Effect of Paired Associative Stimulation on Corticomotor Excitability in Chronic Smokers
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Transcranial Magnetic Stimulation
2.3. Paired Associative Stimulation
2.4. Statistical Analysis Procedures
3. Results
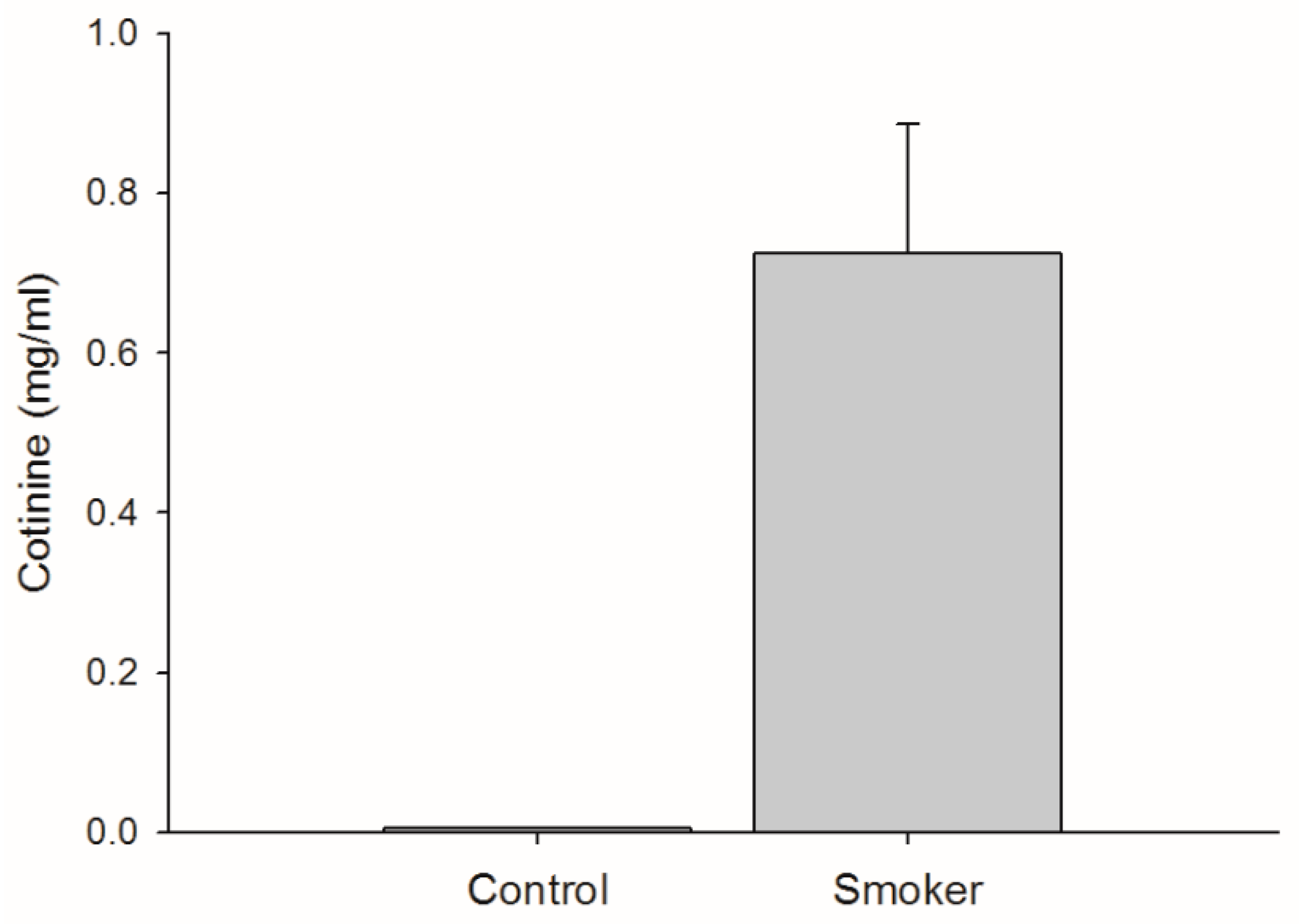
3.1. Cigarette Consumption
3.2. Cortical Stimulation Parameters
3.3. Force Output
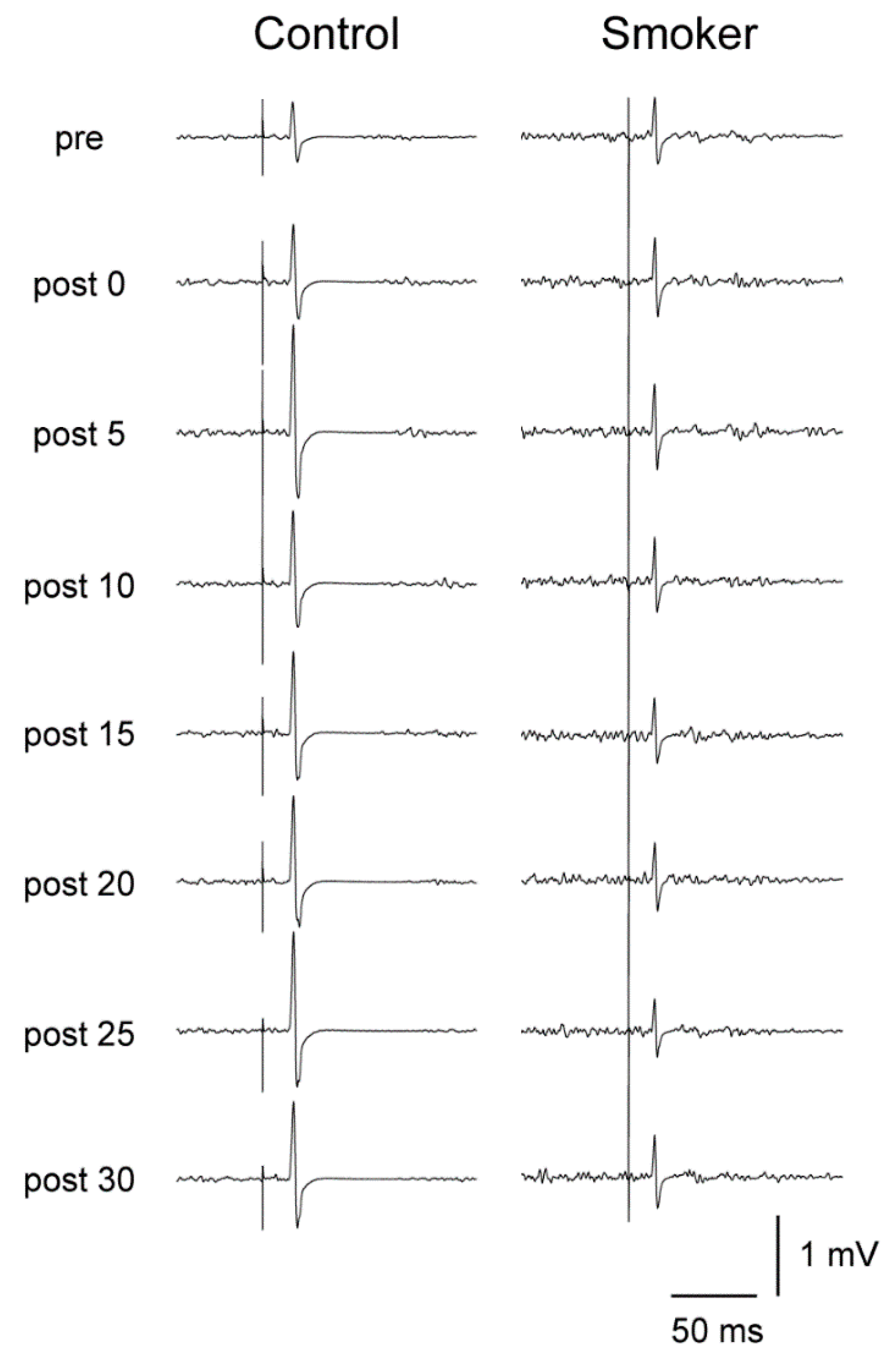
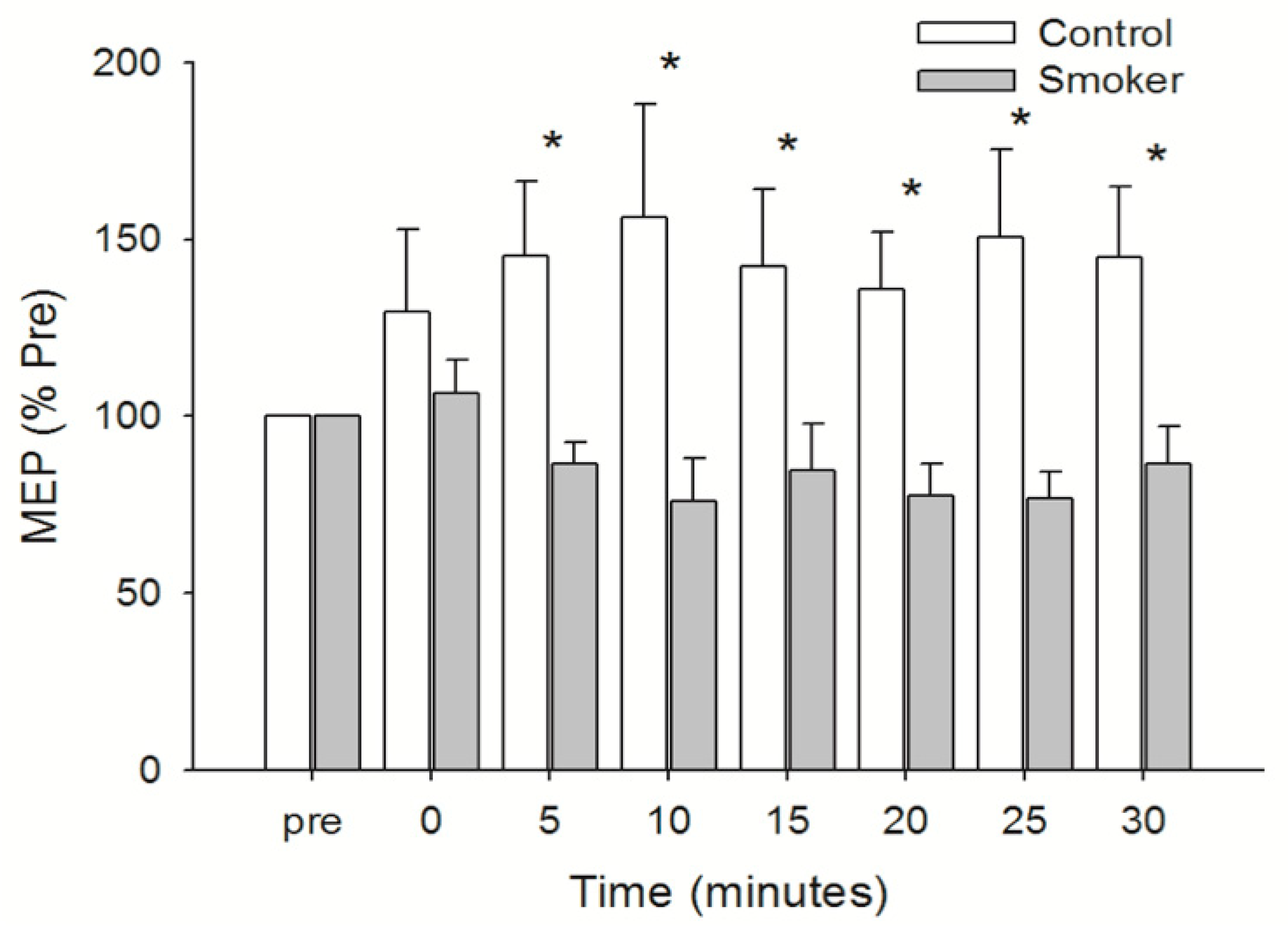
3.4. Motor Cortical Excitability
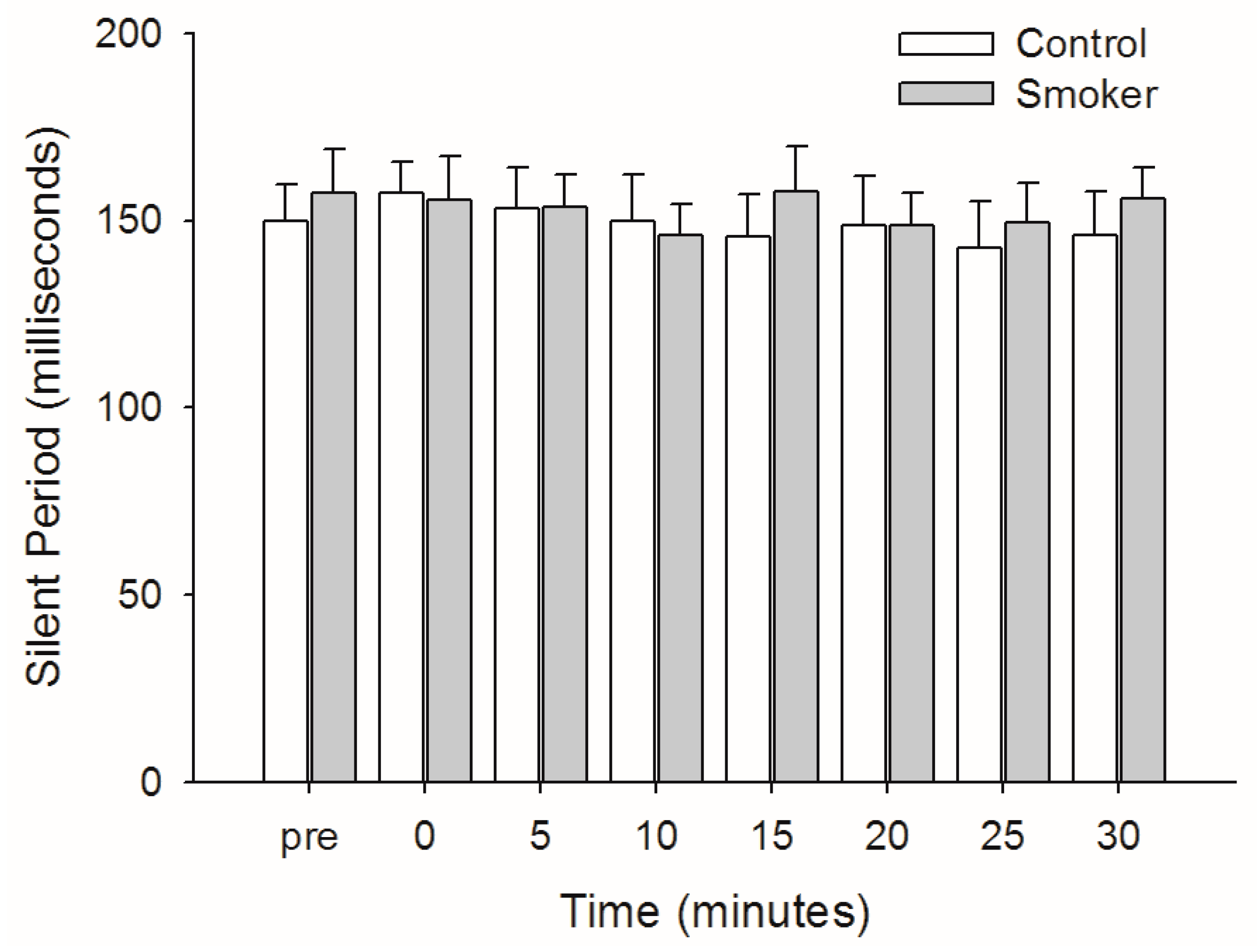
3.5. Silent Period
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Classen, J.; Liepert, J.; Wise, S.P.; Hallett, M.; Cohen, L.G. Rapid plasticity of human cortical movement representation induced by practice. J. Neurophysiol. 1998, 79, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Nudo, R.J.; Milliken, G.W.; Jenkins, W.M.; Merzenich, M.M. Use-dependent alterations of movement representations in primary motor cortex of adult squirrel monkeys. J. Neurosci. Off. J. Soc. Neurosci. 1996, 16, 785–807. [Google Scholar] [CrossRef]
- Ziemann, U.; Ilic, T.V.; Pauli, C.; Meintzschel, F.; Ruge, D. Learning modifies subsequent induction of long-term potentiation-like and long-term depression-like plasticity in human motor cortex. J. Neurosci. Off. J. Soc. Neurosci. 2004, 24, 1666–1672. [Google Scholar] [CrossRef] [PubMed]
- Tecchio, F.; Zappasodi, F.; Pasqualetti, P.; De Gennaro, L.; Pellicciari, M.C.; Ercolani, M.; Squitti, R.; Rossini, P.M. Age dependence of primary motor cortex plasticity induced by paired associative stimulation. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 2008, 119, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Cirillo, J.; Lavender, A.P.; Ridding, M.C.; Semmler, J.G. Motor cortex plasticity induced by paired associative stimulation is enhanced in physically active individuals. J. Physiol. 2009, 587, 5831–5842. [Google Scholar] [CrossRef] [PubMed]
- Hebb, D.O. The Organization of Behavior: A Neuropsychological Theory; Wiley: New York, NY, USA, 1949; 335p. [Google Scholar]
- Srivanitchapoom, P.; Park, J.E.; Thirugnanasambandam, N.; Panyakaew, P.; Ramos, V.F.; Pandey, S.; Wu, T.; Hallett, M. Inducing LTD-like effect in the human motor cortex with low frequency and very short duration paired associative stimulation: An exploratory study. Neural Plast 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Rothwell, J.C. Techniques and mechanisms of action of transcranial stimulation of the human motor cortex. J. Neurosci. Methods 1997, 74, 113–122. [Google Scholar] [CrossRef]
- Rosen, I.; Asanuma, H. Peripheral afferent inputs to the forelimb area of the monkey motor cortex: Input-output relations. Exp. Brain Res. 1972, 14, 257–273. [Google Scholar] [CrossRef] [PubMed]
- Muller-Dahlhaus, F.; Ziemann, U.; Classen, J. Plasticity resembling spike-timing dependent synaptic plasticity: The evidence in human cortex. Front. Synaptic Neurosci. 2010, 2, 34. [Google Scholar] [CrossRef] [PubMed]
- Muller-Dahlhaus, J.F.; Orekhov, Y.; Liu, Y.; Ziemann, U. Interindividual variability and age-dependency of motor cortical plasticity induced by paired associative stimulation. Exp. Brain Res. 2008, 187, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Stefan, K.; Kunesch, E.; Cohen, L.G.; Benecke, R.; Classen, J. Induction of plasticity in the human motor cortex by paired associative stimulation. Brain 2000, 123 Pt 3, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Rodvelt, K.R.; Schachtman, T.R.; Kracke, G.R.; Miller, D.K. NMDA receptor blockade augmented nicotine-evoked dopamine release from rat prefrontal cortex slices. Neurosci. Lett. 2008, 440, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Yang, K.; Wang, H.; Wu, J. Exposure of nicotine to ventral tegmental area slices induces glutamatergic synaptic plasticity on dopamine neurons. Synapse 2011, 65, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Keller, A. Intrinsic connections between representation zones in the cat motor cortex. Neuroreport 1993, 4, 515–518. [Google Scholar] [CrossRef] [PubMed]
- Hess, G.; Donoghue, J.P. Long-term potentiation of horizontal connections provides a mechanism to reorganize cortical motor maps. J. Neurophysiol. 1994, 71, 2543–2547. [Google Scholar] [CrossRef] [PubMed]
- McBain, C.J.; Mayer, M.L. N-methyl-D-aspartic acid receptor structure and function. Physiol. Rev. 1994, 74, 723–760. [Google Scholar] [CrossRef] [PubMed]
- Monyer, H.; Burnashev, N.; Laurie, D.J.; Sakmann, B.; Seeburg, P.H. Developmental and regional expression in the rat brain and functional properties of four NMDA receptors. Neuron 1994, 12, 529–540. [Google Scholar] [CrossRef]
- Monaghan, D.T.; Larsen, H. NR1 and NR2 subunit contributions to N-methyl-D-aspartate receptor channel blocker pharmacology. J. Pharm. Exp. 1997, 280, 614–620. [Google Scholar]
- Trujillo, K.A. Are NMDA receptors involved in opiate-induced neural and behavioral plasticity? A review of preclinical studies. Psychopharmacology 2000, 151, 121–141. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.; Mukherjee, K.; Balhara, Y.P. The role of NMDA receptor antagonists in nicotine tolerance, sensitization, and physical dependence: A preclinical review. Yonsei Med. J. 2008, 49, 175–188. [Google Scholar] [CrossRef] [PubMed]
- Lang, N.; Hasan, A.; Sueske, E.; Paulus, W.; Nitsche, M.A. Cortical hypoexcitability in chronic smokers? A transcranial magnetic stimulation study. Neuropsychopharmacology 2008, 33, 2517–2523. [Google Scholar] [CrossRef] [PubMed]
- Batsikadze, G.; Paulus, W.; Hasan, A.; Grundey, J.; Kuo, M.F.; Nitsche, M.A. Compromised neuroplasticity in cigarette smokers under nicotine withdrawal is restituted by the nicotinic α4β2-receptor partial agonist varenicline. Sci. Rep. 2017, 7, 1387. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, K.; Williamon, A.; Rothwell, J.C. Motorcortical excitability and synaptic plasticity is enhanced in professional musicians. J. Neurosci. Off. J. Soc. Neurosci. 2007, 27, 5200–5206. [Google Scholar] [CrossRef] [PubMed]
- Fino, E.; Deniau, J.M.; Venance, L. Cell-specific spike-timing-dependent plasticity in GABAergic and cholinergic interneurons in corticostriatal rat brain slices. J. Physiol. 2008, 586, 265–282. [Google Scholar] [CrossRef] [PubMed]
- Thirugnanasambandam, N.; Grundey, J.; Adam, K.; Drees, A.; Skwirba, A.C.; Lang, N.; Paulus, W.; Nitsche, M.A. Nicotinergic Impact on Focal and Non-Focal Neuroplasticity Induced by Non-Invasive Brain Stimulation in Non-Smoking Humans. Neuropsychopharmacology 2011, 36, 879. [Google Scholar] [CrossRef] [PubMed]
- Grundey, J.; Thirugnanasambandam, N.; Kaminsky, K.; Drees, A.; Skwirba, A.; Lang, N.; Paulus, W.; Nitsche, M. Rapid Effect of Nicotine Intake on Neuroplasticity in Non-Smoking Humans. Front. Pharmacol. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Grundey, J.; Thirugnanasambandam, N.; Kaminsky, K.; Drees, A.; Skwirba, A.C.; Lang, N.; Paulus, W.; Nitsche, M.A. Neuroplasticity in Cigarette Smokers Is Altered under Withdrawal and Partially Restituted by Nicotine Exposition. J. Neurosci. 2012, 32, 4156. [Google Scholar] [CrossRef] [PubMed]
- Grundey, J.; Thirugnasambandam, N.; Amu, R.; Paulus, W.; Nitsche, M.A. Nicotinic Restoration of Excitatory Neuroplasticity Is Linked to Improved Implicit Motor Learning Skills in Deprived Smokers. Front. Neurol. 2018, 9, 367. [Google Scholar] [CrossRef] [PubMed]
- Heatherton, T.F.; Kozlowski, L.T.; Frecker, R.C.; Fagerstrom, K.O. The Fagerstrom Test for Nicotine Dependence: A revision of the Fagerstrom Tolerance Questionnaire. Br. J. Addict. 1991, 86, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Rossini, P.M.; Burke, D.; Chen, R.; Cohen, L.G.; Daskalakis, Z.; Di Iorio, R.; Di Lazzaro, V.; Ferreri, F.; Fitzgerald, P.B.; George, M.S.; et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: Basic principles and procedures for routine clinical and research application. An updated report from an I.F.C.N. Committee. Clin. Neurophysiol. 2015, 126, 1071–1107. [Google Scholar] [CrossRef] [PubMed]
- Stefan, K.; Wycislo, M.; Classen, J. Modulation of associative human motor cortical plasticity by attention. J. Neurophysiol. 2004, 92, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Kujirai, K.; Kujirai, T.; Sinkjaer, T.; Rothwell, J.C. Associative plasticity in human motor cortex during voluntary muscle contraction. J. Neurophysiol. 2006, 96, 1337–1346. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, K.; Kacar, A.; Rothwell, J.C. Differential modulation of motor cortical plasticity and excitability in early and late phases of human motor learning. J. Neurosci. Off. J. Soc. Neurosci. 2007, 27, 12058–12066. [Google Scholar] [CrossRef] [PubMed]
- Toda, N.; Toda, H. Nitric oxide-mediated blood flow regulation as affected by smoking and nicotine. Eur. J. Pharmacol. 2010, 649, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Winstein, C.J.; Grafton, S.T.; Pohl, P.S. Motor task difficulty and brain activity: Investigation of goal-directed reciprocal aiming using positron emission tomography. J. Neurophysiol. 1997, 77, 1581–1594. [Google Scholar] [CrossRef] [PubMed]
- Sidtis, J.J.; Strother, S.C.; Rottenberg, D.A. The effect of set on the resting state in functional imaging: A role for the striatum? NeuroImage 2004, 22, 1407–1413. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Ma, L.; Wang, B.; Narayana, S.; Duff, E.P.; Egan, G.F.; Fox, P.T. Long-term motor training induced changes in regional cerebral blood flow in both task and resting states. NeuroImage 2009, 45, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Bella, R.; Lanza, G.; Cantone, M.; Giuffrida, S.; Puglisi, V.; Vinciguerra, L.; Pennisi, M.; Ricceri, R.; D’Agate, C.C.; Malaguarnera, G.; et al. Effect of a Gluten-Free Diet on Cortical Excitability in Adults with Celiac Disease. PLoS ONE 2015, 10, e0129218. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, G.; Lanza, G.; Giuffrida, S.; Vinciguerra, L.; Puglisi, V.; Cantone, M.; Pennisi, M.; D’Agate, C.C.; Naso, P.; Aprile, G.; et al. Excitability of the Motor Cortex in De Novo Patients with Celiac Disease. PLoS ONE 2014, 9, e102790. [Google Scholar] [CrossRef] [PubMed]
- Lanza, G.; Cantone, M.; Aricò, D.; Lanuzza, B.; Cosentino, F.I.I.; Paci, D.; Papotto, M.; Pennisi, M.; Bella, R.; Pennisi, G.; et al. Clinical and electrophysiological impact of repetitive low-frequency transcranial magnetic stimulation on the sensory–motor network in patients with restless legs syndrome. Ther. Adv. Neurol. Disord. 2018, 11, 1756286418759973. [Google Scholar] [CrossRef] [PubMed]
- Lanza, G.; Lanuzza, B.; Aricò, D.; Cantone, M.; Cosentino, F.I.I.; Bella, R.; Pennisi, G.; Ferri, R.; Pennisi, M. Impaired short-term plasticity in restless legs syndrome: A pilot rTMS study. Sleep Med. 2018, 46, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Ni, Z.; Gunraj, C.; Chen, R. Short interval intracortical inhibition and facilitation during the silent period in human. J. Physiol. 2007, 583 Pt 3, 971–982. [Google Scholar] [CrossRef]
- Kojima, S.; Onishi, H.; Sugawara, K.; Kirimoto, H.; Suzuki, M.; Tamaki, H. Modulation of the cortical silent period elicited by single- and paired-pulse transcranial magnetic stimulation. BMC Neurosci. 2013, 14, 43. [Google Scholar] [CrossRef] [PubMed]
- Chen, R. Interactions between inhibitory and excitatory circuits in the human motor cortex. Exp. Brain Res. 2004, 154, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, G.; Bella, R.; Lanza, G. Motor cortex plasticity in subcortical ischemic vascular dementia: What can TMS say? Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 2015, 126, 851–852. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, M.; Lanza, G.; Cantone, M.; Ricceri, R.; Spampinato, C.; Pennisi, G.; Di Lazzaro, V.; Bella, R. Correlation between Motor Cortex Excitability Changes and Cognitive Impairment in Vascular Depression: Pathophysiological Insights from a Longitudinal TMS Study. Neural Plast. 2016, 2016, 8154969. [Google Scholar] [CrossRef] [PubMed]
- Bella, R.; Ferri, R.; Lanza, G.; Cantone, M.; Pennisi, M.; Puglisi, V.; Vinciguerra, L.; Spampinato, C.; Mazza, T.; Malaguarnera, G.; Pennisi, G. TMS follow-up study in patients with vascular cognitive impairment-no dementia. Neurosci. Lett. 2013, 534, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Lanza, G.; Bramanti, P.; Cantone, M.; Pennisi, M.; Pennisi, G.; Bella, R. Vascular Cognitive Impairment through the Looking Glass of Transcranial Magnetic Stimulation. Behav. Neurol. 2017, 2017, 1421326. [Google Scholar] [CrossRef] [PubMed]
- Oldfield, R.C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lavender, A.P.; Obata, H.; Kawashima, N.; Nakazawa, K. Effect of Paired Associative Stimulation on Corticomotor Excitability in Chronic Smokers. Brain Sci. 2019, 9, 62. https://doi.org/10.3390/brainsci9030062
Lavender AP, Obata H, Kawashima N, Nakazawa K. Effect of Paired Associative Stimulation on Corticomotor Excitability in Chronic Smokers. Brain Sciences. 2019; 9(3):62. https://doi.org/10.3390/brainsci9030062
Chicago/Turabian StyleLavender, Andrew P., Hiroki Obata, Noritaka Kawashima, and Kimitaka Nakazawa. 2019. "Effect of Paired Associative Stimulation on Corticomotor Excitability in Chronic Smokers" Brain Sciences 9, no. 3: 62. https://doi.org/10.3390/brainsci9030062
APA StyleLavender, A. P., Obata, H., Kawashima, N., & Nakazawa, K. (2019). Effect of Paired Associative Stimulation on Corticomotor Excitability in Chronic Smokers. Brain Sciences, 9(3), 62. https://doi.org/10.3390/brainsci9030062





