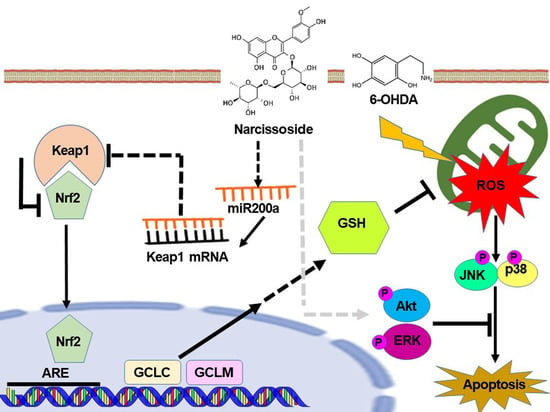
Neuroprotective Capability of Narcissoside in 6-OHDA-Exposed Parkinson’s Disease Models through Enhancing the MiR200a/Nrf-2/GSH Axis and Mediating MAPK/Akt Associated Signaling Pathway
Abstract
:1. Introduction
2. Materials and Methods
2.1. Narcissoside, Chemicals, Maintenance and Pretreatment of SH-SY5Y Cell Line
2.2. Cell Viability Assay
2.3. Mitochondrial Membrane Potential Assay of Cells
2.4. Live Cell Nuclear Staining with Hoechst 33258
2.5. Flow Cytometric Analysis of Apoptosis by FITC-Labeled Annexin-V/Propidium Iodide
2.6. Western Blot of Protein Expression
2.7. Quantitative Determination of Intracellular Reactive Oxygen Species
2.8. Determines Intracellular Glutathione (GSH) Content
2.9. Preparation of Nuclear Extract
2.10. NRF2/ARE Luciferase Reporter Assays
2.11. Electrophoretic Mobility Shift Assay (EMSA)
2.12. Transient Transfection of Small Interfering RNA of Nrf2
2.13. Measurement of MiR-200a Expression Level by qRT–PCR
2.14. Downregulation of miR200a with Anti-miR200a
2.15. C. elegans Culture and Synchronization
2.16. Food Clearance Assay Determines the Concentration of Narcissoside to Treat Nematodes
2.17. Narcissoside Pretreatment and 6-OHDA Exposure in Nematodes
2.18. Analysis of Dopaminergic Neuron Degeneration in Nematodes
2.19. Assessment of Dopamine Neuron Functionality in Nematodes with a Food Sensitivity Behavioral Test
2.20. Lifespan Assessment of Nematodes
2.21. Determining the Content of Reactive Oxygen Species in Nematodes
2.22. Determination of Glutathione Content in Nematodes
2.23. Total RNA Isolation and Quantitative Analysis of Gene Expression in Nematodes
2.24. Measurements of the Autophagy Activity of Nematodes
2.25. Statistical Analysis of the Investigation
3. Results
3.1. Narcissoside (NCS) Exhibits Protective Potential to Prevent SH-SY5Y Cells from Apoptosis Induced by 6-OHDA Exposure
3.2. Pretreatment of Narcissoside (NCS) Enhanced Level of Intracellular Glutathione (GSH) to Diminish Production of Reactive Oxygen Species (ROS) in 6-OHDA-Exposed SH-SY5Y Cells
3.3. Treatment with the GSH Synthetic Inhibitor Buthionine Sulphoximine (BSO) Abrogates the Neuroprotective Ability of Narcissoside (NCS) in 6-OHDA-Treated SH-SY5Y Cells
3.4. The Ability of Narcissoside (NCS) to Alleviate 6-OHDA-Induced Apoptosis of SH-SY5Y Cells May Be through Inhibition of p38 and JNK1/2 Mitogen-Activated Protein Kinase (MAPK) Phosphorylation and Enhancing of the Extracellular-Regulated Kinase (ERK) and Akt Pathway
3.5. The Neuroprotective Property of Narcissoside (NCS) in 6-OHDA-Exposed SH-SY5Y Cells Is Associated with the Elevation of the Activity of the Nuclear Factor Erythroid 2–Related Factor 2 (Nrf2) Pathway
3.6. Knockdown of Nrf2 Abolished the Neuroprotective Ability of Narcissoside (NCS) against Oxidative Stress and Apoptosis of SH-SY5Y Induced by 6-OHDA Exposure
3.7. Narcissoside (NCS) Inhibits Keap1 Level by Augmenting Expression of Endogenous MicroRNA-200a and Thus Enhances Nrf2 Activity
3.8. Narcissoside (NCS) Pretreatment Was Effective in Reducing Dopamine Neuron Degeneration Induced by 6-OHDA Exposure in an Animal Model of Caenorhabditis elegans
3.9. Narcissoside (NCS) Pretreatment Restores Deficits in Dopamine-Mediated Food-Sensitive Behavior in Nematodes Exposed to 6-OHDA
3.10. Narcissoside (NCS) Pretreatment Improves Shortened Lifespan of Nematodes Due to 6-OHDA Toxicity
3.11. Narcissoside (NCS) Pretreatment Declines the Level of Reactive Oxygen Species in 6-OHDA-Exposed Nematodes by Enhancing the GSH Production via Rising Expression of Skn-1, Gcs-1, and E01A2.1
3.12. Narcissoside (NCS) Significantly Reduces α-Synuclein Accumulation by Promoting Autophagy of Nematodes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Calabrese, V.; Santoro, A.; Monti, D.; Crupi, R.; Di Paola, R.; Latteri, S.; Cuzzocrea, S.; Zappia, M.; Giordano, J.; Calabrese, E.J.; et al. Aging and Parkinson’s Disease: Inflammaging, neuroinflammation and biological remodeling as key factors in pathogenesis. Free Radic. Biol. Med. 2018, 115, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Puspita, L.; Chung, S.Y.; Shim, J.W. Oxidative stress and cellular pathologies in Parkinson’s disease. Mol. Brain 2017, 10, 53. [Google Scholar] [CrossRef] [Green Version]
- Muhammad, F.; Liu, Y.; Zhou, Y.; Yang, H.; Li, H. Antioxidative role of Traditional Chinese Medicine in Parkinson’s disease. J. Ethnopharmacol. 2022, 285, 114821. [Google Scholar] [CrossRef] [PubMed]
- Martin, H.L.; Teismann, P. Glutathione—A review on its role and significance in Parkinson’s disease. FASEB J. 2009, 23, 3263–3272. [Google Scholar] [CrossRef] [Green Version]
- Hormann, P.; Delcambre, S.; Hanke, J.; Geffers, R.; Leist, M.; Hiller, K. Impairment of neuronal mitochondrial function by L-DOPA in the absence of oxygen-dependent auto-oxidation and oxidative cell damage. Cell Death Discov. 2021, 7, 151. [Google Scholar] [CrossRef]
- Mendez, J.S.; Finn, B.W. Use of 6-hydroxydopamine to create lesions in catecholamine neurons in rats. J. Neurosurg. 1975, 42, 166–173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jang, H.J.; Chung, K.C. The ubiquitin-proteasome system and autophagy mutually interact in neurotoxin-induced dopaminergic cell death models of Parkinson’s disease. FEBS Lett. 2022. [Google Scholar] [CrossRef]
- Blum, D.; Torch, S.; Lambeng, N.; Nissou, M.; Benabid, A.L.; Sadoul, R.J.; Verna, M. Molecular pathways involved in the neurotoxicity of 6-OHDA, dopamine and MPTP: Contribution to the apoptotic theory in Parkinson’s disease. Prog. Neurobiol. 2001, 65, 135–172. [Google Scholar] [CrossRef]
- Iskusnykh, I.Y.; Zakharova, A.A.; Pathak, D. Glutathione in Brain Disorders and Aging. Molecules 2022, 27, 324. [Google Scholar] [CrossRef]
- Bjorklund, G.; Peana, M.; Maes, M.; Dadar, M.; Severin, B. The glutathione system in Parkinson’s disease and its progression. Neurosci. Biobehav. Rev. 2021, 120, 470–478. [Google Scholar] [CrossRef]
- Lu, S.C. Glutathione synthesis. Biochim. Biophys. Acta 2013, 1830, 3143–3153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ikawa, T.; Sato, M.; Oh-Hashi, K.; Furuta, K.; Hirata, Y. Oxindole-curcumin hybrid compound enhances the transcription of gamma-glutamylcysteine ligase. Eur. J. Pharmacol. 2021, 896, 173898. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.D.; Yoo, J.; Hwang, S.Y.; Cho, S.Y.; Kim, M.; Jang, H.; No, K.O.; Shin, J.C.; Kim, J.H.; Lee, G. Bee Venom Activates the Nrf2/HO-1 and TrkB/CREB/BDNF Pathways in Neuronal Cell Responses against Oxidative Stress Induced by Abeta1-42. Int. J. Mol. Sci. 2022, 23, 1193. [Google Scholar] [CrossRef] [PubMed]
- Tayarani-Najaran, Z.; Hadipour, E.; Seyed Mousavi, S.M.; Emami, S.A.; Mohtashami, L.; Javadi, B. Protective effects of Lavandula stoechas L. methanol extract against 6-OHDA-induced apoptosis in PC12 cells. J. Ethnopharmacol. 2021, 273, 114023. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Zhong, W.; Zhang, M.; Zhang, R.; Hu, W. P38 Mitogen-activated Protein Kinase and Parkinson’s Disease. Transl. Neurosci. 2018, 9, 147–153. [Google Scholar] [CrossRef]
- Peng, J.; Andersen, J.K. The role of c-Jun N-terminal kinase (JNK) in Parkinson’s disease. IUBMB Life 2003, 55, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Ko, Y.H.; Kim, S.K.; Kwon, S.H.; Seo, J.Y.; Lee, B.R.; Kim, Y.J.; Hur, K.H.; Kim, S.Y.; Lee, S.Y.; Jang, C.G. 7,8,4′-Trihydroxyisoflavone, a Metabolized Product of Daidzein, Attenuates 6-Hydroxydopamine-Induced Neurotoxicity in SH-SY5Y Cells. Biomol. Ther. 2019, 27, 363–372. [Google Scholar] [CrossRef]
- Rai, S.N.; Dilnashin, H.; Birla, H.; Singh, S.S.; Zahra, W.; Rathore, A.S.; Singh, B.K.; Singh, S.P. The Role of PI3K/Akt and ERK in Neurodegenerative Disorders. Neurotox. Res 2019, 35, 775–795. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.P.N.; Kumar, M.; Fedele, E.; Bonanno, G.; Bonifacino, T. MicroRNA Alteration, Application as Biomarkers, and Therapeutic Approaches in Neurodegenerative Diseases. Int. J. Mol. Sci. 2022, 23, 4718. [Google Scholar] [CrossRef]
- Khezri, M.R.; Yousefi, K.; Zolbanin, N.M.; Ghasemnejad-Berenji, M. MicroRNAs in the pathophysiology of Alzheimer’s disease and Parkinson’s disease: An overview. Mol. Neurobiol. 2022, 59, 1589–1603. [Google Scholar] [CrossRef]
- Evans, B.; Furlong, H.A.; de Lencastre, A. Parkinson’s disease and microRNAs—Lessons from model organisms and human studies. Exp. Gerontol. 2021, 155, 111585. [Google Scholar] [CrossRef] [PubMed]
- Cenci, M.A.; Skovgard, K.; Odin, P. Non-dopaminergic approaches to the treatment of motor complications in Parkinson’s disease. Neuropharmacology 2022, 210, 109027. [Google Scholar] [CrossRef] [PubMed]
- Bueno, P.C.P.; Abarca, L.F.S.; Anhesine, N.B.; Giffoni, M.S.; Pereira, F.M.V.; Torres, R.B.; de Sousa, R.W.R.; Ferreira, P.M.P.; Pessoa, C.; Cavalheiro, A.J. Infraspecific Chemical Variability and Biological Activity of Casearia sylvestris from Different Brazilian Biomes. Planta Med. 2021, 87, 148–159. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhang, G.; Huang, L.; Pang, H.; Zhang, N.; Chen, Y.; Wang, G. Hepatoprotective Effect of Polyphenol-Enriched Fraction from Folium Microcos on Oxidative Stress and Apoptosis in Acetaminophen-Induced Liver Injury in Mice. Oxidative Med. Cell. Longev. 2017, 2017, 3631565. [Google Scholar] [CrossRef] [Green Version]
- Kozachok, S.; Kolodziejczyk-Czepas, J.; Marchyshyn, S.; Wojtanowski, K.K.; Zgorka, G.; Oleszek, W. Comparison of Phenolic Metabolites in Purified Extracts of Three Wild-Growing Herniaria L. Species and Their Antioxidant and Anti-Inflammatory Activities In Vitro. Molecules 2022, 27, 530. [Google Scholar] [CrossRef]
- Liao, Q.; Chen, Z.; Tao, Y.; Zhang, B.; Wu, X.; Yang, L.; Wang, Q.; Wang, Z. An integrated method for optimized identification of effective natural inhibitors against SARS-CoV-2 3CLpro. Sci. Rep. 2021, 11, 22796. [Google Scholar] [CrossRef]
- Blanco-Salas, J.; Vazquez, F.M.; Hortigon-Vinagre, M.P.; Ruiz-Tellez, T. Bioactive Phytochemicals from Mercurialis spp. Used in Traditional Spanish Medicine. Plants 2019, 8, 193. [Google Scholar] [CrossRef] [Green Version]
- Boubaker, J.; Bhouri, W.; Ben Sghaier, M.; Ghedira, K.; Dijoux Franca, M.G.; Chekir-Ghedira, L. Ethyl acetate extract and its major constituent, isorhamnetin 3-O-rutinoside, from Nitraria retusa leaves, promote apoptosis of human myelogenous erythroleukaemia cells. Cell Prolif. 2011, 44, 453–461. [Google Scholar] [CrossRef]
- Merlin-Lucas, V.; Ordonez-Razo, R.M.; Calzada, F.; Solis, A.; Garcia-Hernandez, N.; Barbosa, E.; Valdes, M. Antitumor Potential of Annona muricata Linn. An Edible and Medicinal Plant in Mexico: In Vitro, In Vivo, and Toxicological Studies. Molecules 2021, 26, 7675. [Google Scholar] [CrossRef]
- Ha, T.; Kim, M.S.; Kang, B.; Kim, K.; Hong, S.S.; Kang, T.; Woo, J.; Han, K.; Oh, U.; Choi, C.W.; et al. Lotus Seed Green Embryo Extract and a Purified Glycosyloxyflavone Constituent, Narcissoside, Activate TRPV1 Channels in Dorsal Root Ganglion Sensory Neurons. J. Agric. Food Chem. 2022, 70, 3969–3978. [Google Scholar] [CrossRef]
- Chen, J.H.; Ou, H.P.; Lin, C.Y.; Lin, F.J.; Wu, C.R.; Chang, S.W.; Tsai, C.W. Carnosic acid prevents 6-hydroxydopamine-induced cell death in SH-SY5Y cells via mediation of glutathione synthesis. Chem. Res. Toxicol. 2012, 25, 1893–1901. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.L.; Hung, H.S.; Tsai, C.W.; Liu, S.P.; Chiang, Y.T.; Kuo, Y.H.; Shyu, W.C.; Lin, S.Z.; Fu, R.H. Peiminine Reduces ARTS-Mediated Degradation of XIAP by Modulating the PINK1/Parkin Pathway to Ameliorate 6-Hydroxydopamine Toxicity and alpha-Synuclein Accumulation in Parkinson’s Disease Models In Vivo and In Vitro. Int. J. Mol. Sci. 2021, 22, 10240. [Google Scholar] [CrossRef]
- Zou, Z.C.; Fu, J.J.; Dang, Y.Y.; Zhang, Q.; Wang, X.F.; Chen, H.B.; Jia, X.J.; Lee, S.M.; Li, C.W. Pinocembrin-7-Methylether Protects SH-SY5Y Cells Against 6-Hydroxydopamine-Induced Neurotoxicity via Modulating Nrf2 Induction Through AKT and ERK Pathways. Neurotox. Res. 2021, 39, 1323–1337. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Yang, J.W.; Kim, M.R.; Roh, S.H.; Kim, H.G.; Lee, K.Y.; Jeong, H.G.; Kang, K.W. Increased expression of Nrf2/ARE-dependent anti-oxidant proteins in tamoxifen-resistant breast cancer cells. Free Radic. Biol. Med. 2008, 45, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.H.; Alfatah, M.; Sin, M.F.; Sim, H.M.; Verma, C.S.; Lane, D.P.; Arumugam, P. A yeast two-hybrid system for the screening and characterization of small-molecule inhibitors of protein-protein interactions identifies a novel putative Mdm2-binding site in p53. BMC Biol. 2017, 15, 108. [Google Scholar] [CrossRef] [PubMed]
- Ezcurra, M.; Tanizawa, Y.; Swoboda, P.; Schafer, W.R. Food sensitizes C. elegans avoidance behaviours through acute dopamine signalling. EMBO J. 2011, 30, 1110–1122. [Google Scholar] [CrossRef] [Green Version]
- Silver, D. Impact of functional age on the use of dopamine agonists in patients with Parkinson disease. Neurologist 2006, 12, 214–223. [Google Scholar] [CrossRef]
- Arvanitis, M.; Li, D.D.; Lee, K.; Mylonakis, E. Apoptosis in C. elegans: Lessons for cancer and immunity. Front. Cell. Infect. Microbiol. 2013, 3, 67. [Google Scholar] [CrossRef] [Green Version]
- Bodhicharla, R.; Nagarajan, A.; Winter, J.; Adenle, A.; Nazir, A.; Brady, D.; Vere, K.; Richens, J.; O’Shea, P.; Bell, D.R.; et al. Effects of alpha-synuclein overexpression in transgenic Caenorhabditis elegans strains. CNS Neurol. Disord. Drug Targets 2012, 11, 965–975. [Google Scholar] [CrossRef] [Green Version]
- Frias, D.P.; Gomes, R.L.N.; Yoshizaki, K.; Carvalho-Oliveira, R.; Matsuda, M.; Junqueira, M.S.; Teodoro, W.R.; Vasconcellos, P.C.; Pereira, D.C.A.; Conceicao, P.R.D.; et al. Nrf2 positively regulates autophagy antioxidant response in human bronchial epithelial cells exposed to diesel exhaust particles. Sci. Rep. 2020, 10, 3704. [Google Scholar] [CrossRef]
- Sahoo, S.; Padhy, A.A.; Kumari, V.; Mishra, P. Role of Ubiquitin-Proteasome and Autophagy-Lysosome Pathways in alpha-Synuclein Aggregate Clearance. Mol. Neurobiol. 2022, 59, 5379–5407. [Google Scholar] [CrossRef]
- Chen, Y.; Scarcelli, V.; Legouis, R. Approaches for Studying Autophagy in Caenorhabditis elegans. Cells 2017, 6, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maegawa, H.; Niwa, H. Generation of Mitochondrial Toxin Rodent Models of Parkinson’s Disease Using 6-OHDA, MPTP, and Rotenone. Methods Mol. Biol. 2021, 2322, 95–110. [Google Scholar] [PubMed]
- Satoh, T.; McKercher, S.R.; Lipton, S.A. Nrf2/ARE-mediated antioxidant actions of pro-electrophilic drugs. Free Radic. Biol. Med. 2013, 65, 645–657. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chakkittukandiyil, A.; Sajini, D.V.; Karuppaiah, A.; Selvaraj, D. The principal molecular mechanisms behind the activation of Keap1/Nrf2/ARE pathway leading to neuroprotective action in Parkinson’s disease. Neurochem. Int. 2022, 156, 105325. [Google Scholar] [CrossRef]
- Boas, S.M.; Joyce, K.L.; Cowell, R.M. The NRF2-Dependent Transcriptional Regulation of Antioxidant Defense Pathways: Relevance for Cell Type-Specific Vulnerability to Neurodegeneration and Therapeutic Intervention. Antioxidants 2021, 11, 8. [Google Scholar] [CrossRef]
- Shilovsky, G.A. Lability of the Nrf2/Keap/ARE Cell Defense System in Different Models of Cell Aging and Age-Related Pathologies. Biochemistry 2022, 87, 70–85. [Google Scholar] [CrossRef]
- Panda, H.; Wen, H.; Suzuki, M.; Yamamoto, M. Multifaceted Roles of the KEAP1-NRF2 System in Cancer and Inflammatory Disease Milieu. Antioxidants 2022, 11, 538. [Google Scholar] [CrossRef]
- Asmar, A.J.; Beck, D.B.; Werner, A. Control of craniofacial and brain development by Cullin3-RING ubiquitin ligases: Lessons from human disease genetics. Exp. Cell Res. 2020, 396, 112300. [Google Scholar] [CrossRef]
- Yang, X.X.; Yang, R.; Zhang, F. Role of Nrf2 in Parkinson’s Disease: Toward New Perspectives. Front. Pharmacol. 2022, 13, 919233. [Google Scholar] [CrossRef]
- Yang, X.; Duan, J.; Wu, L. Research advances in NQO1-responsive prodrugs and nanocarriers for cancer treatment. Future Med. Chem. 2022, 14, 363–383. [Google Scholar] [CrossRef] [PubMed]
- Elko, E.A.; Cunniff, B.; Seward, D.J.; Chia, S.B.; Aboushousha, R.; van de Wetering, C.; van der Velden, J.; Manuel, A.; Shukla, A.; Heintz, N.H.; et al. Peroxiredoxins and Beyond; Redox Systems Regulating Lung Physiology and Disease. Antioxid. Redox Signal. 2019, 31, 1070–1091. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, B.; Arner, E.S.J. Thioredoxin-related protein of 14 kDa as a modulator of redox signalling pathways. Br. J. Pharmacol. 2019, 176, 544–553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kerins, M.J.; Ooi, A. The Roles of NRF2 in Modulating Cellular Iron Homeostasis. Antioxid. Redox Signal. 2018, 29, 1756–1773. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, C.J.; Gao, S.L.; Lin, T.K.; Chu, P.Y.; Lin, H.Y. Ferroptosis as a Major Factor and Therapeutic Target for Neuroinflammation in Parkinson’s Disease. Biomedicines 2021, 9, 1679. [Google Scholar] [CrossRef]
- Segura-Aguilar, J.; Munoz, P.; Inzunza, J.; Varshney, M.; Nalvarte, I.; Mannervik, B. Neuroprotection against Aminochrome Neurotoxicity: Glutathione Transferase M2-2 and DT-Diaphorase. Antioxidants 2022, 11, 296. [Google Scholar] [CrossRef]
- Gupta, R.; Ambasta, R.K.; Pravir, K. Autophagy and apoptosis cascade: Which is more prominent in neuronal death? Cell Mol. Life Sci. 2021, 78, 8001–8047. [Google Scholar] [CrossRef]
- Bekker, M.; Abrahams, S.; Loos, B.; Bardien, S. Can the interplay between autophagy and apoptosis be targeted as a novel therapy for Parkinson’s disease? Neurobiol. Aging 2021, 100, 91–105. [Google Scholar] [CrossRef]
- Ouyang, M.; Shen, X. Critical role of ASK1 in the 6-hydroxydopamine-induced apoptosis in human neuroblastoma SH-SY5Y cells. J. Neurochem. 2006, 97, 234–244. [Google Scholar] [CrossRef]
- Nakaso, K.; Ito, S.; Nakashima, K. Caffeine activates the PI3K/Akt pathway and prevents apoptotic cell death in a Parkinson’s disease model of SH-SY5Y cells. Neurosci. Lett. 2008, 432, 146–150. [Google Scholar] [CrossRef]
- Sun, Z.; Huang, Z.; Zhang, D.D. Phosphorylation of Nrf2 at multiple sites by MAP kinases has a limited contribution in modulating the Nrf2-dependent antioxidant response. PLoS ONE 2009, 4, e6588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naidu, S.; Vijayan, V.; Santoso, S.; Kietzmann, T.; Immenschuh, S. Inhibition and genetic deficiency of p38 MAPK up-regulates heme oxygenase-1 gene expression via Nrf2. J. Immunol. 2009, 182, 7048–7057. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, H.; Zhang, D.; Gao, Z.; Li, H.; Zhang, B.; Zhang, Q.; Li, L.; Cheng, Q.; Pei, D.; Zheng, J. MDA-7/IL-24 inhibits Nrf2-mediated antioxidant response through activation of p38 pathway and inhibition of ERK pathway involved in cancer cell apoptosis. Cancer Gene Ther. 2014, 21, 416–426. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.M.; Lee, K.M.; Lee, H.J.; Yun, J.H.; Nho, C.W. Physalin A regulates the Nrf2 pathway through ERK and p38 for induction of detoxifying enzymes. BMC Complement. Altern. Med. 2019, 19, 101. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Teng, H.; Zhang, K.Y.; Skalicka-Wozniak, K.; Georgiev, M.I.; Xiao, J. Agrimonolide and Desmethylagrimonolide Induced HO-1 Expression in HepG2 Cells through Nrf2-Transduction and p38 Inactivation. Front. Pharmacol. 2016, 7, 513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahn, K.S.; Sethi, G.; Jain, A.K.; Jaiswal, A.K.; Aggarwal, B.B. Genetic deletion of NAD(P)H:quinone oxidoreductase 1 abrogates activation of nuclear factor-kappaB, IkappaBalpha kinase, c-Jun N-terminal kinase, Akt, p38, and p44/42 mitogen-activated protein kinases and potentiates apoptosis. J. Biol. Chem. 2006, 281, 19798–19808. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; He, Z.; Guo, J.; Li, Z.; Wang, X.; Yang, C.; Cui, X. Sulfiredoxin-1 protects against simulated ischaemia/reperfusion injury in cardiomyocyte by inhibiting PI3K/AKT-regulated mitochondrial apoptotic pathways. Biosci. Rep. 2016, 36, e00325. [Google Scholar] [CrossRef]
- Zhu, P.; Qian, J.; Xu, Z.; Meng, C.; Liu, J.; Shan, W.; Zhu, W.; Wang, Y.; Yang, Y.; Zhang, W.; et al. Piperlonguminine and Piperine Analogues as TrxR Inhibitors that Promote ROS and Autophagy and Regulate p38 and Akt/mTOR Signaling. J. Nat. Prod. 2020, 83, 3041–3049. [Google Scholar] [CrossRef]
- Ma, Y.; Pan, C.; Tang, X.; Zhang, M.; Shi, H.; Wang, T.; Zhang, Y. MicroRNA-200a represses myocardial infarction-related cell death and inflammation by targeting the Keap1/Nrf2 and beta-catenin pathways. Hell. J. Cardiol. 2021, 62, 139–148. [Google Scholar] [CrossRef]
- Hu, X.; Liu, H.; Wang, Z.; Hu, Z.; Li, L. miR-200a Attenuated Doxorubicin-Induced Cardiotoxicity through Upregulation of Nrf2 in Mice. Oxidative Med. Cell. Longev. 2019, 2019, 1512326. [Google Scholar] [CrossRef]
- Sun, X.; Zuo, H.; Liu, C.; Yang, Y. Overexpression of miR-200a protects cardiomyocytes against hypoxia-induced apoptosis by modulating the kelch-like ECH-associated protein 1-nuclear factor erythroid 2-related factor 2 signaling axis. Int. J. Mol. Med. 2016, 38, 1303–1311. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.J.; Tao, H.; Hu, W.; Liu, L.P.; Shi, K.H.; Deng, Z.Y.; Li, J. MicroRNA-200a controls Nrf2 activation by target Keap1 in hepatic stellate cell proliferation and fibrosis. Cell. Signal. 2014, 26, 2381–2389. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.J.; Yu, H.W.; Yang, Y.Z.; Wu, W.Y.; Chen, T.Y.; Jia, K.K.; Kang, L.L.; Jiao, R.Q.; Kong, L.D. Polydatin prevents fructose-induced liver inflammation and lipid deposition through increasing miR-200a to regulate Keap1/Nrf2 pathway. Redox Biol. 2018, 18, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Mao, L.; Wang, S.G.; Chen, F.L.; Ji, F.; Fei, H.D. MicroRNA-200a activates Nrf2 signaling to protect osteoblasts from dexamethasone. Oncotarget 2017, 8, 104867–104876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Ye, L.; Zhang, K.; Gao, L.; Xiao, J.; Zhang, Y. Upregulation of microRNA-200a in bone marrow mesenchymal stem cells enhances the repair of spinal cord injury in rats by reducing oxidative stress and regulating Keap1/Nrf2 pathway. Artif. Organs 2020, 44, 744–752. [Google Scholar] [CrossRef]
- Kabaria, S.; Choi, D.C.; Chaudhuri, A.D.; Jain, M.R.; Li, H.; Junn, E. MicroRNA-7 activates Nrf2 pathway by targeting Keap1 expression. Free Radic. Biol. Med. 2015, 89, 548–556. [Google Scholar] [CrossRef] [Green Version]
- Xiao, X.; Lu, Z.; Lin, V.; May, A.; Shaw, D.H.; Wang, Z.; Che, B.; Tran, K.; Du, H.; Shaw, P.X. MicroRNA miR-24-3p Reduces Apoptosis and Regulates Keap1-Nrf2 Pathway in Mouse Cardiomyocytes Responding to Ischemia/Reperfusion Injury. Oxidative Med. Cell. Longev. 2018, 2018, 7042105. [Google Scholar] [CrossRef]
- Xiang, S.; Li, J.; Zhang, Z. miR-26b inhibits isoproterenol-induced cardiac fibrosis via the Keap1/Nrf2 signaling pathway. Exp. Ther. Med. 2020, 19, 2067–2074. [Google Scholar] [CrossRef]
- Zhou, L.; Xu, D.Y.; Sha, W.G.; Shen, L.; Lu, G.Y.; Yin, X.; Wang, M.J. High glucose induces renal tubular epithelial injury via Sirt1/NF-kappaB/microR-29/Keap1 signal pathway. J. Transl. Med. 2015, 13, 352. [Google Scholar] [CrossRef] [Green Version]
- Lv, X.; Huang, J.; Wang, H. MiR-30a-3p ameliorates oxidative stress in rheumatoid arthritis synovial fibroblasts via activation of Nrf2-ARE signaling pathway. Immunol. Lett. 2021, 232, 1–8. [Google Scholar] [CrossRef]
- Huang, R.; Ma, J.; Niu, B.; Li, J.; Chang, J.; Zhang, Y.; Liu, P.; Luan, X. MiR-34b Protects Against Focal Cerebral Ischemia-Reperfusion (I/R) Injury in Rat by Targeting Keap1. J. Stroke Cerebrovasc. Dis. 2019, 28, 1–9. [Google Scholar] [CrossRef]
- Cheng, L.B.; Li, K.R.; Yi, N.; Li, X.M.; Wang, F.; Xue, B.; Pan, Y.S.; Yao, J.; Jiang, Q.; Wu, Z.F. miRNA-141 attenuates UV-induced oxidative stress via activating Keap1-Nrf2 signaling in human retinal pigment epithelium cells and retinal ganglion cells. Oncotarget 2017, 8, 13186–13194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, B.; Liu, H.Y.; Zhu, B.L.; Yue, A.X. MicroRNA-141 protects PC12 cells against hypoxia/reoxygenation-induced injury via regulating Keap1-Nrf2 signaling pathway. J. Bioenerg. Biomembr. 2019, 51, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.Z.; Li, K.R.; Yu, Q.; Jiang, Q.; Yao, J.; Cao, C. Activation of Nrf2 by Ginsenoside Rh3 protects retinal pigment epithelium cells and retinal ganglion cells from UV. Free Radic. Biol. Med. 2018, 117, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Kong, X.; Ma, D. miR-141-3p inhibits vascular smooth muscle cell proliferation and migration via regulating Keap1/Nrf2/HO-1 pathway. IUBMB Life 2020, 72, 2167–2179. [Google Scholar] [CrossRef]
- Ding, X.; Jian, T.; Wu, Y.; Zuo, Y.; Li, J.; Lv, H.; Ma, L.; Ren, B.; Zhao, L.; Li, W.; et al. Ellagic acid ameliorates oxidative stress and insulin resistance in high glucose-treated HepG2 cells via miR-223/keap1-Nrf2 pathway. Biomed. Pharmacother. 2019, 110, 85–94. [Google Scholar] [CrossRef]
- Xiao, Z.; Zheng, Y.B.; Dao, W.X.; Luo, J.F.; Deng, W.H.; Yan, R.C.; Liu, J.S. MicroRNA-328-3p facilitates the progression of gastric cancer via KEAP1/NRF2 axis. Free Radic. Res. 2021, 55, 720–730. [Google Scholar] [CrossRef]
- Duan, F.G.; Wang, M.F.; Cao, Y.B.; Dan, L.; Li, R.Z.; Fan, X.X.; Khan, I.; Lai, H.L.; Zhang, Y.Z.; Hsiao, W.W.; et al. MicroRNA-421 confers paclitaxel resistance by binding to the KEAP1 3’UTR and predicts poor survival in non-small cell lung cancer. Cell Death Dis. 2019, 10, 821. [Google Scholar] [CrossRef] [Green Version]
- Akdemir, B.; Nakajima, Y.; Inazawa, J.; Inoue, J. miR-432 Induces NRF2 Stabilization by Directly Targeting KEAP1. Mol. Cancer Res. 2017, 15, 1570–1578. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Zhou, H.; Zhang, B.; Hu, Q. Exosomal miR-512-3p derived from mesenchymal stem cells inhibits oxidized low-density lipoprotein-induced vascular endothelial cells dysfunction via regulating Keap1. J. Biochem. Mol. Toxicol. 2021, 35, 1–11. [Google Scholar] [CrossRef]
- Xu, X.Z.; Tang, Y.; Cheng, L.B.; Yao, J.; Jiang, Q.; Li, K.R.; Zhen, Y.F. Targeting Keap1 by miR-626 protects retinal pigment epithelium cells from oxidative injury by activating Nrf2 signaling. Free Radic. Biol. Med. 2019, 143, 387–396. [Google Scholar] [CrossRef]
- Li, S.P.; Cheng, W.N.; Li, Y.; Xu, H.B.; Han, H.; Li, P.; Zhang, D.X. Keap1-targeting microRNA-941 protects endometrial cells from oxygen and glucose deprivation-re-oxygenation via activation of Nrf2 signaling. Cell Commun. Signal. 2020, 18, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reziwan, K.; Sun, D.; Zhang, B.; Zhao, Z. MicroRNA-1225 activates Keap1-Nrf2-HO-1 signalling to inhibit TNFalpha-induced osteoclastogenesis by mediating ROS generation. Cell Biochem. Funct. 2019, 37, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Kageyama, S.; Saito, T.; Obata, M.; Koide, R.H.; Ichimura, Y.; Komatsu, M. Negative Regulation of the Keap1-Nrf2 Pathway by a p62/Sqstm1 Splicing Variant. Mol. Cell Biol. 2018, 38, e00642-17. [Google Scholar] [CrossRef] [Green Version]
- Singh, A.; Yadawa, A.K.; Chaturvedi, S.; Wahajuddin, M.; Mishra, A.; Singh, S. Mechanism for antiParkinsonian effect of resveratrol: Involvement of transporters, synaptic proteins, dendrite arborization, biochemical alterations, ER stress and apoptosis. Food Chem. Toxicol. 2021, 155, 112433. [Google Scholar] [CrossRef]
- Betharia, S.; Rondomicronn-Ortiz, A.N.; Brown, D.A. Disubstituted Dithiolethione ACDT Exerts Neuroprotective Effects Against 6-Hydroxydopamine-Induced Oxidative Stress in SH-SY5Y Cells. Neurochem. Res. 2019, 44, 1878–1892. [Google Scholar] [CrossRef] [PubMed]
- Del Fabbro, L.; Rossito Goes, A.; Jesse, C.R.; de Gomes, M.G.; Cattelan Souza, L.; Lobo Ladd, F.V.; Lobo Ladd, A.A.B.; Nunes Arantes, R.V.; Reis Simionato, A.; Oliveira, M.S.; et al. Chrysin protects against behavioral, cognitive and neurochemical alterations in a 6-hydroxydopamine model of Parkinson’s disease. Neurosci. Lett. 2019, 706, 158–163. [Google Scholar] [CrossRef]
- Jaisin, Y.; Ratanachamnong, P.; Kuanpradit, C.; Khumpum, W.; Suksamrarn, S. Protective effects of gamma-mangostin on 6-OHDA-induced toxicity in SH-SY5Y cells. Neurosci. Lett. 2018, 665, 229–235. [Google Scholar] [CrossRef]
- Wang, L.; Yang, Y.F.; Chen, L.; He, Z.Q.; Bi, D.Y.; Zhang, L.; Xu, Y.W.; He, J.C. Compound Dihuang Granule Inhibits Nigrostriatal Pathway Apoptosis in Parkinson’s Disease by Suppressing the JNK/AP-1 Pathway. Front. Pharmacol. 2021, 12, 621359. [Google Scholar] [CrossRef]
- Tang, Z.; Hu, B.; Zang, F.; Wang, J.; Zhang, X.; Chen, H. Nrf2 drives oxidative stress-induced autophagy in nucleus pulposus cells via a Keap1/Nrf2/p62 feedback loop to protect intervertebral disc from degeneration. Cell Death Dis. 2019, 10, 510. [Google Scholar] [CrossRef]
- Liu, T.; Cao, L.; Zhang, T.; Fu, H. Molecular docking studies, anti-Alzheimer’s disease, antidiabetic, and anti-acute myeloid leukemia potentials of narcissoside. Arch. Physiol. Biochem. 2020, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Vignatelli, L.; Baccari, F.; Belotti, L.M.B.; Zenesini, C.; Baldin, E.; Calandra-Buonaura, G.; Cortelli, P.; Descovich, C.; Giannini, G.; Guarino, M.; et al. The Indirect Impact of COVID-19 on Major Clinical Outcomes of People With Parkinson’s Disease or Parkinsonism: A Cohort Study. Front. Neurol. 2022, 13, 873925. [Google Scholar] [CrossRef] [PubMed]



















| Genes of C. elegans (Human) | Primer Sequences (5′-3′) | (Start→End) Size (bp) |
|---|---|---|
| Skn-1 (Nrf-2) | Forward: 5′-TCCACCAGCATCTCCATTC-3′ Reverse: 5′-ACTTCTCCATAGCACATCAATC-3′ | (480–600) 121 |
| gcs-1 (Gclc) | Forward: 5′-GTTACAAGCCGAAGAGCAG-3′ Reverse: 5′-TGAAGCAGCGATGGACC-3′ | (231–361) 131 |
| E01A2.1 (Gclm) | Forward: 5′-CACCAATCCAAACCTCTACTC-3′ Reverse: 5′-TCAAAAGTGGCAGCAATAGC-3′ | (882–1019) 138 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, R.-H.; Tsai, C.-W.; Liu, S.-P.; Chiu, S.-C.; Chen, Y.-C.; Chiang, Y.-T.; Kuo, Y.-H.; Shyu, W.-C.; Lin, S.-Z. Neuroprotective Capability of Narcissoside in 6-OHDA-Exposed Parkinson’s Disease Models through Enhancing the MiR200a/Nrf-2/GSH Axis and Mediating MAPK/Akt Associated Signaling Pathway. Antioxidants 2022, 11, 2089. https://doi.org/10.3390/antiox11112089
Fu R-H, Tsai C-W, Liu S-P, Chiu S-C, Chen Y-C, Chiang Y-T, Kuo Y-H, Shyu W-C, Lin S-Z. Neuroprotective Capability of Narcissoside in 6-OHDA-Exposed Parkinson’s Disease Models through Enhancing the MiR200a/Nrf-2/GSH Axis and Mediating MAPK/Akt Associated Signaling Pathway. Antioxidants. 2022; 11(11):2089. https://doi.org/10.3390/antiox11112089
Chicago/Turabian StyleFu, Ru-Huei, Chia-Wen Tsai, Shih-Ping Liu, Shao-Chih Chiu, Yen-Chuan Chen, Yu-Ting Chiang, Yun-Hua Kuo, Woei-Cherng Shyu, and Shinn-Zong Lin. 2022. "Neuroprotective Capability of Narcissoside in 6-OHDA-Exposed Parkinson’s Disease Models through Enhancing the MiR200a/Nrf-2/GSH Axis and Mediating MAPK/Akt Associated Signaling Pathway" Antioxidants 11, no. 11: 2089. https://doi.org/10.3390/antiox11112089