1. Introduction
The main advantage of hair as a testing matrix is the ability to provide information relating to historical drug exposure. Hair analysis has many applications within forensic (e.g., drug-related deaths, drug-facilitated crimes (DFCs), child protection) and clinical toxicology (e.g., drug rehabilitation programs, workplace drug testing) [
1,
2,
3,
4].
The stability of drugs in hair, however, is affected by exposure to sunlight and weathering, cosmetic chemical treatments (i.e., oxidative dyeing, bleaching, or permanent wave), and physical damage [
5,
6,
7]. In particular, exposure to sunlight and/or artificial light for many hours per day can induce photodegradation (i.e., photolysis) of licit/illicit drugs through the formation of free radicals (produced by the drug itself or formed by eumelanin and pheomelanin, and their oxidative products oxyeumelanin, and oxypheomelanin) or photosensitization reactions by intermolecular energy transfer. [
8]
Previous studies on this matter have been published by Skopp et al. [
9] in which cannabinoids detected in hair and affected by solar radiation were shown to reduce in concentration. More recently, Favretto et al. [
10] evaluated the effect of light exposure on methadone, cocaine, and heroin metabolites in hair.
In order to better understand the role and underlying mechanisms of solar light exposure on decreasing concentrations of drugs in hair, and following our previous [
10] photodegradation studies on UVA- and UVB-induced changes, the aim of the present work was to evaluate the photodegradation of several common stimulant drugs (i.e., amphetamines and ketamine (KET)) in true positive hair samples exposed to the whole spectrum of sunlight in a solar simulator. The use of a solar simulator, including visible light (400–800 nm) and part of UV radiation from 400 nm to 3 µnm (UVA = 400–315 nm, UVB = 315–280 nm), mimics exposure to environmental sunlight.
Amphetamine-based drugs are illegal synthetic stimulants that share a common structural backbone. The four amphetamines considered in the present work are the most used in Europe: amphetamine (AMF), methamphetamine (MA), 3,4-methylenedioxymethamphetamine (MDMA), and 3,4-methylenedioxyamphetamine (MDA).
Amphetamine can be metabolized along two pathways: either by hydroxylation of the aromatic ring to 4-hydroxyamphetamine or by deamination of the side chain to benzyl methyl ketone, which can then be degraded to benzoic acid. Methamphetamine is metabolized by cytochrome P450 (CYP), mainly by the CYP2D and CYP3A subfamilies, leading to the production of 4-hydroxyamphetamine and AMF. The half-life of MA is about 10 h, and 35–45% of a dose is excreted unchanged in the urine over a period of several days. Other metabolites, such as 4-hydroxymethamphetamine, norephedrine, and 4-hydroxynorephedrine, can also be found in urine in substantial quantities [
11,
12,
13].
In addition, MDMA (i.e., 3,4-methylenedioxymethamphetamine, ecstasy) is a widely abused psychostimulant drug that acts as a powerful releaser and/or reuptake inhibitor of serotonin (5-HT), dopamine (DA), and norepinephrine (NE). Its metabolism depends on the following main metabolic pathways: (1)
O-demethylenation followed by catechol-
O-methyltransferase (COMT) methylation and/or glucuronide/sulfate conjugation; and (2)
N-dealkylation, deamination, and oxidation. MDMA
N-demethylation gives rise to 3,4-methylenedioxyamphetamine (MDA). The elimination half-life of MDMA is about 8–9 h, lower than those reported for MA (10–12 h) or AMF (12–15 h) [
14,
15].
Ketamine is a dissociative anesthetic drug that functions as an antagonist of the
N-methyl-
d-aspartate receptor and enhances the antinociceptive effects of conventional opioid analgesia, binding to μ opioid and σ receptors. Ketamine is increasingly misused as a recreational and “club drug” because of its hallucinogenic and stimulant effects, and also as a “date-rape” drug (to facilitate sexual assault). Ketamine is metabolized in the liver by the P450 system, and CYP3A4 is the main enzyme responsible for transforming
N-demethylation into norketamine (NKET), 4-hydroxy-ketamine, and 6-hydroxy-ketamine. The elimination half-life is about 2 h, and the predominant metabolite of urinary excretion over a 72 h period is dehydronorketamine (DHNK) (16%), along with conjugates of hydroxylated ketamine metabolites [
16,
17].
The consumption of alcohol and/or drugs is associated with an increased risk of being the victim of a sexual assault. A retrospective case series in London on 1014 cases of claimed drug-facilitated sexual assaults (DFSAs) showed that in 34% of samples (blood and/or urine), an illicit drug (with or without alcohol) was found, of which 10.8% contained cocaine, 4.6% “ecstasy” (MDMA), and 2.3% AMF [
18,
19]. In DFC cases, alleged victims are often unconscious leading up to the assault due to the amnesiac effects of the drug(s) administered, thus a considerable amount of time may be spent before the victim reports the incident. Consequently, hair analysis is of primary importance compared to blood and urine analysis, particularly when the occasional consumption is sufficiently high; however, exposure to the environment may affect the stability of drugs in the keratin matrix.
For these reasons, reliable interpretation of the analytical results is fundamental for a correct interpretation of a positive or negative result, and general knowledge of the photostability of drug analytes in the biological matrix must be considered. However, no data have been published until now on the effect of light on KET and amphetamines, and their respective metabolites in the keratin matrix; only morphine and 6-monoacetylmorphine (6-MAM) photodegradation have been studied by UVA and UVB irradiation [
20].
In the present paper, levels of AMF, MA, MDMA, MDA, KET, and NKET were determined by means of liquid chromatography high-resolution mass spectrometry (HPLC-HRMS) [
21,
22] in authentic hair samples from drugs users before and after irradiation under the whole spectrum of sunlight in a solar simulator.
Although the photodegradation of a molecule not only depends on the wavelength used, but also on its physical state (solid or liquid) and the environment in which it is irradiated (i.e., solvent, polarity, pH, and the presence of salts, oxygen, and other compounds in the sample), we tested the same analytes in a pure methanol and water solution irradiated under the whole spectrum of sunlight to gather general information on their behavior and to study their kinetics of photodegradation.
2. Experiments
2.1. Chemicals
Amphetamine, MA, MDMA, MDA, KET, and NKET were purchased from LGC Promochem Cerilliant (Teddington, Middlesex, UK) as pure solutions in methanol at 1.0 mg/mL. The internal standards (IS) MA-D5, AMF-D5, MDMA-D5, MDA-D5, and KET-D3 were methanol solutions also from LGC Promochem at 1.0 mg/mL.
Methanol, dichloromethane, and acetonitrile (Merck, Darmstadt, Germany) were high-performance liquid chromatography (HPLC) grade. Trifluoroacetic acid (TFA) was from Sigma (Sigma-Aldrich, Milan, Italy). High-performance liquid chromatography water was prepared using a Milli-Q Plus (Millipore, Molsheim, France) system. All other reagents were from Sigma-Aldrich (St Louis, MO, USA).
2.2. Hair Samples
Authentic positive hairs were selected from samples at the laboratory that had previously tested positive for amphetamines and/or KET. Hair samples were collected with scissors from the posterior vertex and cut as close to the scalp as possible, wrapped in aluminum foil, and kept at room temperature until analysis. As recorded in donor interviews, all samples had not been submitted to previous cosmetic or chemical treatments.
2.3. Irradiation Procedure
Irradiation was performed in a Suntest CPS+ (Atlas, Linsengericht, Germany) equipped with a 1.8 kW xenon lamp and a glass filter (cut-off 310 nm) according to Option 1 of ICH Guideline Q1B (European Medicines Agency, London, UK, ICH Topic Q1B–Photostability testing of new active substances and medical products, 1998). The dark samples were maintained at solar box temperature during irradiation.
2.4. Photolysis Experiments in Solution (In Vitro Study)
Solutions of the compounds at concentrations ranging from 10−5 to 10−4 M in methanol and water were irradiated in the Suntest CPS+ with times increasing from 1 h up to 3 h.
Photolysis was evaluated using Cary 50 UV-Vis spectrophotometer (Varian, Milan, Italy) analyzing the change in the original spectrum upon irradiation, as already described [
20], and by high-resolution mass spectrometry (HRMS). At selected UV doses, the solutions were diluted to 10
−6 M in methanol and water, and analyzed by direct injection HRMS to measure the photodegradation of the analytes by recording the decrease of the ion signal of protonated molecules obtained by electrospray ionization (ESI) of solutions kept in the dark. The results are the mean of at least three experiments.
2.5. Photolysis Experiments in Hair Samples (In Vivo Study)
Hairs 5–7 cm long were divided into two approximately identical strands: the former was put between two 5 × 5 cm optical glasses and exposed at 765 W/m2 (spectrum of irradiance: 310–800 nm) for 48 h in the solar simulator to an endpoint corresponding to two months exposure under the sunlight, and the latter was kept as a dark control in the same chamber of irradiation covered with an aluminum foil.
2.6. Hair Sample Preparation and Extraction
Hair samples were decontaminated with 3 mL of water and 3 mL of methanol. Pulverization was applied by the automatic homogenizer Precellys® Evolution (Bertin Technologies, Genoa, Italy) at a speed of 6000 RPM, cycle 9 × 30 s, pause 30 s. Solid phase extraction (SPE) was performed by Oasis MCX (Waters, Milford, MA, USA) cartridges. Sample preparation consisted of the addition of 2 ng/mg of IS and 3 mL of a methanol/TFA (90:10, v/v) solution to 25 mg powdered hair samples, ultrasonication for 1 h, and incubation overnight at 45 °C. After centrifugation, the methanolic solutions were dried under a stream of nitrogen at 40 °C. The residues were reconstituted in 3 mL of 0.1 M phosphate buffer pH 6 and subjected to SPE. After, cartridges of the sample conditioned with 3 mL methanol and 3 mL 0.1 M phosphate buffer pH 6 were loaded. Cleanup was accomplished by sequential washes with 3 mL of water, 3 mL of 0.1 N hydrochloric acid, and 3 mL of methanol. Cartridges were dried for 10 min under vacuum before elution with 2 mL of dichloromethane: 2-propanol (80:20, v/v) 2% ammonium hydroxide. Eluates were evaporated to dryness with nitrogen at 40 °C, reconstituted in 200 µL of water (0.1% formic acid)/acetonitrile 9:1 mixture, and 25 µL were injected into the LC-HRMS system.
2.7. High-Resolution Mass Spectrometry (HRMS)
All measurements were performed on an LTQ-Orbitrap (Thermo Fisher Scientific, Bremen, Germany) high-accuracy, high-resolution mass spectrometer operating in positive ESI mode and equipped with a Surveyor MS Pump.
2.8. Electrospray Ionization HRMS
For the analysis of pure standard solutions, direct injection analysis was performed with a syringe pump delivering solutions at 10 μL/min directly into the ESI source. The positive ion ESI parameters were as follows: capillary voltage 10 V, sheath gas flow rate 20 (arbitrary units, a.u.), auxiliary gas (N2) flow rate 5 (a.u.), sweep gas flow rate 5 (a.u.), and capillary temperature 275 °C. Profile full-scan mass spectra were acquired in the Orbitrap in the m/z range 120–700 with a target mass resolution of 100,000 (FWHM as defined at 400 m/z) and a scan time of 0.65 s.
2.9. High-Performance Liquid Chromatography HRMS
For the determination of drug concentrations in hair and in standard solutions, 25 μL of solutions or extracts were injected into an Atlantis T3 (150 × 1.0 mm, 3 µm) column (Waters Corporation, Milford, MA, USA). High-performance liquid chromatography separation was achieved by gradient elution at a constant flow rate of 300 μL/min. High-performance liquid chromatography conditions: A (water, 0.1% formic acid) and B (methanol, 0.1% HCOOH); initial conditions 10% B, linear gradient to 25% B in 4 min, 25% B hold from 4 min to 7 min, then ramped to 40% B in 5 min, to 60% B in 4 min and to 90% B in 2 min; 90% B hold from 18 min to 26 min. The column temperature was 40 °C. Mass spectrometry conditions were: positive ion ESI; capillary voltage 10 V, sheath gas flow rate 50 (arbitrary units, a.u.), auxiliary gas (N2) flow rate 5 (a.u.), sweep gas flow rate 5 (a.u.), and capillary temperature 275 °C. Profile full-scan mass spectra were acquired in the Orbitrap in the m/z range 120–700 with a target mass resolution of 60,000 (FWHM as defined at 400 m/z) and a scan time of 0.45 s. Detection of the analytes and the IS was based on retention time, accurate mass measurements of MH+ ions, and correspondence of the observed isotopic pattern to the calculated one. Drug concentrations were determined from peak area ratios of analyte to its IS compared to calibrator curves of peak area ratios to concentrations. The method was fully validated exhibiting a linear range from 0.1 ng/mg to 50 ng/mg (determined from regression with 1/x2 weighting utilizing six calibration points), lower limits of quantification of 0.01 ng/mg and limits of detection of 0.005 ng/mg for all the target analytes, intra-day imprecision, inaccuracy always lower than 19% and 20%, and inter-day imprecision and inaccuracy always lower than 21% and 22%, respectively. Extraction efficiency was determined in the range of 85–100% for the different substances.
4. Discussion
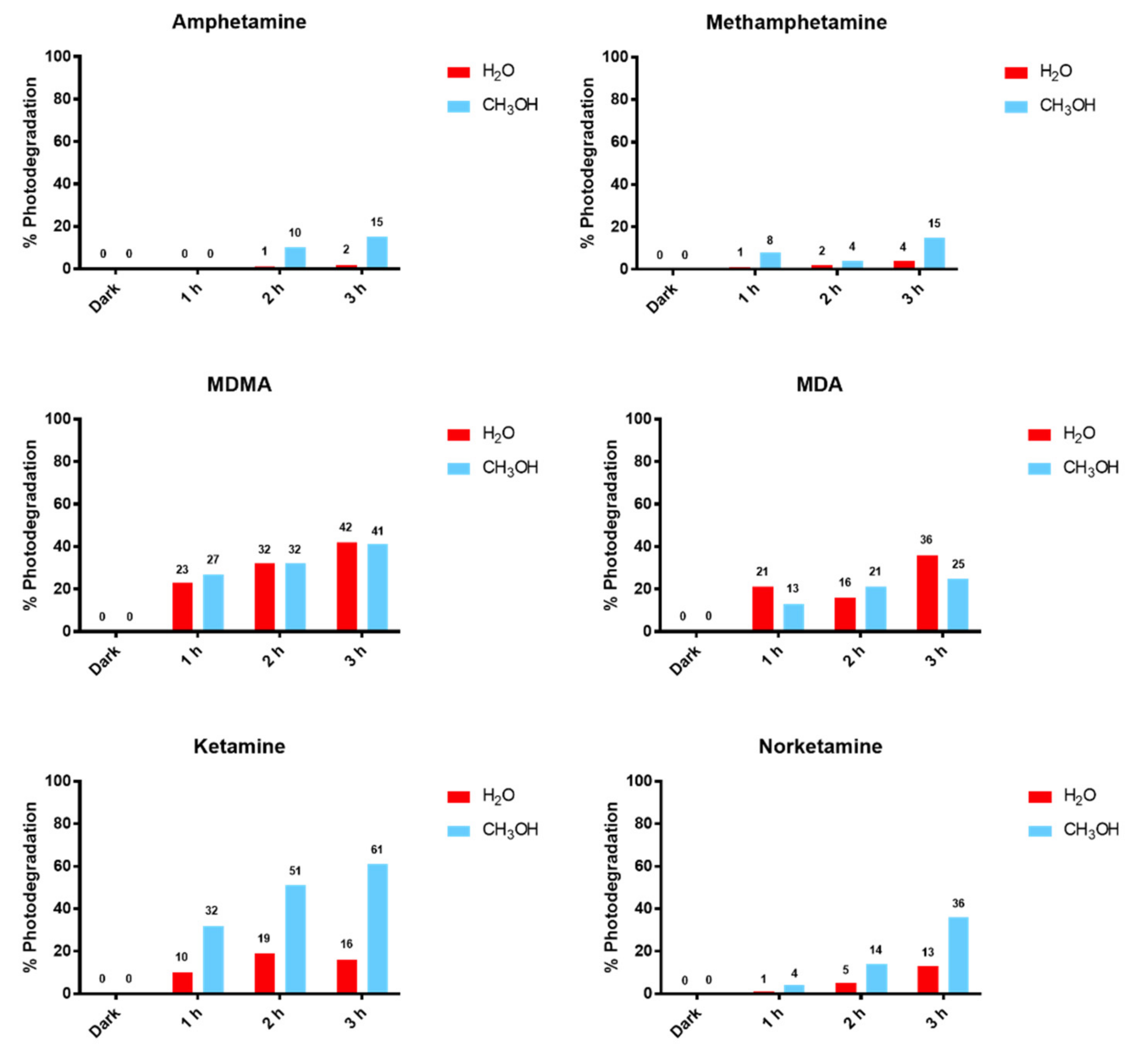
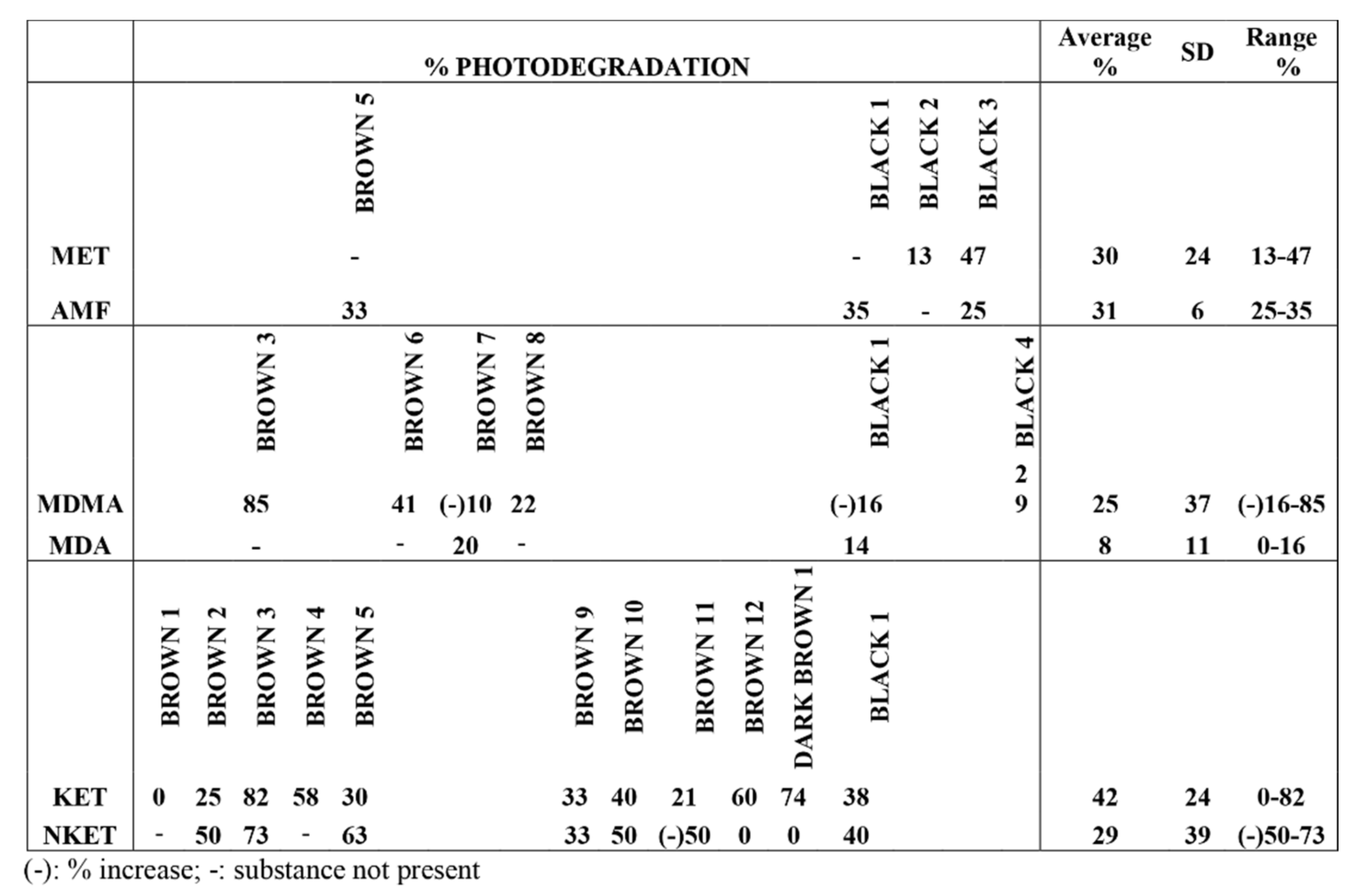
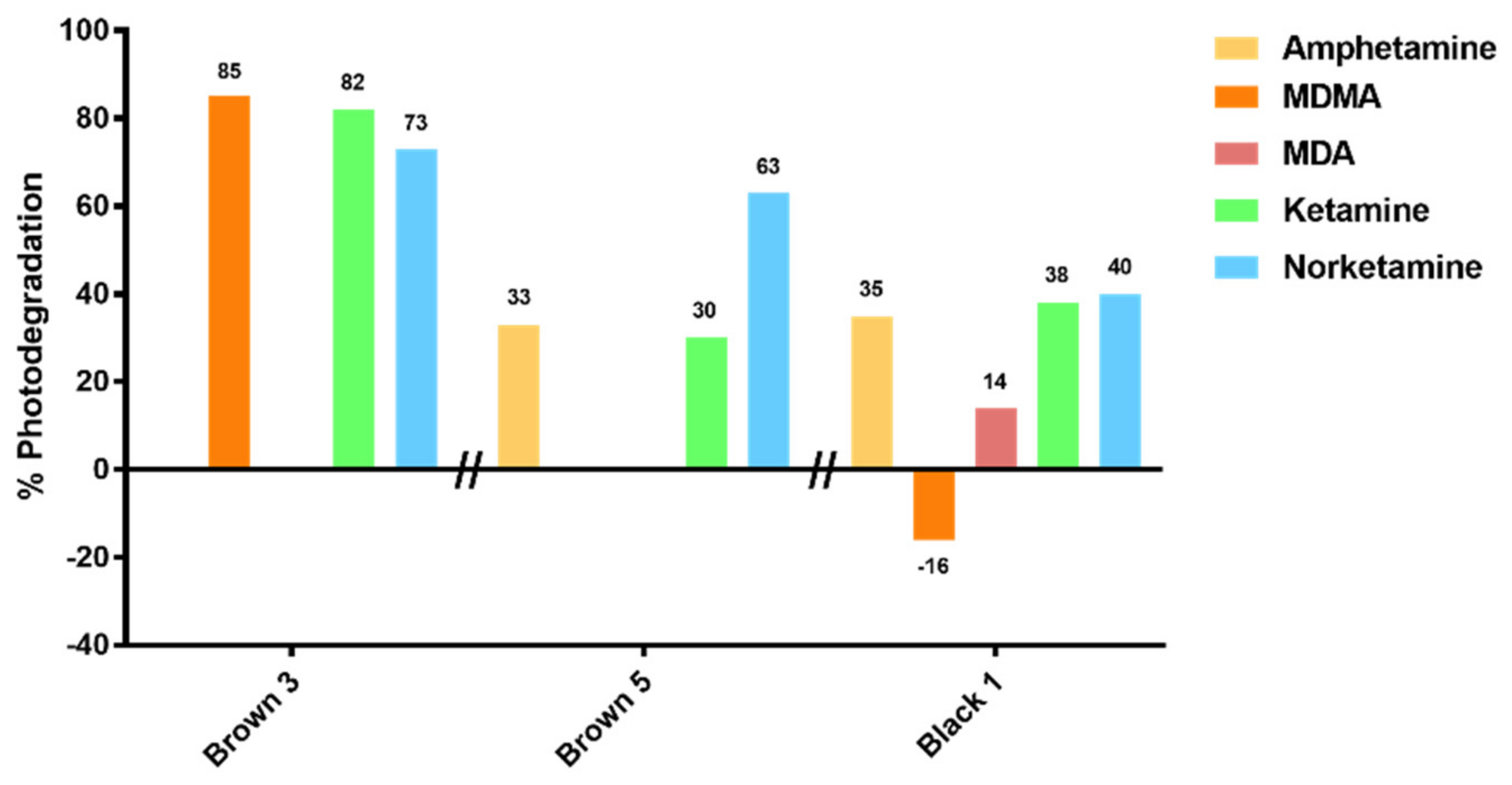
From our results, a clearly different behavior of KET and NKET was evidenced when compared to all the other drugs and metabolites under study. Indeed, these drugs seem photo-unstable, both in solution and in hair matrix. In particular, for the experiments in solution, KET and NKET photo-induced products were identified; in hair samples their degradation was higher (average 42% and 29%) than all the other compounds (average 8–30%).
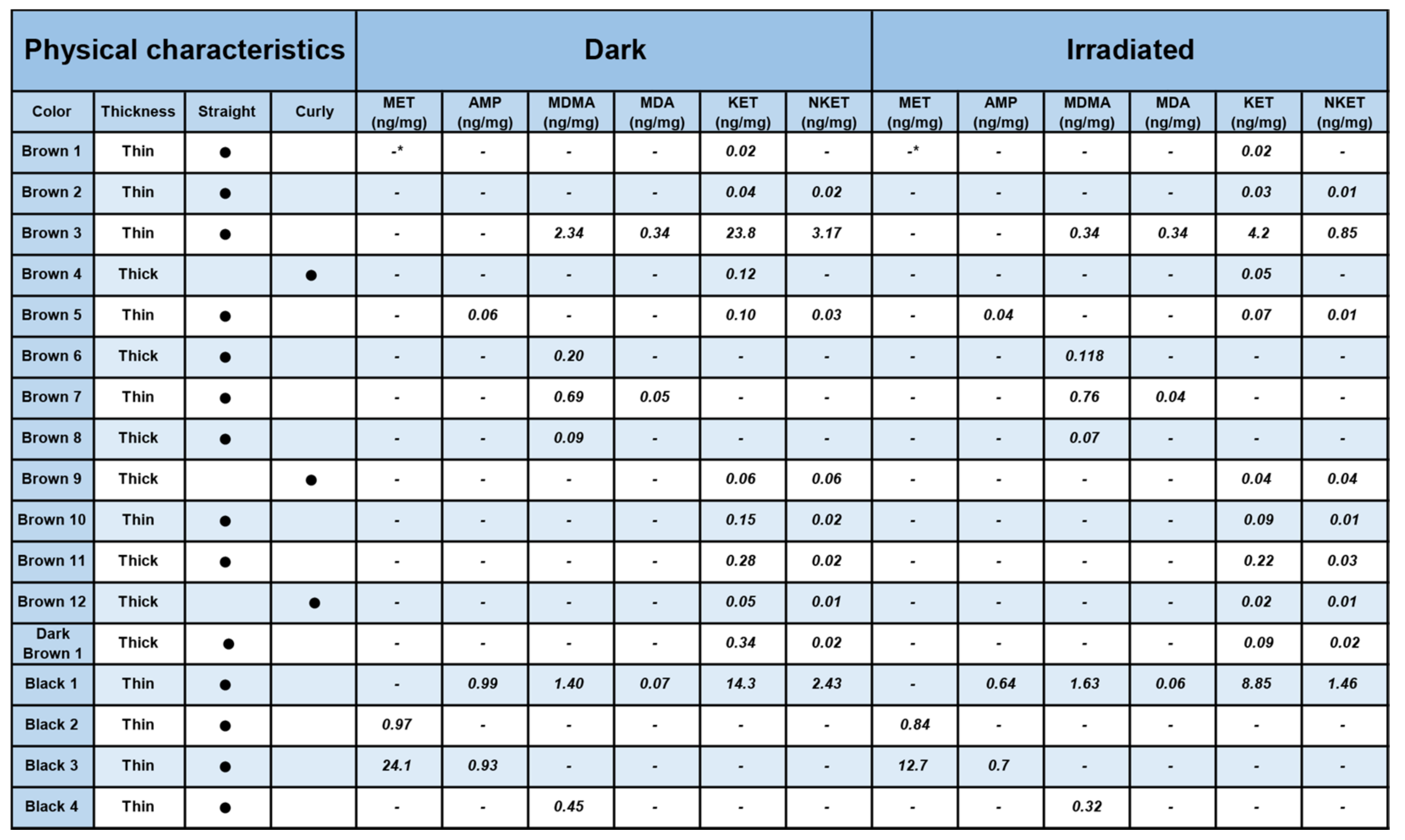
No color effect seems to be present, although no fair hair samples were present in this study, and the number of samples was limited. It is well known that the color of hair depends on the relative amount of pheomelanin (red) and eumelanin (black), the first defense against UV in human hair and skin [
23,
24,
25,
26]. Generally, a part of the light is absorbed by the hair matrix itself without any photochemical effect. In dark hair, the eumelanin can protect the drugs/metabolites with a higher degree than in fair hair. However, melanin may also react with oxygen under irradiation, producing reactive species, such as superoxide anion, that can induce photolysis of melanin itself [
27], thus weakening the photoprotective effect of the pigment. In this context, no clear-cut interpretation of the role of hair color can be made.
Regarding MA and AMF, on the basis of the in vitro experiments, they were expected to photodegrade less readily than KET and NKET. Unexpectedly, the experiments in hair revealed an average degradation of 30% and 31% for MA and AMF, respectively, with a range of 13–47% and 25–35%.
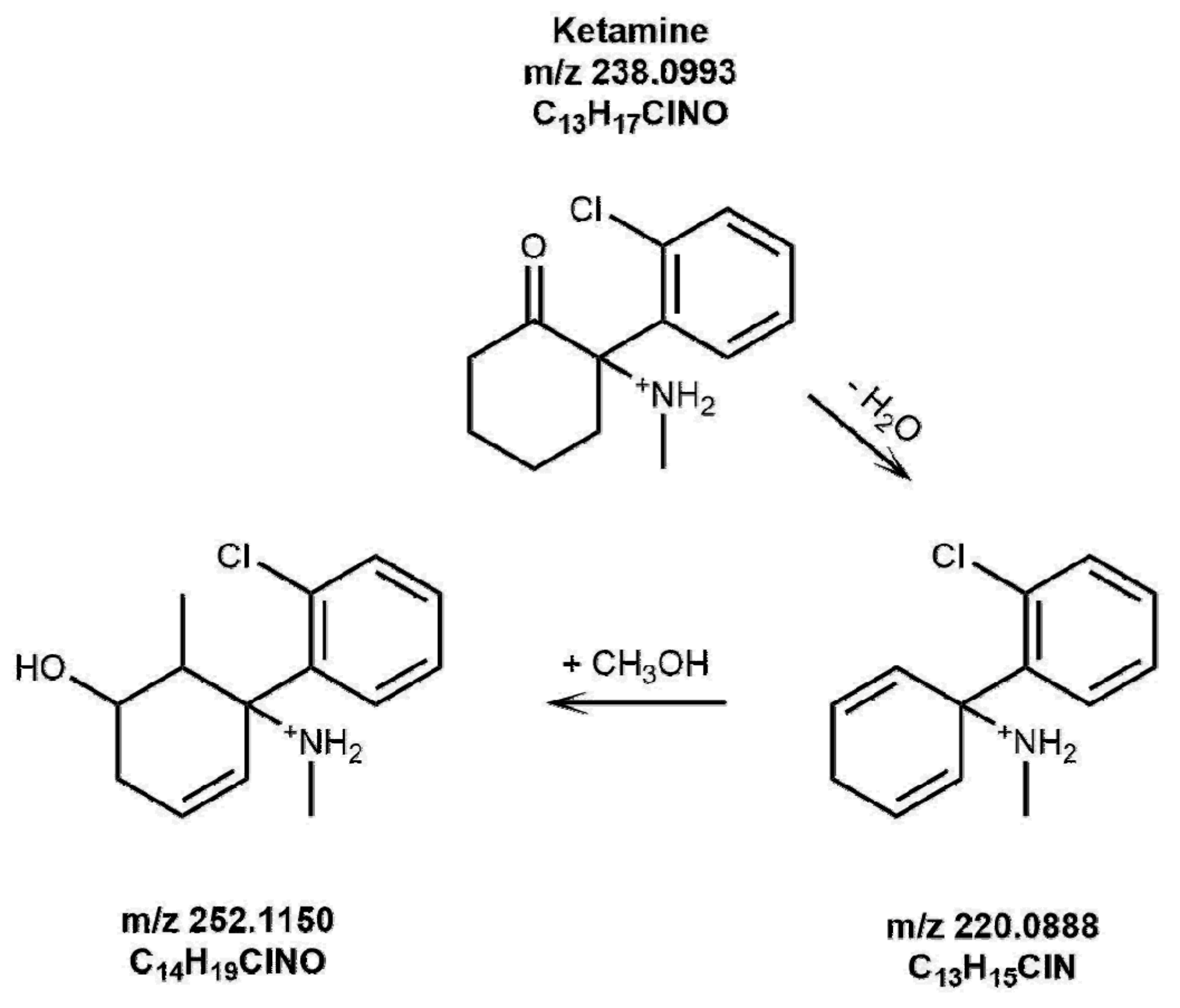
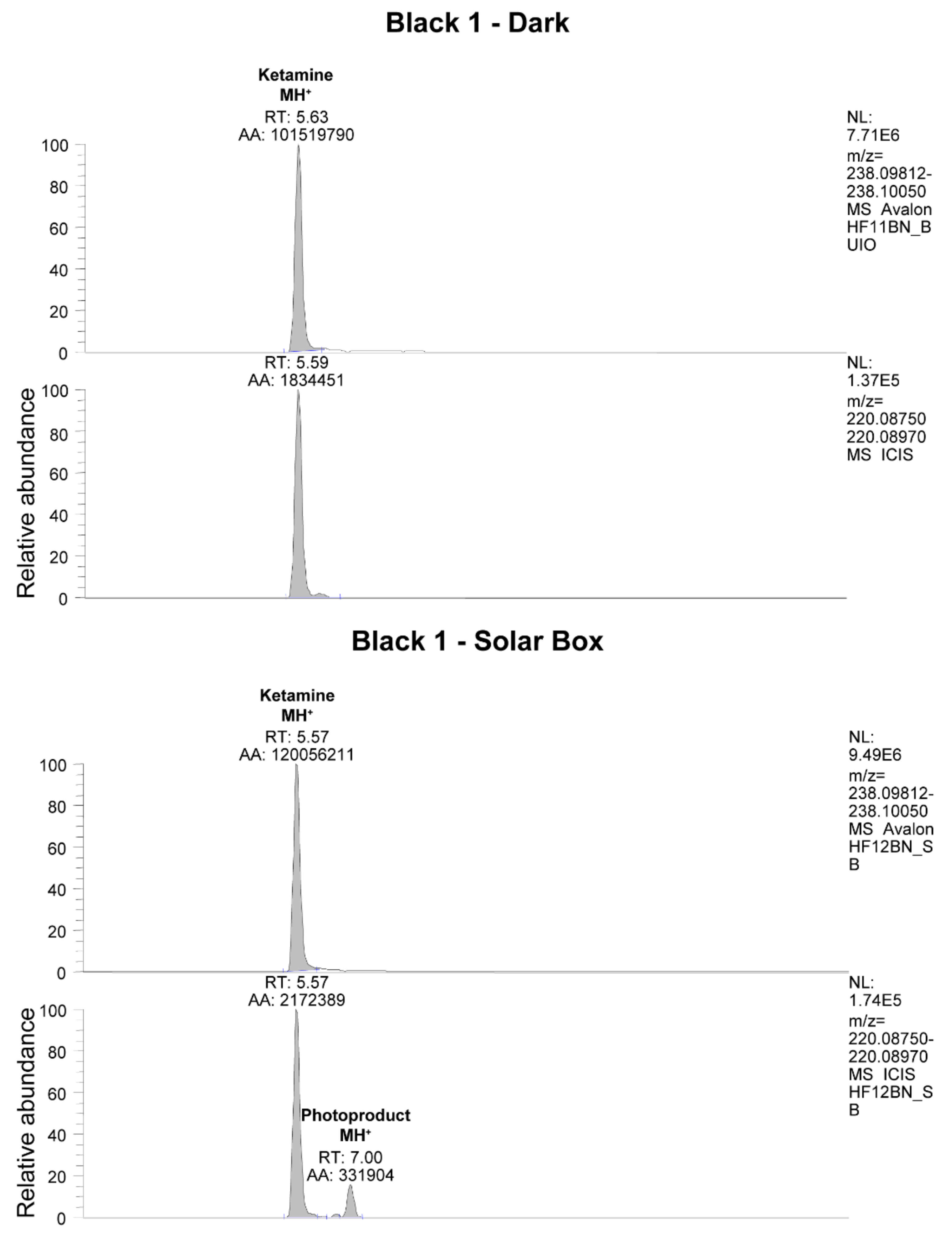
The presence of photoproducts was also investigated in irradiated hair samples. In one sample, the species at 220.08750
m/
z, corresponding to the photoproduct of KET shown in
Figure 1, could be identified by LC-HRMS as evidenced in
Figure 8.
The importance of this finding must be highlighted, since no previous study has identified stable photoproducts in hair for KET, nor for any other substance previously investigated (i.e., cannabinoids, cocaine, opiates, methadone [
9,
10,
20,
21]).
The “apparent production” of NKET upon irradiation of sample Brown 11 was at first sight surprising, but could be reasonably related to the physical decomposition of the matrix, leaving KET more labile during the irradiation experiment and favoring its transformation to the metabolite. Furthermore, the increase of MDMA in sample Black 1 and Brown 7 upon irradiation can be rationalized (as already demonstrated in previous studies) by a greater lability of the keratin matrix with consequent greater yield during the extraction of the analytes in the liquid acid phase with access to deeper layers of the hair structure.
5. Conclusions
Amphetamine, MA MDA, MDMA, KET, and NKET incorporated in hair undergo degradation when irradiated by artificial sunlight, suggesting that they can suffer photodegradation under natural sunlight. With this work, for the first time, the presence of a photoproduct of KET was evidenced in one true positive sample.
Since the detection of drugs in hair is often used as evidence of illegal acts with consequences on the freedom of persons, the UV-Vis effects on the integrity of the drugs and their metabolites should be considered when single administration needs to be evidenced. When decisional cut-offs are applied to hair analysis (e.g., for granting a driving license, a job, or a child custody), it must be taken into account that hair exposed to sunlight may produce false negative results and lead to misjudgment. When possible, the detection of photoproducts of a drug under investigation can be a key factor in a case management.