Optimization of the Microwave-Assisted Extraction Process of Bioactive Compounds from Annatto Seeds (Bixa orellana L.)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Design
2.3. Characterization of Optimal Annatto Seed Extracts
2.3.1. Total Polyphenol Concentration
2.3.2. Quantification of Bixin
- = 3090 (1 g/100 mL)−1 × 1 cm−1 (specific absorptivity coefficient of bixin in acetone) [20];
- A = Absorbance value of the sample; and
- V = Dilution volume (mL) of the sample.
2.3.3. Antioxidant Activity (ABTS+ Method)
2.3.4. Ferric Reducing Antioxidant Power (FRAP)
2.3.5. Antioxidant Activity (DPPH Method)
2.3.6. Antimicrobial Activity of Annatto Seed Extracts
2.4. Statistical Analysis
3. Results
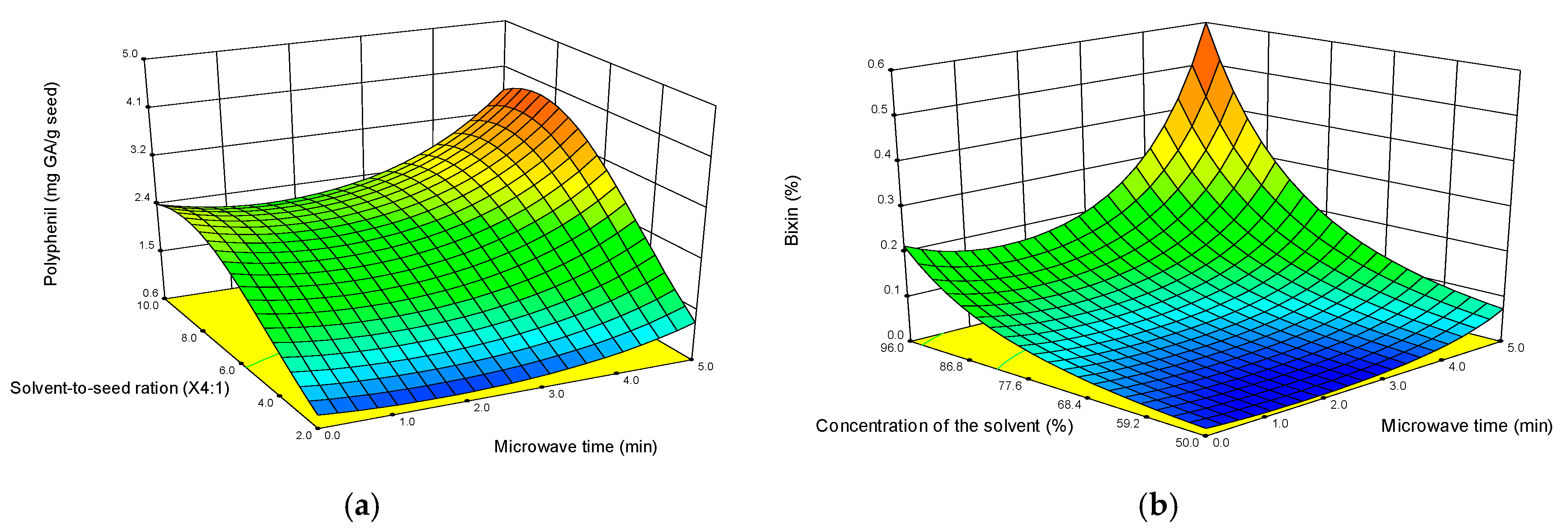
3.1. Experimental Design
3.2. Effect of the MAE on the Antimicrobial and Antioxidant Activities of the Extract
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Viuda, M.; Ciro, G.L.; Ruiz, Y.; Zapata, J.E.; Sendra, E.; Pérez-Álvarez, J.A.; Fernández-López, J. In vitro Antioxidant and Antibacterial Activities of Extracts from Annatto (Bixa orellana L.) Leaves and Seeds. J. Food Saf. 2012, 32, 399–406. [Google Scholar] [CrossRef]
- Zhang, H.; Xue, L.; Li, B.; Tian, H.; Zhang, Z.; Tao, S. Therapeutic potential of bixin in PM2.5 particles-induced lung injury in an Nrf2-dependent manner. Free Radic. Biol. Med. 2018, 126, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Campos, R.; Yamashita, F.; Cesar, F.; Zerlotti, A. Simultaneous extraction and analysis by high performance liquid chromatography coupled to diode array and mass spectrometric detectors of bixin and phenolic compounds from annatto seeds. J. Chromatogr. A 2011, 1218, 57–63. [Google Scholar] [CrossRef]
- Butnariu, M. Methods of Analysis (Extraction, Separation, Identification and Quantification) of Carotenoids from Natural Products. J. Ecosyst. Ecography 2016, 6, 1–19. [Google Scholar] [CrossRef]
- Shahid-ul-Islam, L.J.; Mohammad, F. Phytochemistry, biological activities and potential of annatto in natural colorant production for industrial applications—A review. J. Adv. Res. 2016, 7, 499–514. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Tomita, Y.; Qiu, Y.; He, J.; Morii, E.; Noguchi, S.; Aozasa, K. E74-like factor 2 regulates valosin-containing protein expression. Biochem. Biophys. Res. Commun. 2007, 356, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Yolmeh, M.; Habibi Najafi, M.B.; Farhoosh, R. Optimisation of ultrasound-assisted extraction of natural pigment from annatto seeds by response surface methodology (RSM). Food Chem. 2014, 155, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Lianfu, Z.; Zelong, L. Optimization and comparison of ultrasound/microwave assisted extraction (UMAE) and ultrasonic assisted extraction (UAE) of lycopene from tomatoes. Ultrason. Sonochem. 2008, 15, 731–737. [Google Scholar] [CrossRef]
- Alcázar-Alay, S.C.; Osorio-Tobón, J.F.; Forster-Carneiro, T.; Meireles, M.A.A. Obtaining bixin from semi-defatted annatto seeds by a mechanical method and solvent extraction: Process integration and economic evaluation. Food Res. Int. 2017, 99, 393–402. [Google Scholar] [CrossRef]
- Taham, T.; Silva, D.O.; Barrozo, M.A.S. Improvement of bixin extraction from annatto seeds using a screen-topped spouted bed. Sep. Purif. Technol. 2016, 158, 313–321. [Google Scholar] [CrossRef]
- Rodrigues, L.M.; Alcázar-Alay, S.C.; Petenate, A.J.; Meireles, M.A.A. Bixin extraction from defatted annatto seeds. C. R. Chim. 2014, 17, 268–283. [Google Scholar] [CrossRef]
- Soumya, V.; Venkatesh, P.; Kothandam, H.P. Microwave facilitated extraction of bixin from Bixa orellana and it’s in-vitro antioxidant activity. Der Pharm. Lett. 2010, 2, 479–485. [Google Scholar]
- Zou, T.B.; Jia, Q.; Li, H.W.; Wang, C.X.; Wu, H.F. Response surface methodology for ultrasound-assisted extraction of astaxanthin from Haematococcus pluvialis. Mar. Drugs 2013, 11, 1644–1655. [Google Scholar] [CrossRef] [PubMed]
- Teng, H.; Jo, I.H.; Choi, Y.H. Optimization of ultrasonic-assisted extraction of phenolic compounds from chinese quince (Chaenomeles sinensis) by response surface methodology. J. Appl. Biol. Chem. 2010, 53, 618–625. [Google Scholar] [CrossRef]
- Da Porto, C.; Porretto, E.; Decorti, D. Comparison of ultrasound-assisted extraction with conventional extraction methods of oil and polyphenols from grape (Vitis vinifera L.) seeds. Ultrason. Sonochem. 2013, 20, 1076–1080. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.; Akter, M.S.; Eun, J.B. Optimization conditions for anthocyanin and phenolic content extraction form purple sweet potato using response surface methodology. Int. J. Food Sci. Nutr. 2011, 62, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Ghafoor, K.; Choi, Y.H.; Jeon, J.Y.; Jo, I.H. Optimization of ultrasound-assisted extraction of phenolic compounds, antioxidants, and anthocyanins from grape (Vitis vinifera) seeds. J. Agric. Food Chem. 2009, 57, 4988–4994. [Google Scholar] [CrossRef]
- Barbosa, M.; Borsarelli, C.; Mercadante, A. Light stability of spray-dried bixin encapsulated with different edible polysaccharide preparations. Food Res. Int. 2005, 38, 989–994. [Google Scholar] [CrossRef]
- Swain, T.; Hillis, W.E. The phenolic constituents of Prunus domestica. I.—The quantitative analysis of phenolic constituents. J. Sci. Food Agric. 1959, 10, 63–68. [Google Scholar] [CrossRef]
- FAO/WHO. Compendium of Food Additive Specifications; FAO: Rome, Italy, 2006; Volume 67, pp. 3–4. ISBN 9251055599. [Google Scholar]
- Contreras-Calderón, J.; Calderón-Jaimes, L.; Guerra-Hernández, E.; García-Villanova, B. Antioxidant capacity, phenolic content and vitamin C in pulp, peel and seed from 24 exotic fruits from Colombia. Food Res. Int. 2011, 44, 2047–2053. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Nurhuda, H.H.; Maskat, M.Y.; Mamot, S.; Afiq, J.; Aminah, A. Effect of blanching on enzyme and antioxidant activities of rambutan (Nephelium lappaceum) peel. Int. Food Res. J. 2013, 20, 1725–1730. [Google Scholar]
- Quanhong, L.; Caili, F. Application of response surface methodology for extraction optimization of germinant pumpkin seeds protein. Food Chem. 2005, 92, 701–706. [Google Scholar] [CrossRef]
- Sinha, K.; Chowdhury, S.; Saha, P.D.; Datta, S. Modeling of microwave-assisted extraction of natural dye from seeds of Bixa orellana (Annatto) using response surface methodology (RSM) and artificial neural network (ANN). Ind. Crops Prod. 2013, 41, 165–171. [Google Scholar] [CrossRef]
- Da Costa, C.L.S.; Chaves, M.H. Extração de pigmentos das sementes de Bixa orellana L.: Uma alternativa para disciplinas experimentais de química orgânica. Quim. Nova 2005, 28, 149–152. [Google Scholar] [CrossRef]
- Novoa Vidal, A.; Motidome, M.; Mancini-filho, J.; Linares Fallarero, A.; Tanae, M.; Torres Brando, M.L.; Lapa, A.J. Actividad antioxidante y ácidos fenólicos del alga marina Bryothamnion triquetrum (S.G. Gmelim) Howe. Rev. Bras. Cienc. Farm. 2001, 37, 373–382. [Google Scholar]
- Rubio-Senent, F.; Fernández-Bolaños, J.; García-Borrego, A.; Lama-Muñoz, A.; Rodríguez-Gutiérrez, G. Influence of pH on the antioxidant phenols solubilised from hydrothermally treated olive oil by-product (alperujo). Food Chem. 2017, 219, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.L.; Xu, M.; Wang, Y.M.; Wu, D.M.; Chen, J.H. Optimizing ultrasonic ellagic acid extraction conditions from infructescence of platycarya strobilacea using response surface methodology. Molecules 2010, 15, 7923–7932. [Google Scholar] [CrossRef]
- Yolmeh, M.; Habibi-najafi, M.B.; Shakouri, S.; Hosseini, F.; Resource, N.; Branch, S. Comparing Antibacterial and Antioxidant Activity of Annatto Dye Extracted by Conventional and Ultrasound-Assisted Methods. Zahedan J. Res. Med. Sci. 2015, 17, 29–33. [Google Scholar] [CrossRef]
| Independent Variables | Symbol | Coded Levels | ||
|---|---|---|---|---|
| −1 | 0 | +1 | ||
| Treatment time (min) | X1 | 0 | 2.5 | 5 |
| pH | X2 | 4 | 7.5 | 11 |
| Solvent concentration (ethanol) (%) | X3 | 50 | 73 | 96 |
| Solvent-to-seed ratio (X4:1) | X4 | 2 | 6 | 10 |
| Treatment Number | MAE (min) | pH | Solvent Concentration (Ethanol) (%) | Solvent-to-Seed Ratio (X4:1) | MAE | |
|---|---|---|---|---|---|---|
| Polyphenols (mg GA/g Seed) n = 3 | Bixin (%) n = 3 | |||||
| 1 | 2.5 | 7.5 | 73.0 | 10.0 | 2.83 ± 0.04 | 0.13 ± 0.00 |
| 2 | 2.5 | 4.0 | 50.0 | 6.0 | 1.53 ± 0.22 | 0.03 ± 0.00 |
| 3 | 2.5 | 7.5 | 50.0 | 10.0 | 2.85 ± 0.19 | 0.05 ± 0.00 |
| 4 | 5.0 | 4.0 | 96.0 | 6.0 | 1.96 ± 0.06 | 0.33 ± 0.04 |
| 5 | 2.5 | 7.5 | 73.0 | 6.0 | 1.96 ± 0.15 | 0.05 ± 0.01 |
| 6 | 5.0 | 11.0 | 73.0 | 6.0 | 2.01 ± 0.08 | 0.13 ± 0.02 |
| 7 | 5.0 | 11.0 | 73.0 | 6.0 | 1.35 ± 0.05 | 0.32 ± 0.01 |
| 8 | 2.5 | 11.0 | 96.0 | 6.0 | 3.94 ± 0.00 | 0.51 ± 0.01 |
| 9 | 5.0 | 7.5 | 50.0 | 6.0 | 3.79± 0.07 | 0.04 ± 0.01 |
| 10 | 2.5 | 7.5 | 50.0 | 2.0 | 0.53 ± 0.08 | 0.27 ± 0.03 |
| 11 | 2.5 | 4.0 | 73.0 | 6.0 | 4.16 ± 0.24 | 0.19 ± 0.00 |
| 12 | 2.5 | 4.0 | 73.0 | 2.0 | 0.83 ± 0.06 | 0.12 ± 0.01 |
| 13 | 2.5 | 7.5 | 73.0 | 6.0 | 1.89 ± 0.14 | 0.05 ± 0.01 |
| 14 | 2.5 | 7.5 | 96.0 | 6.0 | 1.72 ± 0.15 | 0.19 ± 0.00 |
| 15 | 2.5 | 11.0 | 73.0 | 10.0 | 3.25 ± 0.57 | 0.04 ± 0.01 |
| 16 | 2.5 | 11.0 | 50.0 | 6.0 | 4.36 ± 0.04 | 0.03 ± 0.00 |
| 17 | 2.5 | 7.5 | 96.0 | 6.0 | 2.43 ± 0.01 | 0.58 ± 0.00 |
| 18 | 2.5 | 7.5 | 96.0 | 2.0 | 0.66 ± 0.01 | 0.38 ± 0.03 |
| 19 | 0.0 | 7.5 | 73.0 | 6.0 | 1.96 ± 0.08 | 0.06 ± 0.01 |
| 20 | 5.0 | 7.5 | 96.0 | 10.0 | 2.10 ± 0.01 | 0.13 ± 0.00 |
| 21 | 2.5 | 7.5 | 73.0 | 2.0 | 0.70 ± 0.01 | 0.07 ± 0.02 |
| 22 | 0.0 | 7.5 | 73.0 | 6.0 | 1.90 ± 0.23 | 0.04 ± 0.00 |
| 23 | 2.5 | 11.0 | 73.0 | 2.0 | 0.68 ± 0.03 | 0.17 ± 0.01 |
| 24 | 0.0 | 7.5 | 73.0 | 10.0 | 2.15 ± 0.16 | 0.10 ± 0.01 |
| 25 | 2.5 | 4.0 | 73.0 | 6.0 | 2.28 ± 0.01 | 0.12 ± 0.03 |
| 26 | 0.0 | 7.5 | 73.0 | 6.0 | 2.02 ± 0.03 | 0.08 ± 0.00 |
| 27 | 0.0 | 7.5 | 50.0 | 6.0 | 3.90 ± 0.30 | 0.08 ± 0.00 |
| 28 | 2.5 | 7.5 | 73.0 | 2.0 | 1.57 ± 0.30 | 0.25 ± 0.01 |
| 29 | 0.0 | 4.0 | 73.0 | 10.0 | 1.96 ± 0.15 | 0.33 ± 0.01 |
| 30 | 5.0 | 7.5 | 73.0 | 6.0 | 1.91 ± 0.18 | 0.06 ± 0.02 |
| Variable | Polyphenols (mg GA/g Seed) | Bixin (%) |
|---|---|---|
| p-Value | p-Value | |
| Model | <0.0001 | <0.0001 |
| X1, treatment time (min) | <0.0001 | <0.0001 |
| X2, pH | 0.532 | 0.112 |
| X3, solvent concentration (%) | <0.0001 | <0.0001 |
| X4, solvent-to-seed ratio (X4:1) | <0.0001 | 0.677 |
| X2X3 | 0.001 | ˃0.050 |
| X2X4 | 0.044 | 0.001 |
| X3X4 | ˃0.050 | 0.004 |
| X2X2 | ˃0.050 | <0.0001 |
| X4X4 | <0.0001 | ˃0.050 |
| X1X1 | <0.0001 | <0.0001 |
| Lack of fit | 0.0405 | 0.137 |
| r2 | 0.931 | 0.914 |
| r2-adj | 0.901 | 0.875 |
| pH | Solvent (Ethanol) (%) | Solvent-to-Seed Ratio (X4:1) | Treatment Time (min) | Polyphenol (mg GA/g Seed) | Bixin (%) |
|---|---|---|---|---|---|
| 7.00 | 96 | 5.95 | 5.00 | 2.69 | 0.58 |
| Experimental result | 3.08 ± 0.01 | 0.58 ± 0.02 | |||
| Relative error | −0.39 | 0.00 | |||
| Absolute error (%) | 14.41 | 0.55 | |||
| Extract | MAE | Leaching | |
|---|---|---|---|
| Bixin | (%) | 0.576 b ± 0.015 | 0.165 a ± 0.002 |
| Polyphenols | (mg GA/g seed) | 3.078 b ± 0.012 | 0.343 a ± 0.003 |
| ABTS | (µM Trolox/L extract) | 577 b ± 5 | 174 a ± 8 |
| FRAP | (µM Trolox/L extract) | 316 b ± 10 | 127 a ± 2 |
| DPPH | (µM Trolox/L extract) | 1043 b ± 50 | 811 a ± 5 |
| Bacillus cereus | pH 11 (mg/L) | 16 a | 128 b |
| pH 7 (mg/L) | 16 a | 128 b | |
| pH 4 (mg/L) | 16 a | 128 b | |
| Staphylococcus aureus | pH 11 (mg/L) | 8 a | 32 b |
| pH 7 (mg/L) | 8 a | 32 b | |
| pH 4 (mg/L) | 8 a | 32 b | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quintero Quiroz, J.; Celis Torres, A.; Muñoz Ramirez, L.; Silva Garcia, M.; Ciro Gomez, G.; Rojas Camargo, J. Optimization of the Microwave-Assisted Extraction Process of Bioactive Compounds from Annatto Seeds (Bixa orellana L.). Antioxidants 2019, 8, 37. https://doi.org/10.3390/antiox8020037
Quintero Quiroz J, Celis Torres A, Muñoz Ramirez L, Silva Garcia M, Ciro Gomez G, Rojas Camargo J. Optimization of the Microwave-Assisted Extraction Process of Bioactive Compounds from Annatto Seeds (Bixa orellana L.). Antioxidants. 2019; 8(2):37. https://doi.org/10.3390/antiox8020037
Chicago/Turabian StyleQuintero Quiroz, Julian, Angélica Celis Torres, Luisa Muñoz Ramirez, Mariluz Silva Garcia, Gelmy Ciro Gomez, and John Rojas Camargo. 2019. "Optimization of the Microwave-Assisted Extraction Process of Bioactive Compounds from Annatto Seeds (Bixa orellana L.)" Antioxidants 8, no. 2: 37. https://doi.org/10.3390/antiox8020037
APA StyleQuintero Quiroz, J., Celis Torres, A., Muñoz Ramirez, L., Silva Garcia, M., Ciro Gomez, G., & Rojas Camargo, J. (2019). Optimization of the Microwave-Assisted Extraction Process of Bioactive Compounds from Annatto Seeds (Bixa orellana L.). Antioxidants, 8(2), 37. https://doi.org/10.3390/antiox8020037




