Antioxidant Efficacy of Litchi (Litchi chinensis Sonn.) Pericarp Extract in Sheep Meat Nuggets
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Litchi Fruit Pericarp Extract
2.2. Sheep Meat Nugget Preparation
2.3. Analysis of Litchi Fruit Pericarp Extract
2.3.1. Analysis of Total Phenolics Content
2.3.2. Radical Scavenging Activity Using the DPPH Assay
2.3.3. Ferric Reducing Antioxidant Power Assay
2.4. Analysis of Cooked Sheep Meat Nuggets
2.4.1. pH and Cooking Yield of Nuggets
2.4.2. Sensory Evaluation of Sheep Meat Nuggets
2.4.3. Lipid Peroxidation of Nuggets during Storage
2.5. Statistical Analysis
3. Results and Discussion
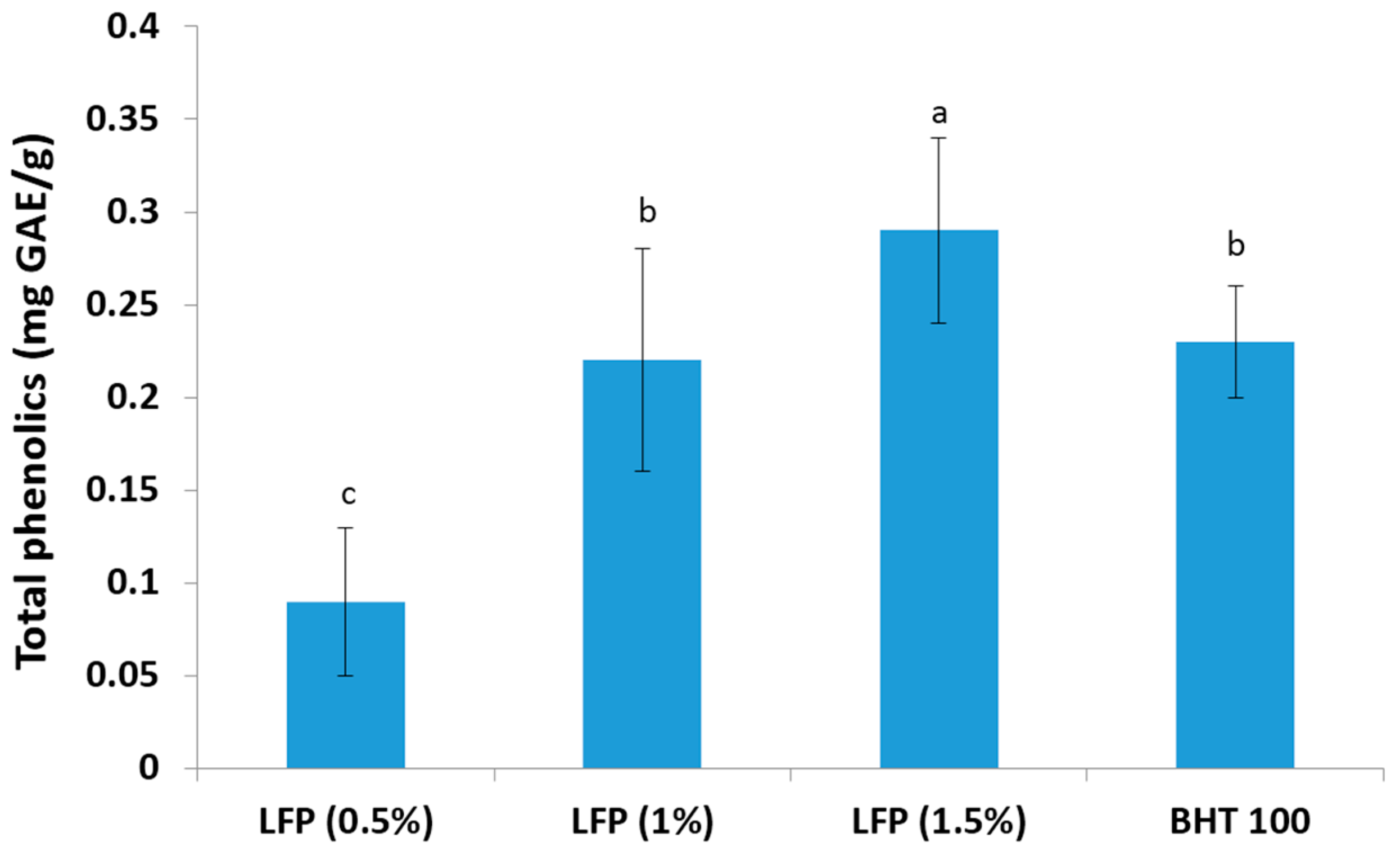
3.1. Total Phenolics Content of Litchi Pericarp Extract
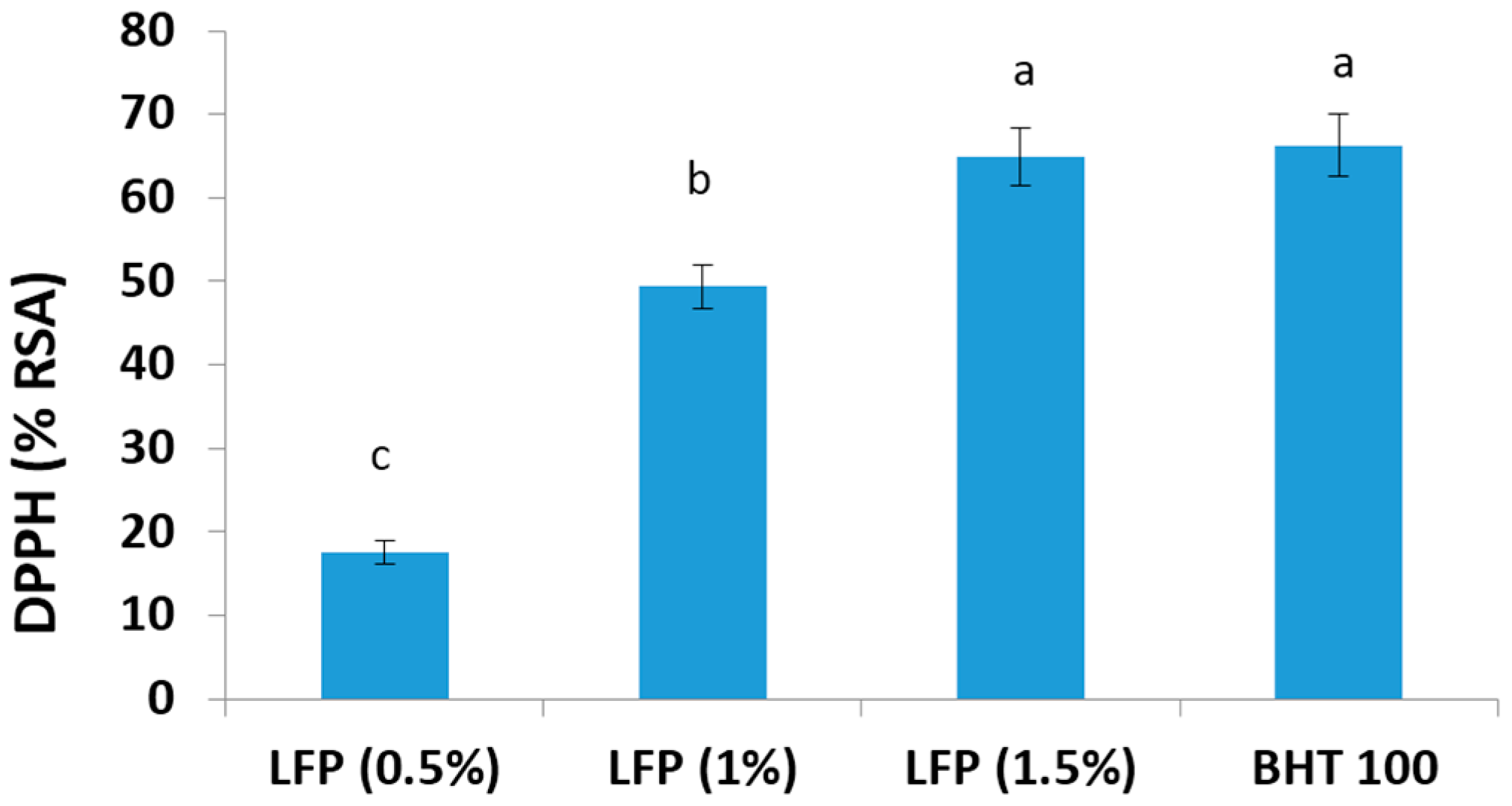
3.2. DPPH Free Radical Scavenging Activity
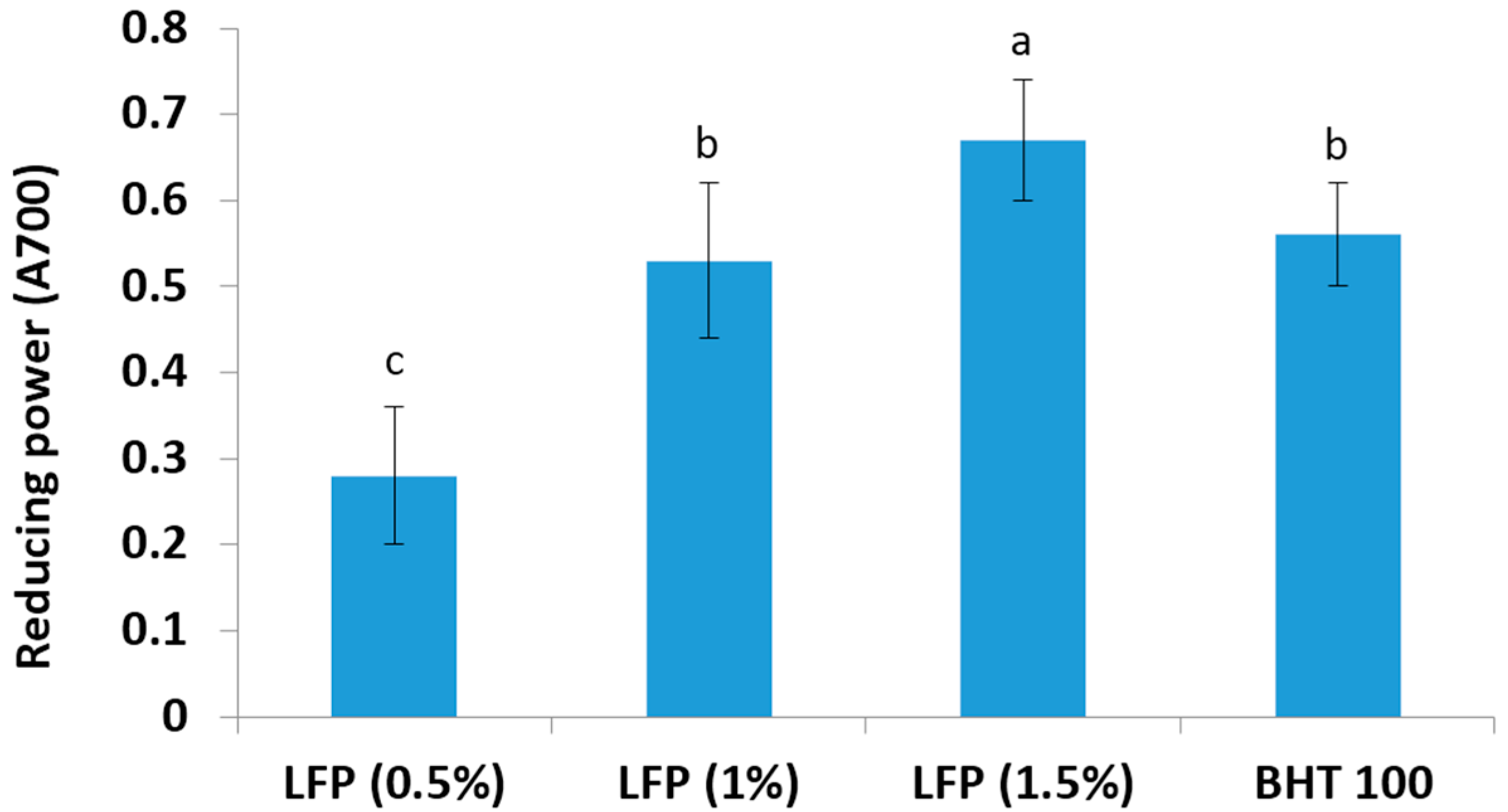
3.3. Ferric Reducing Antioxidant Power Assay
3.4. Total Phenolics, pH and Cooking Yield of Nuggets
3.5. Sensory Analysis
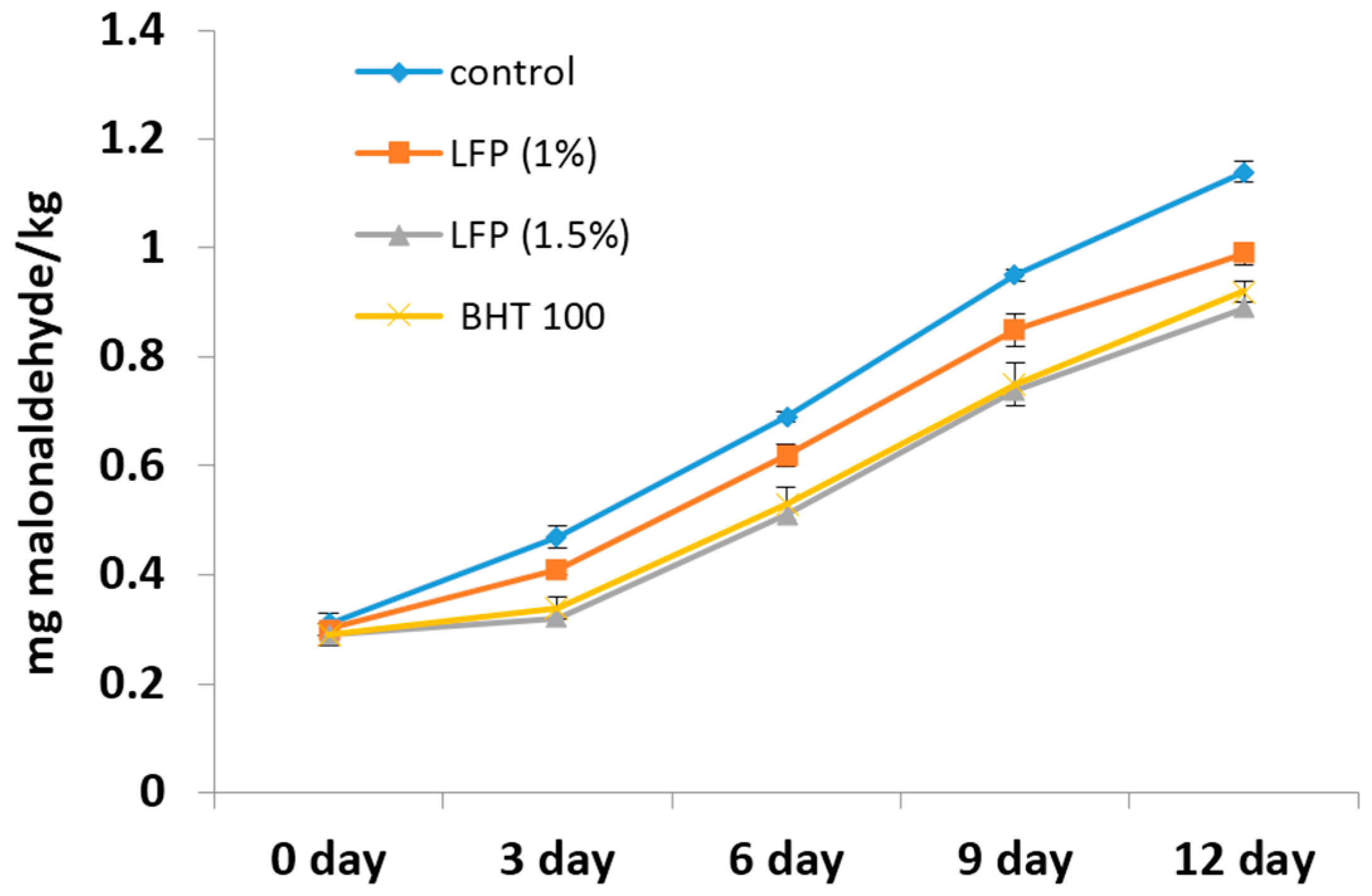
3.6. Lipid Oxidation in Sheep Meat Nuggets
4. Conclusions
Acknowledgments
Author Contributions
Conflict of Interest
References
- Maqsood, S.; Benjakul, S.; Abushelaibi, A.; Alam, A. Phenolic compounds and plant phenolic extracts as natural antioxidants in prevention of lipid oxidation in seafood: A detailed review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1125–1140. [Google Scholar] [CrossRef]
- Lapidot, T.; Granit, R.; Kanner, J. Lipid peroxidation by “free” iron ions and myoglobin as affected by dietary antioxidants in simulated gastric fluids. J. Agric. Food Chem. 2005, 53, 3383–3390. [Google Scholar] [CrossRef] [PubMed]
- Tsuruga, M.; Matsuoka, A.; Hachimori, A.; Sugawara, Y.; Shikama, K. The molecular mechanism of autoxidation for human oxyhemoglobin: Tilting of the distal histidine causes nonequivalent oxidation in the β chain. J. Biol. Chem. 1998, 273, 8607–8615. [Google Scholar] [CrossRef] [PubMed]
- Boyd, L.; Green, D.; Giesbrecht, F.; King, M. Inhibition of oxidative rancidity in frozen cooked fish flakes by tert-butylhydroquinone and rosemary extract. J. Sci. Food Agric. 1993, 61, 87–93. [Google Scholar] [CrossRef]
- Rossi, R.; Pastorelli, G.; Cannata, S.; Tavaniello, S.; Maiorano, G.; Corino, C. Effect of long term dietary supplementation with plant extract on carcass characteristics meat quality and oxidative stability in pork. Meat Sci. 2013, 95, 542–548. [Google Scholar] [CrossRef] [PubMed]
- Pokorný, J. Are natural antioxidants better-and safer-than synthetic antioxidants? Eur. J. Lipid Sci. Technol. 2007, 109, 629–642. [Google Scholar] [CrossRef]
- Das, A.K.; Rajkumar, V.; Verma, A.K.; Swarup, D. Moringa oleiferia leaves extract: A natural antioxidant for retarding lipid peroxidation in cooked goat meat patties. Int. J. Food Sci. Technol. 2012, 47, 585–591. [Google Scholar] [CrossRef]
- Mielnik, M.; Olsen, E.; Vogt, G.; Adeline, D.; Skrede, G. Grape seed extract as antioxidant in cooked, cold stored turkey meat. LWT Food Sci. Technol. 2006, 39, 191–198. [Google Scholar] [CrossRef]
- Ahmad, S.; Gokulakrishnan, P.; Giriprasad, R.; Yatoo, M. Fruit-based natural antioxidants in meat and meat products: A review. Crit. Rev. Food Sci. Nutr. 2015, 55, 1503–1513. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, R.; Verma, A.K.; Das, A.K.; Rajkumar, V.; Shewalkar, A.; Narkhede, H. Antioxidant effects of broccoli powder extract in goat meat nuggets. Meat Sci. 2012, 91, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Qi, S.; Huang, H.; Huang, J.; Wang, Q.; Wei, Q. Lychee (Litchi chinensis Sonn.) seed water extract as potential antioxidant and anti-obese natural additive in meat products. Food Control 2015, 50, 195–201. [Google Scholar] [CrossRef]
- Devatkal, S.K.; Narsaiah, K.; Borah, A. Anti-oxidant effect of extracts of kinnow rind, pomegranate rind and seed powders in cooked goat meat patties. Meat Sci. 2010, 85, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Wu, G.; Jiang, Y. Evaluation of the antioxidant properties of litchi fruit phenolics in relation to pericarp browning prevention. Molecules 2007, 12, 759–771. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Lin, S.; Wen, L.; Jiang, Y.; Zhao, M.; Chen, F.; Prasad, K.N.; Duan, X.; Yang, B. Identification of a novel phenolic compound in litchi (Litchi chinensis Sonn.) pericarp and bioactivity evaluation. Food Chem. 2013, 136, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Jiang, Y.; Su, X.; Zhang, Z.Y.; Shi, J. Antioxidant properties of anthocyanins extracted from litchi (Litchi chinenesis Sonn.) fruit pericarp tissues in relation to their role in the pericarp browning. Food Chem. 2007, 101, 1365–1371. [Google Scholar] [CrossRef]
- Zhao, M.; Yang, B.; Wang, J.; Li, B.; Jiang, Y. Identification of the major flavonoids from pericarp tissues of lychee fruit in relation to their antioxidant activities. Food Chem. 2006, 98, 539–544. [Google Scholar] [CrossRef]
- Li, W.; Liang, H.; Zhang, M.-W.; Zhang, R.-F.; Deng, Y.-Y.; Wei, Z.-C.; Zhang, Y.; Tang, X.-J. Phenolic profiles and antioxidant activity of litchi (Litchi chinensis Sonn.) fruit pericarp from different commercially available cultivars. Molecules 2012, 17, 14954–14967. [Google Scholar] [CrossRef] [PubMed]
- Prasad, N.K.; Yang, B.; Zhao, M.; Wang, B.S.; Chen, F.; Jiang, Y. Effects of high-pressure treatment on the extraction yield, phenolic content and antioxidant activity of litchi (Litchi chinensis Sonn.) fruit pericarp. Int. J. Food Sci. Technol. 2009, 44, 960–966. [Google Scholar] [CrossRef]
- Das, A.K.; Rajkumar, V.; Verma, A.K. Bael pulp residue as a new source of antioxidant dietary fiber in goat meat nuggets. J. Food Process. Preserv. 2015, 39, 1626–1635. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A.J. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Escarpa, A.; Gonzalez, M.C. Approach to the content of total extractable phenolic compounds from different food samples by comparison of chromatographic and spectrophotometric methods. Anal. Chim. Acta 2001, 427, 119–127. [Google Scholar] [CrossRef]
- Fargere, T.; Abdennadher, M.; Delmas, M.; Boutevin, B. Determination of peroxides and hydroperoxides with 2,2-diphenyl-1-picrylhydrazyl (DPPH). Application to ozonized ethylene vinyl acetate copolymers (EVA). Eur. Polym. J. 1995, 31, 489–497. [Google Scholar] [CrossRef]
- Oyaizu, M. Studies on products of the browning reaction. Antioxidative activities of browning reaction products prepared from glucosamine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Witte, V.C.; Krause, G.F.; Bailey, M.F. A new extraction method for determining 2-thiobarbituric acid values of pork and beef during storage. J. Food Sci. 1970, 35, 582–585. [Google Scholar] [CrossRef]
- Zhang, R.; Zeng, Q.; Deng, Y.; Zhang, M.; Wei, Z.; Zhang, Y.; Tang, X. Phenolic profiles and antioxidant activity of litchi pulp of different cultivars cultivated in Southern China. Food Chem. 2013, 136, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Zielinski, H.; Kozlowska, H. Antioxidant activity and total phenolics in selected cereal grains and their different morphological fractions. J. Agric. Food Chem. 2000, 48, 2008–2016. [Google Scholar] [CrossRef] [PubMed]
- Yu, L. Free radical scavenging properties of conjugated linoleic acids. J. Agric. Food Chem. 2001, 49, 3452–3456. [Google Scholar] [CrossRef] [PubMed]
- Robards, K.; Prenzler, P.D.; Tucker, G.; Swatsitang, P.; Glover, W. Phenolic compounds and their role in oxidative processes in fruits. Food Chem. 1999, 66, 401–436. [Google Scholar] [CrossRef]
- Meir, S.; Kanner, J.; Akiri, B.; Philosoph-Hadas, S. Determination and involvement of aqueous reducing compounds in oxidative defense systems of various senescing leaves. J. Agric. Food Chem. 1995, 43, 1813–1819. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.K.; Rajkumar, V.; Banerjee, R.; Biswas, S.; Das, A.K. Guava (Psidium guajava L.) powder as an antioxidant dietary fibre in sheep meat nuggets. Asian-Australas. J. Anim. Sci. 2013, 26, 886–895. [Google Scholar] [CrossRef] [PubMed]
- Naveena, B.; Sen, A.; Vaithiyanathan, S.; Babji, Y.; Kondaiah, N. Comparative efficacy of pomegranate juice, pomegranate rind powder extract and bht as antioxidants in cooked chicken patties. Meat Sci. 2008, 80, 1304–1308. [Google Scholar] [CrossRef] [PubMed]
- Jovanovic, S.V.; Steenken, S.; Tosic, M.; Marjanovic, B.; Simic, M.G. Flavonoids as antioxidants. J. Am. Chem. Soc. 1994, 116, 4846–4851. [Google Scholar] [CrossRef]
- Kondo, K.; Kurihara, M.; Miyata, N.; Suzuki, T.; Toyoda, M. Mechanistic studies of catechins as antioxidants against radical oxidation. Arch. Biochem. Biophys. 1999, 362, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Schlesier, K.; Harwat, M.; Böhm, V.; Bitsch, R. Assessment of antioxidant activity by using different in vitro methods. Free Radic. Res. 2002, 36, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Jongberg, S.; Skov, S.H.; Tørngren, M.A.; Skibsted, L.H.; Lund, M.N. Effect of white grape extract and modified atmosphere packaging on lipid and protein oxidation in chill stored beef patties. Food Chem. 2011, 128, 276–283. [Google Scholar] [CrossRef] [PubMed]
| Ingredients | Control | LFP (1%) | LFP (1.5%) | BHT (100) |
|---|---|---|---|---|
| Meat (%) | 71.1 | 70.1 | 69.5 | 71.1 |
| Salt (%) | 1.8 | 1.8 | 1.8 | 1.8 |
| Ice flakes (%) | 10 | 10 | 10 | 10 |
| Refined oil (%) | 8 | 8 | 8 | 8 |
| Condiments (%) | 4 | 4 | 4 | 4 |
| Polyphosphate (%) | 0.3 | 0.3 | 0.3 | 0.3 |
| Dry spice mix (%) | 1.8 | 1.8 | 1.8 | 1.8 |
| Na nitrite (ppm) (%) | 150 | 150 | 150 | 150 |
| Wheat flour (%) | 3 | 3 | 3 | 3 |
| LFP extract (%) | 0.00 | 1.00 | 1.5 | - |
| BHT (100 ppm) | - | - | - | 100 |
| Measurements | Control | LFP (1%) | LFP (1.5%) | BHT (100) |
|---|---|---|---|---|
| pH | 6.21 ± 0.03 | 6.20 ± 0.02 | 6.22 ± 0.02 | 6.19 ± 0.02 |
| Cooking yield (%) | 93.62 ± 0.48 | 93.29 ± 0.36 | 94.12 ± 0.43 | 93.75 ± 0.39 |
| Total phenolics (GAE) mg/g | 0.05 ± 0.01 c | 0.13 ± 0.01 b | 0.17 ± 0.01 a | 0.16 ± 0.01 a |
| Sensory Attributes | Control | LFP (1%) | LFP (1.5%) | BHT (100) |
|---|---|---|---|---|
| Appearance | 7.23 ± 0.05 | 7.22 ± 0.06 | 7.18 ± 0.06 | 7.12 ± 0.04 |
| Flavor | 7.06 ± 0.6 | 7.05 ± 0.08 | 7.03 ± 0.06 | 6.94 ± 0.07 |
| Texture | 7.15 ± 0.08 | 7.03 ± 0.07 | 7.01 ± 0.06 | 7.02 ± 0.06 |
| Juiciness | 7.07 ± 0.07 | 7.13 ± 0.04 | 7.15 ± 0.05 | 7.17 ± 0.05 |
| Overall acceptability | 7.18 ± 0.05 | 7.08 ± 0.06 | 7.11 ± 0.06 | 7.05 ± 0.07 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, A.K.; Rajkumar, V.; Nanda, P.K.; Chauhan, P.; Pradhan, S.R.; Biswas, S. Antioxidant Efficacy of Litchi (Litchi chinensis Sonn.) Pericarp Extract in Sheep Meat Nuggets. Antioxidants 2016, 5, 16. https://doi.org/10.3390/antiox5020016
Das AK, Rajkumar V, Nanda PK, Chauhan P, Pradhan SR, Biswas S. Antioxidant Efficacy of Litchi (Litchi chinensis Sonn.) Pericarp Extract in Sheep Meat Nuggets. Antioxidants. 2016; 5(2):16. https://doi.org/10.3390/antiox5020016
Chicago/Turabian StyleDas, Arun K., Vincent Rajkumar, Pramod K. Nanda, Pranav Chauhan, Soubhagya R. Pradhan, and Subhasish Biswas. 2016. "Antioxidant Efficacy of Litchi (Litchi chinensis Sonn.) Pericarp Extract in Sheep Meat Nuggets" Antioxidants 5, no. 2: 16. https://doi.org/10.3390/antiox5020016
APA StyleDas, A. K., Rajkumar, V., Nanda, P. K., Chauhan, P., Pradhan, S. R., & Biswas, S. (2016). Antioxidant Efficacy of Litchi (Litchi chinensis Sonn.) Pericarp Extract in Sheep Meat Nuggets. Antioxidants, 5(2), 16. https://doi.org/10.3390/antiox5020016