Antioxidant, Anti-Inflammatory, and Anti-Aging Potential of a Kalmia angustifolia Extract and Identification of Some Major Compounds
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and Extract Preparation
2.2. Isolation of Major Compounds of the K. angustifolia Extract
2.3. NMR and GC-MS Analysis
2.4. Biopsies and Cell Extraction
2.5. Cell Culture
2.6. Cytotoxicity Assay
2.7. Oxygen Radical Absorbance Capacity (ORAC) Assay
2.8. Antioxidant Activity Assessed Using a Cell-Based Assay
2.9. Anti-Inflammatory Activity Assessed by Nitrite Quantification
2.10. Skin Substitute Production
2.11. Histological Analyses
2.12. Immunofluorescence Staining
2.13. Statistical Analysis
3. Results
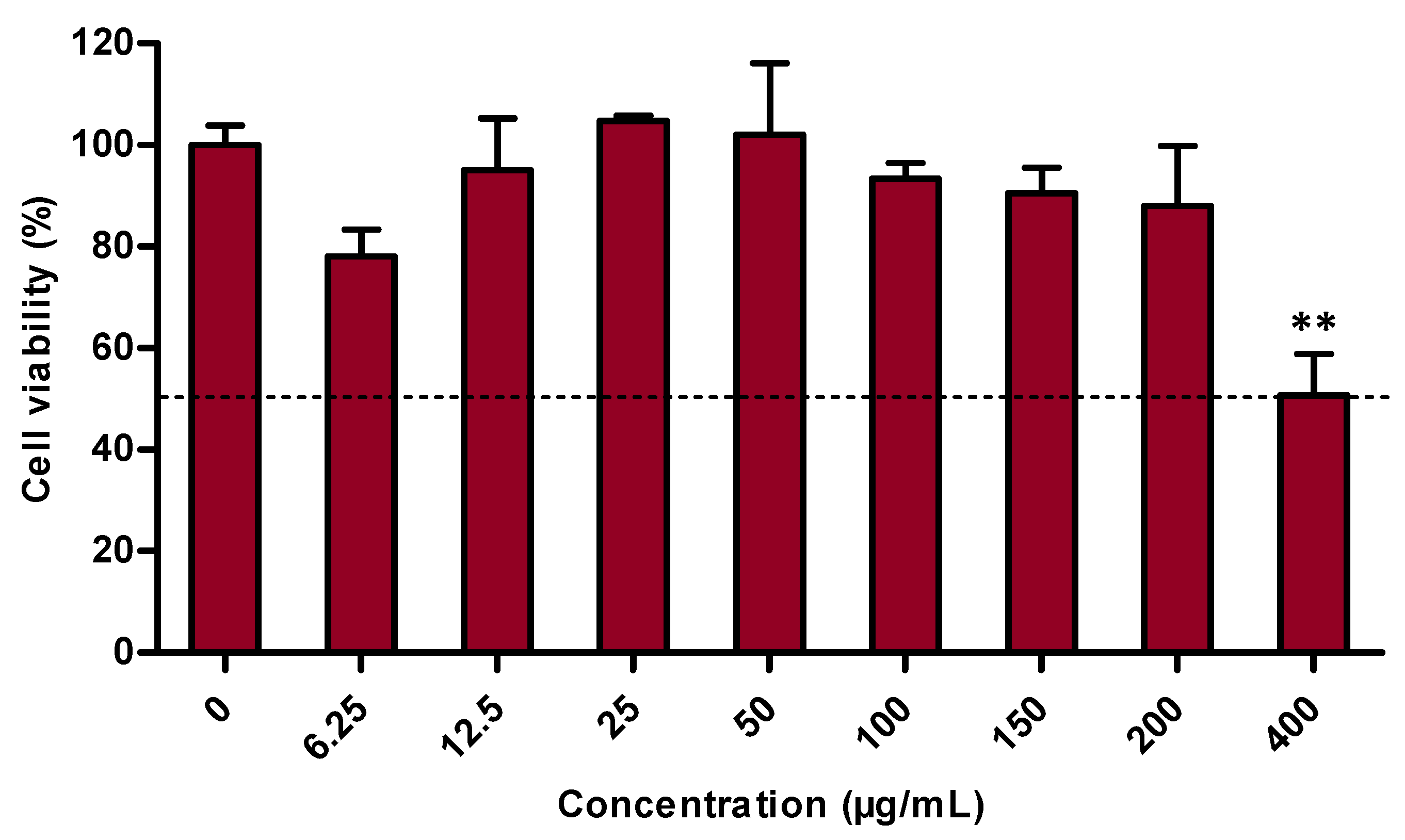
3.1. Cell Viability Evaluation
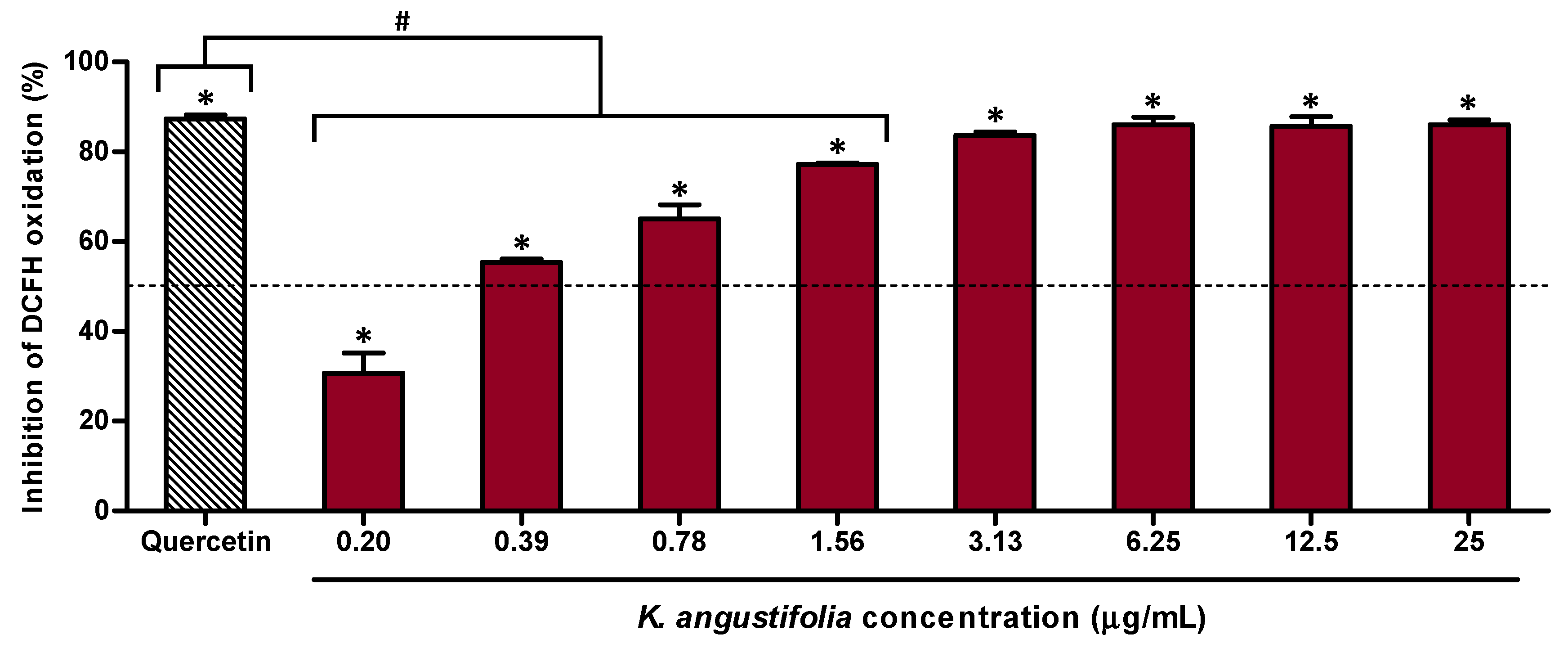
3.2. Antioxidant Capacity
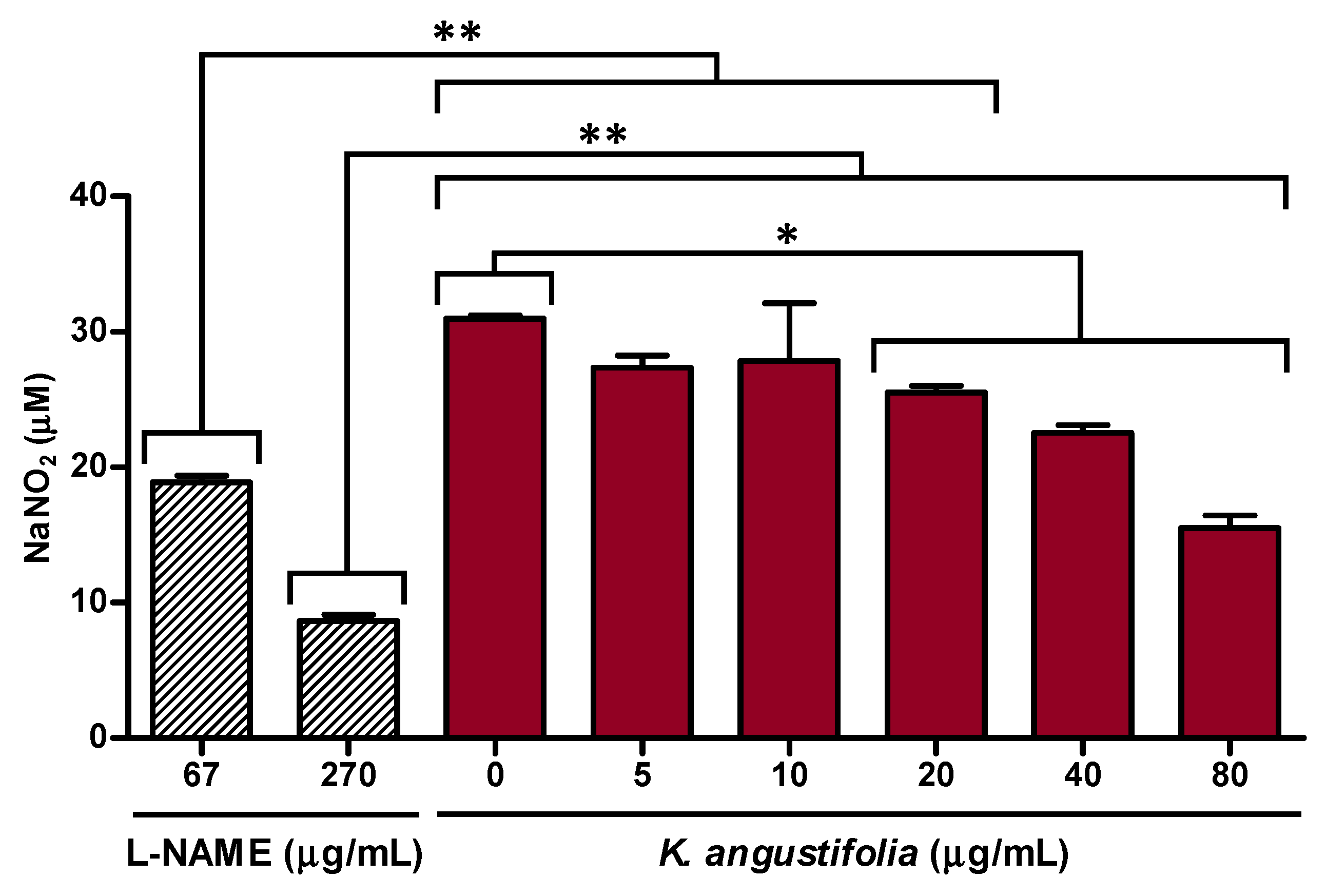
3.3. Anti-Inflammatory Potential
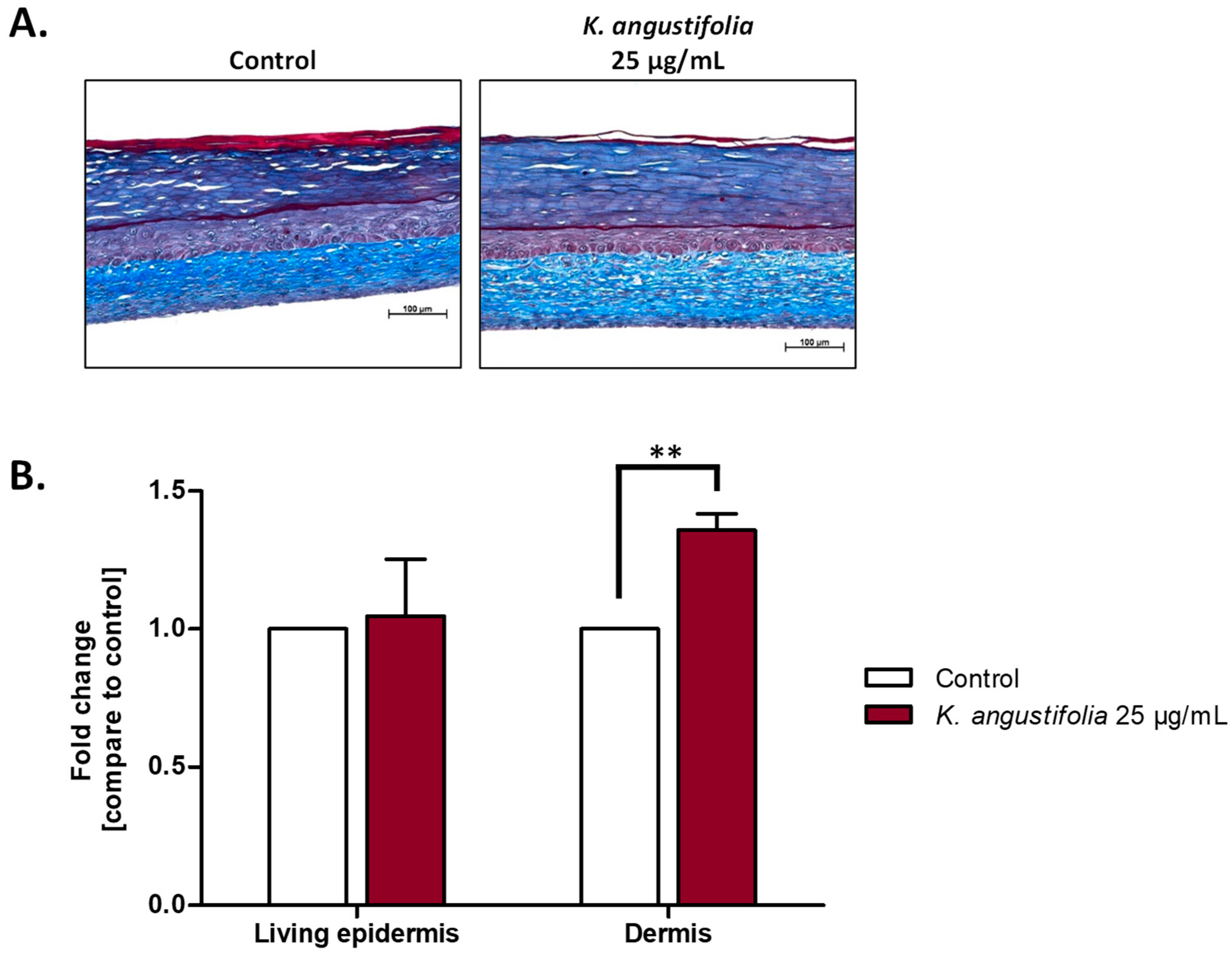
3.4. Histological Analyses
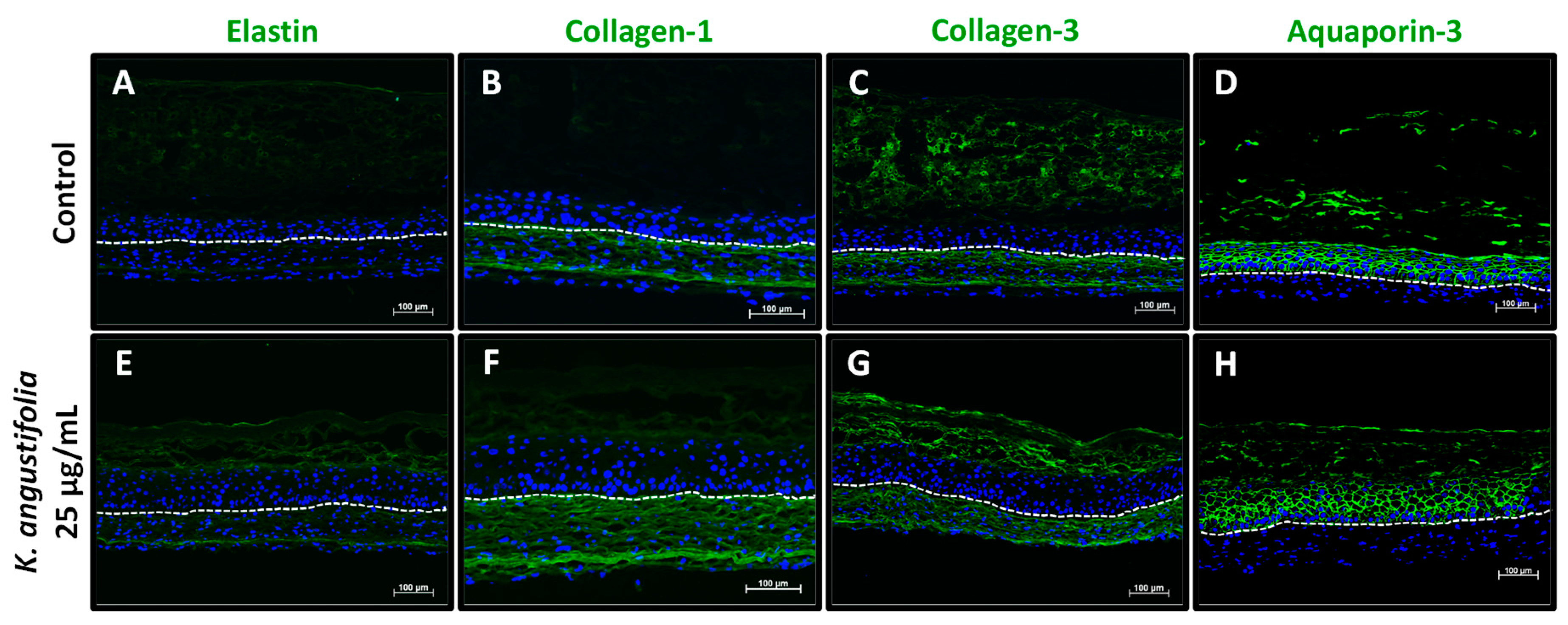
3.5. Immunofluorescence Staining
3.6. Isolation and Identification of Some Major Compounds of the K. angustifolia Extract
3.7. Biological Activities of the Identified Compounds of the K. angustifolia Extract
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, S.; Duan, E. Fighting against Skin Aging: The Way from Bench to Bedside. Cell Transpl. 2018, 27, 729–738. [Google Scholar] [CrossRef]
- Tobin, D.J.; Veysey, E.C.; Finlay, A.Y. Aging and the Skin. In Textbook of Geriatric Medicine and Gerontology; Fillit, H.M., Rockwood, K., Young, J.B., Eds.; Elsevier: London, UK, 2016. [Google Scholar]
- Titus, B.D.; Sidhu, S.S.; Mallik, A.U. A summary of Some Studies on Kalmia angustifolia L.: A Problem Species in Newfoundland Forestry; Government of Canada, Natural Resources Canada, Canadian Forest Service: St. John’s, NL, Canada, 1995; pp. 1–68.
- Erichsen-Brown, C. Medicinal and Other Uses of North American Plants: A Historical Survey with Special Reference to the Eastern Indian Tribes; Dover Publications: Mineola, NY, USA, 1989; p. 544. [Google Scholar]
- Jansen, S.A.; Kleerekooper, I.; Hofman, Z.L.M.; Kappen, I.F.P.M.; Stary-Weinzinger, A.; van der Heyden, M.A.G. Grayanotoxin poisoning: ‘mad honey disease’ and beyond. Cardiovasc. Toxicol. 2012, 12, 208–215. [Google Scholar] [CrossRef] [Green Version]
- Gunduz, A.; Turedi, S.; Uzun, H.; Topbas, M. Mad honey poisoning. Am. J. Emerg. Med. 2006, 24, 595–598. [Google Scholar] [CrossRef]
- Jacobs, M.L.; Lloyd, W.R. A phytochemical study of kalmia angustifolia L. J. Am. Pharm. Assoc. 1939, 28, 408–412. [Google Scholar] [CrossRef]
- Maeda, K.; Fukuda, M. Arbutin: Mechanism of its depigmenting action in human melanocyte culture. J. Pharmacol. Exp. Ther. 1996, 276, 765–769. [Google Scholar]
- Saleem, A.; Harris, C.S.; Asim, M.; Cuerrier, A.; Martineau, L.; Haddad, P.S.; Arnason, J.T. A RP-HPLC-DAD-APCI/MSD method for the characterisation of medicinal Ericaceae used by the Eeyou Istchee Cree First Nations. Phytochem. Anal. 2010, 21, 328–339. [Google Scholar] [CrossRef]
- Zhu, H.; Mallik, A.U. Interactions between Kalmia and black spruce: Isolation and identification of allelopathic compounds. J. Chem. Ecol. 1994, 20, 407–421. [Google Scholar] [CrossRef] [PubMed]
- Mallik, A.U. Can Kalmia angustifolia interference to black spruce (Picea mariana) be explained by allelopathy? For. Ecol. Manag. 2002, 160, 75–84. [Google Scholar] [CrossRef]
- Zeng, R.S.; Mallik, A.U. Selected Ectomycorrhizal Fungi of Black Spruce (Picea mariana) can Detoxify Phenolic Compounds of Kalmia angustifolia. J. Chem. Ecol. 2006, 32, 1473–1489. [Google Scholar] [CrossRef]
- Nierop, K.G.J.; Preston, C.M.; Kaal, J. Thermally Assisted Hydrolysis and Methylation of Purified Tannins from Plants. Anal. Chem. 2005, 77, 5604–5614. [Google Scholar] [CrossRef]
- Burke, J.W.; Doskotch, R.W. High Field 1H- and 13C-nmr Assignments of Grayanotoxins I, IV, and XIV Isolated from Kalmia angustifolia. J. Nat. Prod. 1990, 53, 131–137. [Google Scholar] [CrossRef]
- Burke, J.W.; Doskotch, R.W.; Ni, C.Z.; Clardy, J. Kalmanol, a pharmacologically active diterpenoid with a new ring skeleton from Kalmia angustifolia L. J. Am. Chem. Soc. 1989, 111, 5831–5833. [Google Scholar] [CrossRef]
- Netzlaff, F.; Lehr, C.M.; Wertz, P.W.; Schaefer, U.F. The human epidermis models EpiSkin®, SkinEthic® and EpiDerm®: An evaluation of morphology and their suitability for testing phototoxicity, irritancy, corrosivity, and substance transport. Eur. J. Pharm. Biopharm. 2005, 60, 167–178. [Google Scholar] [CrossRef]
- Spielmann, H.; Hoffmann, S.; Liebsch, M.; Botham, P.; Fentem, J.H.; Eskes, C.; Roguet, R.; Cotovio, J.; Cole, T.; Worth, A.; et al. The ECVAM International Validation Study on in Vitro Tests for Acute Skin Irritation: Report on the Validity of the EPISKIN and EpiDerm Assays and on the Skin Integrity Function Test. Altern. Lab. Anim. 2007, 35, 559–601. [Google Scholar] [CrossRef] [Green Version]
- Bataillon, M.; Lelièvre, D.; Chapuis, A.; Thillou, F.; Autourde, J.B.; Durand, S.; Boyera, N.; Rigaudeau, A.-S.; Besné, I.; Pellevoisin, C. Characterization of a New Reconstructed Full Thickness Skin Model, T-Skin™, and its Application for Investigations of Anti-Aging Compounds. Int. J. Mol. Sci. 2019, 20, 2240. [Google Scholar] [CrossRef] [Green Version]
- Meloni, M.; Farina, A.; de Servi, B. Molecular modifications of dermal and epidermal biomarkers following UVA exposures on reconstructed full-thickness human skin. Photochem. Photobiol. Sci. 2010, 9, 439–447. [Google Scholar] [CrossRef]
- Larouche, D.; Jean, J.; Berthod, F.; Germain, L.; Pouliot, R. Markers for an in vitro skin substitute. In Methods in Bioengineering: Alternative Technologies to Animal Testing; Maguire, T., Novik, E., Eds.; Artech House: Boston, MA, USA, 2010; pp. 183–203. [Google Scholar]
- Jean, J.; García-Pérez, M.-E.; Pouliot, R. Bioengineered Skin: The Self- Assembly Approach. J. Tissue Sci. Eng. 2011, S5, 1. [Google Scholar] [CrossRef]
- Germain, L.; Rouabhia, M.; Guignard, R.; Carrier, L.; Bouvard, V.; Auger, F.A. Improvement of human keratinocyte isolation and culture using thermolysin. Burns 1993, 19, 99–104. [Google Scholar] [CrossRef]
- Auger, F.A.; López Valle, C.A.; Guignard, R.; Tremblay, N.; Noel, B.; Goulet, F.; Germain, L. Skin equivalent produced with human collagen. Vitr. Cell. Dev. Biol. -Anim. 1995, 31, 432–439. [Google Scholar] [CrossRef]
- Ou, B.; Hampsch-Woodill, M.; Prior, R.L. Development and Validation of an Improved Oxygen Radical Absorbance Capacity Assay Using Fluorescein as the Fluorescent Probe. J. Agric. Food Chem. 2001, 49, 4619–4626. [Google Scholar] [CrossRef]
- Girard-Lalancette, K.; Pichette, A.; Legault, J. Sensitive cell-based assay using DCFH oxidation for the determination of pro- and antioxidant properties of compounds and mixtures: Analysis of fruit and vegetable juices. Food Chem. 2009, 115, 720–726. [Google Scholar] [CrossRef]
- Legault, J.; Girard-Lalancette, K.; Grenon, C.; Dussault, C.; Pichette, A. Antioxidant Activity, Inhibition of Nitric Oxide Overproduction, and In Vitro Antiproliferative Effect of Maple Sap and Syrup from Acer saccharum. J. Med. Food 2010, 13, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Sylvester, P.W. Optimization of the Tetrazolium Dye (MTT) Colorimetric Assay for Cellular Growth and Viability. In Drug Design and Discovery: Methods and Protocols; Satyanarayanajois, S.D., Ed.; Humana Press: Totowa, NJ, USA, 2011; 9p. [Google Scholar]
- Morgan, D.M.L. Tetrazolium (MTT) Assay for Cellular Viability and Activity. In Polyamine Protocols; Morgan, D.M.L., Ed.; Humana Press: Totowa, NJ, USA, 1998; pp. 179–184. [Google Scholar]
- Fujimori, K.; Shibano, M. Avicularin, a plant flavonoid, suppresses lipid accumulation through repression of C/EBPα-activated GLUT4-mediated glucose uptake in 3T3-L1 cells. J. Agric. Food Chem. 2013, 61, 5139–5147. [Google Scholar] [CrossRef] [PubMed]
- de Bruyne, T.; Pieters, L.A.C.; Dommisse, R.A.; Kolodziej, H.; Wray, V.; Domke, T.; Vlietinck, A.J. Unambiguous assignments for free dimeric proanthocyanidin phenols from 2D NMR. Phytochemistry 1996, 43, 265–272. [Google Scholar] [CrossRef]
- Lou, H.; Yamazaki, Y.; Sasaki, T.; Uchida, M.; Tanaka, H.; Oka, S. A-type proanthocyanidins from peanut skins. Phytochemistry 1999, 51, 297–308. [Google Scholar] [CrossRef]
- Nkengfack, A.E.; Van Heerden, F.R.; Fuendjiep, V.; Fomum, Z.T. Asebotin, a dihydrochalcone glucoside from Guibourtia tessmannii. Fitoterapia 2001, 72, 834–836. [Google Scholar] [CrossRef]
- Mancini, S.D.; Edwards, J.M. Cytotoxic principles From the Sap of Kalmia latifolia. J. Nat. Prod. 1979, 42, 483–488. [Google Scholar] [CrossRef]
- Dorato, S. Chapter 1—General Concepts: Current Legislation on Cosmetics in Various Countries. In Analysis of Cosmetic Products, 2nd ed.; Salvador, A., Chisvert, A., Eds.; Elsevier: Boston, MA, USA, 2018; pp. 3–37. [Google Scholar] [CrossRef]
- Akbarsha, M.A.; Mascarenhas, B. Cosmetic Regulation and Alternatives to Animal Experimentation in India. In Alternatives to Animal Testing; Kojima, H., Seidle, T., Spielmann, H., Eds.; Springer: Singapore, 2019; pp. 57–62. [Google Scholar]
- Taylor, K.; Rego Alvarez, L. Regulatory drivers in the last 20 years towards the use of in silico techniques as replacements to animal testing for cosmetic-related substances. Comput. Toxicol. 2020, 13, 100112. [Google Scholar] [CrossRef]
- de Ávila, R.I.; Valadares, M.C. Brazil Moves Toward the Replacement of Animal Experimentation. Altern. Lab. Anim. 2019, 47, 71–81. [Google Scholar] [CrossRef]
- Almeida, A.; Sarmento, B.; Rodrigues, F. Insights on in vitro models for safety and toxicity assessment of cosmetic ingredients. Int. J. Pharm. 2017, 519, 178–185. [Google Scholar] [CrossRef]
- Dufour, D.; Pichette, A.; Mshvildadze, V.; Bradette-Hébert, M.-E.; Lavoie, S.; Longtin, A.; Laprise, C.; Legault, J. Antioxidant, anti-inflammatory and anticancer activities of methanolic extracts from Ledum groenlandicum Retzius. J. Ethnopharmacol. 2007, 111, 22–28. [Google Scholar] [CrossRef]
- Ayeleso, A.O.; Oguntibeju, O.O.; Brooks, N.L. In vitro study on the antioxidant potentials of the leaves and fruits of Nauclea latifolia. Sci. World J. 2014, 2014, 437081. [Google Scholar] [CrossRef] [PubMed]
- Grauzdytė, D.; Pukalskas, A.; Viranaicken, W.; El Kalamouni, C.; Venskutonis, P.R. Protective effects of Phyllanthus phillyreifolius extracts against hydrogen peroxide induced oxidative stress in HEK293 cells. PLoS ONE 2018, 13, e0207672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Y.; Fisher, G.J. Ultraviolet (UV) light irradiation induced signal transduction in skin photoaging. J. Dermatol. Sci. Suppl. 2005, 1, S1–S8. [Google Scholar] [CrossRef]
- Morita, A. Tobacco smoke causes premature skin aging. J. Dermatol. Sci. 2007, 48, 169–175. [Google Scholar] [CrossRef]
- Parrado, C.; Mercado-Saenz, S.; Perez-Davo, A.; Gilaberte, Y.; Gonzalez, S.; Juarranz, A. Environmental Stressors on Skin Aging. Mechanistic Insights. Front Pharm. 2019, 10, 759. [Google Scholar] [CrossRef] [PubMed]
- Rinnerthaler, M.; Bischof, J.; Streubel, M.K.; Trost, A.; Richter, K. Oxidative stress in aging human skin. Biomolecules 2015, 5, 545–589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borg, M.; Brincat, S.; Camilleri, G.; Schembri-Wismayer, P.; Brincat, M.; Calleja-Agius, J. The role of cytokines in skin aging. Climacteric 2013, 16, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Kang, S.; Varani, J.; Bata-Csorgo, Z.; Wan, Y.; Datta, S.; Voorhees, J.J. Mechanisms of photoaging and chronological skin aging. Arch. Dermatol. 2002, 138, 1462–1470. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Quan, T.; Shao, Y.; Voorhees, J.J.; Fisher, G.J. Oxidative exposure impairs TGF-β pathway via reduction of type II receptor and SMAD3 in human skin fibroblasts. Age 2014, 36, 9623. [Google Scholar] [CrossRef] [Green Version]
- Kammeyer, A.; Luiten, R.M. Oxidation events and skin aging. Ageing Res. Rev. 2015, 21, 16–29. [Google Scholar] [CrossRef]
- Fisher, G.J.; Wang, Z.; Datta, S.C.; Varani, J.; Kang, S.; Voorhees, J.J. Pathophysiology of Premature Skin Aging Induced by Ultraviolet Light. N. Engl. J. Med. 1997, 337, 1419–1429. [Google Scholar] [CrossRef]
- Gritsenko, D.A.; Orlova, O.A.; Linkova, N.S.; Khavinson, V.K. Transcription factor p53 and skin aging. Adv. Gerontol. 2017, 7, 114–119. [Google Scholar] [CrossRef]
- Rinnerthaler, M.; Streubel, M.K.; Bischof, J.; Richter, K. Skin aging, gene expression and calcium. Exp. Gerontol. 2015, 68, 59–65. [Google Scholar] [CrossRef]
- Raschke, C.; Elsner, P. Skin Aging: A Brief Summary of Characteristic Changes. In Textbook of Aging Skin; Farage, M.A., Miller, K.W., Maibach, H.I., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 37–43. [Google Scholar]
- Scharffetter–Kochanek, K.; Brenneisen, P.; Wenk, J.; Herrmann, G.; Ma, W.; Kuhr, L.; Meewes, C.; Wlaschek, M. Photoaging of the skin from phenotype to mechanisms. Exp. Gerontol. 2000, 35, 307–316. [Google Scholar] [CrossRef]
- Mora Huertas, A.C.; Schmelzer, C.E.H.; Luise, C.; Sippl, W.; Pietzsch, M.; Hoehenwarter, W.; Heinz, A. Degradation of tropoelastin and skin elastin by neprilysin. Biochimie 2018, 146, 73–78. [Google Scholar] [CrossRef]
- McDaniel, D.; Farris, P.; Valacchi, G. Atmospheric skin aging—Contributors and inhibitors. J. Cosmet. Dermatol. 2018, 17, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Freitas-Rodríguez, S.; Folgueras, A.R.; López-Otín, C. The role of matrix metalloproteinases in aging: Tissue remodeling and beyond. Biochim. Et Biophys. Acta (BBA)-Mol. Cell Res. 2017, 1864, 2015–2025. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Wen, X.; Hao, D.; Zhang, N.; He, G.; Jiang, X. NF-κB signaling in skin aging. Mech. Ageing Dev. 2019, 184, 111160. [Google Scholar] [CrossRef]
- Davidson, J.M.; LuValle, P.A.; Zoia, O.; Quaglino, D.; Giro, M. Ascorbate Differentially Regulates Elastin and Collagen Biosynthesis in Vascular Smooth Muscle Cells and Skin Fibroblasts by Pretranslational Mechanisms. J. Biol. Chem. 1997, 272, 345–352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Clerck, Y.A.; Jones, P.A. The effect of ascorbic acid on the nature and production of collagen and elastin by rat smooth-muscle cells. Biochem. J. 1980, 186, 217–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Sample | ORAC Values a (µmol TE/mg) |
|---|---|
| Quercetin | 21 ± 2 |
| Catechin | 20 ± 2 |
| K. angustifolia | 16 ± 3 |
| Compound | ORAC Values a (µmol TE/µmol) | Cell-Based Antioxidant Capacity IC50 (µM) | Anti-Inflammatory Activity IC50 (µM) |
|---|---|---|---|
| Epicatechin | 4.6 ± 0.6 | 1.22 ± 0.06 | >100 |
| Catechin | 6.7 ± 0.3 | 0.8 ± 0.3 | >100 |
| Avicularin | 4.1 ± 0.3 | 0.40 ± 0.03 | >100 |
| Proanthocyanidin A2 | 4.5 ± 0.8 | 0.30 ± 0.03 | >100 |
| Proanthocyanidin Ax b | 5.3 ± 0.6 | 0.24 ± 0.02 | >100 |
| Asebotin | 9 ± 1 | 1.70 ± 0.09 | >100 |
| Quercetin | 7.6 ± 0.7 | 0.21 ± 0.06 | - |
| Trolox | 0.91 ± 0.11 | 0.024 ± 0.002 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grenier, A.; Legault, J.; Pichette, A.; Jean, L.; Bélanger, A.; Pouliot, R. Antioxidant, Anti-Inflammatory, and Anti-Aging Potential of a Kalmia angustifolia Extract and Identification of Some Major Compounds. Antioxidants 2021, 10, 1373. https://doi.org/10.3390/antiox10091373
Grenier A, Legault J, Pichette A, Jean L, Bélanger A, Pouliot R. Antioxidant, Anti-Inflammatory, and Anti-Aging Potential of a Kalmia angustifolia Extract and Identification of Some Major Compounds. Antioxidants. 2021; 10(9):1373. https://doi.org/10.3390/antiox10091373
Chicago/Turabian StyleGrenier, Alexe, Jean Legault, André Pichette, Lorry Jean, Audrey Bélanger, and Roxane Pouliot. 2021. "Antioxidant, Anti-Inflammatory, and Anti-Aging Potential of a Kalmia angustifolia Extract and Identification of Some Major Compounds" Antioxidants 10, no. 9: 1373. https://doi.org/10.3390/antiox10091373
APA StyleGrenier, A., Legault, J., Pichette, A., Jean, L., Bélanger, A., & Pouliot, R. (2021). Antioxidant, Anti-Inflammatory, and Anti-Aging Potential of a Kalmia angustifolia Extract and Identification of Some Major Compounds. Antioxidants, 10(9), 1373. https://doi.org/10.3390/antiox10091373