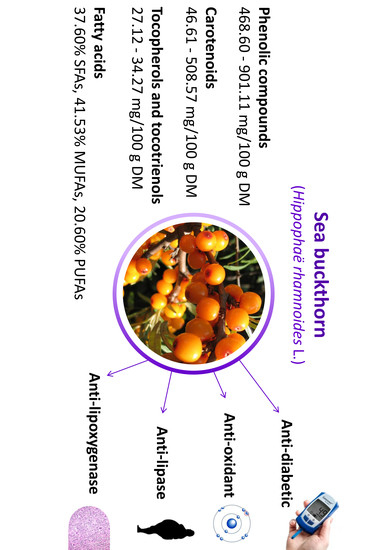
Anti-Oxidant and Anti-Enzymatic Activities of Sea Buckthorn (Hippophaë rhamnoides L.) Fruits Modulated by Chemical Components
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Plant Materials
2.3. Basic Chemical Composition
2.4. Analysis of Phenolic Compounds
2.5. Analysis of Carotenoids, Tocopherols, and Tocotrienols
2.6. Analysis of Fatty Acids
2.7. Determintation of Biological Activity: Anti-Oxidant, Anti-α-Amylase, Anti- α-Glucosidase, Anti-Lipase, and Anti-Lipoxygenase
2.8. Statistical Analysis
3. Results and Discussion
3.1. Basic Chemical Composition of Sea Buckthorn Cultivars
3.2. Analysis of Phenolic Compounds of Sea Buckthorn Cultivars
3.3. Analysis of Carotenoids, Tocopherols, and Tocotrienols of Sea Buckthorn Cultivars
3.4. Analysis of Fatty Acids of Sea Buckthorn Cultivars
3.5. Analysis of Biological Activity of Sea Buckthorn Cultivars: Anti-Oxidant, Anti-α-Amylase, Anti-α-Glucosidase, Anti-Lipase, and Anti-Lipoxygenase Effects
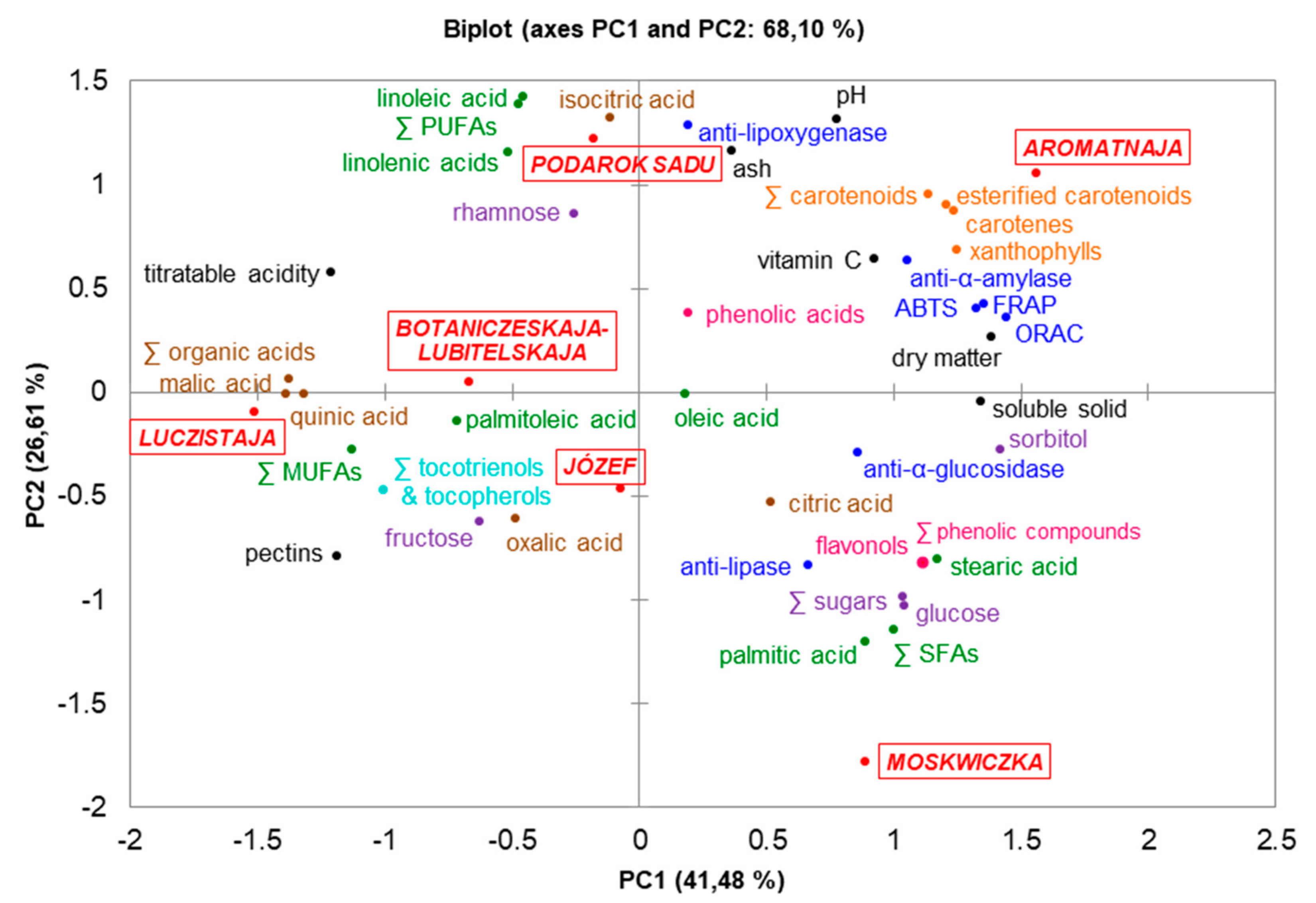
3.6. Pearson’s Correlation and Principal Component Analysis (PCA)
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bal, L.M.; Meda, V.; Naik, S.; Satya, S. Sea buckthorn berries: A potential source of valuable nutrients for nutraceuticals and cosmoceuticals. Food Res. Int. 2011, 44, 1718–1727. [Google Scholar] [CrossRef]
- Rafalska, A.; Abramowicz, K.; Krauze, M. Sea buckthorn (Hippophaë rhamnoides L.) as a plant for universal application. World Sci. News 2017, 72, 123–140. [Google Scholar]
- Ruan, C.-J.; Rumpunen, K.; Nybom, H. Advances in improvement of quality and resistance in a multipurpose crop: Sea buckthorn. Crit. Rev. Biotechnol. 2013, 33, 126–144. [Google Scholar] [CrossRef] [PubMed]
- Piłat, B.; Bieniek, A.; Zadernowski, R. Common sea buckthorn (Hippophaë rhamnoides L.) as an alternative orchard plant. Pol. J. Nat. Sci. 2015, 30, 417–430. [Google Scholar]
- Arimboor, R.; Kumar, K.S.; Arumughan, C. Simultaneous estimation of phenolic acids in sea buckthorn (Hippophaë rhamnoides) using RP-HPLC with DAD. J. Pharm. Biomed. Anal. 2008, 47, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Andersson, S.C.; Rumpunen, K.; Johansson, E.; Olsson, M.E. Tocopherols and tocotrienols in sea buckthorn (Hippophaë rhamnoides L.) berries during ripening. J. Agric. Food Chem. 2008, 56, 6701–6706. [Google Scholar] [CrossRef]
- Guo, R.; Guo, X.; Li, T.; Fu, X.; Liu, R.H. Comparative assessment of phytochemical profiles, antioxidant and antiproliferative activities of Sea buckthorn (Hippophaë rhamnoides L.) berries. Food Chem. 2017, 221, 997–1003. [Google Scholar] [CrossRef]
- Dulf, F.V. Fatty acids in berry lipids of six sea buckthorn (Hippophaë rhamnoides L., subspecies carpatica) cultivars grown in Romania. Chem. Cent. J. 2012, 6, 106. [Google Scholar] [CrossRef]
- Kim, J.-S.; Kwon, Y.-S.; Sa, Y.-J.; Kim, M.-J. Isolation and identification of sea buckthorn (Hippophaë rhamnoides) phenolics with antioxidant activity and α-glucosidase inhibitory effect. J. Agric. Food Chem. 2010, 59, 138–144. [Google Scholar] [CrossRef]
- Patel, C.A.; Divakar, K.; Santani, D.; Solanki, H.K.; Thakkar, J.H. Remedial prospective of Hippophaë rhamnoides Linn.(sea buckthorn). ISRN Pharmacol. 2012, 2012. [Google Scholar] [CrossRef]
- Andersson, S.C.; Olsson, M.E.; Johansson, E.; Rumpunen, K. Carotenoids in sea buckthorn (Hippophaë rhamnoides L.) berries during ripening and use of pheophytin a as a maturity marker. J. Agric. Food Chem. 2008, 57, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Teleszko, M.; Wojdyło, A.; Rudzińska, M.; Oszmiański, J.; Golis, T. Analysis of lipophilic and hydrophilic bioactive compounds content in sea buckthorn (Hippophaë rhamnoides L.) berries. J. Agric. Food Chem. 2015, 63, 4120–4129. [Google Scholar] [CrossRef] [PubMed]
- Chung, L.Y.; Soo, W.K.; Chan, K.Y.; Mustafa, M.R.; Goh, S.H.; Imiyabir, Z. Lipoxygenase inhibiting activity of some Malaysian plants. Pharm. Biol. 2009, 47, 1142–1148. [Google Scholar] [CrossRef]
- Pijanowski, E.; Mrożewski, S.; Horubała, A.; Jarczyk, A. Technologia Produktów Owocowych i Warzywnych, 3rd ed.; PWRiL: Warszawa, Poland, 1973. [Google Scholar]
- Wojdyło, A.; Nowicka, P.; Bąbelewski, P. Phenolic and carotenoid profile of new goji cultivars and their anti-hyperglycemic, anti-aging and antioxidant properties. J. Funct. Foods 2018, 48, 632–642. [Google Scholar] [CrossRef]
- Nowicka, P.; Wojdyło, A.; Laskowski, P. Inhibitory potential against digestive enzymes linked to obesity and type 2 diabetes and content of bioactive compounds in 20 cultivars of the peach fruit grown in Poland. Plant Foods Hum. Nutr. 2018, 73, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Nowacki, D.; Martynowicz, H.; Skoczyńska, A.; Wojakowska, A.; Turczyn, B.; Bobak, Ł.; Trziszka, T.; Szuba, A. Lecithin derived from ω-3 PUFA fortified eggs decreases blood pressure in spontaneously hypertensive rats. Sci. Rep. 2017, 7, 12373. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Ou, B.; Huang, D.; Hampsch-Woodill, M.; Flanagan, J.A.; Deemer, E.K. Analysis of antioxidant activities of common vegetables employing oxygen radical absorbance capacity (ORAC) and ferric reducing antioxidant power (FRAP) assays: A comparative study. J. Agric. Food Chem. 2002, 50, 3122–3128. [Google Scholar] [CrossRef]
- Podsędek, A.; Majewska, I.; Redzynia, M.; Sosnowska, D.; Koziołkiewicz, M. In vitro inhibitory effect on digestive enzymes and antioxidant potential of commonly consumed fruits. J. Agric. Food Chem. 2014, 62, 4610–4617. [Google Scholar] [CrossRef]
- Zheng, J.; Yang, B.; Trépanier, M.; Kallio, H. Effects of genotype, latitude, and weather conditions on the composition of sugars, sugar alcohols, fruit acids, and ascorbic acid in sea buckthorn (Hippophaë rhamnoides ssp. mongolica) berry juice. J. Agric. Food Chem. 2012, 60, 3180–3189. [Google Scholar] [CrossRef]
- Tiitinen, K.M.; Hakala, M.A.; Kallio, H.P. Quality components of sea buckthorn (Hippophaë rhamnoides) varieties. J. Agric. Food Chem. 2005, 53, 1692–1699. [Google Scholar] [CrossRef]
- Yang, B. Sugars, acids, ethyl β-D-glucopyranose and a methyl inositol in sea buckthorn (Hippophaë rhamnoides) berries. Food Chem. 2009, 112, 89–97. [Google Scholar] [CrossRef]
- Kawecki, Z.; Szalkiewicz, M.; Bieniek, A. The common sea buckthorn-a valuable fruit. J. Fruit Ornam. Plant Res. 2004, 12, 183–193. [Google Scholar]
- Rösch, D.; Bergmann, M.; Knorr, D.; Kroh, L.W. Structure−antioxidant efficiency relationships of phenolic compounds and their contribution to the antioxidant activity of sea buckthorn juice. J. Agric. Food Chem. 2003, 51, 4233–4239. [Google Scholar] [CrossRef] [PubMed]
- Kallio, H.; Yang, B.; Peippo, P. Effects of different origins and harvesting time on vitamin C, tocopherols, and tocotrienols in sea buckthorn (Hippophaë rhamnoides) berries. J. Agric. Food Chem. 2002, 50, 6136–6142. [Google Scholar] [CrossRef] [PubMed]
- Olsson, M.E.; Gustavsson, K.-E.; Andersson, S.; Nilsson, Å.; Duan, R.-D. Inhibition of cancer cell proliferation in vitro by fruit and berry extracts and correlations with antioxidant levels. J. Agric. Food Chem. 2004, 52, 7264–7271. [Google Scholar] [CrossRef] [PubMed]
- Zadernowski, R.; Naczk, M.; Czaplicki, S.; Rubinskiene, M.; Szałkiewicz, M. Composition of phenolic acids in sea buckthorn (Hippophaë rhamnoides L.) berries. J. Am. Oil Chem. Soc. 2005, 82, 175–179. [Google Scholar] [CrossRef]
- Pop, R.M.; Socaciu, C.; Pintea, A.; Buzoianu, A.D.; Sanders, M.G.; Gruppen, H.; Vincken, J.P. UHPLC/PDA–ESI/MS analysis of the main berry and leaf flavonol glycosides from different Carpathian Hippophaë rhamnoides L. varieties. Phytochem. Anal. 2013, 24, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Laaksonen, O.; Zheng, J.; Yang, W.; Trépanier, M.; Kallio, H.; Yang, B. Flavonol glycosides in berries of two major subspecies of sea buckthorn (Hippophaë rhamnoides L.) and influence of growth sites. Food Chem. 2016, 200, 189–198. [Google Scholar] [CrossRef]
- Chen, C.; Zhang, H.; Xiao, W.; Yong, Z.-P.; Bai, N. High-performance liquid chromatographic fingerprint analysis for different origins of sea buckthorn berries. J. Chromatogr. A 2007, 1154, 250–259. [Google Scholar] [CrossRef]
- Zheng, J.; Kallio, H.; Yang, B. Sea buckthorn (Hippophaë rhamnoides ssp. rhamnoides) berries in Nordic environment: Compositional response to latitude and weather conditions. J. Agric. Food Chem. 2016, 64, 5031–5044. [Google Scholar] [CrossRef] [PubMed]
- Pop, R.M.; Weesepoel, Y.; Socaciu, C.; Pintea, A.; Vincken, J.-P.; Gruppen, H. Carotenoid composition of berries and leaves from six Romanian sea buckthorn (Hippophaë rhamnoides L.) varieties. Food Chem. 2014, 147, 1–9. [Google Scholar] [CrossRef]
- Kruczek, M.; Swiderski, A.; Mech-Nowak, A.; Król, K. Antioxidant capacity of crude extracts containing carotenoids from the berries of various cultivars of Sea buckthorn (Hippophaë rhamnoides L.). Acta Biochim. Pol. 2012, 59. [Google Scholar] [CrossRef]
- Yang, B.; Kallio, H.P. Fatty acid composition of lipids in sea buckthorn (Hippophaë rhamnoides L.) berries of different origins. J. Agric. Food Chem. 2001, 49, 1939–1947. [Google Scholar] [CrossRef]
- Vescan, L.A.; Pamfil, D.; Constantin, B.; Matea, C.; Sisea, C.R. Several lipophilic components of five elite genotypes of Romanian seabuckthorn (Hippophaë rhamnoides subs. carpatica). Not. Bot. Horti Agrobot. Cluj-Napoca 2010, 38, 114–122. [Google Scholar] [CrossRef]
- Sharma, U.K.; Sharma, K.; Sharma, N.; Sharma, A.; Singh, H.P.; Sinha, A.K. Microwave-assisted efficient extraction of different parts of Hippophaë rhamnoides for the comparative evaluation of antioxidant activity and quantification of its phenolic constituents by reverse-phase high-performance liquid chromatography (RP-HPLC). J. Agric. Food Chem. 2007, 56, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Ohlander, M.; Jeppsson, N.; Björk, L.; Trajkovski, V. Changes in antioxidant effects and their relationship to phytonutrients in fruits of sea buckthorn (Hippophaë rhamnoides L.) during maturation. J. Agric. Food Chem. 2000, 48, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- Lehtonen, H.; Järvinen, R.; Linderborg, K.; Viitanen, M.; Venojärvi, M.; Alanko, H.; Kallio, H. Postprandial hyperglycemia and insulin response are affected by sea buckthorn (Hippophaë rhamnoides ssp. turkestanica) berry and its ethanol-soluble metabolites. Eur. J. Clin. Nutr. 2010, 64, 1465. [Google Scholar] [CrossRef]
- Mortensen, M.W.; Spagner, C.; Cuparencu, C.; Astrup, A.; Raben, A.; Dragsted, L.O. Sea buckthorn decreases and delays insulin response and improves glycaemic profile following a sucrose-containing berry meal: A randomised, controlled, crossover study of Danish sea buckthorn and strawberries in overweight and obese male subjects. Eur. J. Nutr. 2018, 57, 2827–2837. [Google Scholar] [CrossRef]
- Xue, Y.; Miao, Q.; Zhao, A.; Zheng, Y.; Zhang, Y.; Wang, P.; Kallio, H.; Yang, B. Effects of sea buckthorn (Hippophaë rhamnoides) juice and L-quebrachitol on type 2 diabetes mellitus in db/db mice. J. Funct. Foods 2015, 16, 223–233. [Google Scholar] [CrossRef]
- Linderborg, K.M.; Lehtonen, H.-M.; Järvinen, R.; Viitanen, M.; Kallio, H. The fibres and polyphenols in sea buckthorn (Hippophaë rhamnoides) extraction residues delay postprandial lipemia. Int. J. Food Sci. Nutr. 2012, 63, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Zadernowski, R.; Naczk, M.; Nowak-Polakowska, H.; Nesterowicz, J. Effect of sea buckthorn (Hippophaë rhamnoides L.) berry extracts on the activity of lipase and lipoxygenase. J. Food Lipids 2002, 9, 249–258. [Google Scholar] [CrossRef]
- Mudgil, D.; Barak, S. Composition, properties and health benefits of indigestible carbohydrate polymers as dietary fiber: A review. Int. J. Biol. Macromol. 2013, 61, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Weickert, M.O.; Pfeiffer, A.F. Impact of dietary fiber consumption on insulin resistance and the prevention of type 2 diabetes. J. Nutr. 2018, 148, 7–12. [Google Scholar] [CrossRef]
- Wang, C.; Chang, S.; Inbaraj, B.S.; Chen, B. Isolation of carotenoids, flavonoids and polysaccharides from Lycium barbarum L. and evaluation of antioxidant activity. Food Chem. 2010, 120, 184–192. [Google Scholar] [CrossRef]
- Wojdyło, A.; Nowicka, P.; Carbonell-Barrachina, Á.A.; Hernández, F. Phenolic compounds, antioxidant and antidiabetic activity of different cultivars of Ficus carica L. fruits. J. Funct. Foods 2016, 25, 421–432. [Google Scholar] [CrossRef]
- Ado, M.; Abas, F.; Mohammed, A.; Ghazali, H. Anti-and pro-lipase activity of selected medicinal, herbal and aquatic plants, and structure elucidation of an anti-lipase compound. Molecules 2013, 18, 14651–14669. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Antioxidant activity of carotenoids. Mol. Asp. Med. 2003, 24, 345–351. [Google Scholar] [CrossRef]

| Components | Aromatnaja | Botaniczeskaja- -Lubitelskaja | Józef | Luczistaja | Moskwiczka | Podarok Sadu |
|---|---|---|---|---|---|---|
| Dry matter (%) | 13.08 ± 0.23 a | 11.88 ± 0.44 b | 12.03 ± 0.38 b | 11.78 ± 0.19 b | 12.84 ± 0.34 ab | 12.71 ± 0.40 ab |
| Soluble solid (°Bx) | 7.2 ± 0.01 a | 6.4 ± 0.00 a | 7.1 ± 0.01 a | 5.7 ± 0.01 a | 7.2 ± 0.01 a | 7.0 ± 0.00 a |
| pH | 2.95 ± 0.01 a | 2.90 ± 0.00 a | 2.90 ± 0.01 a | 2.90 ± 0.02 a | 2.89 ± 0.00 a | 2.93 ± 0.01 a |
| Titratable acidity (g malic acid/100 g) FW) | 2.44 ± 0.01 c | 2.62 ± 0.00 b | 2.59 ± 0.08 b | 2.71 ± 0.11 ab | 2.48 ± 0.03 bc | 2.79 ± 0.00 a |
| Ash (%) | 0.43 ± 0.01 a | 0.39 ± 0.01 a | 0.40 ± 0.02 a | 0.35 ± 0.04 a | 0.31 ± 0.01 a | 0.38 ± 0.05 a |
| Pectins (%) | 0.34 ± 0.04 b | 0.67 ± 0.16 a | 0.21 ± 0.17 c | 0.68 ± 0.18 a | 0.64 ± 0.06 a | 0.58 ± 0.18 ab |
| Vitamin C (mg/100 g FW) | 158.81 ± 0.78 a | 78.52 ± 0.64 b | 64.92 ± 1.00 d | 80.93 ± 2.32 b | 71.32 ± 3.67 c | 61.02 ± 0.21 d |
| Sugars (g/100 g FW) | ||||||
| Rhamnose | 0.05 ± 0.02 b | nd | 0.04 ± 0.01 c | 0.06 ± 0.00 a | nd | 0.04 ± 0.02 c |
| Fructose | 0.05 ± 0.00 d | 0.04 ± 0.02 e | 0.08 ± 0.02 b | 0.10 ± 0.02 a | 0.08 ± 0.02 b | 0.07 ± 0.01 c |
| Sorbitol | 0.14 ± 0.01 a | 0.07 ± 0.00 c | 0.11 ± 0.02 b | 0.08 ± 0.00 c | 0.13 ± 0.04 a | 0.09 ± 0.02 bc |
| Glucose | 1.96 ± 0.02 b | 1.21 ± 0.02 d | 1.73 ± 0.14 bc | 1.49 ± 0.28 c | 2.66 ± 0.65 a | 1.29 ± 0.08 cd |
| ∑ sugars | 2.20 ± 0.01 b | 1.34 ± 0.03 d | 1.96 ± 0.19 c | 1.70 ± 0.25 cd | 2.87 ± 0.71 a | 1.49 ± 0.13 cd |
| Organic acids (g/100 g FW) | ||||||
| Oxalic acid | 0.01 ± 0.00 b | 0.01 ± 0.00 b | 0.02 ± 0.00 a | 0.02 ± 0.01 a | 0.02 ± 0.02 a | 0.02 ± 0.00 a |
| Citric acid | 0.05 ± 0.00 b | 0.01 ± 0.00 d | 0.10 ± 0.05 a | 0.03 ± 0.01 c | 0.05 ± 0.01 b | 0.02 ± 0.00 cd |
| Isocitric acid | 0.21 ± 0.0 a | 0.11 ± 0.00 a | 0.02 ± 0.00 b | 0.17 ± 0.01 a | nd | 0.16 ± 0.00 a |
| Malic acid | 0.96 ± 0.07 cd | 1.95 ± 0.00 b | 1.84 ± 0.09 b | 2.87 ± 0.21 a | 0.82 ± 0.03 d | 1.17 ± 0.13 c |
| Quinic acid | 0.09 ± 0.19 c | 0.99 ± 0.03 a | 0.27 ± 0.15 b | 1.14 ± 0.22 a | 0.07 ± 0.01 c | 0.16 ± 0.09 c |
| ∑ organic acids | 1.33 ± 0.11 d | 3.09 ± 0.27 b | 2.25 ± 0.20 c | 4.22 ± 0.13 a | 0.96 ± 0.13 e | 1.54 ± 0.14 d |
| Sugar: organic acid ratio | 1.65 b | 0.43 d | 0.87 c | 0.40 d | 2.99 a | 0.97 c |
| Phenolic compounds (mg/100 g DM) | ||||||
| Phenolic acids | 6.11 ± 1.98 c | 5.18 ± 1.52 d | 6.11 ± 1.88 c | 5.46 ± 1.07 d | 7.19 ± 2.52 b | 8.94 ± 2.74 a |
| Flavonols | 655.21 ± 46.16 b | 484.22 ± 24.80 c | 691.45 ± 56.36 b | 463.14 ± 30.48 c | 893.92 ± 54.96 a | 637.22 ± 42.75 b |
| ∑ phenolic compounds | 661.32 ± 48.14 b | 491.20 ± 26.71 c | 697.56 ± 58.34 b | 468.60 ± 31.55 c | 901.11 ± 57.48 a | 646.16 ± 45.52 b |
| Carotenoids (mg/100 g DM) | ||||||
| Xanthophylls | 80.73 ± 10.22 a | 45.71 ± 4.62 cd | 65.04 ± 5.33 b | 37.76 ± 4.77 d | 51.13 ± 7.18 c | 60.27 ± 8.11 b |
| Carotenes | 225.42 ± 12.27 a | 16.03 ± 2.93 e | 69.78 ± 4.60 c | 8.85 ± 1.52 f | 56.18 ± 4.27 d | 115.62 ± 8.11 b |
| Esterified carotenoids | 202.42 ± 7.02 a | nd | 23.11 ± 2.04 c | nd | 14.78 ± 1.35 d | 70.37 ± 3.20 b |
| ∑ carotenoids | 508.57 ± 29.54 a | 61.73 ± 7.55 e | 157.93 ± 11.97 c | 46.61 ± 6.29 e | 122.09 ± 12.80 d | 246.26 ± 19.42 b |
| ∑ tocopherols and tocotrienols (mg/100 g DM) | 27.12 ± 1.31 b | 27.68 ± 1.42 b | 28.23 ± 1.39 b | 34.27 ± 2.00 a | 29.29 ± 1.78 b | 27.58 ± 1.55 b |
| Fatty acids (%) | ||||||
| Palmitic (C16:0) | 34.29 ± 0.01 b | 33.24 ± 0.01 b | 33.45 ± 0.01 b | 32.82 ± 0.01 b | 38.19 ± 0.01 a | 32.00 ± 0.01 b |
| Palmitoleic (C16:1 n-7) | 25.84 ± 0.02 b | 23.67 ± 0.01 b | 25.87 ± 0.01 b | 31.25 ± 0.01 a | 26.40 ± 0.01 b | 26.17 ± 0.01 b |
| Stearic (C18:0) | 4.14 ± 0.01 a | 3.71 ± 0.01 ab | 3.89 ± 0.01 a | 2.65 ± 0.01 b | 4.49 ± 0.01 a | 2.72 ± 0.01 b |
| Oleic (C18:1 n-9) | 14.90 ± 0.01 ab | 17.83 ± 0.01 a | 15.03 ± 0.01 ab | 12.91 ± 0.01 b | 14.81 ± 0.01 b | 14.49 ± 0.01 b |
| Linoleic (C18:2 n-6) | 17.42 ± 0.03 b | 17.60 ± 0.01 b | 16.74 ± 0.01 b | 16.93 ± 0.01 b | 13.16 ± 0.01 c | 20.13 ± 0.01 a |
| Linolenic (C18:3 n-3) | 3.43 ± 0.01 b | 3.94 ± 0.01 ab | 3.40 ± 0.01 b | 3.44 ± 0.01 b | 2.95 ± 0.01 c | 4.49 ± 0.01 a |
| ∑SFAs | 38.42 ± 0.04 b | 36.95 ± 0.02 c | 37.34 ± 0.01 bc | 35.47 ± 0.02 c | 42.68 ± 0.02 a | 34.71 ± 0.01 c |
| ∑MUFAs | 40.73 ± 0.05 b | 41.50 ± 0.05 b | 40.90 ± 0.06 b | 44.16 ± 0.04 a | 41.21 ± 0.04 b | 40.66 ± 0.01 b |
| ∑PUFAs | 20.84 ± 0.03 b | 21.54 ± 0.04 b | 20.14 ± 0.01 b | 20.37 ± 0.02 b | 16.11 ± 0.01 c | 24.62 ± 0.01 a |
| Properties | Aromatnaja | Botaniczeskaja- -Lubitelskaja | Józef | Luczistaja | Moskwiczka | Podarok Sadu |
|---|---|---|---|---|---|---|
| Anti-oxidant activity | ||||||
| ABTS | 3.58 ± 0.36 a | 1.27 ± 0.10 d | 1.12 ± 0.40 d | 1.28 ± 0.01 d | 2.22 ± 0.04 b | 1.69 ± 0.12 c |
| FRAP | 4.70 ± 0.14 a | 1.84 ± 0.17 c | 1.98 ± 0.12 c | 1.85 ± 0.29 c | 2.89 ± 0.09 b | 2.29 ± 0.04 bc |
| ORAC | 34.68 ± 2.14 a | 18.41 ± 0.79 d | 20.04 ± 0.62 c | 15.47 ± 2.38 e | 28.71 ± 0.41 b | 27.30 ± 1.15 b |
| Enzyme inhibitory activity | ||||||
| anti-α-amylase | 26.83 ± 0.22 a | 32.84 ± 0.09 c | 35.12 ± 0.11 d | 32.93 ± 0.48 c | 29.62 ± 0.41 bc | 28.49 ± 0.34 b |
| anti-α-glucosidase | 44.45 ± 0.35 ab | 41.79 ± 0.42 a | 54.76 ± 0.72 c | 60.32 ± 0.87 d | 46.26 ± 0.31 b | 58.89 ± 0.11 cd |
| anti-lipase | 4.55 ± 0.16 a | 9.20 ± 0.20 c | 6.07 ± 0.19 b | 10.07 ± 0.11 d | 4.19 ± 0.17 a | 14.02 ± 0.10 e |
| anti-lipoxygenase | 100.00 a | 92.22 d | 94.10 c | 97.43 b | 92.01 d | 100.00 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tkacz, K.; Wojdyło, A.; Turkiewicz, I.P.; Bobak, Ł.; Nowicka, P. Anti-Oxidant and Anti-Enzymatic Activities of Sea Buckthorn (Hippophaë rhamnoides L.) Fruits Modulated by Chemical Components. Antioxidants 2019, 8, 618. https://doi.org/10.3390/antiox8120618
Tkacz K, Wojdyło A, Turkiewicz IP, Bobak Ł, Nowicka P. Anti-Oxidant and Anti-Enzymatic Activities of Sea Buckthorn (Hippophaë rhamnoides L.) Fruits Modulated by Chemical Components. Antioxidants. 2019; 8(12):618. https://doi.org/10.3390/antiox8120618
Chicago/Turabian StyleTkacz, Karolina, Aneta Wojdyło, Igor Piotr Turkiewicz, Łukasz Bobak, and Paulina Nowicka. 2019. "Anti-Oxidant and Anti-Enzymatic Activities of Sea Buckthorn (Hippophaë rhamnoides L.) Fruits Modulated by Chemical Components" Antioxidants 8, no. 12: 618. https://doi.org/10.3390/antiox8120618
APA StyleTkacz, K., Wojdyło, A., Turkiewicz, I. P., Bobak, Ł., & Nowicka, P. (2019). Anti-Oxidant and Anti-Enzymatic Activities of Sea Buckthorn (Hippophaë rhamnoides L.) Fruits Modulated by Chemical Components. Antioxidants, 8(12), 618. https://doi.org/10.3390/antiox8120618