) | Female | 12 | 4.3 (0.4) | 4.4 (0.5) | 4.3 (0.5) | 0.564 | 0.800 |
| Male | 6 | 4.2 (0.4) | 4.6 (0.3) | 4.4 (0.1) | 0.038 | ||
| iAUC (mmol/L·3 h) | Female | 12 | 410 (160) | 394 (127) | 385 (168) | 0.529 | 0.506 |
| Male | 6 | 412 (149) | 443 (132) | 473 (170) | 0.556 | ||
| Peak concentration (mmol/L) | Female | 12 | 8.4 (1.6) | 8.4 (1.2) a | 7.9 (1.4) a | 0.018 | 0.040 |
| Male | 6 | 9.2 (1.1) | 10.0 (1.5) a | 9.8 (1.3) a | 0.168 | ||
| Outcome | Group | n | Rice Flour | Cellulose | Polyphenol-Rich Extract | p-Value 1 |
|---|---|---|---|---|---|---|
| Plasma insulin for whole group | ||||||
| Fasting (pmol/L) | Total | 18 | 15 (33) | 15 (39) | 20 (59) | 0.978 |
| iAUC (pmol/L·3 h) | Total | 18 | 32,891 (21,899) | 27,709 (37,172) | 30,085 (47,622) | 0.801 |
| Late phase iAUC (pmol/L·90 min) | Total | 18 | 20,483 (17,574) | 17,868 (22,933) | 20,008 (34,148) | 0.846 |
| Peak concentration (pmol/L) | Total | 18 | 331 (299) | 318 (351) | 348 (401) | 0.411 |
| Plasma insulin by ethnicity | ||||||
| Fasting (pmol/L) | Caucasian | 13 | 7 (22) | 7 (32) | 7 (60) | 0.661 |
| Asian | 5 | 44 (108) | 41 (118) | 43 (83) | 0.627 | |
| p-value 2 | 0.059 | 0.143 | 0.336 | |||
| iAUC (pmol/L·3 h) | Caucasian | 13 | 27,621 (16,297) | 23,526 (16,158) | 24,526 (26,432) | 0.926 |
| Asian | 5 | 59,288 (46,230) | 69,746 (52,200) | 70,866 (44,792) | 0.549 | |
| p-value 2 | 0.003 | 0.019 | 0.007 | |||
| Late phase iAUC (pmol/L·90 min) | Caucasian | 13 | 19,087 (8339) | 16,405 (7799) | 17,745 (14,203) | 0.794 |
| Asian | 5 | 40,609 (37,736) | 50,127 (41,054) | 51,391 (30,825) | 1.000 | |
| p-value 2 | 0.007 | 0.035 | 0.019 | |||
| Peak concentration (pmol/L) | Caucasian | 13 | 283 (114) | 280 (210) | 317 (222) | 0.500 |
| Asian | 5 | 714 (377) | 615 (623) | 708 (521) | 0.819 | |
| p-value 2 | 0.002 | 0.046 | 0.007 | |||
| Plasma insulin by sex | ||||||
| Fasting (pmol/L) | Female | 12 | 27 (36) | 29 (49) | 42 (61) | 0.558 |
| Male | 6 | 7 (40) | 7 (49) | 7 (39) | 0.135 | |
| p-value 2 | 0.291 | 0.291 | 0.213 | |||
| iAUC (pmol/L·3 h) | Female | 12 | 35,679 (31,396) | 30,795 (41,403) | 30,085 (47,116) | 0.920 |
| Male | 6 | 23,880 (23,073) | 20,070 (39,441) | 30,935 (44,374) | 0.607 | |
| p-value 2 | 0.151 | 0.151 | 0.616 | |||
| Late phase iAUC (pmol/L·90 min) | Female | 12 | 20,811 (16,029) | 20,181 (28,424) | 20,008 (33,292) | 0.920 |
| Male | 6 | 16,967 (19,008) | 14,970 (31,963) | 22,825 (34,836) | 0.513 | |
| p-value 2 | 0.250 | 0.291 | 0.616 | |||
| Peak concentration (pmol/L) | Female | 12 | 333 (327) | 377 (339) | 348 (406) | 0.717 |
| Male | 6 | 257 (311) | 219 (481) | 379 (553) | 0.513 | |
| p-value 2 | 0.213 | 0.151 | 0.616 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murray, M.; Dordevic, A.L.; Ryan, L.; Bonham, M.P. A Single-Dose of a Polyphenol-Rich Fucus Vesiculosus Extract is Insufficient to Blunt the Elevated Postprandial Blood Glucose Responses Exhibited by Healthy Adults in the Evening: A Randomised Crossover Trial. Antioxidants 2019, 8, 49. https://doi.org/10.3390/antiox8020049
Murray M, Dordevic AL, Ryan L, Bonham MP. A Single-Dose of a Polyphenol-Rich Fucus Vesiculosus Extract is Insufficient to Blunt the Elevated Postprandial Blood Glucose Responses Exhibited by Healthy Adults in the Evening: A Randomised Crossover Trial. Antioxidants. 2019; 8(2):49. https://doi.org/10.3390/antiox8020049
Chicago/Turabian StyleMurray, Margaret, Aimee L. Dordevic, Lisa Ryan, and Maxine P. Bonham. 2019. "A Single-Dose of a Polyphenol-Rich Fucus Vesiculosus Extract is Insufficient to Blunt the Elevated Postprandial Blood Glucose Responses Exhibited by Healthy Adults in the Evening: A Randomised Crossover Trial" Antioxidants 8, no. 2: 49. https://doi.org/10.3390/antiox8020049
APA StyleMurray, M., Dordevic, A. L., Ryan, L., & Bonham, M. P. (2019). A Single-Dose of a Polyphenol-Rich Fucus Vesiculosus Extract is Insufficient to Blunt the Elevated Postprandial Blood Glucose Responses Exhibited by Healthy Adults in the Evening: A Randomised Crossover Trial. Antioxidants, 8(2), 49. https://doi.org/10.3390/antiox8020049






