Antioxidant Capacity, Cytotoxicity and Antimycobacterial Activity of Madeira Archipelago Endemic Helichrysum Dietary and Medicinal Plants
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Instruments
2.3. Plant Material
2.4. Antimycobacterial Activity
2.4.1. Mycobacterium Strains
2.4.2. Inoculum Preparation for Biological Assays
2.4.3. Antimycobacterial Screening by Microplate Alamar Blue Assay
2.5. Artemia Salina Toxicity Evaluation
2.6. Determination of Total Phenolic Content (TPC)
2.7. Determination of Total Flavonoid Content (TFC)
2.8. Antioxidant Assays
2.8.1. 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Radical Scavenging Activity
2.8.2. 2,2′-Azinobis-(3-Ethylbenzthiazoline-6-Sulfonic Acid) (ABTS•+) Radical Scavenging Activity
2.8.3. Ferric Reducing Activity (FRAP Assay)
2.8.4. β-Carotene (BC) Bleaching Assay
2.9. Statistical Analysis
3. Results and Discussion
3.1. Extraction Yield
3.2. Antimycobacterial Activity
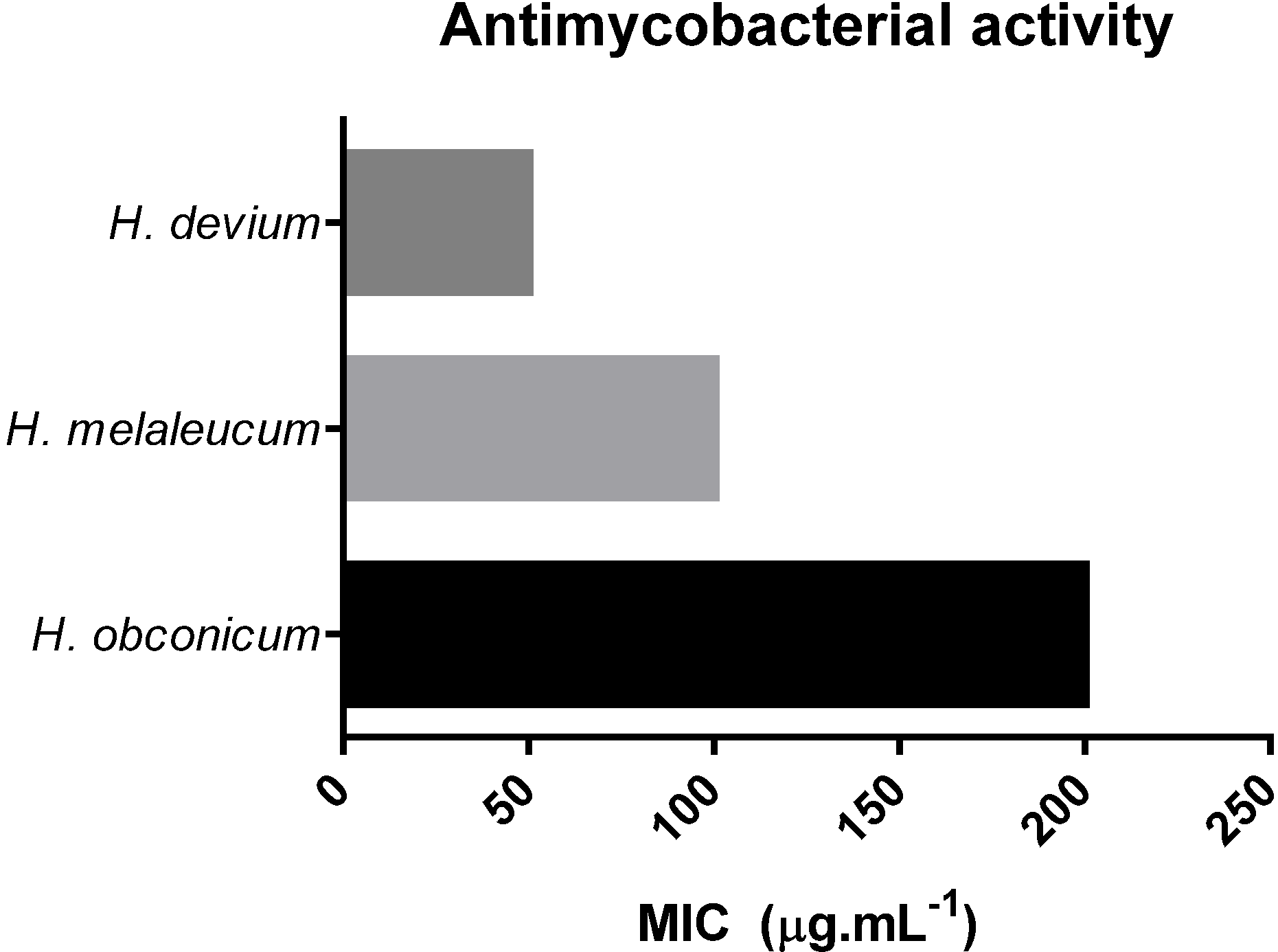
| Plant Extracts | TPC (mg GAE 100 g−1) | TFC (mg RUE 100 g−1) | DPPH | ABTS | FRAP mmol Fe(II) mg−1 | BC (%) | Toxicity Activity LD50 (μg mL−1) | Extraction Yield (%) | |
|---|---|---|---|---|---|---|---|---|---|
| μmol eq. Trolox 100 g−1 | |||||||||
| H. devium | n-Hexane | 221.4 ± 1.72 c | 44.95 ± 0.193 d | 289.7 ± 1.03 e | 116.4 ± 0.434 f | 694.9 ± 3.89 b | 62.82 ± 1.58 f | 2.36 d | 216.7 ± 10.4 |
| Chloroform | 234.6 ± 3.61 d | 40.27 ± 0.429 c | 234.8 ± 1.09 d | 84.57 ± 0.0700 d | 852.4 ± 11.1 e | 60.62 ± 2.25 f | 3.63 e | 5025 ± 63.2 | |
| Ethyl acetate | 312.7 ± 1.73 f | 180.3 ± 10.6 f | 236.4 ± 4.41 d | 86.12 ± 0.0707 d | 2140.0 ± 14.6 g | 52.34 ± 1.53 d | 3.61 e | * | |
| Methanol | 399.6 ± 1.52 g | 302.8 ± 1.19 g | 235.4 ± 1.63 d | 107.1 ± 0.0706 e | 2431.8 ± 12.9 h | 46.82 ± 1.75 c | 4.85 f | 479.0 ± 8.20 | |
| H. melaleucum | n-Hexane | 258.9 ± 0.631 e | 46.26 ± 0.477 d | 137.6 ± 2.66 c | 56.94 ± 0.0658 b | 354.8 ± 1.35 a | 61.02 ± 1.45 f | 1.14 c | 322.9 ± 11.9 |
| Chloroform | 106.5 ± 0.373 b | 56.61 ± 0.663 e | 119.2 ± 0.691 b | 71.97 ± 0.0348 c | 831.0 ± 3.09 d | 56.06 ± 1.29 e | 1.35 c | 752.0 ± 36.6 | |
| Ethyl acetate | NA | NA | NA | NA | NA | NA | 0.18 a | 3349 ± 102.3 | |
| Methanol | 1214 ± 2.11 i | 816.9 ± 1.96 i | 812.9 ± 1.65 g | 286.9 ± 1.28 g | 1581.7 ± 7.71 f | 22.85 ± 0.29 a | 7.64 g | * | |
| H. obconicum | Hexane | 39.75 ± 0.214 a | 19.20 ± 0.251 a | NI | NI | NI | NI | 1.23 c | 934.5 ± 56.2 |
| Chloroform | NA | NA | NA | NA | NA | NA | NA | NA | |
| Ethyl acetate | 42.05 ± 0.134 a | 23.61 ± 0.195 b | 23.78 ± 0.754 a | 5.780 ± 0.031 a | 741.08 ± 19.9 c | 36.43 ± 0.58 b | 0.57 b | 2,410 ± 88.3 | |
| Methanol | 773.4 ± 14.3 h | 703.03 ± 2.98 h | 638.8 ± 1.50 f | 687.9 ± 7.25 h | 19,918.3 ± 75.4 i | 37.96 ± 0.59 b | 15.0 h | 1075 ± 71.5 | |
3.3. Artemia Salina Toxicity
3.4. Total Phenolic Content (TPC) and Total Flavonoid Content (TFC)
3.5. Antioxidant Assays
3.5.1. DPPH• Radical Scavenging Activity
3.5.2. ABTS•+ Radical Scavenging Activity
3.5.3. Ferric Reducing Activity (FRAP Assay)
3.5.4. β-Carotene (BC) Bleaching Assay
3.5.5. Correlations between Antioxidant Assays
| (a) | R2 | DPPH | ABTS | FRAP | BC | ||
| H. devium | 0.140 | 0.054 | 0.952 | 0.980 | |||
| H. melalecum | 0.995 | 0.983 | 0.867 | 0.970 | |||
| (b) | R2 | DPPH | ABTS | FRAP | |||
| ABTS | FRAP | BC | FRAP | BC | BC | ||
| H. devium | 0.755 | 0.380 | 0.036 | 0.891 | 0.068 | −0.984 | |
| H. melaleucum | 0.997 | 0.914 | 0.990 | 0.994 | 0.998 | 0.926 | |
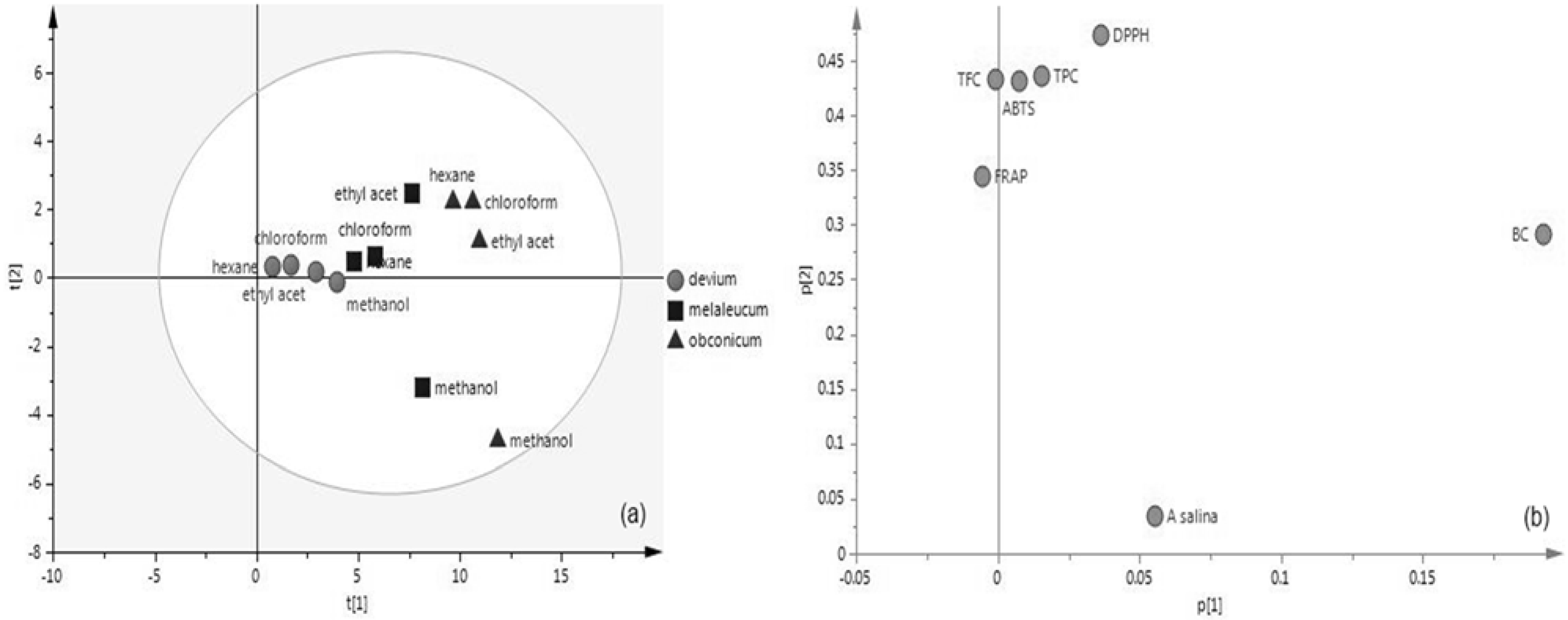
3.6. Principal Component Analysis (PCA)
| Variable | PC1 | PC2 |
|---|---|---|
| TPC | 0.01524 | 0.4357 |
| TFC | −0.00098 | 0.4327 |
| DPPH | 0.03629 | 0.4742 |
| ABTS | 0.00767 | 0.4315 |
| FRAP | −0.00560 | 0.34415 |
| BC | 0.1924 | 0.29152 |
| A. salina | 0.1924 | 0.03366 |
| R2 (cum) | 0.879 | 0.945 |
| eigenvalue | 5.07 | 1.63 |
| Q2 (Cum) | 0.214 | 0.397 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tsao, R.; Deng, Z. Separation Procedures for Naturally Occurring Antioxidant Phytochemicals. J. Chromatogr. B 2004, 812, 85–99. [Google Scholar] [CrossRef]
- Fu, L.; Xu, B.-T.; Xu, X.-R.; Gan, R.-Y.; Zhang, Y.; Xia, E.-Q.; Li, H.-B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. [Google Scholar] [CrossRef]
- Michiels, J.A.; Kevers, C.; Pincemailc, J.; Defraignec, J.O.; Dommesb, J. Extraction conditions can greatly influence antioxidant capacity assays in plant food matrices. Food Chem. 2012, 130, 986–993. [Google Scholar] [CrossRef]
- Atoui, A.K.; Atoui, A.K.; Mansouri, A.; Boskou, G.; Kefalas, P. Tea and herbal infusions: Their antioxidant activity and phenolic profile. Food Chem. 2005, 89, 27–36. [Google Scholar] [CrossRef]
- McGaw, L.J.; Lallb, N.; Meyer, J.J.M.; Eloff, J.N. The potential of South African plants against Mycobacterium infections. J. Ethnopharmacol. 2008, 119, 482–500. [Google Scholar] [CrossRef] [PubMed]
- Copp, B.R. Antimycobacterial natural products. Nat. Prod. Rep. 2003, 20, 535–557. [Google Scholar] [CrossRef] [PubMed]
- Okunade, A.L.; Elvin-Lewis, M.P.F.; Lewis, W.H. Natural antimycobacterial metabolites: Current status. Phytochemistry 2004, 65, 1017–1032. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, S. Plants as a source of bacterial resistance modulators and anti-infective agents. Phytochem. Rev. 2005, 4, 63–78. [Google Scholar] [CrossRef]
- Pauli, G.F.; Case, R.J.; Inui, T.; Wang, Y.; Cho, S.; Fischer, N.H.; Franzblau, S.G. New perspectives on natural products in TB drug research. Life Sci. 2005, 78, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Frothingham, R.; Stout, J.E.; Hamilton, C.D. Current issues in global tuberculosis control. Int. J. Infect. Dis. 2005, 9, 297–311. [Google Scholar] [CrossRef] [PubMed]
- Newton, S.M.; Lau, C.; Wright, C.W. A review of antimycobacterial natural products. Phytother. Res. 2000, 14, 303–322. [Google Scholar] [CrossRef] [PubMed]
- Newton, S.M.; Lau, C.; Gurcha, S.S.; Besra, G.S.; Wright, C.W. The evaluation of forty-three plant species for in vitro antimycobacterial activities; isolation of active constituents from Psoralea corylifolia and Sanguinaria canadensis. J. Ethnopharmacol. 2002, 79, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, S.C.; Castilho, P.C. Analysis of phenolic compounds from different morphological parts of Helichrysum devium by liquid chromatography with on-line UV and electrospray ionization mass spectrometric detection. Rapid Commun. Mass Spectrom. 2009, 23, 3939–3953. [Google Scholar] [CrossRef] [PubMed]
- Carini, M.; Aldini, G.; Furlanetto, S.; Stefani, R.; Facino, R.M. LC coupled to ion-trap MS for the rapid screening and detection of polyphenol antioxidants from Helichrysum stoechas. J. Pharm. Biomed. Anal. 2001, 24, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Matic, I.; Aljančić, I.; Žižak, Ž.; Vajs, V.; Jadranin, M.; Milosavljević, S.; Juranić, Z.D. In vitro antitumor actions of extracts from endemic plant Helichrysum zivojinii. BMC Complement. Altern. Med. 2013, 13, 36. [Google Scholar] [CrossRef] [PubMed]
- Rivera, D.; Obón, C. The ethnopharmacology of Madeira and Porto Santo Islands, a review. J. Ethnopharmacol. 1995, 46, 73–93. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, S.C.; Castilho, P.C. Characterization of phenolic compounds in Helichrysum melaleucum by high-performance liquid chromatography with on-line ultraviolet and mass spectrometry detection. Rapid Commun. Mass Spectrom. 2010, 24, 1851–1868. [Google Scholar] [CrossRef]
- Gouveia, S.C.; Castilho, P.C. Characterisation of phenolic acid derivatives and flavonoids from different morphological parts of Helichrysum obconicum by a RP-HPLC-DAD-(-)-ESI-MSn method. Food Chem. 2011, 129, 333–344. [Google Scholar] [CrossRef]
- Jimenez-Arellanes, A.; Meckes, M.; Ramirez, R.; Torres, J.; Luna-Herrera, J. Activity against multidrug-resistant Mycobacterium tuberculosis in Mexican plants used to treat respiratory diseases. Phytother. Res. 2003, 17, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Hostettmann, K. Methods In Plant Biochemistry—Assays For Bioactivity; Academic Press: London, UK, 1991; Volume 6. [Google Scholar]
- Zheng, W.; Wang, S.Y. Antioxidant activity and phenolic compounds in selected herbs. J. Agric. Food Chem. 2001, 49, 5165–5170. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, S.C.; Castilho, P.C. Helichrysum monizii Lowe: Phenolic composition and antioxidant potential. Phytochem. Anal. 2012, 23, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Gordon, M.H.; Paiva-Martins, F.; Almeida, M. Antioxidant activity of hydroxytyrosol acetate compared with that of other olive oil polyphenols. J. Agric. Food Chem. 2001, 49, 2480–2485. [Google Scholar] [CrossRef] [PubMed]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, S.C.; Castilho, P.C. Antioxidant potential of Artemisia argentea L’Hér alcoholic extract and its relation with the phenolic composition. Food Res. Int. 2011, 44, 1620–1631. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. Studies on antioxidant activities of mucuna seed (Mucuna pruriens var utilis) extract and various non-protein amino/imino acids through in vitro models. J. Sci. Food Agric. 2003, 83, 1517–1524. [Google Scholar] [CrossRef]
- Lall, N.; Hussein, A.A.; Meyer, J.J.M. Antiviral and antituberculous activity of Helichrysum melanacme constituents. Fitoterapia 2006, 77, 230–232. [Google Scholar] [CrossRef] [PubMed]
- Leite, A.M.; de O. Lima, E.; de Souza, E.L.; de F.F.M. Diniz, M.; Leite, S.P.; Xavier, A.L.; de Medeiros, I.A. Preliminary study of the molluscicidal and larvicidal properties of some essential oils and phytochemicals from medicinal plants. Rev. Bras. Farmacogn. 2009, 19, 842–846. [Google Scholar]
- Santos Pimenta, L.P.; Pinto, G.B.; Takahashi, J.A.; e Silva, L.G.; Boaventura, M.A. Biological screening of annonaceous brazilian medicinal plants using Artemia salina (Brine shrimp test). Phytomedicine 2003, 10, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Cuadra, P.; Furrianca, M.; Oyarzún, A.; Yáñez, E.; Gallardo, A.; Fajardo, V. Biological activity of some Patagonian plants. Fitoterapia 2005, 76, 718–721. [Google Scholar] [CrossRef] [PubMed]
- Albayrak, S.; Aksoy, A.; Sagdic, O.; Hamzaoglu, E. Compositions, antioxidant and antimicrobial activities of Helichrysum (Asteraceae) species collected from Turkey. Food Chem. 2010, 119, 114–122. [Google Scholar] [CrossRef]
- Süntar, I.; Küpeli Akkol, E.; Keles, H.; Yesilada, E.; Sarker, S.D. Exploration of the wound healing potential of Helichrysum graveolens (Bieb.) Sweet: Isolation of apigenin as an active component. J. Ethnopharmacol. 2013, 149, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Ou, B.; Prior, R.L. The Chemistry behind Antioxidant Capacity Assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef] [PubMed]
- Stratil, P.; Klejdus, B.; Kubáň, V. Determination of total content of phenolic compounds and their antioxidant activity in vegetables evaluation of spectrophotometric methods. J. Agric. Food Chem. 2006, 54, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Siddhuraju, P. Antioxidant activity of polyphenolic compounds extracted from defatted raw and dry heated Tamarindus indica seed coat. LWT Food Sci. Technol. 2007, 40, 982–990. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gouveia-Figueira, S.C.; Gouveia, C.A.; Carvalho, M.J.; Rodrigues, A.I.; Nording, M.L.; Castilho, P.C. Antioxidant Capacity, Cytotoxicity and Antimycobacterial Activity of Madeira Archipelago Endemic Helichrysum Dietary and Medicinal Plants. Antioxidants 2014, 3, 713-729. https://doi.org/10.3390/antiox3040713
Gouveia-Figueira SC, Gouveia CA, Carvalho MJ, Rodrigues AI, Nording ML, Castilho PC. Antioxidant Capacity, Cytotoxicity and Antimycobacterial Activity of Madeira Archipelago Endemic Helichrysum Dietary and Medicinal Plants. Antioxidants. 2014; 3(4):713-729. https://doi.org/10.3390/antiox3040713
Chicago/Turabian StyleGouveia-Figueira, Sandra C., Carla A. Gouveia, Maria J. Carvalho, Ana I. Rodrigues, Malin L. Nording, and Paula C. Castilho. 2014. "Antioxidant Capacity, Cytotoxicity and Antimycobacterial Activity of Madeira Archipelago Endemic Helichrysum Dietary and Medicinal Plants" Antioxidants 3, no. 4: 713-729. https://doi.org/10.3390/antiox3040713
APA StyleGouveia-Figueira, S. C., Gouveia, C. A., Carvalho, M. J., Rodrigues, A. I., Nording, M. L., & Castilho, P. C. (2014). Antioxidant Capacity, Cytotoxicity and Antimycobacterial Activity of Madeira Archipelago Endemic Helichrysum Dietary and Medicinal Plants. Antioxidants, 3(4), 713-729. https://doi.org/10.3390/antiox3040713




