Antioxidant Content, Antioxidant Activity, and Antibacterial Activity of Five Plants from the Commelinaceae Family
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Chemicals and Reagents
2.3. Extraction of Samples
2.4. Determination of Total Phenolic Content (TPC)
2.5. DPPH Free Radical Scavenging (FRS) Assay
2.6. Ferric Reducing Power (FRP) Assay
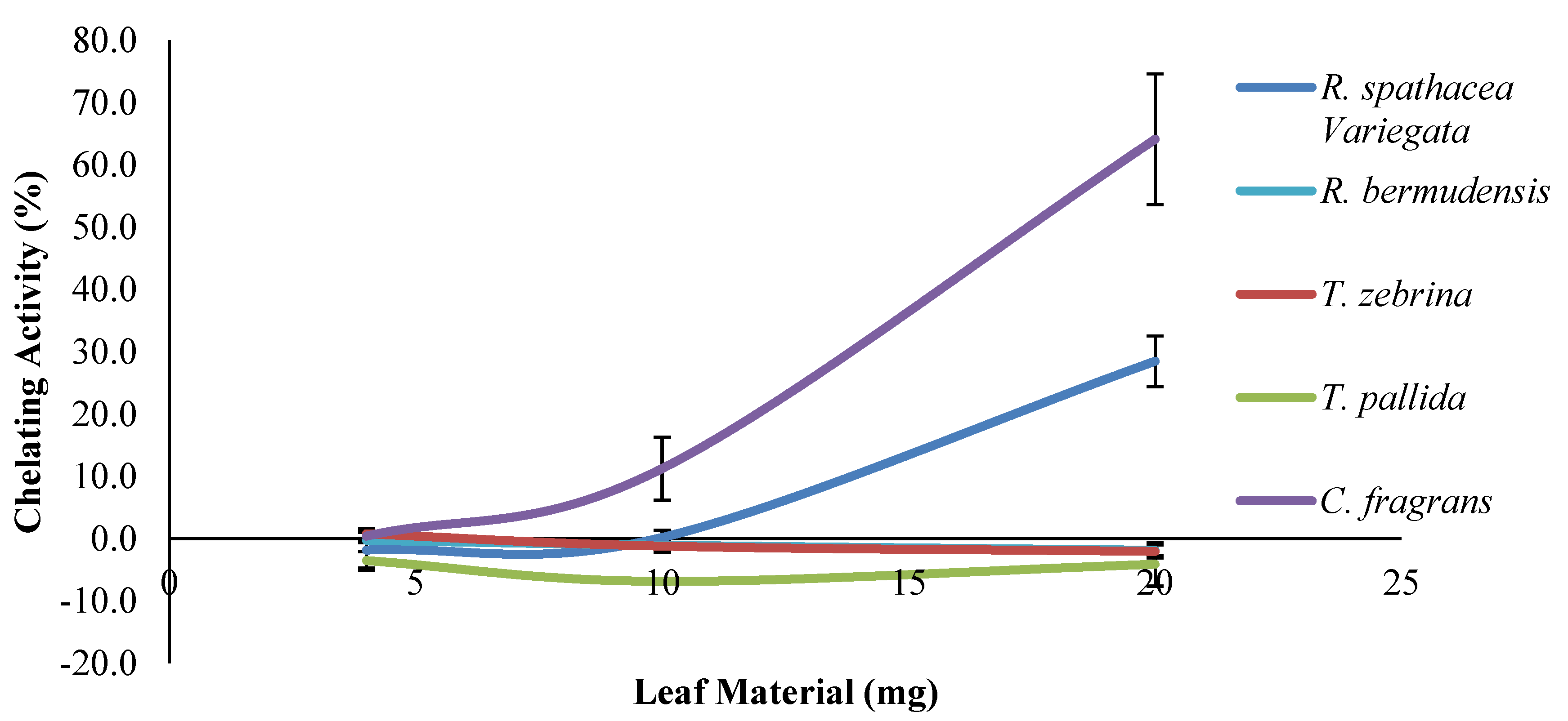
2.7. Ferrous Ion Chelating (FIC) Assay
2.8. Determination of Total Flavonoid Content (TFC)
2.9. Total Tannin Content (TTC)
2.10. Determination of Antimicrobial Activity
2.11. Statistical Analysis
3. Results and Discussion
| Species | TPC (mg GAE/100 g) * | TTC (mg TAE/100 g) * | TFC (mg RE/100 g) | FRS (mg AA/100 g) | FRP (mg GAE/g) |
|---|---|---|---|---|---|
| R. spathacea variegata | 203.9 ± 16.3 a,d (24.8 ± 2.2) a | 20.6 ± 2.3 a | 10.8 ± 2.9 a | 177.3 ± 15.3 a | 1.4 ± 0.1 a |
| R. bermudensis | 296.6 ± 21.7 b (33.5 ± 2.4) b | 11.5 ± 1.7 b | 18.4 ± 2.4 b | 462.2 ± 97.3 b | 2.7 ± 0.3 b |
| T. zebrina | 620.9 ± 39.7 c (70.1 ± 4.5) c | 57.6 ± 3.5 c | 17.1 ± 2.8 b | 906.5 ± 88.2 c | 4.8 ± 0.3 c |
| T. pallida | 153.1 ± 21.8 d (17.8 ± 2.3) d | 13.6 ± 2.1 b | 10.6 ± 4.0 a | 103.0 ± 36.9 a | 0.9 ± 0.1 a |
| C. fragrans | 269.3 ± 33.8 a,b (29.3 ± 2.9) a,b | 19.7 ± 1.5 a | 2.5 ± 1.4 a | 262.5 ± 67.2 a | 1.5 ± 0.3 a |
| Bacterium | R. spathacea Variegata | R. bermudensis | T. zebrina | T. pallida | C. fragrans |
|---|---|---|---|---|---|
| Gram-Positive | |||||
| Bacillus cereus (ATCC 14579) | >10 | 10 | 5 | 5 | 5 |
| Bacillus subtilis (ATCC 8188) | >10 | 10 | 10 | 10 | 5 |
| Micrococcus luteus (ATCC 4698) | >10 | 10 | 5 | 10 | 10 |
| Methicillin-Resistant Staphylococcus aureus (ATCC 33591) | >10 | 10 | 5 | 10 | 10 |
| Staphylococcus epidermidis (ATCC 12228) | 5 | 10 | >10 | >10 | >10 |
| Enterococcus faecalis (ATCC 29212) | 10 | >10 | >10 | 5 | >10 |
| Gram-Negative | |||||
| Aeromonas hydrophila (ATCC 49140) | 5 | >10 | 5 | 5 | 5 |
| Proteus vulgaris (Clinical) | >10 | >10 | 5 | 5 | >10 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Edeoga, H.O.; Ogbebor, N.O. Distribution of calcium oxalate crystals in some Nigerian species of Aneilema R. Br. (Commelinaceae). Plant Biosyst. 1999, 133, 193–198. [Google Scholar] [CrossRef]
- Satterfield, S.K.; Mertens, T.R. Rhoeo spathacea: A tool for teaching meiosis and mitosis. J. Hered. 1972, 63, 375–378. [Google Scholar]
- Faden, R.B. Commelinaceae. In Flowering Plants—Monocotyledons; Kubitzki, K., Ed.; Springer: Berlin-Heidelberg, Germany, 1998; Volume 4, pp. 109–128. [Google Scholar]
- Wilson, A.K. Commelinaceae—A review of the distribution, biology and control of the important weeds belonging to this family. Trop. Pest Manag. 1981, 27, 405–418. [Google Scholar] [CrossRef]
- González-Avila, M.; Arriaga-Alba, M.; de la Garza, M.; del Carmen HernándezPretelín, M.; Domínguez-Ortíz, M.A.; Fattel-Fazenda, S.; Villa-Treviño, S. Antigenotoxic, antimutagenic and ROS scavenging activities of a Rhoeo discolor ethanolic crude extract. Toxicol. In Vitro 2003, 17, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Arriaga-Alba, M.; Blasco, J.L.; Ruíz-Pérez, N.J.; Sánchez-Navarrete, J.; Rivera-Sánchez, R.; González-Avila, M. Antimutagenicity mechanisms of the Rhoeo discolor ethanolic extract. Exp. Toxicol. Pathol. 2011, 63, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Mensah, A.Y.; Houghton, P.J.; Dickson, R.A.; Fleischer, T.C.; Heinrich, M.; Bremner, P. In vitro evaluation of effects of two Ghanaian plants relevant to wound healing. Phytother. Res. 2006, 20, 941–944. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Castro, A.J.; Villarreal, M.L.; Salazar-Olivo, L.A.; Gomez-Sanchez, M.; Dominguez, F.; Garcia-Carranca, A. Mexican medicinal plants used for cancer treatment: Pharmacological, phytochemical and ethnobotanical studies. J. Ethnopharmacol. 2011, 133, 945–972. [Google Scholar] [CrossRef] [PubMed]
- Perry, C.C. An overview of silica in biology: Its chemistry and recent technological advances. In Biosilica in Evolution, Morphogenesis, and Nanobiotechnology; Müller, W.E.G., Grachev, M.A., Eds.; Springer: Berlin-Heidelberg, Germany, 2009; Volume 47, pp. 295–313. [Google Scholar]
- Tan, J.B.L.; Lim, Y.Y.; Lee, S.M. Antioxidant and antibacterial activity of Rhoeo spathacea (Swartz) Stearn leaves. J. Food Sci. Technol. 2013. [Google Scholar] [CrossRef]
- Xeriscape Plants. Available online: http://www.ctahr.hawaii.edu/oc/freepubs/pdf/OF-42.pdf (accessed on 10 May 2014).
- Golczyk, H. Cytogenetics of the permanent translocation heterozygote Rhoeo spathacea var. variegata. Implications for complex chromosome rearrangements in Rhoeo. Caryologia 2011, 64, 325–334. [Google Scholar] [CrossRef]
- Paiva, E.A.S.; Isaias, R.M.S.; Vale, F.H.A.; Queiroz, C.G.S. The influence of light intensity on anatomical structure and pigment contents of Tradescantia pallida (Rose) Hunt. cv. purpurea Boom (Commelinaceae) leaves. Braz. Arch. Biol. Technol. 2003, 46, 617–624. [Google Scholar] [CrossRef]
- Rainho, C.R.D.; Kaezer, A.; Aiub, C.A.F.; Felzenszwalb, I. Ability of Allium cepa L. root tips and Tradescantia pallida var. purpurea in N-nitrosodiethylamine genotoxicity and mutagenicity evaluation. An. Acad. Bras. Ciênc. 2010, 82, 925–932. [Google Scholar] [CrossRef]
- Misik, M.; Ma, T.H.; Nersesyan, A.; Monarca, S.; Kim, J.K.; Knasmueller, S. Micronucleus assays with Tradescantia pollen tetrads: An update. Mutagenesis 2011, 26, 215–221. [Google Scholar] [CrossRef]
- Thewes, M.R.; Junior, D.E.; Droste, A. Genotoxicity biomonitoring of sewage in two municipal wastewater treatment plants using the Tradescantia pallida var. purpurea bioassay. Genet. Mol. Biol. 2011, 34, 689–693. [Google Scholar] [CrossRef]
- Yang, D.S.; Pennisi, S.V.; Son, K.C.; Kays, S.J. Screening indoor plants for volatile organic pollutant removal efficiency. HortScience 2009, 44, 1377–1381. [Google Scholar]
- Li, T.S.C. Taiwanese Native Medicinal Plant: Phytopharmacology and Therapeutic Values. CRC Press: Boca Raton, FL, USA, 2006; pp. 1–328. [Google Scholar]
- Faden, R.B. The author and typification of Tradescantia zebrina (Commelinaceae). Kew Bull. 2008, 63, 679–680. [Google Scholar] [CrossRef]
- Yanzhi, M. Pigment content and anatomical structure of leaves of several species of red-leafed plants. J. N. E. Forest. Univ. 2009, 37, 51–53. [Google Scholar]
- Glimn-Lacy, J.; Kaufman, P.B. Spiderwort family (Commelinaceae). In Botany Illustrated; Springer US: New York, NY, USA, 2006; pp. 121–121. [Google Scholar]
- Amaral, F.M.M.; Ribeiro, M.N.S.; Barbosa-Filho, J.M.; Reis, A.S.; Nascimento, F.R.F.; Macedo, R.O. Plants and chemical constituents with giardicidal activity. Braz. J. Pharmacog. 2006, 16, 696–720. [Google Scholar]
- González-Coloma, A.; Reina, M.; Sáenz, C.; Lacret, R.; Ruiz-Mesia, L.; Arán, V.J.; Sanz, J.; Martinez-Diaz, R.A. Antileishmanial, antitrypanosomal, and cytotoxic screening of ethnopharmacologically selected Peruvian plants. Parasitol. Res. 2012, 110, 1381–1392. [Google Scholar] [CrossRef] [PubMed]
- Alaba, C.S.M.; Chichioco-Hernandez, C.L. 15-Lipoxygenase inhibition of Commelina benghalensis, Tradescantia fluminensis, Tradescantia zebrina. Asian Pac. J. Trop. Biomed. 2014, 4, 184–188. [Google Scholar] [PubMed]
- Chernenko, T.V.; Ul’chenko, N.T.; Glushenkova, A.I.; Redzhepov, D. Chemical investigation of Callisia fragrans. Chem. Nat. Compd. 2007, 43, 253–255. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Ibragimov, T.A.; Zilfikarov, I.N.; Chelombit’ko, V.A. Chemical composition of Callisia fragrans juice 1. Phenolic compounds. Chem. Nat. Compd. 2008, 44, 776–777. [Google Scholar] [CrossRef]
- Misin, V.M.; Sazhina, N.N. Content and activity of low-molecular antioxidants in juices of medicinal plants. Russ. J. Phys. Chem. B 2010, 4, 797–800. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Zilfikarov, I.N.; Toropova, A.A.; Ibragimov, T.A. Chemical composition of Callisia fragrans wood. juice and its antioxidative activity (in vitro). Chem. Plant Raw Mater. 2008, 4, 95–100. [Google Scholar]
- Malaysian Meteorological Department. Available online: http://www.met.gov.my/index.php?option=com_content&task=view&id=34&Itemid=1586 (accessed on 22 October 2014).
- Kähkönen, M.P.; Hopia, A.I.; Vuorela, H.J.; Rauha, J.P.; Pihlaja, K.; Kujala, T.S.; Heinonen, M. Antioxidant activity of plant extracts containing phenolic compounds. J. Agric. Food Chem. 1999, 47, 3954–3962. [Google Scholar] [CrossRef] [PubMed]
- Leong, L.P.; Shui, G. An investigation of antioxidant capacity of fruits in Singapore markets. Food Chem. 2002, 76, 69–75. [Google Scholar] [CrossRef]
- Miliauskas, G.; Venskutonis, P.R.; van Beek, T.A. Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem. 2004, 85, 231–237. [Google Scholar] [CrossRef]
- Chan, E.W.C.; Lim, Y.Y.; Chong, K.L.; Tan, J.B.L.; Wong, S.K. Antioxidant properties of tropical and temperate herbal teas. J. Food Comp. Anal. 2010, 23, 185–189. [Google Scholar] [CrossRef]
- Tan, Y.P.; Chan, E.W.C. Antioxidant, antityrosinase and antibacterial properties of fresh and processed leaves of Anacardium occidentale and Piper betle. Food Biosci. 2014, 6, 17–23. [Google Scholar] [CrossRef]
- Mau, J.L.; Lai, E.Y.C.; Wang, N.P.; Chen, C.C.; Chang, C.H.; Chyau, C.C. Composition and antioxidant activity of the essential oil from Curcuma zedoaria. Food Chem. 2003, 82, 583–591. [Google Scholar] [CrossRef]
- Singh, N.; Rajini, P.S. Free radical scavenging activity of an aqueous extract of potato peel. Food Chem. 2004, 85, 611–616. [Google Scholar] [CrossRef]
- Chew, Y.L.; Goh, J.K.; Lim, Y.Y. Assessment of in vitro antioxidant capacity and polyphenolic composition of selected medicinal herbs from Leguminosae family in peninsular Malaysia. Food Chem. 2009, 116, 13–18. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Siddhuraju, P.; Becker, K. Plant Secondary Metabolites; Humana Press: Totowa, NJ, USA, 2007; pp. 67–82. [Google Scholar]
- Clinical and Laboratory Standards Institute. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved standard—Eight edition. CLSI Doc. 2009, 29, 15–18. [Google Scholar]
- Michel, T.; Destandau, E.; Le Floch, G.; Lucchesi, M.E.; Elfakir, C. Antimicrobial, antioxidant and phytochemical investigations of sea buckthorn (Hippophaë rhamnoides L.) leaf, stem, root and seed. Food Chem. 2012, 131, 754–760. [Google Scholar] [CrossRef]
- Lai, H.Y.; Lim, Y.Y. Evaluation of antioxidant activities of the methanolic extracts of selected ferns in Malaysia. Int. J. Environ. Sci. Dev. 2011, 2, 442–447. [Google Scholar] [CrossRef]
- Russell, A.D. Bacterial resistance to disinfectants: Present knowledge and future problems. J. Hosp. Infect. 1999, 43, S57–68. [Google Scholar] [CrossRef] [PubMed]
- Frei, B.; Heinrich, M.; Bork, P.M.; Herrmann, D.; Jaki, B.; Kato, T.; Kuhnt, M.; Schmitt, J.; Schühly, W.; Volken, C.; et al. Multiple screening of medicinal plants from Oaxaca, Mexico: Ethnobotany and bioassays as a basis for phytochemical investigation. Phytomedicine 1998, 5, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Tene, V.; Malagón, O.; Finzi, P.V.; Vidari, G.; Armijos, C.; Zaragoza, T. An ethnobotanical survey of medicinal plants used in Loja and Zamora-Chinchipe, Ecuador. J. Ethnopharmacol. 2007, 111, 63–81. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, J.B.L.; Yap, W.J.; Tan, S.Y.; Lim, Y.Y.; Lee, S.M. Antioxidant Content, Antioxidant Activity, and Antibacterial Activity of Five Plants from the Commelinaceae Family. Antioxidants 2014, 3, 758-769. https://doi.org/10.3390/antiox3040758
Tan JBL, Yap WJ, Tan SY, Lim YY, Lee SM. Antioxidant Content, Antioxidant Activity, and Antibacterial Activity of Five Plants from the Commelinaceae Family. Antioxidants. 2014; 3(4):758-769. https://doi.org/10.3390/antiox3040758
Chicago/Turabian StyleTan, Joash Ban Lee, Wei Jin Yap, Shen Yeng Tan, Yau Yan Lim, and Sui Mae Lee. 2014. "Antioxidant Content, Antioxidant Activity, and Antibacterial Activity of Five Plants from the Commelinaceae Family" Antioxidants 3, no. 4: 758-769. https://doi.org/10.3390/antiox3040758
APA StyleTan, J. B. L., Yap, W. J., Tan, S. Y., Lim, Y. Y., & Lee, S. M. (2014). Antioxidant Content, Antioxidant Activity, and Antibacterial Activity of Five Plants from the Commelinaceae Family. Antioxidants, 3(4), 758-769. https://doi.org/10.3390/antiox3040758




